
WO 2015155704, An improved process for the preparation of pramipexole dihydrochloride monohydrate
PIRAMAL ENTERPRISES LIMITED [IN/IN]; Piramal Tower, Ganpatrao Kadam Marg Lower Parel Mumbai 400013 (IN)
|





PROUD Indian WORLD RECORD VIEWS holder on THIS BLOG, ………live, by DR ANTHONY MELVIN CRASTO, Worldpeaceambassador, Worlddrugtracker, Helping millions, 100 million hits on google, pushing boundaries,2.5 lakh plus connections worldwide, 45 lakh plus VIEWS on this blog in 227 countries, 7 CONTINENTS ……A 90 % paralysed man in action for you, I am suffering from transverse mylitis and bound to a wheel chair, [THIS BLOG HOLDS WORLD RECORD VIEWS ] A PROUD INDIAN

Mavatrep; UNII-F197218T99; Mavatrep (USAN); JNJ-39439335; 956274-94-5;
2-(2-(2-(2-(4-trifluoromethylphenyl)vinyl)-1H-benzimidazol-5-yl)phenyl)propan-2-ol
(E)-2-(2-{2-[2-(4-trifluoromethyl-phenyl)-vinyl]-1H-benzimidazol-5-yl}-phenyl)-propan-2-ol
(E)-2-(2-(2-(4-(Trifluoromethyl)styryl)-1H-benzo[d]imidazol-5-yl)phenyl)-propan-2-ol Hydrochloride
Phase I Musculoskeletal pain; Pain
| PHASE 1 Johnson & Johnson Pharmaceutical Research & Development, L.L.C. |
|
| Public title: | A Clinical Study to Investigate the Effect on Pain Relief of a Single Dose of JNJ-39439335 in Patients With Chronic Osteoarthritis Pain of the Knee |
http://clinicaltrials.gov/ct2/show/NCT01006304
http://apps.who.int/trialsearch/trial.aspx?trialid=NCT00933582
http://www.ama-assn.org/resources/doc/usan/mavatrep.pdf SEE STRUCTURE IN THIS FILE
MAVATREP IS JNJ-39439335
—
(E)-2-(2-(2-(4-(trifluoromethyl)styryl)-1H-benzo[d]imidazol-6-yl)phenyl)propan-2-ol hydrochloride

956282-89-6 CAS NO OF HCl SALT

CLICK ON IMAGE FOR CLEAR VIEW
Example 10 (E)-2-(2-{2-[2-(4-trifluoromethyl-phenyl)-vinyl]-1H-benzimidazol-5-yl}-phenyl)-propan-2-ol(Cpd 18)
Step A. 3-(4-trifluoromethyl-phenyl)-acrylic acid
Step B. (E)-5-bromo-2-[2-(4-trifluoromethyl-phenyl)-vinyl]-1H-benzimidazole
Step C. 2-(2-bromo-phenyl)-propan-2-ol
Step D. 3,3-dimethyl-3H-benzo[c][1,2]oxaborol-1-ol
Step E. (E)-2-(2-{2-[2-(4-trifluoromethyl-phenyl)-vinyl]-1H-benzimidazol-5-yl}-phenyl)-propan-2-ol
Example 10.1 Scale Up Preparation of (E)-2-(2-{2-[2-(4-trifluoromethyl-phenyl)-vinyl]-1H-benzimidazol-5-yl}-phenyl)-propan-2-ol (Cpd 18) Step A. 3-(4-trifluoromethyl-phenyl)-acrylic acid
Step B. (E)-5-bromo-2-[2-(4-trifluoromethyl-phenyl)-vinyl]-1H-benzimidazole
Step C. 2-(2-bromo-phenyl)-propan-2-ol
Step D. 3,3-dimethyl-3H-benzo[c][1,2]oxaborol-1-ol
Step E. (E)-2-(2-{2-[2-(4-Trifluoromethyl-phenyl)-vinyl]-1H-benzimidazol-5-yl}-phenyl)-propan-2-ol
Example 10.2 (E)-2-(2-{2-[2-(4-trifluoromethyl-phenyl)-vinyl]-1H-benzimidazol-5-yl}-phenyl)-propan-2-ol monosodium salt (Cpd 18)
Example 10.3 (E)-2-(2-{2-[2-(4-trifluoromethyl-phenyl)-vinyl]-1H-benzimidazol-5-yl}-phenyl)-propan-2-ol hydrochloride salt (Cpd 18)


Pfizer Inc., Oncology Institute Of Southern Switzerland INNOVATOR
http://chem.sis.nlm.nih.gov/chemidplus/rn/635715-01-4
FDA grants breakthrough status for Pfizer’s leukaemia drug inotuzumab ozogamicin
The US Food and Drug Administration (FDA) has granted breakthrough therapy designation for Pfizer’s investigational antibody-drug conjugate (ADC) inotuzumab ozogamicin to treat acute lymphoblastic leukaemia (ALL).
The US Food and Drug Administration (FDA) has granted breakthrough therapy designation for Pfizer’s investigational antibody-drug conjugate (ADC) inotuzumab ozogamicin to treat acute lymphoblastic leukaemia (ALL).
The breakthrough status was based on data from the Phase III INO-VATE ALL trial, which enrolled 326 adult patients with relapsed or refractory CD22-positive ALL and compared inotuzumab ozogamicin to standard of care chemotherapy………….http://www.pharmaceutical-technology.com/news/newsfda-grants-breakthrough-status-pfizer-leukaemia-drug-inotuzumab-ozogamicin-4697877?WT.mc_id=DN_News

PFIZER



| Besponsa | FDA
8/17/2017 |
To treat adults with relapsed or refractory acute lymphoblastic leukemia Press Release Drug Trials Snapshot |
Inotuzumab ozogamicin (CMC-544) is an antibody-drug conjugate for the treatment of cancers.[1] It consists of the humanized monoclonal antibody inotuzumab (for CD22), linked to a cytotoxic agent from the class of calicheamicins (which is reflected by ‘ozogamicin‘ in the drug’s name).[2]
This drug is being developed by Pfizer and UCB.
It is undergoing numerous clinical trials,[3] including two phase II trials for Non-Hodgkin lymphoma (NHL).
A phase III trial in patients with follicular b-cell NHL has been terminated due to poor enrollment.[4] A Phase III trial in patients with relapsed or refractory CD22+ aggressive non-Hodgkin lymphoma (NHL) who were not candidates for intensive high-dose chemotherapy was terminated for futility.[5]
Monoclonal antibodies (mAbs) and derivatives are currently the fastest growing class of therapeutic molecules. More than 30 G-type immunoglobulins (IgG) and related agents have been approved over the past 25 years mainly for cancers and inflammatory diseases. In oncology, mAbs are often combined with cytotoxic drugs to enhance their therapeutic efficacy. Alternatively, small anti-neoplastic molecules can be chemically conjugated to mAbs, used both as carriers (increased half-life) and as targeting agents (selectivity). Potential benefits of antibody-drug conjugates (ADCs), strategies, and development challenges are discussed in this review. Several examples of ADCs are presented with emphasis on three major molecules currently in late clinical development as well as next generation thio-mAbs conjugates with improved therapeutic index.

PATENT
http://www.google.com/patents/WO2013088304A1?cl=en
Inotuzumab ozogamicin:
is described in U.S. Patent Application No. 10/428894
U.S. Patent Application No. 10/428894



Structure of inotuzumab ozogamicin. ABOVE
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Humanized (from mouse) |
| Target | CD22 |
| Identifiers | |
| CAS Registry Number | 635715-01-4 |
| ATC code | None |
| UNII | P93RUU11P7 |
| KEGG | D08933 |
| Chemical data | |
| Formula | C6518H10002N1738O2036S42 |
| Molecular mass | 150,000 Daltons |
//////////

Regulation of Herbal (Traditional) Medicinal Products in the European Union
| Introduction | |
| The European Union (EU) regulatory framework for medicinal products is complex and is based on the need of a marketing authorization before placing medicines in the market. The main objective is to protect public health by assuring quality, efficacy and safety. The requirements and procedures to obtain a marketing authorization are laid down in regulations, directives and scientific guidelines which are contained in the “Rules Governing Medicinal Products in the European Union”. Several volumes are included which are supported by other publications with complementary information such as scientific or Good Manufacturing Practice (GMP) guidelines, between others [1]. | |
 |
|
| Medicinal plants have been used since Ancient times in all parts of the world. Nonetheless, regulation of herbal medicines in a legal environment was introduced in the 20th century. The EU regulatory framework includes specific requirements for herbal medicinal products (HMP) which are independent from their legal status: traditional herbal medicinal product (THMP) or products based on clinical evidence – well established use (WEU). | |
| Before a HMP is placed in the market, it must be approved by a MS or by the European Commission by one of the existing types of application: full marketing authorization application, well-established use marketing authorization application or Traditional use marketing registration (Table 1).
|
|
| The applicant has to submit adequate quality, non-clinical and clinical documentation of the product, irrespectively of the procedure used. Quality requirements of the pharmaceutical product are the same, regardless of the type of application, while efficacy documentation differs between them. The full marketing application is chosen for new medicinal products (new chemical entity) and it has to be completed with the results of pharmaceutical tests (quality documentation), nonclinical (toxicological and pharmacological) studies and clinical trials. Safety data have to be of sufficient size according the existing guidelines; efficacy is demonstrated by results from the clinical trials which have to be in conformity with the guidelines of the corresponding therapeutic area. This type of application is open for HMP, but only a few examples of herbal products have obtained a marketing authorization in the EU in this way. | |
| EU Pharmaceutical Legislation for Herbal Medicinal Products for Human Use | |
 |
|
| Quality requirements | |
| The principles to assure quality of medicinal products are defined mainly in two Directives of volume 1: Directive 2001/83/EC (which was emended by Directive 2004/24/EC) and Directive 2003/63/EC. | |
| The basic legislation lay down in Directive 2001/83/EC describes the general requirements and provides legal definitions of herbal substances, herbal preparations and herbal medicinal products (Table 2). These concepts are essential for setting quality standards for HMP, as they are by definition complex in nature and so quality requirements set for purified compounds are not suitable for herbal products. | |
| According to the Directive 2001/83/EC, monographs in the European Pharmacopoeia (Eur. Ph.) are legally binding and applicable to all substances which are included in it. For substances which do not have a Eur. Ph. monograph, each Member State (MS) may apply its own national pharmacopoeia. Constituents which are not given in any pharmacopoeia shall be described in the form of a monograph under the same headings included in any monograph in the Eur. Ph., i.e., the name of the substance supplemented by any trade or scientific synonyms; the definition of the substance, set down in a form similar to that used in the European Pharmacopoeia; methods of identification and purity tests. | |
| Moreover, all medicinal products have to be manufactured according to the principles and guidelines of GMP for medicinal products. GMP are applicable to both finished HMP and active substances and, according to Article 46 (f) of Directive 2001/83/EC as amended, marketing authorization holders are required to use as starting materials only active substances which have been manufactured in accordance with the guidelines on the GMP for starting materials as adopted by the Community and distributed in accordance with good distribution practices for active substances. | |
| |
|
| Additional requirements are found in the Directive 2003/63/EC, as Herbal medicinal products differ substantially from conventional medicinal products in so far as they are intrinsically associated with the very particular notion of herbal substances and herbal preparations. It is therefore appropriate to determine specific requirements in respect of these products with regards to the standardized marketing authorization requirements. Then, detailed information on the herbal medicinal product, herbal substances and herbal preparations has to be included, such as the name, address and responsibility of each herbal substance supplier or description of the plant production process, geographical source or drying and storage conditions. The application dossier of a HMP should include specifications and details of all the analytical methods used for testing herbal substances and herbal preparations, results of batch analyses and analytical validation, together with the justification for the specifications. | |
| Most of the quality requirements for HMP are laid down in soft laws (considered as EU measures such as scientific guidelines) which do not have legal force but provide practical harmonization between the MS and the European Medicines Agency (EMA). | |
| The guideline on quality of HMP/THMP covers the general quality aspects of HMP for human and veterinary use, including THMP for human use (EMA, 2014) and indicates which information has to be included in the application dossier. It provides definitions to be taken in account such as genuine (native) herbal preparations, markers, drug to extract ratio (DER) and specifications. Which is more important, it states that the herbal substance or herbal preparation is considered as the whole active substance. In consequence, the quality control of these products has to include appropriate fingerprint analysis to cover not only the content of markers or constituents with known therapeutic activity but also a wider range of chemical constituents. | |
| |
|
| Efficacy requirements | |
| Before a HMP is placed in the market, it must be approved by a MS or by the European Commission by one of the existing types of application: full authorization application, well-established use authorization application or Traditional use registration (Table 3). | |
| The applicant has to submit adequate quality, non-clinical and clinical documentation of the product, irrespectively of the procedure used. Quality requirements of the pharmaceutical product are the same, regardless of the type of application, while efficacy documentation differs between them. The full marketing application is chosen for new medicinal products (new chemical entity) and it has to be completed with the results of pharmaceutical tests (quality documentation), nonclinical (toxicological and pharmacological) studies and clinical trials. Safety data have to be of sufficient size according the existing guidelines; efficacy is demonstrated by results from the clinical trials which have to be in conformity with the guidelines of the corresponding therapeutic area. This type of application is open for HMP, but only a few examples of herbal products have obtained a marketing authorization in the EU in this way. | |
| The well-established medicinal use (WEU) in the EU can be applied to medicinal products for which there exists a wide clinical experience within the EU (not only HMP). The assessment may be based in published controlled clinical trials, non-clinical studies and epidemiological studies. In this type of application, there are no limitations to the therapeutic indication, as this will be derived from the available documentation. | |
| |
|
| (Traditional) Herbal Medicinal Products | |
| Under the Traditional use registration for herbal medicinal product (article 16e), there exist some herbal products that not fulfill the efficacy requirements for a marketing authorization but are endorsed with a long tradition of use. In this case, no clinical trials on these products have been conducted and the efficacy is based on the long-standing use and experience. This simplified registration procedure is limited to products which are intended for use without medical supervision, with a specified strength and posology, to be used by oral, external or inhalation ways, and which can demonstrate a period of use equal or superior to 30 years, including at least 15 years within the EU. In this case, therapeutic indications are limited to those which can be considered safe for use without the supervision of a physician such as minor disorders or symptoms that are benign or self- limiting. In case the applicant should consider another kind of indication, the product must be documented with results of clinical and non-clinical studies, so a full application would be necessary. | |
| Simplified registration of THMP is described in Chapter 2a of Directive 2004/24/EC with three main objectives: a) to protect public health by allowing access to safe and high-quality HMP; b) to allow European citizens the access to medicines of their choice, even those HMP with a long tradition of use and which efficacy hasn’t been proved by clinical trials performed according the modern standards; c) to facilitate movement of medicinal products on the European market. | |
| Directive 2004/24/EC has two different dimensions: the evaluation by National Competent Authorities (NCA) of applications submitted by companies at any MS in the EU and at the EMA, and the establishment of advisory scientific opinions on the medicinal use of herbal substances or preparations. The directive on THMP also established a new scientific committee, the Herbal Medicinal Products Committee (HMPC) at the EMA in London, in 2004, to replace the previous Working Party on Herbal Medicinal Products (CPMP) with the following aims: to elaborate Community monographs and List entries for herbal substances/preparations; to publish scientific guidelines useful for the application of European legal framework; to publish its scientific opinion on questions related to herbal medicinal products and coordinate its work with the European Quality group. The HMPC is made up by 33 members, one member (and one alternate) nominated by each MS of the EU and by Iceland and Norway (the EFAEFTA states). Among them, also five experts are included, representing specific fields of expertise as clinical and non-clinical pharmacology, toxicology or pediatrician medicine. | |
| The guidelines and the monographs developed and approved by the HMPC are accepted by both companies and NCAs and are used for TUR and WEU marketing authorizations. This committee plays a key role in the harmonization of the regulation of HMP whereby Community herbal monographs have a fundamental role. | |
| Usage of Community herbal monographs in the EU regulation of traditional HMP | |
| These documents are established for HMP with regards to bibliographic applications (art. 10 a Directive 2001/83/EC) as well as THPMs. Community monographs reflect the scientific opinion of the HMPC on safety and efficacy data concerning a herbal substance. Any single plant or herbal preparation is assessed individually, according to the available information and includes qualitative and quantitative composition, pharmaceutical form(s), therapeutic indication(s), posology and method of administration, contraindications, special warnings and precautions of use, interactions, use in special population (pregnancy, lactation), effects on ability to drive and use machines, undesirable effects, overdose, pharmacological,pharmacodynamics, pharmacokinetic properties and preclinical safety data. | |
| Community list entry | |
| In the EU, a community list of herbal substances, preparations and combinations thereof for use in THMPs has been established. This list is based in the proposals form HMPC and is gradually developed. Substances or preparations which are included in the list have the main advantage that applicants do not need to provide evidence on the safe or traditional use for its registration at the NCA in the intended use and indication. | |
| A Public statement for one herbal substance/preparation is published because of safety reasons or lack of data to comply with the conditions in the Directive 2004/24/EC (the assessment work didn’t allow a monograph to be published) [2]. | |
| Community monographs are published by the EMA while list entries are approved and published by the European Commission because they are endorsed with a wider legal status: list entries are legally binding and NCAs should not request additional data on safety and traditional use. | |
| The establishment of monographs and list entries is based on the assessment of the published scientific data, together with the existing products in the market. Most of the assessment work is developed by the Monograph and List Working Party (MLWP) at the HMPC, which was established in 2006. In this working group, a member is designed as rapporteur and is responsible of drafting a monograph and/or list entry which will be later on considered and approved by the HPC and then, by the EMA. The documents are published on the EMA website: Community monographs have to be taken in account by the MS when assessing the application of any company. Monographs are note legally binding and MS are not obliged to follow the monographs. | |
| More than 100 species are included in the priority list with the following data: a) scientific data being assessed (R- Rapporteur assigned); b) evaluation report in progress and discussion in the MLWP (D- Draft under discussion); c) scientific opinion under public consultation (PDraft Published); d) comments after public consultation period being evaluated (PF- Assessment close to finalization – pre-final); e) final opinion adopted (F- Final opinion adopted). | |
| MLWP is also responsible of developing guidelines related to legal requirements for TU and WEU, as well as evaluating hazards and problems related to HMP. For the latter, coordination is established with the Safety Working Party (SWP) from the Committee for Medicinal Products for Human Use (CHMP). | |
| Community herbal monographs to support HMPC authorization | |
| A community monograph reflects the scientific opinion from the HMPC in relation to safety and efficacy of one herbal substance/ preparation for medicinal use. AS stated before, a community monograph may be used by a company for a TU or WEU application. That’s the reason why monographs are divided in to two columns: Well Established Use and Traditional Use (simplified application) (Figure 1). WEU is based in the existence of safety data of sufficient size and efficacy data derived from good-quality clinical trials. Traditional use is accepted for those applications which fulfill the criteria shown in the Directive 2004/24/EC. | |
| Each herbal substance/preparation is assessed individually, as the available information may be different for each one. As a result, some substances/preparations may be included in the WEU side, while others will be included in the TU side. If no enough data are available for the substance/preparation, it won’t be included in the monograph. | |
| The approved draft art he HMPC is published for public consultation for 3 month at the EMA website. Comments received are discussed and taken in account when necessary to achieve the final version of the monograph which will be finally published at the MA website. | |
| By the end of 2014, 126 monographs have been adopted and published by the EMA: 104 of them for TU only; 9 of the monographs refer only to WEU (Aloe vera, Cimicifuga racemosa, Rhamnus frangula, Plantago ovata – seed and tegumentum-, Plantago afra, Rheum palmatum, Cassia senna – leaves and fruits-.among them 13 monograph include both TU and WEU. | |
| The main application of a community monograph is to serve as a reference material for the marketing application, both for TU or WEU. Simplified registration is carried on at a national level, so the company gives the dossier to the NCA. With the aim of improving harmonization, the other MSs should recognize the first authorization granted in the first MS, considering that this is based in the European list. | |
| Directive 2004/24/CE established an adaptation period for those herbal products which were on the European market at the moment the Directive was approved. This seven-year period finalized last April 30th, 2011 and implies that nowadays those herbal preparations that not fulfill the actual legislation will not be marketed any more. | |
| In the public report form the EMA last June 2014, the status of updating the medicines registration in the EU was shown. The number Traditional use registrations (TUR) and Well-established use marketing authorizations (WEU) grouped for mono component and combination products has increased in the last years (Figure 2). | |
| The European market for HMP is increasing during the last years and even exceeds prescription medicines. The indications approved cover a wide range of therapeutic areas, most of them characteristic of self-medication diseases: the main therapeutic areas are respiratory tract disorders (cough and cold), mental stress and mood disorders, urinary tract and gynecology disorders, sleep disorders and temporary insomnia (Figure 3). Most of the approved THMP until now were updates of existing authorizations and were based on Community monographs. The Summary of Product Characteristics (SoPC) reflects the items in the corresponding monograph [3]. | |
| A good correlation between the HMPC work and the evaluation of the dossiers from the companies was detected. The relevance of these documents (as shown by the accepted dossiers) is reflected in the HMPC working plan; as an example, last December 2012, 54 among the 56 species with more than 3 marketing authorizations were listed in the priority list. | |
| Conclusión | |
| European legal framework for medicinal products does also include herbal medicinal products to assure their quality, efficacy and safety. The specific characteristics of these products led to the development of a simplified procedure to assure pharmaceutical quality, while keeping safety and efficacy criteria according to marketing authorization granted. | |
| Although the starting point was quite different for the MS, nowadays there exist Community monographs for most of the herbal substances/ preparations that are used in the European market and which form the basis for a harmonization scenario. Moreover, HMPC acts as an International Regulatory Body for herbal medicinal products in order to achieve global standards for this type of medicines, according to other International organization such as the International Conference on Harmonization (ICH). The main tasks the HMPC has to face are those related to herbal medicinal products which have been previously marketed abroad the EU and the increasing existence of combination products within the MS. | |
| References | |
|
|
|
|
||||||||||||
| Table 1: Types of applications for marketing authorization for a HMP in the EU according the Directive 2001/83/EC. |
|
||||||
| Table 2: Definitions applicable to herbal medicinal products (Directive 2001/83/EC). |
|
||||||
| Table 3: Definitions applicable to herbal medicinal products (Directive 2001/83/EC). |
 |
| Figure 1: European Community Monograph for Valeriana officinalis L., radix, for WEU and TU |
Ruiz-Poveda OMP*
Department of Pharmacology, Faculty of Pharmacy, Universidad Complutense de Madrid, 28040 Madrid, Spain
Ruiz-Poveda OMP
Department of Pharmacology, Faculty of Pharmacy
Universidad Complutense de Madrid, Madrid
Ciudad Universitaria s/n. 28040 Madrid, Spain
Tel: 913 941 767
Fax: 913 941 726
E-mail: olgapalomino@farm.ucm.es
Citation: Ruiz-Poveda OMP (2015) Regulation of Herbal (Traditional) Medicinal Products in the European Union. Pharmaceut Reg Affairs 4:142. doi: 10.4172/2167-7689.1000142
/////////Herbal medicines, Good manufacturing practices, Traditional uses
EMA: European Medicines Agency; DER: Drug to Extract Ratio; HMP: Herbal Medicinal Products; THMP: Traditional Herbal Medicinal Products

Dr. Ashok Kumar
The 25-year process patent regime allowed a large number of generic companies in India to reap rich dividends, but there were few who believed in the need to go beyond the horizon of process development to tap into unexplored terrains.
In the year 2000, when Dr Ashok Kumar joined the board of Mumbai-based IPCA Labs, he was determined to implement a different strategy to accelerate IPCA’s R&D initiatives. Having seen IPCA grow from a 350-crore company to one clocking an annual turnover of over 2,500 crore, Dr Ashok Kumar, president- Center for Research and Development, IPCA Labs, is now leaving no stone unturned to exploit the biotech and drug discovery space.

