Home » 2013 (Page 30)
Yearly Archives: 2013
Vortioxetine ボルチオキセチン 臭化水素酸塩 – FDA Approves Brintellix to Treat Major Depressive Disorder
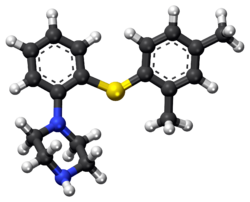
Vortioxetine
ボルチオキセチン 臭化水素酸塩
1-[2-(2,4-dimethylphenyl)sulfanylphenyl]piperazine
Lu AA21004
| VORTIOXETINE; CAS 508233-74-7;
1-(2-((2,4-Dimethylphenyl)thio)phenyl)piperazine; Lu AA21004; UNII-3O2K1S3WQV; C18H22N2S; |
|
| Molecular Formula: | C18H22N2S |
|---|---|
| Molecular Weight: | 298.44568 g/mol |
Vortioxetine Hydrobromide

C18H22N2S.HBr : 379.36
[960203-27-4] HYDROBROMIDE
Vortioxetine is an atypical antipsychotic and antidepressant indicated for the treatment of major depressive disorder (MDD). It is classified as a serotonin modulator and simulator (SMS) as it has a multimodal mechanism of action towards the serotonin neurotransmitter system whereby it simultaneously modulates one or more serotonin receptors and inhibits the reuptake of serotonin. More specifically, vortioxetine acts via the following biological mechanisms: as a serotonin reuptake inhibitor (SRI) through inhibition of the serotonintransporter, as a partial agonist of the 5-HT1B receptor, an agonist of 5-HT1A, and an antagonist of the 5-HT3, 5-HT1D, and 5-HT7 receptors. SMSs were developed because there are many different subtypes of serotonin receptors, however, not all of these receptors appear to be involved in the antidepressant effects of SRIs. Some serotonin receptors seem to play a relatively neutral or insignificant role in the regulation of mood, but others, such as 5-HT1A autoreceptors and 5-HT7 receptors, appear to play an oppositional role in the efficacy of SRIs in treating depression.
Sept. 30, 2013 — The U.S. Food and Drug Administration today approved Brintellix (vortioxetine) to treat adults with major depressive disorder.
Major depressive disorder (MDD),
 Commonly referred to as depression, is a mental disorder characterized by mood changes and other symptoms that interfere with a person’s ability to work, sleep, study, eat and enjoy once-pleasurable activities. Episodes of depression often recur throughout a person’s lifetime, although some may experience a single occurrence.
Commonly referred to as depression, is a mental disorder characterized by mood changes and other symptoms that interfere with a person’s ability to work, sleep, study, eat and enjoy once-pleasurable activities. Episodes of depression often recur throughout a person’s lifetime, although some may experience a single occurrence.
READ ALL AT
http://www.drugs.com/newdrugs/fda-approves-brintellix-major-depressive-disorder-3918.html
The disease: Major depression
The developers: Lundbeck, Takeda
Vortioxetine (vor-tye-OX-e-teen, code name Lu AA21004) is an experimental drug currently under development by Lundbeck and Takeda for the treatment of major depressive disorder (MDD) and generalized anxiety disorder (GAD).Commercial names chosen are Brintellix and Rexulti.
Regulatory approval for the treatment of MDD for the European market has been filed in September 2012, for the United States in October 2012, and filing for Canada should follow. Filing for the Japanese market is expected in 2013.
Depression
In May 22 2011, Lundbeck presented the results of four phase III trials on vortioxetine at the 2011 Annual Meeting of the American Psychiatric Association. A statistically significant effect was shown in two of the studies (one for active treatment using the Hamilton Depression Rating Scale (HAM-D), the second as a maintenance treatment), vortioxetine failed to prove superiority over placebo in a third (again using the HAM-D) and the fourth was nullified by an exceptionally high placebo response (according to the Montgomery-Åsberg Depression Rating Scale (MADRS)).
In July 2011, Lundbeck published the results of a double-blind, randomized, placebo-controlled clinical trial with venlafaxine as an active reference. It was found to be superior to placebo in treating MDD while having fewer side effects than venlafaxine. Similarly, in May 2012, Lundbeck published the results of a double-blind, randomized, placebo-controlled clinical trial with duloxetine evaluating vortioxetine in elderly depressed patients, and it was found superior to placebo, with fewer side effects than duloxetine.
In May 2012, Lundbeck disclosed the results of three phase III clinical trials, showing vortioxetine’s superiority over placebo according to the MADRS.