Dr Kumar completed his M Sc in Chemistry from Kumaun University, now in Uttarakhand. He then decided to pursue his PhD in organic chemistry and joined Banaras Hindu University (BHU), but opted out three months later to do PhD from the Central Drug Research Institute (CDRI), Lucknow, under the guidance of the then director of the CDRI, Dr Nityanand.
Dr Kumar did his post doctoral studies from the University of Sussex, UK. “During the 1980s, jobs in scientific research were not available in India. It was always good to go for higher studies abroad,” he says about the reason for going abroad. “Dr Nityanand taught me to be explorative and think of new ways to approach a subject. I still follow that process,” he says. In 1984, Dr Kumar decided to return to India and took up a job at the Imperial Chemical Laboratories (ICI), Mumbai. In 1994, he joined Lupin Labs where he was once again involved in process development of small molecules.

In 2000, he joined IPCA Labs where he immediately focused on bringing about two changes – introducing a library and bringing in systems like a nuclear magnetic resonance (NMR), which at that time was the costliest instrument. “I understood the importance of high-end technologies since my PhD years at the CDRI (which housed a couple of NMRs) and then in the UK. You do not enjoy organic chemistry without an NMR. My main objective at IPCA was cost reduction along with process development.”
In the last few years, IPCA’s R&D team has brought out over 100 products. Under Dr Kumar, IPCA has an R&D center in Mumbai and another parallel R&D center in Ratlam, Madhya Pradesh. “In Mumbai, we have around 60 people. The Mumbai team takes care of basic chemistry and small-scale development. Scaling up is done in Ratlam,” he explains. IPCA is also coming up with a facility at Vadodara, Gujarat, that will look into large-scale manufacture of both organic and biotech drugs. The facility will have a strategic importance for the company. “We are growing at a rate of 20 per cent year-on-year and, next year, we intend to add 500 crore to our revenue. For that, we need more products to come to the market and more volume,” he adds.
Apart from organic chemistry, Dr Kumar is currently aligning his attention to two promising but high risk segments – fermentation-based products and biosimilars. “We are working on five-to-six molecules, mainly active metabolites that are intermediates or biotech drugs,” he adds. IPCA has also collaborated with two companies in India for the development of biosimilars. Currently, there are three biosimilar products in the pipeline.
The R&D team at IPCA ventured into drug discovery three years ago. It has two products in the pipeline; one anti-malarial and the other anti-thrombotic. “We will be filing the investigational new drug application for one molecule this year and for the other next year. The success rate here is 99 per cent,” adds Dr Kumar. IPCA has also joined hands with the CDRI and licensed two molecules in the anti-malarial space. One of these molecules is currently in phase-I stage.
Innogen summit India 2016, 18-19 Aug, Mumbai, India, HOTEL HOLIDAY INN, Mumbai International Airport,Organised by Inventicon Business Intelligence Pvt. Ltd………topic is Supergenerics, Innovation in Generics, commercialization, regulatory, other insights,
Dr. Ashok Kumar, President – Centre for Research & Development, Ipca Laboratories Ltd, at Innogen summit India 2016, 18-19 Aug, Mumbai, India,, HOTEL HOLIDAY INN, Mumbai International Airport,Organised by Inventicon Business Intelligence Pvt. Ltd — with DR ASHOK KUMAR OF IPCA at Holiday Inn-Mumbai Intl Airport.

PANEL DISCUSSION, Dr. Ashok Kumar, President – Centre for Research & Development, Ipca Laboratories Ltd , Dr. Nilima A. Kshirsagar, National Chair Clinical Pharmacology, ICMR Government of India, Yugal Sikri, Chairman – Pharmaceutical Management, School of Business Management, SVKM’s Narsee Monjee Institute of Management Studies — with Yugal Sikri,, Nilima A. Kshirsagarand ASHOK KUMAR OF IPCA at Holiday Inn-Mumbai Intl Airport.




2005 – Present (10 years)
chlorthalidone is 3-hydroxy-3-(3′-sulfamyI-4′- chlorophenyl)phtalimidine and is represented by the structural formula shown below.
(Scheme 2). The starting material, 2-(4′-chlorobenzoyl) benzoic acid, of Formula (2) and its preparation was reported earlier for example in patents US 4500636, US 30555904, US4379092, US 3764664.
Formula 9 CIS03H
Chlorthalidone Formula 10 Formula 1
https://www.google.co.in/patents/US7847094Quetiapine and its process for preparation is first disclosed in the patent specification EP0240228 and various other processes for the preparation are disclosed in EP0282236, WO0155125, WO9906381, WO2004076431.
http://www.google.com/patents/US20080009635
. The chemical name of Ondansetron is 1,2,3,9-tetrahydro-9-methyl-3-[(2-methyl)-1H-imidazole-1-yl)methyl]-4H-carbazol-4-one and is represented by the structural formula given below:




Chemical Research & Development Centre
123-AB, Kandivli Industrial Estate, Kandivli (West)
Mumbai 400 067, Maharashtra







October 20, 2015
The U.S. Food and Drug Administration today approved Coagadex, Coagulation Factor X (Human), for hereditary Factor X (10) deficiency. Until today’s orphan drug approval, no specific coagulation factor replacement therapy was available for patients with hereditary Factor X deficiency.
In healthy individuals, the Factor X protein activates enzymes to help with normal blood clotting in the body. Factor X deficiency is an inherited disorder, affecting men and women equally, where the blood does not clot as it should. Patients with the disorder are usually treated with fresh-frozen plasma or plasma-derived prothrombin complex concentrates (plasma products containing a combination of vitamin K-dependent proteins) to stop or prevent bleeding. The availability of a purified Factor X concentrate increases treatment options for patients with this rare bleeding disorder.
“The approval of Coagadex is a significant advancement for patients who suffer from this rare but serious disease,” said Karen Midthun, M.D., director of the FDA’s Center for Biologics Evaluation and Research.
Coagadex, which is derived from human plasma, is indicated for individuals aged 12 and older with hereditary Factor X deficiency for on-demand treatment and control of bleeding episodes, and for perioperative (period extending from the time of hospitalization for surgery to the time of discharge) management of bleeding in patients with mild hereditary Factor X deficiency.
The safety and efficacy of Coagadex was evaluated in a multi-center, non-randomized study involving 16 participants (208 bleeding episodes) for treatment of spontaneous, traumatic and heavy menstrual (menorrhagic) bleeding episodes. Coagadex was demonstrated to be effective in controlling bleeding episodes in participants with moderate to severe hereditary Factor X deficiency. Coagadex was also evaluated in five participants with mild to severe Factor X deficiency who were undergoing surgery. The five individuals received Coagadex for perioperative management of seven surgical procedures. Coagadex was demonstrated to be effective in controlling blood loss during and after surgery in participants with mild deficiency. No individuals with moderate or severe Factor X deficiency received Coagadex for perioperative management of major surgery, and no safety concerns were identified in either study.
The FDA granted Coagadex orphan product designation for these uses. Orphan product designation is given to drugs intended to treat rare diseases in order to promote their development. Coagadex was also granted fast track designation and priority review.
Coagadex is manufactured by Bio Products Laboratory Limited in Elstree, Hertfordshire, United Kingdom.

TAK 272
C27 H41 N5 O4 . Cl H, 536.106
CAS.1202269-24-6. MonoHCl
1202265-90-4 DIHCL
Base cas…1202265-63-1
Metanesulfonate…1202266-34-9
Takeda Pharmaceutical Company Limited, INNOVATOR
see……….http://www.allfordrugs.com/2015/10/21/tak-272-for-hypertension-takedas-next-sartan/
1-(4-methoxybutyl)-N-(2-methylpropyl)-N-[(3S,5R)-5-(morpholin-4-ylcarbonyl)-piperidin-3-yl]-1H-benzimidazole-2-carboxamide
1- (4-methoxybutyl) -N- (2-methylpropyl) -N- [ (3S, 5R) -5- (morpholin-4-ylcarbonyl) piperidin-3-yl] -lH-benzimidazole-2-carboxamide dihydrochloride
N-Isobutyl-1-(4-methoxybutyl)-N-[5(R)-(morpholin-4-ylcarbonyl)piperidin-3(S)-yl]-1H-benzimidazole-2-carboxamide hydrochloride
1- (4-methoxybutyl) -N- (2- methylpropyl) -N – [(3S, 5R) -5- (morpholin-4-ylcarbonyl) piperidine-3 – yl] -1H- benzimidazole-2-carboxamide hydrochloride,
The compound is used as renin inhibitor for treating diabetic nephropathy and hypertension
Takeda’s TAK-272, was reported to be in phase II in October 2015), an oral renin inhibitor, for treating diabetic nephropathy and hypertension

In the above method, the acid anhydride (BANC) from chiral dicarboxylic acid monoester ((-) – BMPA) were synthesized and then the carboxylic acid after conversion and hydrolysis reaction of the Z amine by the Curtius rearrangement of the carboxylic acid (BAPC) and it was then performs amidation by the condensation reaction with the amine (morpholine), is synthesized heterocyclic amide compound (BMPC). Further, Patent Document 2, the preparation of compounds useful as synthetic intermediates of the above heterocyclic compounds are disclosed.
(Wherein each symbol is as described in Patent Document 2.)
WO2009154300
https://www.google.co.in/patents/WO2009154300A2?cl=en
INTERMEDIATES FOR CONSTRUCTION
Reference Example 31 tert-butyl (3S,5R)-3-[{ [1- (4-methoxybutyl) -lH-benzimidazol-2- yl] carbonyl} (2-methylpropyl) amino] -5- (morpholin-4- ylcarbonyl)piperidine-l-carboxylate and 1- (4-methoxybutyl) -N-
(2-methylpropyl) -N- [ (3S, 5R) -5- (morpholin-4- ylcarbonyl)piperidin-3-yl]-lH-benzimidazole-2-carboxamide
tert-Butyl (3S, 5R) -3-{ [ ( {2- [ (4- methoxybutyl) amino] phenyl}amino) (oxo) acetyl] (2- methylpropyl) amino} -5- (morpholin-4-ylcarbonyl) piperidine-1- carboxylate (9.11 g) was dissolved in acetic acid (50 ml), and the mixture was stirred at 😯0C for 15 hr. The reaction mixture was cooled to room temperature and concentrated under reduced pressure, the residue was diluted with aqueous sodium bicarbonate, and the mixture was extracted with ethyl acetate. The extract was washed with saturated brine, and dried over anhydrous magnesium sulfate. The solvent was evaporated under reduced pressure. The residue was subjected to basic silica gel column chromatography, and a fraction eluted with ethyl acetate was concentrated under reduced pressure to give tert- butyl (3S, 5R) -3- [ { [1- (4-methoxybutyl) -lH-benzimidazol-2- yl] carbonyl } (2-methylpropyl) amino] -5- (morpholin-4- ylcarbonyl)piperidine-l-carboxylate (5.85 g) , and a fraction eluted with ethyl acetate-methanol (85:15) was concentrated under reduced pressure to give 1- (4-methoxybutyl) -N- (2- methylpropyl) -N- [ (3S, 5R) -5- (morpholin-4-ylcarbonyl) piperidin- 3-yl] -lH-benzimidazole-2-carboxamide (580 mg) . [0424] tert-butyl (3S,5R)-3-[{ [1- (4-methoxybutyl) -lH-benzimidazol-2- yl] carbonyl} (2-methylpropyl) amino] -5- (morpholin-4- ylcarbonyl ) piperidine-1-carboxylate 1H-NMR (CDCl3) δ 0.63-0.80 (2H, m) , 0.89-1.07 (4H, m) , 1.41- 1.59 (9H, m) , 1.59-1.80 (2H, m) , 1.87-2.23 (4H, m) , 2.30-2.98 (3H, m) , 3.21-3. 46 ( 6H, m) , 3.49-3. 91 (1OH, m) , 3. 95-4 . 47 (5H, m) , 7 . 18-7 . 51 (3H, m) , 7. 56-7 . 84 ( IH, m) .
MS (ESI+, m/e) 600 (M+l )
1- (4-methoxybutyl) -N- (2-methylpropyl) -N- [ (3S, 5R) -5- (morpholin- 4-ylcarbonyl)piperidin-3-yl] -lH-benzimidazole-2-carboxamide BASE
1H-NMR (CDCl3) δ 0.64-0.74 (2H, m) , 0.95-1.07 (4H, m) , 1.43-
1.74 (3H, m) , 1.84-2.41 (4H, m) , 2.48-2.67 (IH, m) , 2.67-3.01
(3H, m), 3.03-3.44 (8H, m) , 3.47-3.78 (9H, m) , 4.06-4.46 (3H, m) , 7.28-7.47 (3H, m) , 7.62-7.81 (IH, m) . MS (ESI+, m/e) 500 (M+l)
Example 10
1- (4-methoxybutyl) -N- (2-methylpropyl) -N- [ (3S, 5R) -5- (morpholin-
4-ylcarbonyl) piperidin-3-yl] -lH-benzimidazole-2-carboxamide dihydrochloride
tert-Butyl (3S,5R)-3-[{ [1- (4-methoxybutyl) -IH- benzimidazol-2-yl] carbonyl} (2-methylpropyl) amino] -5-
(morpholin-4-ylcarbonyl)piperidine-l-carboxylate (5.85 g) was dissolved in methanol (20 ml) , 4M hydrogen chloride-ethyl acetate (20 ml) was added, and the mixture was stirred at room temperature for 15 hr. The reaction mixture was concentrated, and the residue was diluted with aqueous sodium bicarbonate, and the mixture was extracted with ethyl acetate. The extract was washed with saturated brine, and dried over anhydrous magnesium sulfate. The solvent was evaporated under reduced pressure. The residue was subjected to basic silica gel column chromatography, and a fraction eluted with ethyl acetate- methanol (9:1) was concentrated under reduced pressure to give 1- (4-methoxybutyl) -N- (2-methylpropyl) -N- [ (3S, 5R) -5- (morpholin- 4-ylcarbonyl) piperidin-3-yl] -lH-benzimidazole-2-carboxamide (4.40 g) . The obtained 1- (4-methoxybutyl) -N- (2-methylpropyl) – N- [ (3S, 5R) -5- (morpholin-4-ylcarbonyl) piperidin-3-yl] -IH- benzimidazole-2-carboxamide (2.20 g) was dissolved in ethyl acetate (20 ml) , 4M hydrogen chloride-ethyl acetate (5 ml) and methanol (20 ml) were added, and the mixture was stirred at room temperature for 5 min. The reaction mixture was concentrated under reduced pressure to give the object product (2.52 g).
dihydrochloride
1H-NMR (DMSO-d6) δ 0.63-0.76 (2H, m) , 0.85-1.00 (4H, m) , 1.40-
1.60 (2H, m) , 1.68-1.89 (2H, m) , 1.93-2.17 (2H, m) , 2.20-2.44
(2H, m) , 2.81-3.81 (2OH, m) , 4.19-4.39 (3H, m) , 7.23-7.46 (2H, m) , 7.57-7.81 (2H, m) , 8.38-9.77 (2H, m) .
MS (ESI+, m/e) 500 (M+l)
Example 252
1- ( 4-methoxybutyl ) -N- ( 2-methylpropyl ) -N- [ ( 3S 1. 5R) -5- (morpholin- 4-ylcarbonyl ) piperidin-3-yl ] -lH-benzimidazole-2-carboxamide methanesulfonate
l-(4-Methoxybutyl) -N- (2-methylpropyl) -N- [ (3S,5R)-5- (morpholin-4-ylcarbonyl) piperidin-3-yl] -lH-benzimidazole-2- carboxamide (208 mg) was dissolved in ethyl acetate (2 ml) , a solution of methanesulfonic acid (40 μl) in ethyl acetate (1 ml) was added at 75°C, hexane (1 ml) was added, and the mixture was heated under reflux and stood at room temperature overnight. The precipitated crystals were collected by filtration, and dried at 7O0C for 3 hr to give the object product (158 mg) . MS (ESI+, m/e) 500 (M+l) melting point : 144.40C
Example 32
methyl (3R, 5S)-5-[{ [1- (4-methoxybutyl) -lH-benzimidazol-2- yl] carbonyl} (2-methylpropyl) amino] piperidine-3-carboxylate dihydrochloride [0675]
MS (ESI+, m/e) 445 (M+l)
Example 33
(3R, 5S) -5- [ { [1- (4-methoxybutyl) -lH-benzimidazol-2- yljcarbonyl} (2-methylpropyl) amino] piperidine-3-carboxylic acid dihydrochloride
MS (ESI+, m/e) 431 (M+l)
Reference Example 29
{ [ ( 3S , 5R) -1- (tert-butoxycarbonyl ) -5- (morpholin-4- ylcarbonyl ) piperidin-3~yl ] ( 2-itιethylpropyl ) amino } (oxo ) acetic acid
To a solution of tert-butyl (3S,5R)~3-{ [ethoxy (oxo) acetyl] (2-methylpropyl) amino}-5- (morpholin-4- ylcarbonyl) piperidine-1-carboxylate (10.3 g) in ethanol (40 ml) was added 2M aqueous sodium hydroxide solution (22 ml) , and the mixture was stirred at room temperature for 6 hr. The reaction mixture was adjusted to pH 7 with IM hydrochloric acid, and extracted with ethyl acetate. The extract was washed with saturated brine, and dried over anhydrous magnesium sulfate. The solvent was evaporated under reduced pressure to give the object product (10.3 g) .
1H-NMR (CDCl3) δ 0.78-0.99 (6H, m) , 1.37-1.52 (9H, m) , 1.79- 2.16 (3H, m) , 2.38-3.86 (14H, m) , 3.93-4.43 (2H, m) . MS (ESI+, m/e) 442 (M+l)
Reference Example 28
tert-butyl (3S, 5R) -3-{ [ethoxy (oxo) acetyl] (2- methylpropyl ) amino } -5- (morpholin-4-ylcarbonyl) piperidine-1- carboxylate
To a solution of tert-butyl (3S, 5R) -3- [ (2- methylpropyl) amino] -5- (morpholin-4-ylcarbonyl) piperidine-1- carboxylate (9.24 g) and diisopropylethylamine (10.5 ml) in DMA (100 ml) was added dropwise ethyl chloroglyoxylate (3.4 ml) at 0°C. The reaction mixture was stirred at room temperature for 15 hr, and the reaction mixture was concentrated. An aqueous sodium bicarbonate solution was added to the residue, and the mixture was extracted with ethyl acetate. The extract was washed with saturated brine, and dried over anhydrous magnesium sulfate. The solvent was evaporated under reduced pressure. The residue was subjected to silica gel column chromatography, and a fraction eluted with ethyl acetate was concentrated under reduced pressure to give the object product (10.3 g) . 1H-NMR (CDCl3) δ 0.84-1.00 (6H, m) , 1.37 (3H, q) , 1.42-1.53 (9H, m) , 1.80-2.19 (3H, m) , 2.26-2.42 (IH, m) , 2.59-2.96 (IH, in) , 2.97-3.30 (3H, m) , 3.37-3.92 (9H, m) , 4.01-4.26 (2H, m) , 4.26- 4.40 (2H, m) . MS (ESI4-, m/e) 470 (M+l) “
Reference Example 22 tert-butyl (3S, 5R) -3- [ (2-methylpropyl) amino] -5- (morpholin-4- ylcarbonyl)piperidine-l-carboxylate
[0369] tert-Butyl (3S,5R)-3-{ [ (benzyloxy) carbonyl] aminoJ-5- (morpholin-4-ylcarbonyl)piperidine-l-carboxylate (58 g) and palladium (II) hydroxide-carbon (5 g) were suspended in methanol (400 ml) and the mixture was stirred under a hydrogen atmosphere (1 atom) at room temperature for 16 hr. The palladium catalyst was filtered off, and the filtrate was concentrated under reduced pressure. The obtained residue and acetic acid (8.8 ml) were dissolved in methanol (400 ml), 2- methylpropanal (14.0 ml) was added, and the mixture was stirred at room temperature for 1 hr. Sodium triacetoxyborohydride (40.4 g) was added to the reaction mixture, and the mixture was stirred at room temperature for 2 hr. The reaction mixture was concentrated under reduced pressure, and the concentrate was basified with 3.5M aqueous potassium carbonate solution, and the mixture was extracted with ethyl acetate. The extract was washed with saturated brine, and dried over anhydrous magnesium sulfate. The solvent was evaporated under reduced pressure. The residue was subjected to basic silica gel column chromatography, and a fraction eluted with ethyl acetate-hexane (1:5) – ethyl acetate-hexane (1:1) was concentrated under reduced pressure to give the object product (33.3 g) .
1H-NMR (CDCl3) δ: 0.90 (6H, d) , 1.46 (9H, s) , 1.54 (IH, d) , 1.69 (IH, dt), 1.96-2.12 (2H, m) , 2.23-2.37 (IH, m) , 2.47 (3H, d) , 2.66 (IH, d) , 3.61 (IH, br s) , 3.55 (2H, d) , 3.69 (5H, ddd) , 4.01-4.46 (2H, m) .
Example 6 1-tert-butyl 3-methyl (3R, 5S) -5-aminopiperidine-l, 3- dicarboxylate [0318]
(3S, 5R) -1- (tert-Butoxycarbonyl) -5-(methoxycarbonyl)piperidine-3-carboxylic acid (2.83 g) was suspended in toluene (36 ml), diphenylphosphoryl azide (2.60 ml) and triethylamine (1.70 ml) were added, and the mixture was stirred at 100°C for 1 hr. The reaction mixture was cooled to room temperature, benzyl alcohol (1.53 ml) and triethylamine (7.00 ml) were added and the mixture was stirred at 80°C for 3 hr. The reaction mixture was concentrated, the residue was dissolved in ethyl acetate, and the solution was washed with water, 0.5M hydrochloric acid, saturated aqueous sodium hydrogen carbonate and saturated brine in this order, and dried over anhydrous magnesium sulfate. The solvent was evaporated under reduced pressure. The residue was subjected to silica gel column chromatography, and a fraction eluted with ethyl acetate-hexane (1:3 – 3:1) was concentrated under reduced pressure. The obtained residue was dissolved in methanol (60 ml), 10% palladium carbon (50% in water) (150 mg) was added and the mixture was stirred under a hydrogen pressurization (5 atom) at ambient temperature and normal pressure for 5 hr. The catalyst was filtered off, and the filtrate was concentrated under reduced pressure to give the object product (1.83 g) as an oil.
1H-NMR (CDCl3) δ 1.22-1.43 (4H, m) , 1.46 (9H, s), 2.27-2.79 (4H, m) , 3.70 (3H, s) , 4.13 (2H, br s) [0320] In the same manner as in the method shown in Reference Example 6, the following compound (Reference Example 7) was obtained.
Reference Example 8
1-tert-butyl 3-methyl (3R, 5S) -5- [ (2- methylpropyl) amino] piperidine-1, 3-dicarboxylate [0325]
1-tert-Butyl 3-methyl (3R, 5S) -5-aminopiperidine-l, 3- dicarboxylate (1.83 g) , isobutyraldehyde (0.78 ml) and acetic acid (0.49 ml) were dissolved in methanol (50 ml), and the mixture was stirred at room temperature for 30 min. Sodium triacetoxyborohydride (3.80 g) was added to the reaction mixture, and the mixture was stirred at room temperature for 7 hr. The reaction mixture was concentrated under reduced pressure, the concentrate was basified with aqueous sodium bicarbonate, and extracted with ethyl acetate. The extract was washed with water and saturated brine, and dried over anhydrous magnesium sulfate. The solvent was evaporated under reduced pressure. The residue was subjected to silica gel column chromatography, and a fraction eluted with ethyl acetate-hexane (1:1) – ethyl acetate 100% – ethyl acetate- methanol (9:1) was concentrated under reduced pressure to give the object product (1.42 g) as an oil.
1H-NMR (CDCl3) δ 0.90 (6H, d) , 1.22-1.38 (3H, m) , 1.46 (9H, s) , 1.69 (IH, dt), 2.23-2.39 (2H, m) , 2.44-2.59 (IH, m) , 2.47 (2H, d) , 2.74 (IH, br s) , 3.69 (3H, s) , 4.18-4.34 (2H, m)
Reference Example 27
N- (4-methoxybutyl) benzene-1, 2-diamine
To a solution of phenylenediamine (10.8 g) and 4- methoxybutyl methanesulfonate (9.11 g) in acetonitrile (100 ml) was added potassium carbonate (20.7 g) , and the mixture was stirred heated under reflux for 15 hr. Water was added to the reaction mixture, and the mixture was extracted twice with ethyl acetate. The extract was washed with saturated brine, and dried over anhydrous magnesium sulfate. The solvent was evaporated under reduced pressure. The residue was subjected to silica gel column chromatography, and a fraction eluted with ethyl acetate-hexane (35:65) was concentrated under reduced pressure to give the object product (5.44 g) . 1H-NMR (CDCl3) δ 1.67-1.82 (4H, m) , 3.13 (2H, t) , 3.24-3.39 (6H, m) , 3 . 38 -3 . 50 ( 2H, m) , 6 . 62 – 6 . 74 ( 3H, m) , 6 . 81 ( IH, in) . MS ( ESI+ , m/e ) 195 (M+l )
Reference Example 146 tert-butyl (3S, 5R) -3- [ { [1- (4-methoxybutyl) -lH-benzimidazol-2- yl]carbonyl} (2-methylpropyl) amino] -5- (morpholin-4- ylcarbonyl)piperidine-l-carboxylate
A solution of tert-butyl (3S, 5R) -3- [ (lH-benzimidazol-2- ylcarbonyl) (2-methylpropyl) amino] -5- (morpholin-4- ylcarbonyl)piperidine-l-carboxylate (200 mg) , 4-itιethoxybutyl methanesulfonate (107 mg) and cesium carbonate (254 mg) in N,N-dimethylacetamide (5 ml) was stirred at 60°C for 15 hr. After cooling to room temperature, the reaction mixture was diluted with water and extracted with ethyl acetate (10 ml*2) . The extract was washed with saturated brine, and dried over anhydrous magnesium sulfate. The solvent was evaporated under reduced pressure. The residue was subjected to silica gel column chromatography, and a fraction eluted with ethyl acetate-hexane (5:95 – 3:7) was concentrated under reduced pressure to give the object product (190 mg) . 1H-NMR (CDCl3) δ 0.63-0.80 (2H, m) , 0.89-1.07 (4H, m) , 1.41- 1.59 (9H, m) , 1.59-1.80 (2H, m) , 1.87-2.23 (4H, m) , 2.30-2.98 (3H, m) , 3.21-3.46 (6H, m) , 3.49-3.91 (1OH, m) , 3.95-4.47 (5H, m) , 7.18-7.51 (3H, m) , 7.56-7.84 (IH, m) . MS (ESI+, m/e) 600 (M+l)
ALTERNATE METHOD IN THIS PATENT



Reference Example 61
2- (trichloromethyl) -lH-benzimidazole
O-Phenylenediamine (25 g) was dissolved in acetic acid (750 ml), and methyl 2, 2, 2-trichloroacetimidate (28.5 ml) was added dropwise over 15 min. After stirring at room temperature for 1 hr, the reaction mixture was concentrated to about 150 ml, and poured into water (1500 ml) . The precipitated crystals were collected by filtration, washed with water (1000 ml) and suspended in toluene (500 ml) . The solvent was evaporated under reduced pressure. The residue was again suspended in toluene (500 ml) and the solvent was evaporated under reduced pressure. The residue was dried under reduced pressure to give the object product (51.8 g) . 1H-NMR (CDCl3) δ 7.31-7.45 (2H, m) , 7.49-7.55 (IH, m) , 7.89 (IH, d) , 9 . 74 ( IH, br s )
Reference Example 64
1-tert-butyl 3-methyl (3R, 5S) -5- [ (lH-benzimidazol-2- ylcarbonyl) (2-methylpropyl) amino] piperidine-1, 3-dicarboxylate
2- (Trichloromethyl) -lH-benzimidazole (19 g) and 1-tert- butyl 3-methyl (3R, 5S) -5- [ (2-methylpropyl) amino] piperidine- 1,3-dicarboxylate (25 g) were dissolved in THF (1200 ml), sodium hydrogen carbonate (67 g) and water (600 ml) were added, and the mixture was stirred at room temperature for 1 hr and at 5O0C for 1 hr. After evaporation of the solvent, the residue was extracted 3 times with ethyl acetate (700 ml) . The extract was washed successively with 10%-aqueous citric acid solution (500 ml) and brine, and dried over anhydrous sodium sulfate. The solvent was evaporated under reduced pressure.
The residue was dissolved in ethyl acetate (1000 ml), subjected to basic silica gel column chromatography, and a fraction eluted with ethyl acetate was concentrated under reduced pressure to give the object product (30.6 g) .
1H-NMR (CDCl3) δ 0.78-1.09 (6 H, m) , 1.17-1.55 (9 H, m) , 1.77-2.95 (5 H, m) , 3.11-3.79 (6 H, m) , 3.99-4.73 (4 H, m) , 7.24- 7.41 (2 H, m) , 7.45-7.59 (1 H, m) , 7.72-7.88 (1 H, m) , 10.66-10.98 (1 H, m)MS (ESI+, m/e) 459 (M+l)
Reference Example 69
1-tert-butyl 3-methyl (3R, 5S) -5- [ { [1- (4-methoxybutyl) -IH- benzimidazol-2-yl] carbonyl} (2-methylpropyl) amino] piperidine-1 , 3-dicarboxylate
1-tert-Butyl 3-methyl (3R, 5S) -5- [ (lH-benzimidazol-2- ylcarbonyl) (2-methylpropyl) amino] piperidine-1, 3-dicarboxylate (30 g) and 4-methoxybutyl methanesulfonate (12.5 g) were dissolved in DMA (600 ml), cesium carbonate (32 g) was added, and the mixture was stirred at 70°C for 12 hr. The reaction mixture was poured into ice water (1000 ml), and the mixture was extracted twice with ethyl acetate (1000 ml) . The extract was washed with brine, and dried over anhydrous sodium sulfate. The solvent was evaporated under reduced pressure. The residue was subjected to silica gel column chromatography, and a fraction eluted with ethyl acetate-hexane (1:4 – 1:1) was concentrated under reduced pressure to give the object product (28.7 g) .
1H-NMR (CDCl3) δ 0.76 (4H, d) , 1.01 (2H, d) , 1.30-1.52 (9H, m) , 1.58-2.07 (4H, m) , 2.10-2.93 (4H, m) , 3.27-3.75 (12H, m) , 4.06-4.57 (5H, m) , 7.26-7.48 (3H, m) , 7.79 (IH, d) MS (ESI+, m/e) 545 (M+l)
Example 71
1- (4-methoxybutyl) -N- (2-methylpropyl) -N- [ (3S, 5R) -5- (morpholin- 4-ylcarbonyl) piperidin-3-yl] -lH-benzimidazole-2-carboxamide
tert-Butyl (3S, 5R) -3- [{ [1- (4-methoxybutyl) -IH- benzimidazol-2-yl] carbonyl} (2-methylpropyl) amino] -5- (morpholin-4-ylcarbonyl)piperidine-l-carboxylate (5.85 g) was dissolved in methanol (20 ml) , 4M hydrogen chloride-ethyl acetate (20 ml) was added, and the mixture was stirred at room temperature for 15 hr. The reaction mixture was concentrated, the residue was diluted with aqueous sodium bicarbonate,…and, the mixture was extracted with ethyl acetate. The extract was washed with saturated brine, and dried over anhydrous sodium sulfate. The solvent was evaporated under reduced pressure. The residue was subjected to basic silica gel column chromatography, and a fraction eluted with ethyl acetate- methanol (9:1) was concentrated under reduced pressure to give the object product (4.40 g) . MS (ESI+, m/e) 500 (M+l)
Example 101
1- (5-methoxypentyl) -N- (2-methylpropyl) -N- [ (3S, 5R) -5- (morpholin-4-ylcarbonyl) piperidin-3-yl] -lH-benzimidazole-2- carboxamide dihydrochloride
[1144] tert-Butyl (3S, 5R) -3- [ { [1- (5-methoxypentyl) -IH- benzimidazol-2-yl] carbonyl} (2-methylpropyl) amino] -5- (morpholin-4-ylcarbonyl)piperidine-l-carboxylate (123 mg) was dissolved in 4M hydrogen chloride-ethyl acetate (5 ml) , and the mixture was stirred at room temperature for 3 hr. The reaction mixture was concentrated, and the residue was subjected to reversed-phase preparative HPLC and the eluted fraction was concentrated under reduced pressure. The residue was diluted with aqueous sodium bicarbonate, and the mixture was extracted with ethyl acetate. The extract was washed with saturated brine, and dried over anhydrous sodium sulfate. 4M Hydrogen chloride-ethyl acetate (1 ml) was added and the mixture was stirred for 5 min. The solvent was evaporated under reduced pressure to give the object product (76 mg) . MS (ESI+, m/e) 514 (M+l)
PATENT
WO2013122260
http://www.google.co.in/patents/WO2013122260A1?cl=en
PATENT
WO 2011158880
http://www.google.co.in/patents/WO2011158880A1?cl=en
Reference Example 1
1- (4-methoxybutyl) -N- (2- methylpropyl) -N – [(3S, 5R) -5- (morpholin-4-ylcarbonyl) piperidin-3-yl] -1H- benzimidazole -2 – carboxamide hydrochloride (A-type crystal)
tert- butyl (3S, 5R) -3 – [{[1- (4- methoxy-butyl) -1H- benzimidazol-2-yl] carbonyl} (2-methylpropyl) amino] -5- (morpholin-4- ylcarbonyl) was suspended dissolved piperidine-1-carboxylate The (300g) in 3N- hydrochloric acid water (1200mL) and Ethyl acetate (60mL), and stirred over 3 h at 25 ~ 35 ℃. After completion of the reaction, it was added ethyl acetate (2400mL) in the same temperature. After the addition, it was added 25% aqueous ammonia (600mL) with cooling. After the addition stirring and extracted the organic layer of 5% aqueous ammonia (600mL) was added and stirred. After stirring, the resulting organic layer it was concentrated until the solvent no longer distilled off. After concentrated, dissolved with ethyl acetate (1500mL), and transferred to solution to the crystallizer vessel, and washed with ethyl acetate (750mL). After washing, it was raised in stirring under 45 ~ 55 ℃. After raising the temperature, at the same temperature 4N- hydrogen chloride – it was dropped ethyl acetate (131.3mL). After dropping, it was to dissolve the precipitate at the same temperature. After dissolution confirmation, it was added heptane (750mL) at 40 ~ 50 ℃, after the addition, then cooled to 25 ~ 35 ℃. After cooling, the addition of A-type crystals of the seed crystals (300mg) which was obtained according to the method described in Example 265 of WO2009 / 154300, and stirred for 30 minutes or more. After stirring, the temperature was raised to 40 ~ 45 ℃, it was dropped heptane (1500mL). After the completion of the dropping, it was stirred at the same temperature. Then gradually cooled to 5 ℃ below, followed by stirring at the same temperature for 1 hour. After stirring, ethyl acetate and filtered crystals – heptane: washed with (1 1,600mL), to obtain a wet crystal. The obtained wet crystals dried under reduced pressure at 50 ℃, 1- (4- methoxybutyl) -N- (2- methylpropyl) -N – [(3S, 5R) -5- (morpholin-4-yl carbonyl) piperidin-3-yl] -1H- obtained a crystalline powder of benzimidazole-2-carboxamide hydrochloride (A-type crystal, 198.82g, 74.1% yield). FINAL PRODUCT
TERT BUTYL DERIVATIVE, N-1
Reference Example 4
tert- butyl (3S, 5R) -3 – [{[1- (4- methoxy-butyl) -1H- benzoimidazol-2-yl] carbonyl} (2-methylpropyl) amino] -5- (morpholin-4- ylcarbonyl) piperidine-1-carboxylate 1)
o- nitro aniline (50.0g, 0.362mol), tetrabutylammonium bromide (58.3g, 0.181mol), potassium bromide (43.1g, 0.362mol) in toluene (500mL ) and it was added. At a temperature of 20 ~ 30 ℃ 1- chloro-4-methoxy-butane (66.6g, 0.543mol) and, I was added to 50w / v% sodium hydroxide solution (145mL, 1.81mol). The reaction was heated to a temperature 85 ~ 95 ℃, and stirred for 6 hours. After cooling to a temperature 20 ~ 30 ℃, the reaction mixture water (250mL), 1N- aqueous hydrochloric acid (250mL × 2), 5w / v% aqueous solution of sodium bicarbonate (250mL), it was washed successively with water (250mL). After concentration under reduced pressure the organic layer to Contents (250mL), was added toluene (100mL), was obtained
N- (4- methoxy-butyl) -2-nitroaniline in toluene (350mL, 100% yield).
1 H-NMR (300MHz, CDCl 3) δ 1.64-1.89 (m, 4H), 3.25-3.39 (m, 2H), 3.35 (s, 3H), 3.44 (t, J = 6.1 Hz, 2H), 6.63 ( ddd, J = 8.5, 6.9, 1.2 Hz, 1H), 6.86 (dd, J = 8.5, 1.2 Hz, 1H), 7.43 (ddd, J = 8.5, 6.9, 1.5 Hz, 1H), 8.07 (br s, 1H ), 8.17 (dd, J = 8.5, 1.5 Hz, 1H).
2) N- (4-methoxy-butyl) -2-10 percent in nitroaniline of toluene solution (350mL) Pd / C (K-type, 50% water-containing product) (10.0g) and toluene (100mL) it was added. Hydrogen pressure of 0.1MPa, it was stirred for 3 hours at a temperature of 20 ~ 30 ℃. A stream of nitrogen, the catalyst was filtered, I was washed with toluene (100mL). After the water in the filtrate was separated off and adding magnesium sulfate (25.0g) at a temperature 20 ~ 30 ℃, and stirred at the same temperature for 30 minutes. Filtered over magnesium sulfate, washed with toluene (100mL), was obtained N- (4- methoxybutyl) -o- toluene solution of phenylenediamine (100% yield).
1 H NMR (500 MHz, CDCl 3) δ1.67-1.78 (m, 4H), 3.12-3.14 (m, 2H), 3.32 (br, 3H), 3.35 (s, 3H), 3.41-3.47 (m, 2H), 6.63-6.69 (m, 2H), 6.69-6.74 (m, 1H), 6.82 (td, J = 7.57, 1.58 Hz, 1H).
3) N- (4- methoxy-butyl) -o- After the toluene solution of phenylenediamine cooled to a temperature 0 ~ 10 ℃, acetic acid (65.2g, 1.09mol) and 2,2,2 trichloroacetimide acid methyl ( 70.3g, 0.398mol) and I were added. After stirring for 30 minutes at a temperature 0 ~ 10 ℃, it was stirred for 3 hours at a temperature of 20 ~ 30 ℃. The reaction was 5w / v% saline (250mL), 2N- aqueous hydrochloric acid / 5w / v% sodium chloride solution: a mixture of (1 1) (250mL × 2), 5w / v% aqueous solution of sodium bicarbonate (250mL), 5w / v It was washed successively with% saline solution (250mL). A stream of nitrogen, was added magnesium sulfate (25.0g) to the organic layer at a temperature 20 ~ 30 ℃, and stirred at the same temperature for 30 minutes. Filtered magnesium sulfate, and washed with toluene (100mL). The filtrate was concentrated under reduced pressure and the amount of contents (150mL). Stir the concentrated solution at a temperature 20 ~ 30 ℃, was allowed to precipitate crystals, was added dropwise heptane (750mL). The crystals bleeding is heated to a temperature 40 ~ 50 ℃, after stirring for 30 min, cooled to a temperature 0 ~ 10 ℃, and the mixture was stirred at the same temperature for 2 hours.The precipitated crystals were collected by filtration, toluene – heptane: was washed with (1 5,150 mL). And dried under reduced pressure at 40 ℃, it was obtained 1- (4-methoxy-butyl) -2-fine brown crystals of trichloromethyl -1H- benzimidazole (96.5g, 82.9% yield from o- nitroaniline).
1 H-NMR (300MHz, CDCl 3) δ: 1.68-1.85 (m, 2H), 1.99-2.17 (m, 2H), 3.37 (s, 3H), 3.48 (t, J = 6.1 Hz, 2H), 4.50 -4.65 (m, 2H), 7.27-7.49 (m, 4H), 7.82-7.93 (m, 1H).
. Anal Calcd for C 13 H 15 Cl 3 N 2 O:. C, 48.55; H, 4.70; N, 8.71; Cl, 33.07 Found: C, 48.30; H, 4.61; N, 8.74; Cl, 33.30.
4) pyridine-3,5-dicarboxylic acid (110g, 0.66mol), it was dropped methanol (660 mL) mixture of concentrated sulfuric acid at a temperature of 50 ℃ or less of (226.0g, 2.30mol). Thereafter, the mixture was stirred and heated to a temperature 55 ~ 65 ℃ 7 hours. The reaction was the temperature 40 ~ 50 ℃, was added water (220mL). And further dropping temperature 40-50 5% aqueous ammonia at ℃ (about 1.10L) was adjusted to pH8.0 ~ 8.5. After stirring at a temperature 40 ~ 50 ℃ 30 minutes and stirred for 1 hour and cooled to a temperature 0 ~ 10 ℃. Was collected by filtration precipitated crystals, methanol – water (1: 3,165mL), and washed successively with water (440mL). To obtain a white crystalline powder pyridine-3,5-dicarboxylic acid dimethyl and dried under reduced pressure at 50 ℃ (105.0g, 82.0% yield).
1 H-NMR (300 MHz, CDCl 3) δ 4.00 (s, 6H), 8.87 (s, 1H), 9.37 (s, 2H).
. Anal Calcd for C 9 H 9 NO 4:. C, 55.39; H, 4.65; N, 7.18; O, 32.79 Found: C, 55.42; H, 4.65; N, 7.16.
5) 1 L autoclave pyridine-3,5-dicarboxylic acid dimethyl (100g, 0.51mol) and was charged with dimethylacetamide (400mL), temperature 30 ℃ below with trifluoroacetic acid (59.2mL, after dropping the 0.77mol), 10% Pd-C (PE-type) the (20.0g) it was added. Hydrogen pressure of 0.5 ~ 0.7MPa, it was stirred for 12 hours at a temperature of 55 ~ 65 ℃. The catalyst was filtered off, it was washed with dimethylacetamide (50mL × 2). Triethylamine and the combined filtrates at a temperature 20 ~ 30 ℃ (77.8g, 0.77mol) was added dropwise, and adjusted to pH9.0 ~ 10.0. Temperature 30 ~ 40 ℃ by di -tert- butyl (134g, 0.614mol) was added dropwise and stirred at the same temperature for 2 hours. After the reaction mixture as a 20 ~ 30 ℃, it was added ethyl acetate (600mL), washed with water (900mL). The aqueous layer it was re-extracted with ethyl acetate (400mL). The combined organic layers 5w / v% citric acid -10w / v% sodium chloride solution (600mL), 3% aqueous sodium bicarbonate (600mL), and washed successively with water (600mL). Contents The organic layer (200mL) until it was concentrated under reduced pressure, methanol (250mL) was added to the concentrated solution, and then concentrated under reduced pressure until Contents (200mL). The addition of methanol (250mL) again concentrate, After concentration under reduced pressure until Contents (200mL), was added methanol (2.40L). The solution in water (18.5g, 1.03mol), cesium carbonate (417g, 1.28mol) was added and stirred for about 24 hours at a temperature 55 ~ 65 ℃. The reaction solution was the temperature 20 ~ 30 ℃, concentrated to Contents (700mL), it was added tetrahydrofuran (500mL). The solution temperature at 15 ~ 35 ℃ 2N- hydrochloric acid solution (1.28L, 2.56mol) was added dropwise and adjusted to pH3.0 ~ 3.5, and the mixture was stirred for 30 minutes at a temperature 20 ~ 30 ℃. Extracted with ethyl acetate (750mL × 2), and the organic layer was washed with 10w / v% aqueous sodium chloride solution (500mL × 3). Contents The organic layer (300mL) until it was concentrated under reduced pressure, to obtain a weight content by adding ethyl acetate (650mL).Heating the concentrate to a temperature of 55 ~ 65 ℃, it was added dropwise heptane (500mL). It cooled to a temperature 20 ~ 30 ℃ and stirred for 1 hour. The precipitated crystals were collected by filtration, ethyl acetate – heptane: was washed with (1 1,120mL). Dried under reduced pressure at 50 ℃ 1- (tert- butoxycarbonyl) to give a white crystalline powder of piperidine-3,5-dicarboxylic acid (113.3g, 80.9% yield).