In August 2012, a randomized, double-blind trial confirms the superiority of vortioxetine over placebo according to all measures, excepted the Sheehan Disability scale.
In September 2012, a randomised, double-blind trial reveals that a dose of 5mg shows superiority over placebo only in patients that suffer from comorbid anxiety.This is consistent with results from another trial published in December 2012, demonstrating that 2.5 mg and 5 mg doses are ineffective.
Anxiety
August 2012, contradictory results of two randomized, double-blind trial were published. While the first demonstrated vortioxetine’s superiority over the placebo, the second showed that the drug had no efficacy, leading the authors to question the designs of the different trials.

United States Patent Number: 7,144,884 , 8,476,279
related to Chinese patent: CN1319958 C , CN1561336 A; CN1319958C, CN1561336A
patent validity: January 9, 2023 (U.S. Patent Number: 7,144,884), October 2, 2022 (U.S. Patent No.: 8,476,279)
peak annual sales (estimated): $ 2 billion
drug companies: Lundbeck (Lundbeck), Takeda (Takeda)
Wal antidepressant drug Paxil (Brintellix, Vortioxetine) for – 1 – Preparation – [2 – (2,4 methyl) phenyl] piperazine process
Method II:
815g of the NaOBut (8,48 mo1), 844 g of piperazine Qin (9,8 mol), 6,6 g of Pd (dba) 2 (11,48 mmol) and 13,6 g of rac-BINAP (21, 84 mmol) was stirred for 50 minutes with 4L ofbenzene. Then, 840 g 2 – bromo – iodobenzene (2,97 mol) and 1.5L of Yue added to the mixture with benzene, and the stirring was continued for 30 minutes. Finally, 390.8g of 2,4 -thiophenol (2,83 mol) was added together with 1.5L toluene. The resulting suspension was heated to reflux and reflux was continued for 5 hours. The reaction mixture was cooled overnight. 2L of water was added and stirred for l hour and then filtered through a filter aid, the resulting mixture. Then, the filtrate was washed with brine 3xlL. Subsequently, the combined aqueous phase extracted with 600ml of benzene. Then, Yue The combined benzene phase was heated to 70 ° C, then adding 329.2ml 48-wt. / HBr (aq.) and 164.6ml water o’s. The mixture was cooled to room temperature overnight. Final product was collected by filtration (l-[2 – (2,4 – di曱group – phenylsulfanyl) – phenyl] – piperazine hydrobromide Qin), and dried under vacuum (60 0 C), to give 895g of product (84% yield).
Method III:
The benzene is placed 500ml three-necked 1L round bottom flask equipped with a mechanical stirrer and add 809mg Pd2dba3 (0.88mmol; 0.5 mol%) and 952 mg DPEPhos (1.77 mmol; 0.5mol-%). The deep red solution was purged with nitrogen for 5 minutes, then add 100g2-bromo-iodobenzene (353 mmol) and 48.9 g 2,4 – bis thiophenol (353 mmol). Add 43.6g KOBut (389 mmol) caused an exothermic reaction, so that the temperature rise of 20 ° C 42 ° C, while forming a non-uniform mixture, and the color changed from deep red to orange / brown. The force of the suspension under nitrogen was heated to port 100 ° C. After only 20 minutes, HPLC showed complete conversion to have l-(2 – bromo – phenylsulfanyl) -2,4 – Yue group – benzene. The mixture was cooled to 40 ° C, was added to 600ml 15-wt% NaCl, and stirred for 5 minutes. The organic phase was separated, and the aqueous phase was washed 2xl00mwith benzene. The combined organic phase was washed with HCl (aq) NaCl and washed with 100ml 2M 100ml 15-wt%, and then Na 2 S04 dried by activated charcoal (10 g) at reflux for 15 minutes, filtered twice and evaporated to 107.3 g of orange-red oil (103%), the oil was found by HPLC purity of 98%.