1 H-NMR (300 MHz, DMSO-d 6) δ 1.40 (s, 9H), 1.44-1.61 (m, 1H), 2.21-2.26 (m, 1H), 2.31-2.41 (m, 2H), 4.10- 4.12 (m, 2H).
. Anal Calcd for C 12 H 19 NO 6:. C, 52.74; H, 7.01; N, 5.13; O, 35.13 Found: C, 52.96; H, 6.99; N, 5.39.
6) Under a nitrogen stream, 1- (tert- butoxycarbonyl) piperidine-3,5-dicarboxylic acid (5.00g, 18.3mmol) was suspended in tetrahydrofuran (10.0mL), trifluoroacetic acid anhydride at a temperature 20 ~ 30 ℃ It was dropping things (3.80mL, 27.5mmol). After the completion of the dropping, it was stirred for 1 hour at a temperature of 20 ~ 30 ℃. It was added dropwise heptane (20.0mL) at a temperature 20 ~ 30 ℃ the reaction solution, and stirred for 3 hours then cooled to a temperature 0 ~ 10 ℃. The precipitated crystals were collected by filtration, and washed with heptane (3.00mL). Dried under reduced pressure at 40 ℃ 2,4- dioxo-3-oxa-7-azabicyclo [3,3,1] white crystalline powder of nonane-7-carboxylic acid tert- butyl was obtained (4.03g, yield 86.1%).
1 H-NMR (300 MHz, CDCl 3) δ 1.43 (s, 9H), 1.93-1.99 (m, 1H), 2.40-2.46 (m, 1H), 3.06-3.11 (m, 4H), 4.50-4.54 ( m, 2H).
. Anal Calcd for C 12 H 17 NO 5:. C, 56.46; H, 6.71; N, 5.49; O, 31.34 Found: C, 56.51; H, 6.63; N, 5.69.
7) Under a nitrogen stream, quinidine (69.9g, 0.215mol) and was charged with tetrahydrofuran (200mL), and cooled to a temperature -5 ~ 5 ℃. At the same temperature 2,4-dioxo-3-oxa-7-azabicyclo [3,3,1] nonane-7-carboxylic acid tert- butyl (50.0g, 0.196mol) was added and washed with tetrahydrofuran (50.0mL) crowded. Temperature -5 ~ 5 methanol at ℃ (9.41g, 0.29 4mol) was added dropwise, and the mixture was stirred for 2 hours at a temperature -5 ~ 5 ℃. Ethyl acetate (350mL) to the reaction mixture, was by adding minute solution 20w / v% citric acid aqueous solution (250mL). The aqueous layer it was re-extracted with ethyl acetate (125mL × 2). The organic layers were combined 20w / v% aqueous solution of citric acid (250mL), I was washed successively with water (250mL × 2). The organic layer it was concentrated under reduced pressure. To the residue ethanol (100mL) was added ethyl acetate (450mL) was heated to a temperature 60 ~ 70 ℃, (R) – was added phenethylamine (23.7g, 0.196mol). Temperature 50-60 for one hour at ℃, 1 hour at a temperature of 20 ~ 30 ℃, it was stirred for 1 hour at a temperature of -5 ~ 5 ℃. The precipitated crystals were collected by filtration, ethanol – ethyl acetate: and washed with (2 9,100mL). And dried under reduced pressure at 50 ℃ (3S, 5R) -1- (tert- butoxycarbonyl) -5- (methoxycarbonyl) piperidin-3 to give a white crystalline powder of the carboxylic acid (1R) -1- phenylethylamine salt It was (55.7g, 69.6% yield).
1 H-NMR (300 MHz, DMSO-d 6) δ 1.42 (s, 9H), 1.43-1.51 (m, 3H), 2.06-2.14 (m, 1H), 2.21-2.26 (m, 1H), 2.39- 2.44 (m, 1H), 2.52-2.53 (m, 1H), 2.57 (br s, 2H), 3.64 (s, 3H), 4.12 (br s, 2H), 4.19-4.26 (m, 1H), 7.30- 7.40 (m, 3H), 7.45-7.48 (m, 2H).
. Anal Calcd for C 21 H 32 N 2 O 6:. C, 61.75; H, 7.90; N, 6.86; O, 23.50 Found: C, 61.54; H, 7.77; N, 6.86.
8) (3S, 5R) -1- (tert- butoxycarbonyl) -5- (methoxycarbonyl) piperidine-3-carboxylic acid (1R) -1- phenylethylamine salt (20.0g, 49.0mmol), methanol (20mL) and it was charged with water (80mL). Temperature 20-30 citric acid at ℃ (11.3g, 58.8mmol) was added dropwise a solution prepared by dissolving in water (20.0mL), and the mixture was stirred 1.5 hours at the same temperature. The precipitated crystals were collected by filtration and washed with water (60mL). And dried under reduced pressure at 50 ℃ (3S, 5R) -1- (tert- butoxycarbonyl) -5- give a white crystalline powder (methoxycarbonyl) piperidine-3-carboxylic acid (13.5g, 96.1% yield ).
1 H-NMR (300 MHz, CDCl 3) δ 1.40 (s, 9H), 1.46-1.59 (m, 1H), 2.22-2.27 (m, 1H), 2.37-2.45 (m, 2H), 2.63-2.73 ( m, 2H), 3.63 (s, 3H), 4.14 (br s, 2H), 12.51 (br s, 1H).
. Anal Calcd for C 13 H 21 NO 6:. C, 54.35; H, 7.37; N, 4.88; O, 33.41 Found: C, 54.14; H, 7.28; N, 4.85.
9) Under a nitrogen stream, (3S, 5R) -1- (tert- butoxycarbonyl) -5- (methoxycarbonyl) piperidine-3-carboxylic acid (30.0g, 104mmol), triethylamine (31.7g, 313mmol) and toluene ( It was charged with 180mL). Diphenylphosphorylazide at a temperature of 15 ~ 35 ℃ (28.7g, 313mmol) I was dropped a toluene (30.0mL) solution. After stirring at a temperature 30 ± 5 ℃ 30 minutes, and the mixture was stirred and heated to a temperature 65 ~ 75 ℃ 30 minutes. Temperature 60 ~ 70 ℃ in the benzyl alcohol (12.4g, 115mmol) it was dropped. To a temperature 80 ~ 90 ℃ was stirred and heated for 3 hours. The reaction mixture was cooled to a temperature 20 ~ 30 ℃, sodium nitrite (7.20g, 104mmol) and after stirring was added a solution prepared by dissolving in water (150mL) 1 hour, the aqueous layer was separated. The organic layer 5w / v% aqueous sodium bicarbonate solution (150mL), 20w / v% aqueous citric acid solution (150mL), washed successively with 5w / v% aqueous sodium chloride solution (150mL), the organic layer was concentrated under reduced pressure. The residue methanol (60.0mL) was added and concentrated under reduced pressure to. The more we went once in the same manner.To the residue was added methanol and the content amount of the (90.0g). Temperature 15 ~ 35 ℃ 2N- aqueous sodium hydroxide (62.6mL, 125mmol) was added and stirred for 1 hour at a temperature 30 ± 5 ℃. Temperature 20 ~ 30 ℃ in methanol (120mL), was added to 20w / v% aqueous citric acid solution (300mL), it was a pH3.0 ~ 3.5. After stirring for 30 minutes at a temperature 50 ~ 60 ℃, cooled to a temperature 20 ~ 30 ℃ and stirred for 1 hour. It was stirred for 1 hour at the temperature 0 ~ 10 ℃. The precipitated crystals were collected by filtration, and washed with water (90.0mL). And dried under reduced pressure at 50 ℃ (3R, 5S) -5 – {[(benzyloxy) carbonyl] amino} -1- (tert- butoxycarbonyl) to yield a white crystalline powder piperidine-3-carboxylic acid (35.0 g, 88.6% yield).
1 H-NMR (300 MHz, DMSO-d 6) δ 1.41 (s, 9H), 2.11 (d, J = 12.4 Hz, 1H), 2.40-2.48 (m, 4H), 2.62 (br s, 1H), 4.08 (t, J = 14.4 Hz, 2H), 5.04 (s, 2H), 7.31-7.41 (m, 5H), 12.53 (br s, 1H).
. Anal Calcd for C 19 H 26 N 2 O 6:. C, 60.30; H, 6.93; N, 7.40; O, 25.37 Found: C, 60.03; H, 6.99; N, 7.41.
10) Under a nitrogen stream, (3R, 5S) -5 – {[(benzyloxy) carbonyl] amino} -1- (tert- butoxycarbonyl) piperidine-3-carboxylic acid (30.0g, 79.3mmol), morpholine (7.60 g, 87.2mmol), 1- hydroxybenzotriazole monohydrate (2.43g, it was charged with 15.9mmol) and dimethylacetamide (90.0mL). Hydrochloride 1-ethyl at a temperature 20 ~ 30 ℃ -3- (3- dimethylaminopropyl) carbodiimide (16.7g, 87.1mmol) after addition and stirred for 1 hour at a temperature 45 ~ 55 ℃. Temperature 45 ~ 55 ℃ with tetrahydrofuran (90.0mL), sequentially dropwise addition of water (210mL), and stirred for 1 hour. After stirring for 1 hour and cooled to a temperature 20 ~ 30 ℃, were collected by filtration the precipitated crystals, tetrahydrofuran – water: washing with (1 3,120mL). And dried under reduced pressure at 50 ℃ tert- butyl piperidine -1- (3S, 5R) -3 – a white crystalline powder of {[(benzyloxy) carbonyl] amino} -5 (morpholin-4-yl-carbonyl) carboxylate It was obtained (32.7g, 92.3% yield).
1 H-NMR (300 MHz, DMSO-d 6) δ 1.41 (s, 9H), 1.49-1.57 (m, 1H), 1.87 (d, J = 12.3 Hz, 1H), 2.43 (br s, 1H), 2.63-2.71 (m, 1H), 2.79-2.83 (m, 1H), 3.37-3.54 (m, 9H), 3.89 (d, J = 11.5 Hz, 1H), 4.06 (br s, 1H), 5.03 (s , 2H), 7.30-7.38 (m, 5H).
. Anal Calcd for C 23 H 33 N 3 O 6:. C, 61.73; H, 7.43; N, 9.39; O, 21.45 Found: C, 61.59; H, 7.50; N, 9.43.
11) tert- Butyl piperidin -1- (3S, 5R) -3 – {[(benzyloxy) carbonyl] amino} -5- (morpholin-4-ylcarbonyl) carboxylate (30.0g, 67.0mmol), isobutyraldehyde (7.25g, 101mmol), it was charged with 10% Pd-C (PE type) (1.50g) and methanol (240mL).Hydrogen pressure of 0.2 ~ 0.3MPa, it was stirred for 4 hours at a temperature of 20 ~ 30 ℃. The catalyst is filtered off and washed with methanol (60.0mL). The filtrate was concentrated under reduced pressure, ethyl acetate was added (60.0mL), and concentrated under reduced pressure again. The residue ethyl acetate was added, followed by the amount of contents (360mL). Temperature 45-55 succinate by heating to ℃ (7.90g, 67.0mmol) was added. After stirring for 1 hour at a temperature 45 ~ 55 ℃, cooled to a temperature 20 ~ 30 ℃, and stirred for 1 hour. The precipitated crystals were collected by filtration, and washed with ethyl acetate (90.0mL). And dried under reduced pressure at 50 ℃ tert- butyl (3S, 5R) -3 – [(2- methyl-propyl) amino] -5- (morpholin-4-yl-carbonyl) piperidine – 1-carboxylate white crystals of alert succinate got sex powder (30.2g, 92.5% yield).
1 H-NMR (300 MHz, D 2 O) δ 1.02 (s, 3H), 1.04 (s, 3H), 1.47 (s, 9H), 1.97-2.09 (m, 2H), 2.26-2.30 (m, 1H ), 2.55 (s, 4H), 2.99 (d, J = 7.0 Hz, 2H), 3.23 (br s, 1H), 3.39-3.45 (m, 2H), 3.53-3.80 (m, 10H), 3.82-3.93 (br s, 1H).
. Anal Calcd for C 23 H 41 N 3 O 8:. C, 56.66; H, 8.48; N, 8.62; O, 26.25 Found: C, 56.48; H, 8.46; N, 8.39.
12) tert- Butyl (3S, 5R) -3 – [(2- methylpropyl) amino] -5- (morpholin-4-ylcarbonyl) piperidine – 1 – carboxylate succinate (30.3g, 62.2mmol), acetonitrile (60.0mL) and, it was charged with water (40.0mL). Then after stirring was added potassium carbonate (34.4g, 0.249mmol) 10 minutes, 1- (4-methoxybutyl) -2-trichloromethyl -1H- benzimidazole (20.0g, 62.2mmol) was added. After stirring for 2 hours at a temperature of 70 ~ 80 ℃, it was added dimethyl sulfoxide (15.0mL), and the mixture was stirred for 6 hours at a temperature 70 ~ 80 ℃. After cooling the reaction mixture to a temperature 20 ~ 30 ℃, water (120mL), it was separated and by adding toluene (240mL). The organic layer 10w / v% sodium chloride solution (100mL), 10w / v% aqueous solution of citric acid (100mL), it was washed sequentially with 10w / v% sodium chloride solution (100mL). The organic layer of activated carbon Shirasagi A a (1.0g) was added, and the mixture was stirred for 30 minutes at a temperature 20 ~ 30 ℃. Activated carbon was filtered, washed with toluene (40.0mL), and concentrated under reduced pressure of the filtrate to 110 mL. By heating to a temperature 35 ~ 45 ℃ was added dropwise heptane (280mL). At a temperature 35 ~ 45 ℃ tert- butyl (3S, 5R) -3 – [{[1- (4- methoxy-butyl) -1H- benzoimidazol-2-yl] carbonyl} (2-methylpropyl) amino] -5 – and the mixture was stirred for 1 hour at (morpholin-4-ylcarbonyl) piperidine-1-carboxylate was added to the same temperature the crystals (10mg) of the acrylate. Heptane (140mL) was stirred and added dropwise to 30 minutes at a temperature 35 ~ 45 ℃. It was cooled to a temperature 20 ~ 30 ℃ and stirred for 2 hours. The precipitated crystals were collected by filtration, toluene – heptane: was washed with (1 5,40.0mL). And dried under reduced pressure at 50 ℃ tert- butyl (3S, 5R) -3 – [{[1- (4- methoxy-butyl) -1H- benzoimidazol-2-yl] carbonyl} (2-methylpropyl) amino] – 5- (morpholin-4-ylcarbonyl) piperidine-1-carboxylate was obtained a pale yellowish crystalline powder of alert (27.7g, 74.2% yield).
1 H-NMR (300 MHz, CDCl 3) δ 0.68-0.80 (m, 3H), 0.96-1.08 (m, 3H), 1.31 (br s, 5H), 1.49 (s, 4H), 1.61-1.71 (m , 2H), 1.71 (br s, 0.5H), 1.92-2.05 (m, 3H), 2.05-2.24 (m, 2H), 2.45 (br s, 1H), 2.60 (br s, 1H), 2.72-2.96 (m, 2H), 3.26-3.35 (m, 3H), 3.35-3.47 (m, 2H), 3.47-3.73 (m, 10H), 4.02-4.26 (m, 2H), 4.26-4.34 (m, 1H) , 4.34-4.47 (m, 0.5H), 7.25-7.29 (m, 1H), 7.29-7.41 (m, 1H), 7.41-7.53 (m, 1H), 7.64 (br s, 0.5H), 7.79 (d , J = 8.2 Hz, 0.5H).
. Anal Calcd for C 32 H 49 N 5 O 6:. C, 64.08; H, 8.23; N, 11.68; O, 16.01 Found: C, 63.82; H, 8.12; N, 11.64.
PATENT
TAKEDA PHARMACEUTICAL COMPANY LIMITED [JP/JP]; 1-1, Doshomachi 4-chome, Chuo-ku, Osaka-shi, Osaka 5410045 (JP)
Provided is a method for producing a synthetic intermediate of a heterocyclic compound having a renin inhibitory activity and effective as a prophylactic or therapeutic drug against diabetic renal disease, hypertension, and the like. A method for producing a compound represented by formula (III-1a), (III-1b), (III-1c), and/or (III-1d) [where the symbols in the formulas are as defined in the description], or a salt thereof, said method characterized in that a compound represented by formula (Ia) or (Ib) [where the symbols in the formulas are as defined in the description] or a salt thereof is reacted with a compound represented by formula (II) [where the symbols in the formula are as defined in the description] or a salt thereof in the presence of an aluminum compound and a chiral amine compound.