To 90 g of the orange-red oil (307 mmol) in 500ml of anhydrous toluene was added 57 g boc-piperazine Qin (307 mmol), degassed with nitrogen for 5 minutes, was added 1.4g Pd2dba3 (1.53 mmol- %; 0.5 mol%) and 2.9g mc-BINAP (4.6 mmol; 1.5 mol-%), degassed and then another 2 minutes, then add 35.4 g of NaOtBu (368 mmol), and heated to 80 ° C for 18 hours. HPLC showed complete conversion to have the reaction mixture was cooled to RT, filtered, and the filter cake was washed with 2 x 100ml of曱benzene. % NaCl, washed twice in Na2S04 dried, added charcoal, refluxed for 30 minutes, filtered twice and evaporated to 140.7 g of a brown oil (4 – – The combined filtrates with 2 x 150ml 15 [2 – (2, 4 – di曱group – phenylsulfanyl) -. phenyl]-BOC-piperazine Qin). The resulting crude oil was dissolved in 300ml MeOH and 200ml 6MHCl (aq.) and refluxed for l hour, after which HPLC showed complete deprotection. After cooling to RT, the vacuum on a rotary evaporator to remove曱alcohol was added 20ml of concentrated NaOH (pH was measured to 13-14), after which the mixture with 1000ml EtOAc – 15 minutes from stirring. The organic phase was collected and dried 300ml 15wtQ /. Saline extraction in Na2S04 dried, and added 46.3 g of fumaric acid in 300mlMeOH (399 mmol) was added. The mixture was heated to reflux, cooled to room temperature and then placed in the tank (-18. C) overnight. The precipitate was collected, washed with 100ml and 100ml of acetone with EtOAc, and dried in vacuo (50 ° C), to give 103.2g of l-[2 – (2,4 – di group – phenylsulfanyl) – phenyl] – piperazine. Qin fumarate (249mmo1), as a white powder, overall yield 81%, determined by LC-MS and the purity was 99% fumarate. Use EtOAc/H20 / concentrated NaOH to the fumarate salt into the free base (l-[2 – (2,4 – dimethyl – phenylsulfanyl) – phenyl] – piperazine Qin), The organic phase was washed with brine, dried over Na 2 S04 sulfate, filtered and to the filtrate was added 34ml48-wto / o of HBr (aq.), to form a white solid precipitated. The solid was collected, and the solid was washed with 1000ml H20 boiling process, the resultant was cooled to room temperature and purified by forming a slurry. The final product was collected by filtration (l-[2 – (2,4 – digroup – phenylsulfanyl) – phenyl] – piperazine hydrobromide Qin Kr), and dried in vacuo (50 ° C), to produce 83g of white powder (total yield 71%).
Source:
1) Bang-Andersen B, Ruhland T, Jørgensen M, Smith G, Frederiksen K, Jensen KG, Zhong H, Nielsen SM, Hogg S, Mørk A, Stensbøl TB “Discovery of 1 -. [2 – (2,4 – dimethylphenylsulfanyl) phenyl] PIPERAZINE (Lu AA21004): a novel multimodal Major Compound for the treatment of depressive disorder. ” Journal of Medicinal Chemistry 54 (9): 3206-21.
2) Thomas Ruhland, Garrick Paul Smith, Benny Bang-Andersen, Ask Puschl, Ejner Knud Moltzen, Kim Andersen,; Phenyl-piperazine derivatives as serotonin reuptake inhibitors; US patent number 7144884 ; also published as CA2462110A1, CA2462110C , CN1319958C, CN1561336A, DE60225162D1, DE60225162T2, DE60233608D1, EP1436271A1, EP1436271B1, EP1749818A2, EP1749818A3, EP1749818B1, US7138407, US7148238, US7683053, US8110567, US8476279, US20050014740, US20060084662, US20060089368, US20070060574, US20110009423, US20120302553, WO2003029232A1; H. Lundbeck A / S;
T · Rouland, G · P · Smith, B · Bang – Anderson, A · Pi Shier, E · K · Moore Cen, K · Anderson; as serotonin reuptake inhibitors phenyl piperazine derivatives matter; CN 1319958 C
T · Rouland, G · P · Smith, B · Bang – Anderson, A · Pi Shier, E · K · Moore Cen, K · Anderson; as serotonin reuptake inhibitors phenyl piperazine derivatives; CN 1561336 A
3) Benny Bang-Andersen; Phenyl-piperazine derivatives as serotonin reuptake inhibitors; US patent number 8476279 B2 ; Also published as CA2462110A1, CA2462110C, CN1319958C, CN1561336A, DE60225162D1, DE60225162T2, DE60233608D1, EP1436271A1, EP1436271B1, EP1749818A2, EP1749818A3, EP1749818B1, US7138407, US7144884, US7148238, US7683053, US8110567, US20050014740, US20060084662, US20060089368, US20070060574, US20110009423, US20120302553, WO2003029232A1; H. Lundbeck A / S;
4) Kim Lasse Christensen; Process for the manufacture of 1 – [2 – (2,4-dimethyl-phenylsulfanyl)-phenyl]-piperazine; PCT application, WO2013102573 A1
5) Benny Bang-Andersen, Joergen Brodersen, Andre Faldt, Rene Holm, Morten Joergensen, De Diego Heidi Lopez, Michael J Mealy, Arne Moerk, Nicholas Moore, Lone Munch Ringgaard, Michael Harold Rock, Tine Bryan Stensboel; 1 – [2 – (2, 4-dimethylphenylsulfanyl)-phenyl] piperazine as a compound with combined serotonin reuptake, 5-ht3 and 5-ht1a activity for the treatment of cognitive impairment; WO2007144005 A1
Updated oct 2015…………….