In the above method, the acid anhydride (BANC) from chiral dicarboxylic acid monoester ((-) – BMPA) were synthesized and then the carboxylic acid after conversion and hydrolysis reaction of the Z amine by the Curtius rearrangement of the carboxylic acid (BAPC) and it was then performs amidation by the condensation reaction with the amine (morpholine), is synthesized heterocyclic amide compound (BMPC). Further, Patent Document 2, the preparation of compounds useful as synthetic intermediates of the above heterocyclic compounds are disclosed.[Formula 3]

(Wherein each symbol is as described in Patent Document 2.)
| WO2010150840A1 | 24 Jun 2010 | 29 Dec 2010 | Dainippon Sumitomo Pharma Co., Ltd. | N-substituted-cyclic amino derivative |
| WO2011158880A1 | 15 Jun 2011 | 22 Dec 2011 | Takeda Pharmaceutical Company Limited | Crystal of amide compound |
| WO2012062687A1 * | 7 Nov 2011 | 18 May 2012 | F. Hoffmann-La Roche Ag | Triazole derivatives and their use for neurological disorders |
| WO2013122260A1 | 14 Feb 2013 | 22 Aug 2013 | Takeda Pharmaceutical Company Limited | Tablet |
| CN103221402B * | 7 Nov 2011 | 17 Jun 2015 | 霍夫曼-拉罗奇有限公司 | 三唑衍生物及其用于神经障碍的用途 |
| US8329691 | 14 Oct 2008 | 11 Dec 2012 | Takeda Pharmaceutical Company Limited | Amide compounds and use of the same |
| US8389511 | 19 Dec 2008 | 5 Mar 2013 | Dainippon Sumitomo Pharma Co., Ltd. | Bicyclic heterocyclic derivative |
| US8658639 | 24 Jun 2010 | 25 Feb 2014 | Dainippon Sumitomo Pharma Co., Ltd | N-substituted-cyclic amino derivative |
| US8742097 | 2 Nov 2011 | 3 Jun 2014 | Hoffmann-La Roche Inc. | Triazole compounds I |
| US9018374 | 15 Jun 2011 | 28 Apr 2015 | Takeda Pharmaceutical Company Limited | Crystal of amide compound |
| US9090601 | 28 Jan 2010 | 28 Jul 2015 | Millennium Pharmaceuticals, Inc. | Thiazole derivatives |
///////////TAK 272, Hypertension



Pramipexole can be synthesized from a cyclohexanone derivative by the following route:
Pramipexole synthesis:[14]
Pramipexole has been evaluated for the treatment of cluster headache and to counteract problems with sexual dysfunction experienced by some users of selective serotonin reuptake inhibitor (SSRI) antidepressants.[15] Pramipexole has shown effects on pilot studies in a placebo-controlled proof of concept study in bipolar disorder.[8][16][17] It is also being investigated for the treatment of clinical depression and fibromyalgia.[18][19][20]
http://pubs.acs.org/doi/abs/10.1021/op1000989

Pramipexole is a dopamine D2 subfamily receptor agonist that is used for the treatment of Parkinson’s disease. We report here on the successful application of the Fukuyama alkylation protocol to the development of a novel and scalable process for synthesis of pramipexole and its pharmaceutically acceptable salts. The synthesis consists of converting the crucial intermediate (S)-2,6-diamino-4,5,6,7-tetrahydrobenzothiazole to (6S)-N-(2-amino-4,5,6,7-tetrahydrobenzothiazole-6-yl)-2-nitrobenzenesulfonamide, which is in turn monoalkylated to (6S)-N-(2-amino-4,5,6,7-tetrahydrobenzothiazole-6-yl)-2-nitro-N-propylbenzenesulfonamide. Deprotection of the latter yields pramipexole base, which is finally converted to a crude pramipexole dihydrochloride monohydrate with a yield of over 50% over four steps. The process allows for the telescoping of the final three steps, has high conversion rates of intermediates, offers ease of purification, and preserves high optical purity throughout all of the stages.
pramipexole dihydrochloride monohydrate 13 (315 g) with a yield of 70% (calculated from 12) and an HPLC purity of 94.4%. 1H NMR (300 MHz, DMSO-d6) δ 0.89 (t, J = 7.5 Hz, 3H), 1.62−1.75 (m, J = 7.6 Hz, 2H), 1.87−2.00 (m, 1H), 2.24−2.28 (m, 1H), 2.55−2.67 (m, 2H), 2.71−2.79 (m, 1H), 2.86−2.89 (m, 2H), 2.99−3.06 (m, 1H), 3.47 (m, 1H), 9.50 (m, 2H). 13C NMR (300 MHz, DMSO-d6) 11.1, 19.1, 20.9, 23.5, 24.8, 46.0, 52.3, 111.0, 132.9, 168.7. FT-IR (cm−1): 3150−3450 (NH2 stretching), 2700−3000 (C−H stretching), 1600−1650 (C═N stretching), 1550−1600 (heteroaromatic ring skeleton).

A NEW, EFFICIENT AND ECONOMIC METHOD FOR PREPARATION OF PRAMIPEXOLE. |
| Roman Balicki , Agnieszka Ciesielska , Michał Odrowąż-Sypniewski |
| Pharmaceutical Research Institute (IF), Rydygiera 8, Warszawa 01-793, Poland |
| Abstract |
|
Pramipexole is a novel nonergot dopamine agonist which has high selectivity for interacting with dopamine D2 receptors. It is effective in early Parkinson,s disease as monotherapy and as adjunctive therapy with L-dopa in advanced stages of the disease. Known, two-steps method for preparation of pramipexole (3) is based on acylation reaction of diamine 1 with propionic anhydride. The obtained amide 2 is subsequently reduced using borane to give final product 3 with 65% yield. Now, we present novel, more economic and safe procedure for obtaining pramipexole. Our one-step method requires only alkylation of 1 using n-propyl tosylate. Dangerous reduction with borane is eliminated and the final compound is obtained with similar yield as in a previous method. |
http://www.google.com/patents/US7741490
Pramipexole, of formula (A)
is a dopaminergic agonist, known from U.S. Pat. No. 4,843,086, used in the treatment of Parkinson’s disease in the form of dihydrochloride monohydrate.
US 2002/0103240 discloses inter alia a method for the resolution or the enrichment of (R,S)-2-amino-6-propylamino-4,5,6,7-tetrahydrobenzothiazole in the single (R) or (S) enantiomers, in particular in the (S) enantiomer. The same application illustrates in detail the synthetic routes known for the preparation of pramipexole, in particular those described in U.S. Pat. No. 4,886,812, EP 186087, EP 207696 and J. Med. Chem. 30. 494 (1987). From what reported it is evident that the synthetic pathways up to now available make use of synthetic steps that do not fulfill the requirements for the production of pramipexole on the industrial scale. Therefore there is the need for an improved process, which is simpler, easier to carry out and meets the requirements for the industrial production of pramipexole.
Example 13 Intermediate (VIII) Ra=H; Pramipexole Free Base
A 2 liter reactor under nitrogen is loaded with 53.3 g of, 33.0 g of (S) N-(6-propionylamino-4,5,6,7-tetrahydro-benzothiazol-2-yl)-amine, 95% sodium borohydride and 260 ml of tetrahydrofuran (THF). A solution of 98.7 g of iodine in 160 ml of THF is dropped therein in about 3 hours, keeping the temperature at approx. 20-25° C. The reaction mixture is kept under stirring for further 2 hours at about 20-25° C. The reaction mixture is poured into a solution of 60.0 g of 37% HCl in 260 ml of water. The mixture is heated to 50-55° C. and left under stirring for an hour. The complete cleavage of the boran-complexes is checked by HPLC. The mixture is added with 250 g of 50% aqueous NaOH, keeping the temperature at about 20-25° C. After that, 315 ml of toluene are added and the mixture is heated to about 30-35° C. Stirring is interrupted and the two phases are separated. The organic phase are washed, concentrated to a residue and dissolved in 420 ml of ethyl acetate.
The solution is concentrated under vacuum at a temperature below 50° C. to about 150 ml volume. The resulting suspension is refluxed, then cooled to about 10-15° C., stirred for further 1-2 hours, then filtered with suction and the precipitate is washed twice with 30 ml of ethyl acetate. The product is dried under vacuum at 40° C. 32 g of (S)-2-amino-6-propylamino-4,5,6,7-tetrahydrobenzothiazole are obtained.
https://www.google.sc/patents/WO2008097203A1?cl=en
Example 1
Synthesis of N-(2-amino-4,5, 6, 7-tetrahydrobenzo[d]thiazole-6-yl)-2- n itrobenzenesulfonam ide
o-Nitrobenzenesulfonyl chloride (8.865 g, 40 mmol) was dissolved in 100 ml of THF and during stirring cooled to -100C (ice + salt). Then, first 3 equiv. of triethylamine (Et3N) (120 mmol, 16.8 ml) and then also 1.1 equiv. of 4,5,6,7-tetrahydrobenzo[J]thiazol-2,6-diamine (7.605 g, 45 mmol) were added. The formed suspension was, during stirring, gradually heated to room temperature and was left standing until the reaction was completed. In the course of the reaction, in addition to a soluble product, also in THF non-soluble Et3NH+Cl“ was formed, which was, at the end of the reaction, filtered off by suction and the reaction mixture was evaporated to dryness on a rotatory evaporator. The residue was poured over with H2O (300 ml), whereby on the bottom of a round-bottom flask an orange viscous liquid was obtained. After rubbing with the glass stick a yellow precipitate (N-(2- amino-4,5,6,7-tetrahydrobenzo[^thiazole-6-yl)-2-nitrobenzenesulfonamide) was formed. The precipitate was filtered off and washed with 100 ml of cold ethylether and dried. The yield of the reaction was 95%.
Example 2
Synthesis of N-(2-amino-4,5,6, 7-tetrahydrobenzo[d]thiazole-6-yl)-2-nitro-N- propylbenzenesulfonamide
Process A:
N-(2-amino-4,5,6,7-tetrahydrobenzo[rf]thiazole-6-yl)-2-nitrobenzenesulfonamide (1.770 g, 5 mmol) and K2CO3 (2.856 g, 20 mmol) were suspended in acetonitrile (40 ml) and was, during stirring, heated to 600C. Then propylbromide (1.65 ml, 18 mmol) was added and the reaction was left to run over night (the course of the reaction was followed by the use of a suitable method).
After the reaction was completed, the present precipitate was filtered off by suction. The reaction mixture was evaporated to dryness and the residue was dissolved in dichloromethane (150 ml). Organic phase was washed with IM NaOH (3 x 50 ml), saturated NaCl solution (2 x 50 ml) and dried with Na2SO4. After evaporation of dichloromethane an orange oily product N-(2-amino-4, 5,6,7- tetrahydrobenzo[^thiazole-6-yl)-2-nitro-Ν-propylbenzenesulfonamide was obtained.
Process B:
N-(2-amino-4,5,6,7-tetrahydrobenzo[rf]thiazole-6-yl)-2-nitrobenzenesulfonamide (1.770 g, 5 mmol), Cs2CO3 (3.909 g, 12 mmol) and KI (0.415 g, 2.5 mmol) was suspended in acetonitrile (40 ml) and heated to 600C. Then propyl bromide (0.9 ml, 10 mmol) was added and the course of the reaction was followed by the use of a suitable method.
The process of the isolation was the same as in the process A.
Example 3
Synthesis of lf-propyl-4,5,6, 7-tetrahydrobenzo[d]thiazole-2,6-diamine
Process A:
K2CO3 (2.073 g, 15 mmol) was suspended in 20 ml of DMF (stored above molecular sieves), thioglycolic acid (SHCH2COOH, 0.6 ml, 7.5 mmol) was added and stirred for 30 minutes. Then N-(2-amino-4,5,6,7-tetrahydrobenzo[d]thiazole-6-yl)-2-nitro-N- propylbenzenesulfonamide ( 0,99 g, 2,5 mmol), dissolved in 20 ml of DMF was added and the reaction was left to run over night. After the reaction was completed, DMF was evaporated, the residue was poured over with H2O (100 ml) and IM NaOH (200 ml). The aqueous phase was then washed with dichloromethane (3 x 80 ml) and the combined organic fractions were dried with z Na2SO4. After the evaporation of the solvent an oily residue of orange-red colour (presence of DMF is possible) was obtained.
Process B:
LiOH (0.5 g, 20 mmol) was suspended in 20 ml of DMF (stored above molecular sieves), thioglycolic acid (SHCH2COOH, 0.6 ml, 7.5 mmol) was added and stirred for 30 minutes. Then N-(2-amino-4,5,6,7-tetrahidrobenzo[cT|thiazole-6-yl)-2-nitro-N- propylbenzenesulfonamide (0.99 g, 2.5 mmol), dissolved in 20 ml of DMF was added and the reaction was left to run over night (the solution was coloured in orange-red).
After the reaction was completed, DMF was evaporated off, the residue was poured over with H2O (100 ml) and IM NaOH (200 ml). The aqueous phase was then washed with dichloromethane (3 x 80 ml) and the combined organic fractions were dried with Na2SO4. After the evaporation of the solvent an oily residue of an orange- red colour was obtained.
Example 4
Pramipexole dihydrochloride monohydrate
(S)-(-)-2-Amino-6-(N-propylamino)-4,5,6,7-tetrahydrobenzothiazole (9.15 g, 43.28 mmol) in 500 ml round-bottom flask was dissolved in 30 ml of ethanol and water (0.78 g, 43.33 mmol) was added. A solution was cooled in an ice bath to 00C and gaseous HCl^)1 was blown through whereby a white precipitate fell out. The round- bottomed flask was sealed and it was stirred over night at room temperature. The next day the precipitate was filtered off by suction and washed with a small amount of anhydrous ethanol. The precipitate was transferred into 100 ml round-bottom flask and anhydrous ethanol (50 ml) was added. The suspension was heated to 45°C and ethanol was evaporated on a rotatory evaporator. The process was repeated for another two times in order to drive out all of the excessive HCl(g). The product was recrystallized from methanol: a salt was dissolved in methanol (70 ml) at 450C, approximately 40 ml of methanol was evaporated and 20 ml of ethanol were added. It was cooled to room temperature and the resulting precipitate was filtered by suction, washed with some cooled anhydrous ethanol and dried in vacuum over P2O5 and NaOH. Yield: 11.631 g (89.01 %)
Example •§
Synthesis of N-(2-amino-4, 5, 6, 7-tetrahydrobenzo[d]thiazole-6-yl)-2- nitrobenzenesulfonamide
2-Nitrobenzenesulfonyl chloride (390 g, 1.76 mol) was dissolved in 4 1 of THF. The solution was cooled to approximately -100C. Triethylamine (Et3N) (740 g, 7.313 mol) and (6S)-4,5,6,7-tetrahydrobenzotiazole-2,6-diamine (327 g, 1.932 mol) were added. The suspension was heated during mixing to approximately 25°C and allowed to react at this temperature for approximately 1 hour.
Precipitated triethylammonium chloride (Et3NH+Cl“) was filtered off. The filtrate was concentrated to about 1/3 of the volume and water (2 1) was added. Again, approximately 1/2 of the solvent was distilled off. Water (2 1) was added, the mixture was cooled to about 25°C and mixed for about 1 hour. The precipitated product ((6S)- N-(2-amino-4,5,6,7-tetrahydrobenzotiazole-6-yl)-2-nitrobenzenesulfonamide) was separated by filtration or centrifuging.
Example g
Synthesis of (S)-(-)-2-Amino-6-(N-propylamino)-4,5,6, 7-tetrahidrobenzo[d]thiazole dihydrochloride hydrate
Potassium carbonate (1890 g, 13.675 mol), (6S)-N-(2-amino-4,5,6,7- tetrahydrobenzotiazole-6-yl)-2-nitrobenzenesulfonamide (590 g, 1.665 mol) and propyl bromide (1.09 1, 12 mol) were suspended in 4.1 1 of acetonitrile. The mixture was heated during stirring to approximately 600C and mixed at this temperature for about 12 hours. The mixture was cooled to about 25°C. Potassium bromide was removed by filtration. The solution was concentrated to about 1/4 of the volume (not exceeding 600C) and cooled to the room temperature. Methylene chloride (2 1) and 1 M NaOH water solution (2.43 1) were added and the mixture was mixed for about 30 minutes. Phases were separated and water phase was washed again with methylene chloride (1.46 1). Organic phases were collected and concentrated to about 1/10 of the volume. 0.83 1 of ethanol was added and the solution was concentrated to 1/10 of the volume. 3.35 1 of ethanol was added and ethanolic solution of (6S)-N-(2-amino- 4,5 ,6,7-tetrahydrobenzotiazole-6-yl)-2-nitro-Ν-propylbenzenesulfonamide was stored for further reaction.
Ethanol (2.35 1) and of LiOH (288 g, 12 mol) were put into the reactor and the suspension was cooled to 0 – 5°C. During about 30 minutes thioglycolic acid (SHCH2COOH) (720 g, 7.816 mol) was added (the temperature must not exceed 25°C). The suspension was heated to about 25°C and mixed for about 45 minutes. Ethanolic solution of (6S)-N-(2-amino-4,5,6,7-tetrahydrobenzotiazole-6-yl)-2-nitro-N- propylbenzenesulfonamide was added. The air in the reactor was replaced by nitrogen. The mixture was heated to about 500C and mixed at this temperature for about 4 hours. The mixture was cooled to about 25°C and filtrated. The filtrate was concentrated at 400C to about 1/4 of the volume and cooled to the ambient temperature. Methylene chloride (4.23 1) and of IM aqueous NaOH solution solution (2.53 1) were added and the mixture was mixed for about 30 minutes. Phases were separated and water phase was washed again with 4.23 1 of methylene chloride. Organic phases containing pramipexole were collected and concentrated to about 1/4 of the volume. 5 1 of ethanol was added.
To the ethanolic solution of pramipexole water was added (27.6 ml, 1.53 mol) and solution was cooled to about -100C. Gaseous HCl(g) was introduced into the solution (200 g). The temperature of the solution and later the suspension must not exceed 250C during addition of gaseous HCl(g) . After the addition the suspension was heated to about 4O0C and concentrated to 2/3 of the volume. 2.65 1 of ethanol was added and the suspension was concentrated to 1/2 of the volume. Again 3.5 1 of ethanol was added and the suspension was concentrated to 1/2 of the volume. The solution was cooled to about -15°C and the product was separated by filtration. The product was dried at 25°C and finally at 400C on air.
http://www.google.com/patents/WO2008041240A1?cl=en
(S)-2-amino-6-propylaminio-4,5,6,7- tetrahydrobenzothiazole of formula (I), which is more commonly known as Pramipexole. Pramipexole is the commercial product marketed, in a form of a dihydrochloride salt in a peroral formulation, under several brand names e.g. Mirapexin[TM].
The compound of formula (I) has one symmetric carbon and they may exist either as a single enantiomer or in a mixed or racemic form. The pharmacological activity of compounds of formula (I) is generally connected only or mainly with one isomer thereof. Accordingly, the dopaminergic activity of the (S) isomer is twice as high as that of the (R) enantiomer.
A general process for the preparation of Pramipexole dihydrochloride has been described in US 4886812, EP 186087 and EP 207696. The process comprises the protection of amino function of 4-aminocyclohexanol to give the intermediate compound wherein, Rl is acyl or alkoxycarbonyl and R2 is hydrogen or Rl and R2 together form an amino protective group such as pthalimido group which on oxidation with an oxidising agent, followed by halogenation (preferably bromination) of protected ketone to give alpha halogenatedketone which on reaction with thiourea, followed by deprotection yielded the racemic 2,6-diaminotetrahydrobenzothiazole. Reductive alkylation of diaminotetrahydrobenzothiazole with n-propanal furnished the racemic pramipexole. Although the (S) isomer of pramipexole is mentioned therein, it is not clear at what stage the chiral resolution has been carried out. The general process steps are indicated in Scheme- Ia below.
H2N
Racemate Resolution
n-Propyl Bromide –
Scheme-la
Another process for preparing optically pure pramipexole dihydrochloride was disclosed in J. Med. Chem. 1987, 30, 494-498, wherein, racemic 2,6-diamino-4,5,6,7- tetrahydrobenzo- thiazole was resolved, using L (+) tartaric acid to give optically pure (S)-2,6-diamino-4,5,6,7-tetrahydrobenzothiazole, which was converted to optically pure pramipexole by reacting (S)- 2,6-diamino-4,5,6,7-tetrahydro benzothiazole with propionic anhydride in THF and followed by reduction with borane THF complex . The reaction steps are shown in Scheme-lb as under:
(VIII) (II)
(CH3CH2CO)2O
2HCl Scheme- Ib
However, the variants of the above general process prepare only racemate.
Thus, the synthesis of pramipexole by the above process yields R,S(±)-2~amino-6- propylamino-4,5,6,7-tetrahydrobenzothiazole. The above-mentioned acknowledge that the produced racemic compound may be resolved into single enantiomers by classical methods such as chromatography on a chiral phase or fractional crystallization of a salt with an optically active acid. However, even though the S(-)-enantiomer of pramipexole was disclosed and characterized therein, no information is provided how it was prepared; i.e. whether it was prepared by a resolution of racemic pramipexole of form some optically active precursor. Further, no information is provided on how to produce the S(-)- enantiomer of pramipexole.
WO 2006/003677 Al discloses the improved process the preparation of biologically active tetrahydrobenzothiazole derivative. The patent application discloses the process that has tried to solve the problems of prior art. However, much improvement over the prior art process has still been achieved. Moreover, the process discloses the formation of 2,6-diamino-4,5,6,7-tetrahydrobenzothiazole via an isolated bromo intermediate, which on reaction with thiourea gets converted to tetrahydrobenzothiazole. The isolation of bromo intermediate can also be avoided. The halogenation of the protected cyclohexanone derivative is performed in presence of Lewis acid catalysts like AICI3, ZnCl2 or SnCl2 etc. which will give aluminous waste though increase the yield during the halognation reaction. Moreover, the overall steps of the reaction will increase by performing isolation and work up for bromo intermediate.
US 6,727,637 B2 discloses the monobasic acid addition salts and the mixed salts of 2-amino-6-propylamino-4,5,6,7-tetrahydrobenzothiazole wherein the monobasic acid includes hydrochloric, hydrobromic, hydroiodic, nitric, benzoic, acetic, methane sulfonic, ethane sulfonic, trifluromethane sulfonic, benzene sulfonic, and p- toluene sulfonic acids. Further the patent US ‘637 B2 discloses the formation of mixed salts like of 2-amino-6-propylamino-4,5,6,7-tetrahydrobenzothiazole monohydrochloride monotartrate, of 2-amino-6-propylamino -4,5,6,7- tetrahydrobenzothiazole monohydrobromide monotartrate or of 2-amino-6- propylamino-4,5,6,7-tetrahydrobenzothiazole. monomethane sulfonate dibenzoyl-D- tartrate. The process as disclosed in US ‘637 B2 converts the racemic pramipexole into monohydrochloride salt of pramipexole which is then resolved with a optically active auxilliary acid to give mixed salt like of 2-amino-6-propylamino-4,5,6,7-tetrahydro- benzothiazole monohydrochloride monotartrate which is then converted to (S)- pramipexole free base and then to the desired pharmaceutically active ingredient (S)- pramipexole dihydrochloride.
US 6,770,761 B2 also discloses the process for preparation of 2-amino-6(alkyl)- amino-4,5,6,7-tetrahydrobenzothiazoles which includes the bromination of 1,4- cyclohexadione by bromine in an alcoholic solvent, followed by treatment of the reaction mixture with thiourea or N-acylthiourea and isolation of compound (A), that is further treated with an amine R1-NH2 or a chiral amine to get an imine intermediate and reducing it by reaction with said reducing agent or by hydrogenation, to yield the compound of formula (B)
(A) (B) Polymorphism is the occurrence of different crystalline forms of a single compound and it is a property of some compounds and complexes. Thus, polymorphs are distinct solids sharing the same molecular formula, yet each polymorph may have distinct physical properties. Therefore, a single compound may give rise to a variety of polymorphic forms where each form has different and distinct physical properties, such as different solubility profiles, different melting point temperatures and/or different x- ray diffraction peaks. Since the solubility of each polymorph may vary, identifying the existence of pharmaceutical polymorphs is essential for providing pharmaceuticals with predicable solubility profiles. It is desirable to investigate all solid-state forms of a drug, including all polymorphic forms, and to determine the stability^ dissolution and flow properties of each polymorphic form. Polymorphic forms of a compound can be distinguished in a laboratory by X-ray diffraction spectroscopy and by other methods such as, infrared spectrometry. For a general review of polymorphs and the pharmaceutical applications of polymorphs see G. M. Wall, Pharm Manuf. 3, 33 (1986); J. K. Haleblian and W. McCrone, J. Pharm. ScL, 58, 911 (1969); and J. K. Haleblian, J. Pharm. ScL, 64, 1269 (1975), all of which are incorporated herein by reference.