Vortioxetine (vor-tye-oks-e-teen, trade name Trintellix) is an atypical antidepressant (a serotonin modulator and stimulator) made by Lundbeck and Takeda.[1]
Vortioxetine [1-[2-(2,4-Dimethylphenyl-sulfanyl)-phenyl]-piperazine] is an orally administered small molecule developed as once-daily treatment of major depressive disorder (MDD) and generalized anxiety disorder (GAD). As a drug, Vortioxetine is a bis-aryl-sulphanyl amine compound that combines serotonin (5-HT) reuptake inhibition with other characteristics, including receptor activity modulation.
Animal and in vitro studies indicate that several neurotransmitter systems may be impacted by vortioxetine, with the drug enhancing levels of 5-HT, noradrenaline, dopamine, acetylcholine and histamine in certain areas of the brain, as well as modulating γ-aminobutyric acid and glutamate neurotransmission. Results from additional animal models suggest vortioxetine may also improve measures of cognitive function, such as memory. In healthy volunteers, single or repeated administration of vortioxetine (10 mg) did not impair cognitive function, psychomotor performance or driving ability in a placebo-controlled study.
Medical use
Vortioxetine is used as first-line treatment for major depressive disorder.[1][2][3][4][5]
Pharmacokinetics
Vortioxetine reaches peak plasma concentration (Cmax) within 7 to 11 hours post-administration (Tmax), and its mean terminal half-life (t½) is ≈ 66 hours. Steady-state plasma concentrations are typically reached within two weeks.[1] It has no active metabolites (i.e. it is not a prodrug).[1]
Research
Vortioxetine has been studied in several clinical trials as a potential treatment for general anxiety disorder but results were inconsistent.[9][10]
History
Vortioxetine was discovered by scientists at Lundbeck who reported the rationale and synthesis for the drug (then called Lu AA21004) in a 2011 paper.[7][11]
In 2007, the compound was in Phase II clinical trials, and Lundbeck and Takeda entered into a partnership in which Takeda paid Lundbeck $40 million upfront, with promises of up to $345 million in milestone payments, and Takeda agreed to pay most of the remaining cost of developing the drug. The companies agreed to co-promote the drug in the US and Japan, and that Lundbeck would receive a royalty on all such sales. The deal included another drug candidate, tedatioxetine (Lu AA24530), and could be expanded to include two other Lundbeck compounds.[12]
Vortioxetine was approved by the U.S. FDA for the treatment of major depressive disorder (MDD) in adults in September, 2013,[13] and it was approved in Europe later that year.[14]
Vortioxetine was previously trademarked as Brintellix in the United States, but on May 2, 2016, the US FDA approved a name change to Trintellix in order to avoid confusion with the blood-thinning medication ticagrelor (Brilinta).[15]
WO2015155153, SYNTHESIS OF VORTIOXETINE VIA (2,4-DIMETHYLPHENYL)(2-IODOPHENYL)SULFANE INTERMEDIATE
| LEK PHARMACEUTICALS D.D. [SI/SI]; Verovskova 57 1526 Ljubljana (SI) | |
| Inventors:ZUPANCIC, Borut; (SI) |
Vortioxetine is disclosed as Example 1 e in WO 2003/029232 A1 and is described as being prepared analogously to Example 1 . The process used to prepare Example 1 involves the preparation of 1 -(2-((2-(trifluoromethyl)phenyl)thio)phenyl)piperazine on a solid polystyrene support, followed by decomplexation using visible light irradiation, and purification by preparative LC-MS and ion-exchange chromatography. The overall yield for the preparation of vortioxetine is described as 17%.
Several alternative palladium catalyzed processes for the preparation of vortioxetine are described in Examples 17 to 25 of WO 2007/144005 A1 . These processes describe the preparation of vortioxetine from 2,4-dimethylthiophenol and 2-bromoiodobenzene (or 1 ,2-dibromobenzene) starting materials via a 1 -(2-bromo-phenylsulfanyl)-2,4-dimethyl-benzene intermediate. Each of these processes involves the use of a palladium catalyst and a phosphine ligand.
The preparation of vortioxetine is also described by Bang-Andersen et al. in J. Med. Chem. (201 1 ), Vol. 54, 3206-3221 . Here, in a first step, te/t-butyl 4-(2-bromophenyl)piperazine-1 -carboxylate intermediate is prepared from Boc-piperazine and 2-bromoiodobenzene in a palladium catalyzed coupling reaction. te/t-Butyl 4-(2-bromophenyl)piperazine-1 -carboxylate is then reacted with 2,4-dimethylthiophenol, again in the presence of palladium catalyst and a phosphine ligand, to provide Boc-protected vortioxetine. In the final step, vortioxetine is deprotected using hydrochloric acid to give vortioxetine hydrochloride.
WO 2013/102573 A1 describes a reaction between 1 -halogen-2,4-dimethyl-phenyl, 2-halogen-thiophenol and an optionally protected piperazine in the presence of a base and a palladium catalyst consisting of a palladium source and a phosphine ligand.
Each of the above processes has disadvantages. The process described in WO 2003/029232 is low yielding and unsuitable for the large scale production of vortioxetine, whereas the processes described in WO 2007/144005 A1 , WO 2013/102573 A1 and by Bang-Andersen et al. require the use of expensive starting materials, palladium catalyst and phosphine ligand. In addition, the toxicity of palladium is well known, Liu et al. Toxicity of Palladium, Toxicology Letters, 4 (1979) 469-473, and the European Medicines Agency’ s Guideline on the Specification for Residues of Metal Catalysts sets clear limits on the permitted daily exposure to palladium arising from palladium residue within drug substances, http://www.ema.europa.eu. Thus it would be desirable to avoid the use of a palladium catalyst in the synthesis of vortioxetine and the subsequent purification steps required to remove palladium residue from the final pharmaceutical product.
The invention is described below in further detail by embodiments, without being limited thereto.
A general concept of the process of the present invention may be represented in Scheme 1 .

Scheme 1 : General representation of the basic synthetic concept of the present invention.
Scheme 2.

X = NH2: lb
Scheme 2: Representation of a particular synthetic embodiment of the present invention.
Compound III can also be prepared from 2,4-dimethylbenzenethiol (II) and 1 -fluoro-2-nitrobenzene (l”‘a) or 1 -chloro-2-nitrobenzene (l'”b). In the first step (2,4-dimethylphenyl)(2- nitrophenyl)sulfane (III’) is formed and in the second reaction step nitro group is reduced to ami

Z = F: l”‘a
Z = CI: l”‘b
Scheme 3: Representation of a particular synthetic embodiment of the present invention.
Example 7: Preparation of 1 -(2-((2,4-dimethylphenyl)thio)phenyl)piperazine vortioxetine, VII)

Mixture of (2,4-dimethylphenyl)(2-iodophenyl)sulfane V (0.34 g, 1 .0 mmol), piperazine VI (0.13 g, 1 .5 mmol), K3P03 (0.42 g, 2.0 mmol), Cul (19 mg, 0.1 mmol), and 2-phenylphenol (68 mg, 0.4 mmol) in dry and degassed DMSO (2 mL) was heated under nitrogen atmosphere at 120°C for 20 h. Water (10 mL) is then added and product is extracted to EtOAc (3 x 10 mL). Combined organic layers were washed with water (3 x 10 mL) and brine (2 x 10 mL) and dried over Na2S04. After evaporation of the solvent crude product is purified by chromatography to afford title compound: H NMR (CDCI3, 500 MHz) δ 1 .63 (br s, 1 H), 2.33 (s, 3H), 2.37 (s, 3H), 3.02-
3.09 (m, 8H), 6.52 (m, 1 H), 6.87 (m, 1 H), 7.04 (m, 1 H), 7.06-7.10 (m, 2H), 7.16 (m, 1 H), 7.39 (d, J= 7.8 Hz, 1 H); MS (ESI) m/z: 299 [MH]+.