Example-1: Preparation of 2-amino-6-phthaIimido-4,5,6,7- tetrahydrobenzothiazole A) Preparation of chromic acid:
0.278 kg of chromium trioxide was added in 0.428 L of water at 150C to 35°C. The reaction mixture was cooled to 50C to 1O0C. 0.198 L of sulfuric acid Was added slowly within 25 to 30 minutes. 1.0 L of water was added to get the clear solution. B) Preparation of 2-amino-6-phthalimido-4,5,6,7-tetrahydrobenzothiazole via 4- (phthalimido)-cyclohexanone
1.0 Kg of 4-(phthalimido)-cyclohexanol was added in 20.0 L of acetone at 250C to 350C. The reaction mixture was cooled to 50C to 100C and treated with chromic acid solution. 0.2 L of isopropanol was added and stirred for 30 min. The reaction mixture was filtered and washed with acetone (1.0 L). The filtrate was treated with 0.4 kg sodium bicarbonate at 250C to 350C and stirred for 1 h. The reaction mass was again filtered, washed with acetone (1.0 L). Excess of acetone was distilled under vacuum. The residue was treated with 0.5 L ethanol followed by distillation of ethanol under vacuum. The reaction mass was cooled and treated with 3.36 L ethanol at 450C to 250C while gradual cooling. The reaction mixture was further cooled to 150C to 2O0C and treated with 0.22 L of bromine and 0.43 Kg of thiourea under stirring for 1 h. The reaction mixture was heated to reflux at 750C to 780C for 6 hrs. The reaction mixture was cooled and stirred for 1 hr at 50C to 1O0C. The product was isolated by centrifuge, washing with ethanol 0.66 L and drying under vacuum at 5O0C t0 550C. (yield: 0.70 Kg).
ExampIe-2: Preparation of 2, 6-diamino-4,5,6,7-tetrahydrobenzothiazole
1.595 kg of 2-amino-6-phthalimido-4,5,6,7-tetrahydrobenzothiazole was treated with 40% aqueous solution of monomethylamine at 250C to 350C. The reaction mass was allowed to stir for 5-10 minutes and heated at 45°C to 5O0C for 1 – 1.5 hr. The reaction mixture was cooled gradually to 50C to 1O0C and maintained for 30 minutes. The product thus obtained was filtered, washed with chilled water and dried at 5O0C to 550C to obtained racemic 2,6-diamino-4,5,6,7-tetrahydrobenzothiazole. (Yield: 0.522 kg)
Example-3: Preparation of 2, 6-diamino-4,5,6,7-tetrahydrobenzothiazoIe tartrate salt
1.0 Kg of 2, 6-diamino-4,5,6,7-tetrahydrobenzothiazole was added in 9.5 L of water and heated at 750C to 850C. 0.888 Kg of L-(+)-tartaric acid was added to the reaction mixture and maintained for 30 minutes. The reaction mixture was fine filtered at high temperature and washed with 0.5 L of water. The filtrate was gradually cooled to 250C to 300C and maintained for 16 hours. The product was centrifuge and washed with 1 L water. The wet cake was treated with 6.0 L water and heated at 😯0C to 9O0C with addition of excess water to ensure clear solution. The reaction mass was fine filtered at high temperature and washed with 0.5 L water. The filtrate thus obtained was gradually cooled to 5°C to 1O0C and maintained for 2 hrs. The product was centrifuge and washed with 1 L chilled water. The wet cake was treated with 6.0 L water and heated at 😯0C to 9O0C with addition of excess water to ensure clear solution. The reaction mass was gradually cooled to 950C to 25°C and maintained for 2 hrs. The product was centrifuge, washed with 1 L chilled water, dried at 5O0C to 550C and cooled to 250C to 350C. (Yield: 0.70 Kg). ExampIe-4: Preparation of (S)-2,6-diamino-4,5,6,7-tetrahydrobenzothiazole
1.0 Kg of 2, 6-diamino-4,5,6,7-tetrahydrobenzothiazole tartrate salt was treated with 1.5 L of water and stirred for 15 minutes at 25°C to 35°C. 0.245 Kg of sodium hydroxide solution in 0.612 L of water was added to adjust the pH 11.0 to 12.0 within 35 to 40 minutes and stirred for 1 hr. The product was centrifuge, washed with 1.0 L water and dried at 500C to 550C. The product was cooled to 20°C-40°C to obtain (S)- 2,6-diamino-4,5,6,7-tetrahydrobenzothiazole. (Yield: 0.37 Kg). Example-5: Preparation of Pramipexole crude
To the solution of 1.0 Kg of (S)-2,6-diamino-4,5,6,7-tetrahydrobenzothiazole and 0.1225 Kg of potassium carbonate in 10.0 L isopropanol was added 0.540 L n- propyl bromide. The reaction mixture was stirred for 15 minutes and heated to reflux on a water bath up to 😯0C and was maintained for 5 hours. 0.3236 L of n-propyl bromide was further added in two portions at 😯0C to 82°C and maintained for 5 hours. The isopropanol was removed completely by distillation under vacuum at 550C to 750C. 7.5 L of process water was added into the reaction mass and stirred for 30 minutes. The reaction mixture was cooled to 250C to 350C. 40% sodium hydroxide solution (0.108 Kg in 0.27 L water) was added to adjust the constant pH 10.0 to 10.5 followed by treatment with 5.0 L methylene dichloride twice and separating the organic layer. The organic layer was treated with 5.0 L of process water and stirred for 30 minutes. The separated organic layer was subjected to distillation to remove methylene dichloride under vacuum. 5.0 L of isopropanol was added at 4O0C to 450C and heated up to 6O0C to 650C. Acidic isopropanol 0.440L was added to adjust the pH 7.0 to 7.5 and stirred for 1 hour. The reaction mass was cooled to 250C to 35°C. The product was obtained by centrifuge, washing with 0.5 L of isopropanol and drying at 5O0C to 550C followed by cooling. (Yield: 1.0 Kg)
ExampIe-6: Preparation of Pramipexole dihydrochloride monohydrate
1.0 Kg of crude Pramipexole was added in 5.0 L of ethanol and heated to reflux using water bath at 800C. The reaction mixture was maintained for 1 hour and cooled to 250C to 35°C and stirred for 1 hour. The product was centrifuge and washed with 0.5 L ethanol. The wet cake thus obtained was further treated with 5.0 L of ethanol and heated to reflux using water bath at 😯0C. The reaction mixture was maintained for 1 hour and cooled to 250C to 350C and stirred for 1 hour. The product was centrifuge and washed with 0.5 L ethanol. The wet cake was treated with 5.0 L isopropanol and heated to 6O0C to 65°C using water bath. Acidic isopropanol 0.35 L was added to adjust the pH 1.7 to 2.3 and maintained for 1 hour. The product was centrifuge and washed with 1 L of isopropanol and dried in hot air oven at 5O0C to 550C to give Pramipexole dihydrochloride pure, which is converted to Pramipexole dihydrochloride monohydrate upon cooling the dried material under airflow. (Purity: 99.5% by HPLC and having known individual impurities less than 0.1% and total impurities less than 1.0%.) Example-7.: Preparation of Pramipexole dihydrochloride monohydrate
1.0 Kg of crude Pramipexole was added in 5.0 L of ethanol and heated to reflux using water bath at 😯0C. The reaction mixture was maintained for 1 hour and cooled to 250C to 350C and stirred for 1 hour. The product was centrifuge and washed with 0.5 L ethanol. The wet cake thus obtained was further treated with 5.0 L of ethanol and heated to reflux using water bath at 800C. The reaction mixture was maintained for 1 hour and cooled to 250C to 350C and stirred for 1 hour. The product was centrifuge and washed with 0.5 L ethanol. The wet cake was treated with 5.0 L isopropanol and heated to 600C to 650C using water bath. Isopropanolic HCl (0.35 L) containing water was added to adjust the pH 1.7 to 2.3 and maintained for 1 hour. The product was centrifuge and washed with 1 L of isopropanol and dried at 4O0C to 5O0C to give Pramipexole dihydrochloride monohydrate

WO 2015155704, An improved process for the preparation of pramipexole dihydrochloride monohydrate
PIRAMAL ENTERPRISES LIMITED [IN/IN]; Piramal Tower, Ganpatrao Kadam Marg Lower Parel Mumbai 400013 (IN)
|





| WO2006003677A1 * | Apr 25, 2005 | Jan 12, 2006 | Alembic Ltd | Improved process for the preparation of biologically active tetrahydrobenzthiazole derivative |
| EP0186087A1 * | Dec 16, 1985 | Jul 2, 1986 | Dr. Karl Thomae GmbH | Tetrahydro-benzothiazoles, their production and their use as intermediates or drugs |
| EP0207696A1 * | Jun 20, 1986 | Jan 7, 1987 | Eli Lilly And Company | Dialkylaminotetrahydrobenzothiazoles and oxazoles |
| EP1731514A1 * | Jun 2, 2005 | Dec 13, 2006 | Sandoz AG | Process for the preparation of Pramipexole |
| BOEHRINGER INGELHEIM: “Mirapex“[Online] 2006, pages 4-31, XP002444888 Retrieved from the Internet: URL:http://www.fda.gov/medwaTCH/safety/200 6/Nov_PIs/Mirapex_PI.pdf> | ||
| 2 | * | SCHNEIDER C S ET AL: “Dopamine autoreceptor agonists: resolution and pharmacological activity of 2,6-diaminotetrahydrobenzothiazole and aminothiazole analogue of apomorphine” JOURNAL OF MEDICINAL CHEMISTRY, AMERICAN CHEMICAL SOCIETY. WASHINGTON, US, vol. 30, no. 3, March 1987 (1987-03), pages 494-498, XP002186199 ISSN: 0022-2623 cited in the application |
| Citing Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| EP2137171A2 * | Mar 14, 2008 | Dec 30, 2009 | Knopp Neurosciences, Inc. | Synthesis of chirally purified substituted benzothiazole diamines |
 |
|
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
(S)-N 6-propyl-4,5,6,7-tetrahydro-1,3-benzothiazole-2,6-diamine
|
|
| Clinical data | |
| Trade names | Mirapex, Mirapexin, Sifrol |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a697029 |
| Pregnancy category |
|
| Legal status |
|
| Routes of administration |
Oral |
| Pharmacokinetic data | |
| Bioavailability | >90% |
| Protein binding | 15% |
| Biological half-life | 8–12 hours |
| Excretion | Urine (90%), Feces(2%) |
| Identifiers | |
| CAS Registry Number | 104632-26-0 |
| ATC code | N04BC05 |
| PubChem | CID: 119570 |
| IUPHAR/BPS | 953 |
| DrugBank | DB00413 |
| ChemSpider | 106770 |
| UNII | 83619PEU5T |
| KEGG | D05575 |
| ChEBI | CHEBI:8356 |
| ChEMBL | CHEMBL301265 |
| Chemical data | |
| Formula | C10H17N3S |
| Molecular mass | 211.324 g/mol |
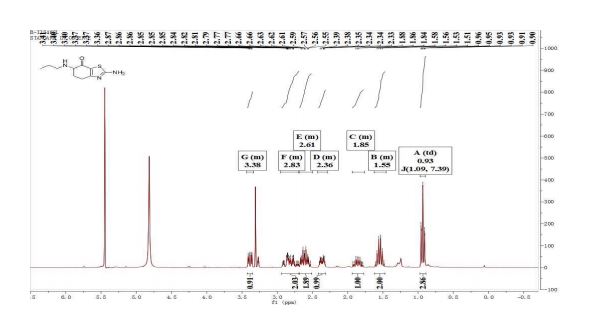


Three impurities of pramipexole dihydrochloride were synthesized, and the possible generation mechanisms and the preparation methods of some impurities were reviewed. The desired configuration at C7 of 3 was built by a Mitsunobu reaction.


N-[5-[(4-ethylpiperazin-1-yl)methyl]pyridin-2-yl]-5-fluoro-4-(7-fluoro-2-methyl-3-propan-2-ylbenzimidazol-5-yl)pyrimidin-2-amine
[5-(4-Ethyl-piperazin-1-ylmethyl)-pyridin-2-yl]-[5-fluoro-4-(7-fluoro-3-isopropyl-2-methyl-3H-benzoimidazol-5-yl)-pyrimidin-2-yl]-amine
| C27H32F2N8 | |
| M.W. | 506.59 |
|---|
Abemaciclib; 1231929-97-7; LY2835219; LY2835219 free base; UNII-60UAB198HK; LY 2835219 (free base);
Abemaciclib is an orally available cyclin-dependent kinase (CDK) inhibitor that targets the CDK4 (cyclin D1) and CDK6 (cyclin D3) cell cycle pathway, with potential antineoplastic activity. Abemaciclib specifically inhibits CDK4 and 6, thereby inhibiting retinoblastoma (Rb) protein phosphorylation in early G1. Inhibition of Rb phosphorylation prevents CDK-mediated G1-S phase transition, thereby arresting the cell cycle in the G1 phase, suppressing DNA synthesis and inhibiting cancer cell growth. Overexpression of theserine/threonine kinases CDK4/6, as seen in certain types of cancer, causes cell cycle deregulation.
LY2835219 is a potent and selective inhibitor of CDK4 and CDK6 with IC50 of 2 nM and 10 nM, respectively.
IC50 Value: 2 nM(CDK4); 10 nM(CDK6)
Target: CDK4/6
in vitro: LY2835219 is an orally available cyclin-dependent kinase (CDK) inhibitor that targets the CDK4 (cyclin D1) and CDK6 (cyclin D3) cell cycle pathway, with potential antineoplastic activity. LY2835219 specifically inhibits CDK4 and 6, thereby inhibiting retinoblastoma (Rb) protein phosphorylation in early G1. Inhibition of Rb phosphorylation prevents CDK-mediated G1-S phase transition, thereby arresting the cell cycle in the G1 phase, suppressing DNA synthesis and inhibiting cancer cell growth. Overexpression of the serine/threonine kinases CDK4/6, as seen in certain types of cancer, causes cell cycle deregulation.
in vivo: LY2835219 saturates BBB efflux with an unbound plasma IC50 of about 95 nM. The percent of dose in brain for LY2835219-MsOH is 0.5–3.9%. In both a subcutaneous and intracranial human glioblastoma model (U87MG), LY2835219-MsOH suppressed tumor growth in a dose-dependent manner both as a single agent, and in combination with temozolomide.

 Methane sulfonate
Methane sulfonate
cas 1231930-82-7, C28H36F2N8O3S, 602.7

a. 1-(6-Bromo-pyridin-3-ylmethyl)-4-ethyl-piperazine
b. 5-(4-Ethyl-piperazin-1-ylmethyl)-pyridin-2-ylamine
c. N-Isopropyl-acetamide
d. N-(4-Bromo-2,6-difluoro-phenyl)-N′-isopropyl-acetamidine
e. 6-Bromo-4-fluoro-1-isopropyl-2-methyl-1H-benzoimidazole
f. 4-Fluoro-1-isopropyl-2-methyl-6-(4,4,5,5-tetramethyl-[1,3,2]dioxaborolan-2-yl)-1H-benzoimidazole
g. 6-(2-Chloro-5-fluoro-pyrimidin-4-yl)-4-fluoro-1-isopropyl-2-methyl-1H-benzoimidazole
h. [5-(4-Ethyl-piperazin-1-ylmethyl)-pyridin-2-yl]-[5-fluoro-4-(7-fluoro-3-isopropyl-2-methyl-3H-benzoimidazol-5-yl)-pyrimidin-2-yl]-amine Crystalline form III


| Patent | Submitted | Granted |
|---|---|---|
| PROTEIN KINASE INHIBITORS [US7855211] | 2010-06-24 | 2010-12-21 |
| Human papilloma virus as predictor of cancer prognosis [US8673972] | 2013-08-02 | 2014-03-18 |
| HUMAN PAPILLOMA VIRUS AS PREDICTOR OF CANCER PROGNOSIS [US2015030587] | 2014-02-11 | 2015-01-29 |
/////////LY 2835219, Abemaciclib, Bemaciclib
CCN1CCN(CC1)Cc2ccc(nc2)Nc3ncc(c(n3)c4cc5c(c(c4)F)nc(n5C(C)C)C)F
Ribociclib
Ribociclib (LEE 011)
CAS: 1211441-98-3
Chemical Formula: C23H30N8O
Exact Mass: 434.25426
7-Cyclopentyl-2-(5-piperazin-1-yl-pyridin-2-ylamino)-7H-pyrrolo[2,3-d]pyrimidine-6-carboxylic acid dimethylamide
FDA UNII
Current developer: Novartis /Astex Pharmaceuticals.
Novartis Ag, Astex Therapeutics Ltd.
NMR.http://file.selleckchem.com/downloads/nmr/S744002-LEE011-2-HNMR-Selleck%20.pdf
http://file.selleckchem.com/downloads/hplc/S744002-LEE011-2-HPLC-Selleck.pdf
Ribociclib (LEE011) is an orally available, and highly specific CDK4/6 inhibitor. Phase 3.
CDK4 AND 6
(Cell-free assay)Product Ingredients
| Kisqali | FDA 3/13/2017 | To treat postmenopausal women with a type of advanced breast cancer Drug Trials Snapshot |

| INGREDIENT | UNII | CAS | INCHI KEY |
|---|---|---|---|
| Ribociclib hydrochloride | 63YF7YKW7E | 1211443-80-9 | JZRSIQPIKASMEV-UHFFFAOYSA-N |
| Ribociclib succinate | BG7HLX2919 | 1374639-75-4 | NHANOMFABJQAAH-UHFFFAOYSA-N |
RIBOCICLIB SUCCINATE
STRUCTURE ….LINK
Ribociclib is in phase III clinical trials by Novatis for the treatment of postmenopausal women with advanced breast cancer.
Phase II clinical trials are also in development for the treatment of liposarcoma, ovarian cancer, fallopian tube cancer, peritoneum cancer, endometrial cancer, and gastrointestinal cancer.
Ribociclib, also known as LEE011, is an orally available cyclin-dependent kinase (CDK) inhibitor targeting cyclin D1/CDK4 and cyclin D3/CDK6 cell cycle pathway, with potential antineoplastic activity. CDK4/6 inhibitor LEE011 specifically inhibits CDK4 and 6, thereby inhibiting retinoblastoma (Rb) protein phosphorylation. Inhibition of Rb phosphorylation prevents CDK-mediated G1-S phase transition, thereby arresting the cell cycle in the G1 phase, suppressing DNA synthesis and inhibiting cancer cell growth. Overexpression of CDK4/6, as seen in certain types of cancer, causes cell cycle deregulation
Orally bioavailable CDK4/6-selective inhibitor that has been tested in Phase III clinical trials for treatment of advanced breast cancer.
CDK full name of cyclin-dependent kinases, there are many other subtypes CDK1-11, capable of binding to cell cycle proteins regulate the cell cycle. Pfizer Palbociclib been submitted for FDA review under phase II clinical data, Novartis Ribociclib (LEE011), Lilly Abemaciclib (LY2835219) the three CDK4 / 6 inhibitors have entered late stage development for the treatment of breast cancer
SYNTHESIS