Example 8: Preparation of 1 -(2-((2,4-dimethylphenyl)thio)phenyl)piperazine (vortioxetine, VII)

Mixture of (2,4-dimethylphenyl)(2-iodophenyl)sulfane V (0.34 g, 1 .0 mmol), piperazine VI (0.13 g, 1 .5 mmol), K3P03 (0.42 g, 2.0 mmol), Cul (19 mg, 0.1 mmol), and N,N-diethyl-2-hydroxybenzamide (39 mg, 0.2 mmol) in dry and degassed DMSO (2 mL) was heated under nitrogen atmosphere at 120 ^ for 20 h. Water (10 mL) is then added and product is extracted to EtOAc (3 x 10 mL). Combined organic layers were washed with water (3 x 10 mL) and brine (2 x 10 mL) and dried over Na2S04. After evaporation of the solvent crude product is purified by chromatography to afford title compound: H NMR (CDCI3, 500 MHz) δ 1 .63 (br s, 1 H), 2.33 (s, 3H), 2.37 (s, 3H), 3.02-3.09 (m, 8H), 6.52 (m, 1 H), 6.87 (m, 1 H), 7.04 (m, 1 H), 7.06-7.10 (m, 2H), 7.16 (m, 1 H), 7.39 (d, J= 7.8 Hz, 1 H); MS (ESI) m/z: 299 [MH]+.
Example 9: Preparation of 1 -(2-((2,4-dimethylphenyl)thio)phenyl)piperazine hydrobromide
(vortioxetine HBr, VII.HBr)

To a solution of vortioxetine VII (1 .80 g, 6.03 mmol) in iPrOAc (20 mL) at room temperature 48% HBr (0.68 mL, 6.03 mmol) was slowly added. Obtained mixture was stirred at room temperature for 1 h, white precipitate was then filtered off, washed with acetone (2 x 20 mL), and dried to afford title compound VII.HBr as a white powder (2.15 g, 94% yield): H NMR (DMSO-d6, 500 MHz) δ 2.23 (s, 3H), 2.32 (s, 3H), 3.15-3.27 (m, 8H), 6.40 (m, 1 H), 6.96 (m, 1 H), 7.08-7.17 (m, 3H), 7.24 (m, 1 H), 7.32 (d, J= 7.8 Hz, 1 H), 8.85 (br, 2H).
Reference Example 1 : Preparation of 1 -(2-((2,4-dimethylphenyl)thio)phenyl)piperazine
(vortioxetine, VII)

Mixture of piperazine (1 .0 g, 1 1 .6 mmol), NaOtBu (1 .37 g, 13.8 mmol), Pddba2 (40 mg, 0.07 mmol), and 1 ,3-bis(2,6-di-i-propylphenyl)imidazolium chloride (24 mg, 0,07 mmol) in dry and degassed toluene (10 mL) is stirred at room temperature for 1 h. (2,4-Dimethylphenyl)(2-iodophenyl)sulfane V (1 .32 g, 3.86 mmol) is then added, reaction mixture is heated to l OO’C and stirred for 24 h. After cooling to room temperature to the reaction mixture water (5 mL) and Celite (0.4 g) is added. After stirring for 20 min salts are filtered off, organic layer is separated, washed with brine (2 x 10 mL), dried over Na2S04 and solvent is evaporated to afford crude product, which is then purified by chromatography to afford title compound as yellowish crystals: H NMR (CDCI3, 500 MHz) δ 1 .63 (br s, 1 H), 2.33 (s, 3H), 2.37 (s, 3H),
Reference Example 2: Preparation of 1 -(2-((2,4-dimethylphenyl)thio)phenyl)piperazine
(vortioxetine, VII)

Mixture of piperazine (1 .29 g, 15.0 mmol), NaOtBu (1 .77 g, 17.8 mmol), Pddba2 (52 mg, 0.09 mmol), and rac-BINAP (93 mg, 0,15 mmol) in dry and degassed toluene (10 mL) was stirred at room temperature for 1 h. (2,4-Dimethylphenyl)(2-iodophenyl)sulfane V (1 .70 g, 5.0 mmol) was then added, reaction mixture was heated to 100°C and stirred for 24 h. After cooled to room temperature to the reaction mixture water (5 mL) and Celite (0.4 g) were added. After stirring for 20 min salts were filtered off, organic layer was separated, washed with brine (2 x 10 mL), dried over Na2S04 and solvent was evaporated to afford product as an orange oil (1 .41 g, 95% yield): H NMR (CDCI3, 500 MHz) δ 1 .63 (br s, 1 H), 2.33 (s, 3H), 2.37 (s, 3H), 3.02-3.09 (m, 8H), 6.52 (m, 1 H), 6.87 (m, 1 H), 7.04 (m, 1 H), 7.06-7.10 (m, 2H), 7.16 (m, 1 H), 7.39 (d, J = 7.8 Hz, 1 H); MS (ESI) m/z: 299 [MH]+.
Comparative Example 1 : Preparation of 1 -(2-((2,4-dimethylphenyl)thio)phenyl)piperazine
(vortioxetine, VII)

Mixture of (2,4-dimethylphenyl)(2-bromohenyl)sulfane V” (0.29 g, 1 .0 mmol), piperazine VI (0.13 g, 1 .5 mmol), K3P03 (0.42 g, 2.0 mmol), Cul (19 mg, 0.1 mmol), and 2-phenylphenol (68 mg, 0.4 mmol) in dry and degassed DMSO (2 mL) was heated under nitrogen atmosphere at 120°C for 20 h. Vortioxetine VII was not formed.
Comparative Example 2: Preparation of 1 -(2-((2,4-dimethylphenyl)thio)phenyl)piperazine
(vortioxetine, VII)

Mixture of (2,4-dimethylphenyl)(2-bromophenyl)sulfane V (0.29 g, 1 .0 mmol), piperazine VI (0.13 g, 1 .5 mmol), K3P03 (0.42 g, 2.0 mmol), Cul (19 mg, 0.1 mmol), and N,N-diethyl-2-hydroxybenzamide (39 mg, 0.2 mmol) in dry and degassed DMSO (2 mL) was heated under nitrogen atmosphere at 120 ^ for 20 h. Vortioxetine VII was not formed.
WO2007144005A1: Industrial process
Discovery of 1-[2-(2,4-Dimethylphenylsulfanyl)phenyl]piperazine (Lu AA21004): A Novel Multimodal Compound for the Treatment of Major Depressive Disorder

The synthesis and structure−activity relationship of a novel series of compounds with combined effects on 5-HT3A and 5-HT1A receptors and on the serotonin (5-HT) transporter (SERT) are described. Compound 5m (Lu AA21004) was the lead compound, displaying high affinity for recombinant human 5-HT1A (Ki = 15 nM), 5-HT1B (Ki = 33 nM), 5-HT3A (Ki = 3.7 nM), 5-HT7 (Ki = 19 nM), and noradrenergic β1 (Ki = 46 nM) receptors, and SERT (Ki = 1.6 nM). Compound 5mdisplayed antagonistic properties at 5-HT3A and 5-HT7 receptors, partial agonist properties at 5-HT1B receptors, agonistic properties at 5-HT1A receptors, and potent inhibition of SERT. In conscious rats, 5m significantly increased extracellular 5-HT levels in the brain after acute and 3 days of treatment. Following the 3-day treatment (5 or 10 (mg/kg)/day) SERT occupancies were only 43% and 57%, respectively. These characteristics indicate that 5m is a novel multimodal serotonergic compound, and 5m is currently in clinical development for major depressive disorder.
1-[2-(2,4-Dimethylphenylsulfanyl)phenyl]piperazine Hydrochloride (5m)
ALERT HYDROCHLORIDE DATA
References
- US Label Last updated July 2014 after review in September, 2014. Versions of label are available at FDA index page Page accessed January 19, 2016
- [No authors listed] Vortioxetine. Aust Prescr. 2015 Jun;38(3):101-2. PMID 26648632Free full text
- “Relative efficacy and tolerability of vortioxetine versus selected antidepressants by indirect comparisons of similar clinical studies.”. Curr Med Res Opin 30: 2589–606. Oct 10, 2014. doi:10.1185/03007995.2014.969566. PMID 25249164.
- Köhler S, Cierpinsky K, Kronenberg G, Adli M. The serotonergic system in the neurobiology of depression: Relevance for novel antidepressants. J Psychopharmacol. 2016 Jan;30(1):13-22. PMID 26464458
- Kelliny M, Croarkin PE, Moore KM, Bobo WV. Profile of vortioxetine in the treatment of major depressive disorder: an overview of the primary and secondary literature. Ther Clin Risk Manag. 2015 Aug 12;11:1193-212. PMID 26316764 Free full text
- “Lundbeck’s “Serotonin Modulator and Stimulator” Lu AA21004: How Novel? How Good? – GLG News”.
- ^ Jump up to:a b c Bang-Andersen B, Ruhland T, Jørgensen M, et al. (May 2011). “Discovery of 1-[2-(2,4-dimethylphenylsulfanyl)phenyl]piperazine (Lu AA21004): a novel multimodal compound for the treatment of major depressive disorder”. Journal of Medicinal Chemistry 54 (9): 3206–21. doi:10.1021/jm101459g. PMID 21486038.
- N. Moore; B. Bang-Andersen; L. Brennum; K. Fredriksen; S. Hogg; A. Mork; T. Stensbol; H. Zhong; C. Sanchez; D. Smith (August 2008). “Lu AA21004: a novel potential treatment for mood disorders”. European Neuropsychopharmacology 18 (Supplement 4): S321.doi:10.1016/S0924-977X(08)70440-1.