WO2010020675
US20120115878
WO2010020675
http://www.google.co.in/patents/WO2010020675A1?cl=en
Example 74
7-Cyclopentyl-2-(5-piperazin-1-yl-pyridin-2-ylamino)-7H-pyrrolo[2,3-d]pyrimidine-6-carboxylic acid dimethylamide
Following Buchwald Method B, then General Procedure A, 2-chloro-7-cyclopentyl-7H- pyrrolo[2,3-d]pyrimidine-6-carboxylic acid dimethylamide (300 mg, 1.02 mmol) and 5-piperazin-1- yl-pyridin-2-ylamine (314 mg, 1.13 mmol) gave 7-cyclopentyl-2-(5-piperazin-1-yl-pyridin-2- ylamino)-7H-pyrrolo[2,3-d]pyrimidine-6-carboxylic acid dimethylamide (142 mg, 36%). MS(ESI) m/z 435.3 (M+H)+
POSTER

SYNTHESIS
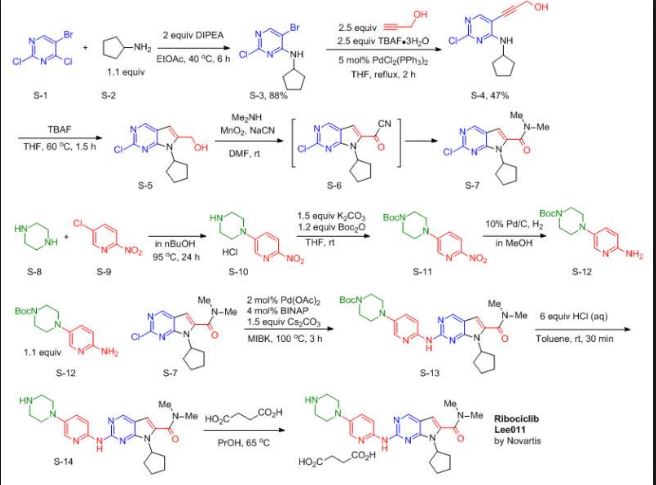


TAKEN FROM ….http://www.joygooo.com/news_71.htm?pageNum=21
PCT Int Appl, WO2012061156.
US Pat Appl Publ, US20120115878
PCT Int Appl, WO2011130232 5) Brain, Christopher Thomas et al; Preparation of pyrrolopyrimidine Derivatives for Use as CDK4 / 6 inhibitors;. PCT Int Appl, WO2011101409.
PCT Int Appl, WO2011101417. 7) Besong, Gilbert et al;.
PCT Int Appl, WO2010020675.
PCT Int Appl, WO2007140222.
Reference:1. WO2012064805A1 / US20120115878A1.
2. WO2010020675A1 / US8415355B2.
3. WO2011130232A1 / US20130035336A1.
Clinical Trial Information( data from http://clinicaltrials.gov, updated on 2015-10-17)
| NCT Number | Recruitment | Conditions | Sponsor /Collaborators |
Start Date | Phases |
|---|---|---|---|---|---|
| NCT02571829 | Not yet recruiting | Liposarcoma|Soft Tissue Sarcoma | Hadassah Medical Organization | December 2015 | Phase 2 |
| NCT02524119 | Not yet recruiting | Hepatocellular Carcinoma | University of Texas Southwestern Medical Center|Novartis …more | November 2015 | Phase 2 |
| NCT02494921 | Recruiting | Prostate Cancer | Rahul Aggarwal|University of California, San Francisco | September 2015 | Phase 1|Phase 2 |
| NCT02420691 | Recruiting | Gastrointestinal Cancer | M.D. Anderson Cancer Center|Novartis | August 2015 | Phase 2 |
| NCT02431481 | Not yet recruiting | Normal Renal Function|Impaired Renal Function | Novartis Pharmaceuticals|Novartis | August 2015 | Phase 1 |
Protocols from literature
|
In vitro protocol:: |
Pharmacologic growth inhibition: Clin Cancer Res. 2013 Nov 15;19(22):6173-82. Cell-cycle analysis: Clin Cancer Res. 2013 Nov 15;19(22):6173-82. Senescence and apoptosis assays: Clin Cancer Res. 2013 Nov 15;19(22):6173-82. |
|
In vivo protocol: |
Xenograft therapeutic trials: Clin Cancer Res. 2013 Nov 15;19(22):6173-82 Immunohistochemistry of xenografted neuroblastomas.Clin Cancer Res. 2013 Nov 15;19(22):6173-82 |
Ribociclib (LEE011) is a Me-Too version of palbociclib. Their structures are compared side-by-side as the following:
 |
Ribociclib (LEE011) is currently being developed by Novartis and Astex. According its Novartis’s website, LEE011 is a novel, orally available, selective inhibitor of CDK4/6 kinases, which induces complete dephosphorylation of Rb and G1 arrest in cancer cells. In preclinical in vitro and in vivo tumor models, LEE011 has been shown active in cancers harboring aberrations that increase CDK4/6 activity, including those directly linked to the kinases as well as activating alterations in the upstream regulators. First-in-human study of LEE011 in patients with solid tumors and lymphoma is currently ongoing. (source: http://www.novartisoncology.us/research/pipeline/lee011.jsp).
Treatment with LEE011 significantly reduced proliferation in 12 of 17 human neuroblastoma-derived cell lines by inducing cytostasis at nanomolar concentrations (mean IC50 = 307 ± 68 nmol/L in sensitive lines). LEE011 caused cell-cycle arrest and cellular senescence that was attributed to dose-dependent decreases in phosphorylated RB and FOXM1, respectively. In addition, responsiveness of neuroblastoma xenografts to LEE011 translated to the in vivo setting in that there was a direct correlation of in vitro IC50 values with degree of subcutaneous xenograft growth delay. Although our data indicate that neuroblastomas sensitive to LEE011 were more likely to contain genomic amplification of MYCN (P = 0.01), the identification of additional clinically accessible biomarkers is of high importance. LEE011 is active in a large subset of neuroblastoma cell line and xenograft models, and supports the clinical development of this CDK4/6 inhibitor as a therapy for patients with this disease. (Clin Cancer Res. 2013 Nov 15;19(22):6173-82)
|
References |
1. Rader J, Russell MR, Hart LS, Nakazawa MS, Belcastro LT, Martinez D, Li Y, Carpenter EL, Attiyeh EF, Diskin SJ, Kim S, Parasuraman S, Caponigro G, Schnepp RW, Wood AC, Pawel B, Cole KA, Maris JM. Dual CDK4/CDK6 inhibition induces cell-cycle arrest and senescence in neuroblastoma. Clin Cancer Res. 2013 Nov 15;19(22):6173-82. doi: 10.1158/1078-0432.CCR-13-1675. Epub 2013 Sep 17. PubMed PMID: 24045179; PubMed Central PMCID: PMC3844928.
2. Caponigro, Giordano; Stuart, Darrin; Kim, Sunkyu; Loo, Alice; Delach, Scott. Pharmaceutical combinations of a CDK4/6 inhibitor and a B-RAF inhibitor for treatment of proliferative diseases such as cancer. PCT Int. Appl. (2014), WO 2014018725 A1 20140130.
3. Kim, Sunkyu; Doshi, Shivang; Haas, Kristy; Kovats, Steven; Huang, Alan Xizhong; Chen, Yan. Combination therapy comprising a cyclin dependent kinase 4/6 (CDK4/6) inhibitor and a phosphatidylinositol 3-kinase (PI3K) inhibitor for use in the treatment of cancer. PCT Int. Appl. (2013), WO 2013006532 A1 20130110
4. Kim, Sunkyu; Doshi, Shivang; Haas, Kristy; Kovats, Steven. Combination of cyclin dependent kinase 4/6 (CDK4/6) inhibitor and fibroblast growth factor receptor (FGFR) kinase inhibitor for the treatment of cancer. PCT Int. Appl. (2013), WO 2013006368 A1 20130110
5. Calienni, John Vincent; Chen, Guang-Pei; Gong, Baoqing; Kapa, Prasad Koteswara; Saxena, Vishal. Salt(s) of 7-cyclopentyl-2-(5-piperazin-1-yl-pyridin-2-ylamino-7H-pyrrolo[2,3-d]pyrimidine-6-carboxylic acid dimethylamide and processes of making thereof. U.S. Pat. Appl. Publ. (2012), US 20120115878 A1 20120510.
6. Borland, Maria; Brain, Christopher Thomas; Doshi, Shivang; Kim, Sunkyu; Ma, Jianguo; Murtie, Josh; Zhang, Hong. Combination comprising a cyclin dependent kinase 4 or cyclin dependent kinase (cdk4/6) inhibitor and an Mtor inhibitor for treating cancer. PCT Int. Appl. (2011), WO 2011130232 A1 20111020
7. Besong, Gilbert; Brain, Christopher Thomas; Brooks, Clinton A.; Congreve, Miles Stuart; Dagostin, Claudio; He, Guo; Hou, Ying; Howard, Steven; Li, Yue; Lu, Yipin; et al. Preparation of pyrrolopyrimidine compounds as CDK inhibitors. PCT Int. Appl. (2010), WO 2010020675 A1 20100225.
CLIP
Cyclin-dependent kinase inhibitors (14 compounds) under clinical evaluation.

LEE-011 is one of the most selective inhibitors for CDK4 and CDK6 [59] and is being developed by Astex Pharmaceuticals™ and Novartis. In January 2014 this inhibitor entered phase III clinical trials for the treatment of breast cancer [60]. Due to encouraging results LEE-011 has now become the main competing drug-candidate with Pfizer’s PD0332991 (palbociclib), see Figure 3 [59].

Upon comparison of the chemical structure of Novartis’ LEE-011 and Pfizer’s PD0332991, the similarity is evident. The major difference lies in the bicyclic core since LEE-011 possesses a pyrrolo-pyrimidine and PD0332991 a pyridopyrimidine. The “east” part of the structure is also modified. The structural similarities make their analogous CDKs inhibition profiles (high selectivity for CDK4 and CDK6) quite obvious Moreover, both derivatives are orally administered which is pretty advantageous compared with dinaciclib, which is also in phase III clinical trials but is administered intravenously.
http://www.mdpi.com/1420-3049/19/9/14366/htm
Sources:
1)Rader, JulieAnn et al.;Dual CDK4/CDK6 Inhibition Induces Cell-Cycle Arrest and Senescence in Neuroblastoma;Clinical Cancer Research (2013), 19(22), 6173-6182
2)Tavares, Francis X. and Strum, Jay C.;Preparation of pyrazinopyrrolopyrimidine derivatives and analogs for use as CDK inhibitors;PCT Int. Appl., WO2012061156
3)Calienni, John Vincent et al.;Salt(s) of 7-cyclopentyl-2-(5-piperazin-1-yl-pyridin-2-ylamino)-7H-pyrrolo[2,3-d]pyrimidine-6-carboxylic acid dimethylamide and processes of making thereof;U.S. Pat. Appl. Publ., US20120115878
4)Borland, Maria et al;Combination comprising a cyclin dependent kinase 4 or cyclin dependent kinase (cdk4/6) inhibitor and an Mtor inhibitor for treating cancer;PCT Int. Appl., WO2011130232
5)Brain, Christopher Thomas et al;Preparation of pyrrolopyrimidine derivatives for use as CDK4/6 inhibitors;PCT Int. Appl., WO2011101409
6)Brain, Christopher Thomas and Perez, Lawrence Blas; Preparation of deuterated pyrrolopyrimidine compounds as inhibitors of CDK4/6 for treating cancer; PCT Int. Appl., WO2011101417
7)Besong, Gilbert et al.;Preparation of pyrrolopyrimidine compounds as CDK inhibitors;PCT Int. Appl., WO2010020675
8)Brain, Christopher Thomas et al.;Preparation of pyrrolopyrimidine compounds as protein kinase inhibitors; PCT Int. Appl., WO2007140222
9)A Randomized Double-blind, Placebo-controlled Study of LEE011 in Combination With Letrozole for the Treatment of Postmenopausal Women With Hormone Receptor Positive, HER2 Negative, Advanced Breast Cancer Who Received no Prior Therapy for Advanced Disease;ClinicalTrials.gov Identifier: NCT01958021
/////////Ribociclib, novartis, LEE011, astex, phase 3, CDK inhibitors
CN(C)C(=O)c1cc2cnc(nc2n1C3CCCC3)Nc4ccc(cn4)N5CCNCC5

VARDENAFIL
224785-90-4 CAS NO
Vardenafil hydrochloride (CAS NO.224785-91-5)
| Formula | C23H32N6O4S |
|---|---|
| Mol. mass | 488.604 g/mol |
4-[2-Ethoxy-5-(4-ethylpiperazin-1-yl)sulfonyl-phenyl]-9-methyl-7-propyl-3,5,6,8-tetrazabicyclo[4.3.0]nona-3,7,9-trien-2-one
Vivanza, Vardenafil (INN), Levitra (TN), STK642629, , LEVITRA
Vardenafil (INN) is a PDE5 inhibitor used for treating erectile dysfunction that is sold under the trade names Levitra (Bayer AG, GSK, and SP) andStaxyn.
Vardenafil was co-marketed by Bayer Pharmaceuticals, GlaxoSmithKline, and Schering-Plough under the trade name Levitra. As of 2005, the co-promotion rights of GSK on Levitra have been returned to Bayer in many markets outside the U.S. In Italy, Bayer sells vardenafil as Levitra and GSK sells it as Vivanza. Thus, because of European Union trade rules, parallel imports might result in Vivanza sold next to Levitra in the EU.
Vardenafil (Levitra) is an oral therapy for the treatment of erectile dysfunction. It is a selective inhibitor of cyclic guanosine monophosphate (cGMP)-specific phosphodiesterase type 5 (PDE5). Penile erection is a hemodynamic process initiated by the relaxation of smooth muscle in the corpus cavernosum and its associated arterioles. During sexual stimulation, nitric oxide is released from nerve endings and endothelial cells in the corpus cavernosum. Nitric oxide activates the enzyme guanylate cyclase resulting in increased synthesis of cyclic guanosine monophosphate (cGMP) in the smooth muscle cells of the corpus cavernosum. The cGMP in turn triggers smooth muscle relaxation, allowing increased blood flow into the penis, resulting in erection. The tissue concentration of cGMP is regulated by both the rates of synthesis and degradation via phosphodiesterases (PDEs). The most abundant PDE in the human corpus cavernosum is the cGMPspecific phosphodiesterase type 5 (PDE5); therefore, the inhibition of PDE5 enhances erectile function by increasing the amount of cGMP.
An orally disintegrating form, marketed as Staxyn, has been gaining approvals in countries such as the United States[1] and Canada.[2]
Vardenafil’s indications and contra-indications are the same as with other PDE5 inhibitors; it is closely related in function to sildenafil citrate (Viagra) and tadalafil (Cialis). The difference between the vardenafil molecule and sildenafil citrate is a nitrogen atom’s position and the change of sildenafil’spiperazine ring methyl group to an ethyl group. Tadalafil is structurally different from both sildenafil and vardenafil. Vardenafil’s relatively short effective time is comparable to but somewhat longer than sildenafil’s.
Beyond its indications for erectile dysfunction, vardenafil may be effective in the treatment of premature ejaculation, where it may significantly increase the time from vaginal penetration to ejaculation.[3]
The common, adverse drug reactions (side-effects) are the same as with other PDE5 inhibitors. The frequent vardenafil-specific side-effect is nausea; the infrequent side-effects are abdominal pain, back pain, photosensitivity, abnormal vision, eye pain, facial edema, hypotension, palpitation,tachycardia, arthralgia, myalgia, rash, itch, and priapism.

One possibly serious, but rare, side-effect with vardenafil is heart attack. Also, in rare cases, vardenafil use may cause priapism, a very painful emergency condition that can cause impotence if left untreated.[4]
On 18 October 2007, the U.S. Food and Drug Administration (FDA) announced that a warning about possible deafness (sudden hearing loss) would be added to the drug labels of Vardenafil, and other PDE5 inhibitors.[5]
Vardenafil, as with all PDE5 inhibitors, should not be used by men taking nitrate medications, because combining them with vardenafil might provoke potentially life-threatening hypotension (low blood pressure).
Further, Vardenafil causing lengthening of the QT interval. Therefore it should not be taken by men taking other medications that affect the QT interval (such as amiodarone).
It is available in 2.5 mg, 5 mg, 10 mg, and 20 mg doses in round orange tablets. The normal starting dose is 10 mg (roughly equivalent to 50 mg of sildenafil). Vardenafil should be taken 1 to 2 hours prior to sexual activity, with a maximum dose frequency of once per day. In some territories, such as the UK, only certain doses may be available.
Vardenafil is also available under the name Staxyn as a tablet which dissolves on the tongue rather than being swallowed in the form of a pill.
STAXYN is an oral therapy for the treatment of erectile dysfunction. This monohydrochloride salt of vardenafil is a selective inhibitor of cyclic guanosine monophosphate (cGMP)-specific PDE5.
Vardenafil HCl is designated chemically as piperazine, 1-[[3-(1,4-dihydro-5-methyl-4-oxo-7-propylimidazo[5,1f][1,2,4]triazin-2-yl)-4-ethoxyphenyl]sulfonyl]-4-ethyl-, monohydrochloride and has the following structural formula:
 |
Vardenafil HCl is a nearly colorless, solid substance with a molecular weight of 579.1 g/mol and a solubility of 0.11 mg/mL in water.
TRIHYDRATE, HCL SALT

vardenafil hydrochloride is piperazine, 1-[[3-(1,4-dihydro-5-methyl-4-oxo-7-propylimidazo[5,1-f][1,2,4]triazin-2-yl)-4-ethoxyphenyl]sulfonyl]-4-ethyl-, mono -hydrochloride and can be structurally represented by Formula I.
The monohydrochloride salt of vardenafil is a selective inhibitor of cyclic guaosine monophosphate (cGMP)-specific phosphodiesterase type 5 (PDE5). It is commercially available in products sold under the brand name LEVITRA formulated as 2.5 mg, 5 mg, 10 mg, 20 mg film-coated tablets.
U.S. Pat. No. 6,362,178 B1 discloses vardenafil, its related compounds and processes for their preparation. The patent describes a process in which vardenafil is obtained by recrystallization in ether in Example 19. Vardenafil produced as per Example 19 is hereinafter referred as “crystalline Form I” of vardenafil. The patent also describes processes for the preparation of its monohydrochloride and dihydrochloride salts, which are formed in a combination of ether and dichloromethane. The patent also describes a process for the preparation of vardenafil monohydrochloride trihydrate.
U.S. Patent Application Publication No. 2005/0203298 also describes a process for the preparation of vardenafil, and its monohydrochloride trihydrate.
Chemical synthesis of vardenafil has mostly been directed to the preparation of the trihydrate of monohydrochloride of vardenafil.
In WO 99/24433, sulphonamide-substituted imidazotriazinones are described as potent inhibitors of either one or more of the cyclic guanosine 3′,5′-monophosphate-metabolizing phosphodiesterases (cGMP PDEs). According to the nomenclature of Beavo and Reifsnyder (Trends in Pharmacol. Sci. 11, 150-155, 1990), these cGMP PDEs are the phosphodiesterase isoenzymes PDE-I, PDE-II and PDE-V.
According to WO 99/24433, the sulphonamide-substituted imidazotriazinones described therein are prepared from corresponding 2-ethoxyphenyl-substituted imidazotriazinones by reaction with chlorosulphonic acid and subsequent reaction with an appropriate amine, as is illustrated by the following scheme (R1 to R6 here have the meanings indicated in WO 99/24433):
In this process, highly reactive chlorosulphonic acid has to be used as a reagent. Moreover, the imidazotriazinonesulphonyl chlorides formed as intermediates are sensitive to hydrolysis, which, in particular in the conversion of this preparation process to the industrial scale, can lead to not inconsiderable yield variations.
It was therefore the object of the present invention to make available a process for the preparation of sulphonamide-substituted imidazotriazinones in which the disadvantages of the above process known from the prior art are avoided.
This object is achieved according to the present invention by a process as in claim 1. In particular, in the process according to the invention as in claim 1 the use of chlorosulphonic acid is avoided by introduction of the sulphonic acid via a reaction with sulphuric acid and subsequent reaction with thionyl chloride. Moreover, the reaction with thionyl chloride and the subsequent reaction with an amine is carried out in a one-pot process, so that the imidazotriazinonesulphonyl chloride intermediate, which is sensitive to hydrolysis, does not need to be isolated. By means of this, yield variations on account of partial hydrolysis of this intermediate can be excluded. As a result of these advantages, the process according to the invention is much simpler to carry out on the industrial scale than the process described in WO 99/24433.
………………….
SYNTHESIS
2-butyrylamino-propionic acid
EXAMPLE 1A 2-Butyrylaminopropionic acid
22.27 g (250 mmol) of D,L-alanine and 55.66 g (550 mmol) of triethylamine are dissolved in 250 ml of dichloromethane, and the solution is cooled to 0° C. 59.75 g (550 mmol) of trimethylsilyl chloride are added dropwise, and the solution is stirred for 1 hour at room temperature and for 1 hour at 40° C. After cooling to −10° C., 26.64 g (250 mmol) of butyryl chloride are added dropwise, and the resulting mixture is stirred for 2 hours at −10° C. and for one hour at room temperature.
With ice-cooling, 125 ml of water are added dropwise and the reaction mixture is stirred at room temperature for 15 minutes. The aqueous phase is evaporated to dryness, the residue is titrated with acetone and the mother liquor is filtered off with suction. The solvent is removed and the residue is chromatographed. The resulting product is dissolved in 3N aqueous sodium hydroxide solution and the resulting solution is evaporated to dryness. The residue is taken up in conc. HCl and once more evaporated to dryness. The residue is stirred with acetone, precipitated solid is filtered off with suction and the solvent is removed under reduced pressure. This gives 28.2 g (71%) of a viscous oil which crystallizes after some time.
200 MHz 1H-NMR (DMSO-d6): 0.84, t, 3H; 1.22, d, 3H; 1.50, hex, 2H; 2.07, t, 2H; 4.20, quin., 1H; 8.09, d, 1H.
EXAMPLE 3A 2-Ethoxybenzonitrile
25 g (210 mmol) of 2-hydroxybenzonitrile are refluxed with 87 g of potassium carbonate and 34.3 g (314.8 mmol) of ethyl bromide in 500 ml of acetone overnight. The solid is filtered off, the solvent is removed under reduced pressure and the residue is distilled under reduced pressure. This gives 30.0 g (97%) of a colourless liquid.
200 MHz 1H-NMR (DMSO-d6): 1.48, t, 3H; 4.15, quart., 2H; 6.99, dt, 2H; 7.51, dt, 2H.
EXAMPLE 4A 2-Ethoxybenzamidine hydrochloride
21.4 g (400 mmol) of ammonium chloride are suspended in 375 ml of toluene, and the suspension is cooled to 0° C. 200 ml of a 2M solution of trimethylaluminium in hexane are added dropwise, and the mixture is stirred at room temperature until the evolution of gas has ceased. After addition of 29.44 g (200 mmol) of 2-ethoxybenzonitrile, the reaction mixture is stirred at 80° C. (bath) overnight.
With ice-cooling, the cooled reaction mixture is added to a suspension of 100 g of silica gel and 950 ml of chloroform, and the mixture is stirred at room temperature for 30 minutes. The mixture is filtered off with suction, and the filter residue is washed with the same amount of methanol. The mother liquor is concentrated, the resulting residue is stirred with a mixture of dichloromethane and methanol (9:1), the solid is filtered off with suction and the mother liquor is concentrated. This gives 30.4 g (76%) of a colourless solid.
200 MHz 1H-NMR (DMSO-d6): 1.36, t, 3H; 4.12, quart., 2H; 7.10, t, 1H; 7.21, d, 1H; 7.52, m, 2H; 9.30, s, broad, 4H.
EXAMPLE 10A 2-(2-Ethoxy-phenyl)-5-methyl-7-propyl-3H-imidazo[5,1-f][1,2,4]triazin-4-one
7.16 g (45 mmol) of 2-butyrylamino-propionic acid and 10.67 g of pyridine are dissolved in 45 ml of THF and, after addition of a spatula tip of DMAP, heated to reflux. 12.29 g (90 mmol) of ethyl oxalyl chloride are slowly added dropwise, and the reaction mixture is refluxed for 3 hours. The mixture is poured into ice-water and extracted three times with ethyl acetate and the organic phase is dried over sodium sulphate and concentrated using a rotary evaporator. The residue is taken up in 15 ml of ethanol and refluxed with 2.15 g of sodium bicarbonate for 2.5 hours. The cooled solution is filtered.
With ice-cooling, 2.25 g (45 mmol) of hydrazine hydrate are added dropwise to a solution of 9.03 g (45 mmol) of 2-ethoxybenzamidine hydrochloride in 45 ml of ethanol, and the resulting suspension is stirred at room temperature for another 10 minutes. The ethanolic solution described above is added to this reaction mixture, and the mixture is stirred at a bath temperature of 70° C. for 4 hours. After filtration, the mixture is concentrated, the residue is partitioned between dichloromethane and water, the organic phase is dried over sodium sulphate and the solvent is removed under reduced pressure.
This residue is dissolved in 60 ml of 1,2-dichloroethane and, after addition of 7.5 ml of phosphorus oxychloride, refluxed for 2 hours. The mixture is diluted with dichloromethane and neutralized by addition of sodium bicarbonate solution and solid sodium bicarbonate. The organic phase is dried and the solvent is removed under reduced pressure. Chromatography using ethyl acetate and crystallization afford 4.00 g (28%) of a colourless solid, Rf=0.42 (dichloromethane/methanol=95:5)
200 MHz 1H-NMR (CDCl3): 1.02, t, 3H; 1.56, t, 3H; 1.89, hex, 2H; 2.67, s, 3H; 3.00, t, 2H; 4.26, quart., 2H; 7.05, m, 2H; 7.50, dt, 1H; 8.17, dd, 1H; 10.00, s, 1H.
EXAMPLE 15A 4-Ethoxy-3-(5-methyl-4-oxo-7-propyl-3,4-dihydro-imidazo[5,1-f][1,2,4]triazin-2-yl)-benzenesulphonyl chloride
At 0° C., 2.00 g (6.4 mmol) of 2-(2-ethoxy-phenyl)-5-methyl-7-propyl-3H-imidazo[5,1-f][1,2,4]triazin-4-one are slowly added to 3.83 ml of chlorosulphonic acid. At room temperature, the reaction mixture is stirred ovemight, and then poured into ice-water and extracted with dichloromethane. This gives 2.40 g (91%) of a colourless foam.
200 MHz 1H-NMR (CDCl3): 1.03, t, 3H; 1.61, t, 2H; 1.92, hex, 2H; 2.67, s, 3H; 3.10, t, 2H; 4.42, quart., 2H; 7.27, t, 1H; 8.20, dd, 1H; 8.67, d, 1H; 10.18, s, 1H.
Example 19 2-[2-Ethoxy-5-(4-ethyl-piperazine-1-sulphonyl)-phenyl]-5-methyl-7-propyl-3H-imidazo[5,1-f][1,2,4]triazin-4-one
470 mg (1.14 mmol) of 4-ethoxy-3-(5-methyl-4-oxo-7-propyl-3,4-dihydro-imidazo[5,1-f][1,2,4]triazin-2-yl)-benzenesulphonyl chloride are dissolved in 20 ml of dichloromethane and cooled to 0° C. 390 mg (3.42 mmol) of N-ethylpiperazine are added, and the reaction mixture is stirred at room temperature overnight. The mixture is diluted with dichloromethane, the organic phase is washed twice with water and dried over sodium sulphate and the solvent is removed under reduced pressure. Crystallization from ether gives 370 mg (66%) of a colourless solid.
400 MHz 1H-NMR (CDCl3): 1.01, t, 3H; 1.59, t, 3H; 1.88, hex, 2H; 2.42, quart., 2H; 2.56, m, 4H; 2.63, s, 3H; 3.00, t, 2H; 3.10, m, 4H; 4.33, quart., 2H, 7.17, d, 1H; 7.88, dd, 1H; 8.44, d, 1H; 9.75, s, 1H.
…………………….
EXAMPLE 7 Preparation of the Trihydrate of Vardenafil Monohydrochloride
14 g of vardenafil hydrochloride was taken into a round bottom flask followed by the addition of 70 ml water and the pH of the reaction mass was adjusted using sodium hydroxide to 11 at 30° C. 280 ml of dichloromethane was added to the above reaction mass and the layers were separated. The organic layer was dried over sodium sulfate and the organic layer was transferred into a round bottom flask and subjected to heating for distillation at 40° C. for 1.5 hours. The solid material was transferred into a round bottom flask and 36 ml of a mixture of acetone and water in 12:1 ratio was added with stirring, then 2.2 ml of 36% aqueous hydrochloric acid was added with stirring. The reaction mass was heated to a temperature of about 45° C. and the undissolved particles were removed by filtration. The filtrate was taken into a round bottom flask and cooled to 5° C., maintained for 45 minutes at 3 to 5° C. followed by the filtration of the solid which was then subjected to suction drying and finally dried at 40° C. to yield 9.0 g of the trihydrate of vardenafil monohydrochloride.
……………………..
STARTING COMPOUNDS
Example I Preparation of 2-(2-ethoxyphenyl)-5-methyl-7-propyl-3H-imidazo-[5,1-f][2,4]triazin-4-oneIa) Preparation of 2-butyrylaminopropionic acid
A solution of 100 kg of D,L-alanine in aqueous sodium hydroxide solution is reacted in the cold with 119 kg of butyryl chloride. After addition of butyl acetate, the mixture is acidified with hydrochloric acid, the organic phase is separated off and the aqueous phase is re-extracted. The organic phase is dried by azeotropic distillation. The crystallizate is isolated, washed with butyl acetate and dried.
Yield: 132.6 kg (68%)
1H-NMR: δ=0.8 (t, 3H), 1.25 (d, 3H), 1.5 (m, 2H), 2.1 (t, 2H), 4.2 (q, 1H), 8.1 (d, NH), 12.0-12.7 (s, COOH)
MS: 336 (2M+NH4, 40), 319 (2M+H, 15), 177 (M+NH4, 100), 160 (M+H, 20)
Ib) Preparation of 2-ethoxybenzonitrile
260 kg of thionyl chloride are added at 85-95° C. to a suspension of 250 kg of 2-ethoxybenzamide in toluene under metering control. The reaction mixture is stirred in the presence of heat. Thionyl chloride and toluene are then distilled off in vacuo. The product is employed in the subsequent stage as a crude product.
Yield: 228.5 kg (crude product)
1H-NMR: δ=1.45 (t, 3H), 4.15 (q, 2H), 7.0 (m, 2H, phenyl), 7.5 (m, 2H, phenyl)
MS: 312 (2M+N4, 35), 165 (M+NH4, 100), 147 (5)
Ic) Preparation of 2-ethoxy-N-hydroxybenzamidine
111 kg of 2-ethoxybenzonitrile (crude product) from Example Ib are heated under reflux with 164 1 of triethylamine and 73 kg of hydroxylamine hydrochloride in isopropanol. The reaction mixture is treated with water and cooled. The crystallizate is isolated, washed and employed in the subsequent stage as a moist product.
Yield: 92.6 kg (moist product)
1H-NMR: δ=1.35 (t, 3H), 4.1 (q, 2H), 5.6 (s, 2H), 6.9-7.4 (4H, phenyl), 9.4 (s, 1H, OH)
MS: 361 (2M+H, 30), 198 (M+N, 30), 181 (M+H, 100)
Id) Preparation of 2-ethoxybenzamidine hydrochloride
135 kg of 2-ethoxy-N-hydroxybenzamidine (moist product) from Example Ic are hydrogenated at 50-60° C. in acetic acid using palladium on carbon as a catalyst. For the work-up, the hydrogenation reaction is freed from the catalyst, treated with hydrochloric acid and concentrated. Residual acetic acid and water are removed by azeotropic distillation with toluene. The crystallizate is isolated and dried in vacuo.
Yield: 136.4 kg
H-NMR: 1.35 (t, 3H), 4.15 (q, 2H), 7.1-7.7 (m, 4H, phenyl), 9.1-9.4 (2×s, 3H), 10.5-10.7 (s, 1H)
MS: 329 (2M+H, 10), 165 (M+H, 100)
Ie) Preparation of 2-(2-ethoxyphenyl)-5-methyl-7-propyl-3H-imidazo[5,1-f][1,2,4]-triazin-4-one
231 kg of 2-butyrylaminopropionic acid from Example Ia are treated in tetrahydrofuran with 341 kg of pyridine, catalytic amounts of 4-N,N-dimethylaminopyridine and 392 kg of ethyl chloroxalate and stirred with heating under reflux. The reaction mixture is taken up in ethyl acetate, washed with water and the ethyl acetate phase is concentrated. The distillation residue is taken up in methanol and reacted with the following solution.
192 kg of 2-ethoxybenzamidine hydrochloride from Example Id are treated in methanol with 47.5 kg of hydrazine hydrate and the mixture is stirred at room temperature. The solution is combined with the solution of 2-butyrylamino-1-ethoxycarbonylpropenyl ethyl oxalate prepared above. The reaction mixture thus obtained is stirred with heating under reflux. Methanol is removed by distillation and replaced by acetic acid.
Option A:
138.6 kg of phosphorus oxychloride are added and stirred in the presence of heat.
Acetic acid is distilled off in vacuo. The residue is treated with water and dichloromethane or optionally methyl isobutyl ketone and rendered neutral using sodium hydroxide solution. The organic phase is concentrated, and the residue is dissolved in acetone and crystallized with cooling. The crystallizate is isolated, washed and dried.
Option B:
At least 190 kg of acetyl chloride are added and stirred in the presence of heat. Acetic acid is distilled off in vacuo. The distillation residue is treated with acetone and water, and the product is crystallized by rendering neutral with sodium hydroxide solution. The product is isolated, washed and dried.
Yield: 90-160 kg
1H-NMR: δ=1.0 (t, 3H), 1.6 (t, 3H), 1.9 (m, 2H), 2.8 (s, 3H), 3.3 (t, 2H), 4.3 (q, 2H), 7.0-8.2 (Ar, 4H), 10.3 (CONH, 1H)
MS: 313 (M+H, 100), 149 (25), 151 (40), 121 (15)
HPLC: Kromasil C-18 phase, neutral phosphate buffer, acetonitrile, 233 nm, linear gradient of 30% acetonitrile ->80% acetonitrile (30 min.): 99 area % (Rt 19.1)
PREPARATION EXAMPLES Example 1a 4-ethoxy-3-(5-methyl-4-oxo-7-propyl-3,4-dihydroimidazo[5,1-fl-][1,2,4]triazin-2-yl)benzenesulphonic acid
194 kg of 2-(2-ethoxyphenyl)-5-methyl-7-propyl-3H-imidazo[5,1-f][1,2,4]triazin-4-one from Example Ie are reacted with 504 kg of concentrated sulphuric acid. The reaction mixture is added to water, cooled, and the crystallizate is isolated and dried in vacuo.
Yield: 195.2 kg
1H-NMR: δ=0.95 (t, 3H), 1.3 (t, 3H), 1.8 (m, 2H), 2.6 (s, 3H), 3.05 (t, 2H), 4.1 (q, 2H), 7.15 (Ar, 1H), 7.75 (m, 2H), 12.3 (SO2OH)
MS: 393 (M+H, 100), 365 (25), 151 (40)
HPLC: X-Terra C-18 phase, aqueous phosphoric acid, acetonitrile, 242 nm, linear gradient of 10% acetonitrile ->90% acetonitrile (20 min.):
98 area % (R, 9.2)
Example 1b) 2-[2-ethoxy-5-(4-ethlylpiperazin-1-sulphonyl)phenyl]-5-methyl-7-propyl-3H-imidazo[5,1-f][1,2,4]triazin-4-one
22.5 kg of 4-ethoxy-3-(5-methyl-4-oxo-7-propyl-3,4-dihydro-imidazo[5,1-f][1,2,4]-triazin-2-yl)benzenesulphonic acid from Example 1a are reacted with 74 kg of thionyl chloride and catalytic amounts of dimethylformamide until the evolution of gas has ended. Xylene is repeatedly added to the reaction mixture and thionyl chloride is distilled off. 15.1 kg of N-ethylpiperazine are added to the suspension and it is stirred. After the addition of water, it is adjusted to pH 1 using hydrochloric acid, and the phases are separated. The aqueous phase is treated with acetone and rendered neutral by addition of sodium hydroxide solution. The mixture is cooled, and the crystallizate is isolated, washed and dried in vacuo.
Yield: 26.1 kg
1H-NMR: δ=1.0 (2×t, 6H), 1.6 (t, 3H), 1.9 (m, 2H), 2.45 (q, 2H), 2.55 (m, 4H), 2.65 (s, 3H), 3.0 (t, 2H), 3.1 (m, 4H), 4.35 (q, 2H), 7.15 (Ar, 1H), 7.9 (Ar, 1H), 8.4 (Ar, 1H), 9.8 (CONH)
MS: 489 (M+H, 100), 345 (10), 313, (10), 285 (10), 113 (20)
HPLC: X-Terra C-18 phase, neutral phosphate buffer, acetonitrile, 242 nm, linear gradient of 20% acetonitrile ->75% acetonitrile (20 min.): 98 area % (Rt 16.3)
1 c) 2-[2-ethoxy-5-(4-ethylpiperazin-1-sulphonyl)phenyl]-5-methyl-7-propyl-3H-imidazo[5,1-fl][1,2,4]triazin-4-one hydrochloride trihydrate
22.5 kg of 2-[2-ethoxy-5-(4-ethylpiperazin-1-sulphonyl)phenyl]-5-methyl-7-propyl-3H-imidazo[5,1-f][1,2,4]triazin-4-one from Example 1b are dissolved in 5.1 kg of concentrated hydrochloric acid and acetone/water (12:1 v/v) in the presence of heat. The clear solution is filtered hot and crystallized by cooling and seeding. The crystallizate is isolated, washed and dried in vacuo at about 30° C. and about 300 mbar.
Yield: 25.4 kg
M.p. (DSC): 192° C.
HPLC: X-Terra C-18 phase, neutral phosphate buffer, acetonitrile, 242 nm, linear gradient of 20% acetonitrile ->75% acetonitrile (20 min.): 99 area % (Rt 16.3)
|
2-1-2013
|
Synthesis of quinoline derivatives: discovery of a potent and selective phosphodiesterase 5 inhibitor for the treatment of Alzheimer’s disease.
|
European journal of medicinal chemistry
|
PATENTS
| US6362178 * | Oct 31, 1998 | Mar 26, 2002 | Bayer Aktiengesellschaft | 2-phenyl substituted imidazotriazinones as phosphodiesterase inhibitors |
| US20050203298 * | May 5, 2005 | Sep 15, 2005 | Bayer Healthcare Aktiengesellschaft | Process for the preparation of sulphonamide-substituted imidazotriazinones |
| US20060111354 * | Jul 3, 2003 | May 25, 2006 | Peter Serno | Medicaments containing vardenafil hydrochloride trihydrate |
| WO2004006894A1 * | Jul 3, 2003 | Jan 22, 2004 | Bayer Healthcare Ag | Medicaments containing vardenafil hydrochloride trihydrate |
|
11-4-2011
|
ROFLUMILAST FOR THE TREATMENT OF DIABETES MELLITUS
|
|
|
9-14-2011
|
Roflumilast for the Treatment of Diabetes Mellitus
|
|
|
8-5-2011
|
N-BUTYRAMIDE, THE PREPARATION METHOD AND USE THEREOF
|
|
|
3-4-2011
|
Fatty Acid Oxidation Inhibitors Treating Hyperglycemia and Related Disorders
|
|
|
1-14-2011
|
2-PHENYL SUBSTITUTED IMIDAZOTRIAZINONES AS PHOSPHODIESTERASE INHIBITORS
|
|
|
9-17-2010
|
SUBSTITUTED PDE5 INHIBITORS
|
|
|
7-16-2010
|
Combination treatment for diabetes mellitus
|
|
|
4-28-2010
|
2-Phenyl substituted imidazotriazinones as phosphodiesterase inhibitors
|
|
|
4-14-2010
|
2-PHENYL SUBSTITUTED IMIDAZOTRIAZINONES AS PHOSPHODIESTERASE INHIBITORS
|
|
|
2-5-2010
|
Heterocyclic Compounds And Uses Thereof In The Treatment Of Sexual Disorders
|
|
12-25-2009
|
Therapeutic Compositions Comprising a Specific Endothelin Receptor Antagonist and a PDE5 Inhibitor
|
|
|
11-27-2009
|
Substituted PDE5 inhibitors
|
|
|
9-4-2009
|
Uses of 2-Phenyl-Substituted Imidazotriazinone Derivatives for Treating Pulmonary Hypertension
|
|
|
8-28-2009
|
Roflumilast for the Treatment of Pulmonary Hypertension
|
|
|
8-7-2009
|
Use of Phosphodiesterase Inhibitor as a Component of Implantable Medical Devices
|
|
|
6-26-2009
|
Method for healing a wound using a phosphodiesterase type five inhibitor
|
|
|
3-20-2009
|
Pde5 inhibitor compositions and methods for immunotherapy
|
|
|
3-6-2009
|
Pde5 inhibitor compositions and methods for treating cardiac indications
|
|
|
10-31-2008
|
Formulations with Controlled Release of Active Ingredient
|
|
|
8-15-2008
|
HIGHLY SELECTIVE and LONG-ACTING PDE5 MODULATORS
|
|
8-8-2008
|
Formulations With Controlled Release Of Active Ingredient
|
|
|
4-11-2008
|
Use of 2-alkoxyphenyl-substituted imidazotriazinones
|
|
|
1-2-2008
|
2-phenyl substituted imidazotriazinones as phosphodiesterase inhibitors, for treatment of hypertension
|
|
|
12-28-2007
|
Novel Uses of 2-Phenyl-Substituted Imidazotriazinone Derivatives
|
|
|
10-3-2007
|
Use of 2-alkoxyphenyl-substituted imidazotriazinones
|
|
|
11-24-2006
|
Methods for synthesizing imidazotriazinones
|
|
|
10-18-2006
|
2-Phenyl substituted imidazotriazinones as phosphodiesterase inhibitors
|
|
|
2-15-2006
|
Process for the preparation of sulphonamide-substituted imidazotriazinones
|
|
|
8-17-2005
|
Use of 2-alkoxyphenol-substituted imidazotriazinones
|
|
|
5-11-2005
|
2-phenyl substituted imidazotriazinones as phosphodiesterase inhibitors
|
|
1-21-2005
|
Process for the preparation of sulphonamide-substituted imidazotriazinones
|
|
|
8-18-2004
|
Process for the preparation of sulphonamide-substituted imidazotriazinones
|
|
|
8-6-2004
|
Novel use of 2-phenyl-substituted imidazotriazinones
|
|
|
7-32-2003
|
Daily treatment for erectile dysfunction using a PDE5 inhibitor
|
|
|
5-21-2003
|
2-phenyl substituted imidatriazinones as phosphodiesterase inhibitors
|
|
|
3-27-2002
|
2-phenyl substituted imidazotriazinones as phosphodiesterase inhibitors
|
|
|
12-21-2001
|
Daily treatment for erectile dysfunction using a PDE5 inhibitor
|
|
|
5-21-1999
|
2-PHENYL SUBSTITUTED IMIDAZOTRIAZINONES AS PHOSPHODIESTERASE INHIBITORS
|