- Pae CU et al. Vortioxetine, a multimodal antidepressant for generalized anxiety disorder: a systematic review and meta-analysis. J Psychiatr Res. 2015 May;64:88-98. PMID 25851751
- Reinhold JA, Rickels K. Pharmacological treatment for generalized anxiety disorder in adults: an update. Expert Opin Pharmacother. 2015;16(11):1669-81. PMID 26159446
- Sanchez C, Asin KE, Artigas F Vortioxetine, a novel antidepressant with multimodal activity: review of preclinical and clinical data. Pharmacol Ther. 2015 Jan;145:43-57. PMID 25016186 Free full text
- Daniel Beaulieu for First Word Pharma. September 5th, 2007 Lundbeck, Takeda enter strategic alliance for mood disorder, anxiety drugs
- FDA approves new drug to treat major depressive disorder, U.S. Food and Drug Administration Press Announcement.
- EMA Brintellix page at EMA site Page accessed January 19, 2016
- Commissioner, Office of the. “Safety Alerts for Human Medical Products – Brintellix (vortioxetine): Drug Safety Communication – Brand Name Change to Trintellix, to Avoid Confusion With Antiplatelet Drug Brilinta (ticagrelor)”. http://www.fda.gov. Retrieved2016-05-02.
| Patent ID | Date | Patent Title |
|---|---|---|
| US7683053 | 2010-03-23 | PHENYL-PIPERAZINE DERIVATIVES AS SEROTONIN REUPTAKE INHIBITORS |
| US7148238 | 2006-12-12 | Phenyl-piperazine derivatives as serotonin reuptake inhibitors |
| US7144884 | 2006-12-05 | Phenyl-piperazine derivatives as serotonin reuptake inhibitors |
| US7138407 | 2006-11-21 | Phenyl-piperazine derivatives as serotonin reuptake inhibitors |
| Patent ID | Date | Patent Title |
|---|---|---|
| US2013184291 | 2013-07-18 | THERAPEUTIC USES OF 1-[2-(2,4-DIMETHYL-PHENYLSUFLANYL)PHENYL]PIPERAZINE |
| US8476279 | 2013-07-02 | Phenyl-piperazine derivatives as serotonin reuptake inhibitors |
| US2013115292 | 2013-05-09 | ENTERIC TABLET |
| US2012189697 | 2012-07-26 | COMPOSITIONS OF 1-[2-(2,4-DIMETHYL-PHENYLSULFANYL)-PHENYL]PIPERAZINE |
| US2012035188 | 2012-02-09 | LIQUID FORMULATIONS OF SALTS OF 1-[2-(2,4-DIMETHYLPHENYLSULFANYL)PHENYL]-PIPERAZINE |
| US8110567 | 2012-02-07 | PHENYL-PIPERAZINE DERIVATIVES AS SEROTONIN REUPTAKE INHIBITORS |
| US2012004409 | 2012-01-05 | Purification of 1-[2-(2,4-dimethylphenylsulfanyl)phenyl]piperazine |
| US2011201617 | 2011-08-18 | Therapeutic Uses Of Compounds Having Combined SERT, 5-HT3 And 5-HT1A Activity |
| US2011009422 | 2011-01-13 | 1- [2-(2,4-DIMETHYLPHENYLSULFANYL)-PHENYL] PIPERAZINE AS A COMPOUND WITH COMBINED SEROTONIN REUPTAKE, 5-HT3 AND 5-HT1A ACTIVITY FOR THE TREATMENT OF PAIN OR RESIDUAL SYMPTOMS IN DEPRESSION RELATING TO SLEEP AND COGNITION |
| US2010297240 | 2010-11-25 | 1- [2- (2,4-DIMETHYLPHENYLSULFANYL)-PHENYL] PIPERAZINE AS A COMPOUND WITH COMBINED SEROTONIN REUPTAKE, 5-HT3 AND 5-HT1A ACTIVITY FOR THE TREATMENT OF COGNITIVE IMPAIRMENT |
| Patent ID | Date | Patent Title |
|---|---|---|
| US2015094316 | 2015-04-02 | LIQUID FORMULATIONS OF SALTS OF 1-[2-(2,4-DIMETHYLPHENYLSULFANYL)PHENYL]PIPERAZINE |
| US8969355 | 2015-03-03 | 1-[2-(2,4 dimethylphenylsulfanyl)-phenyl]piperazine as a compound with combined serotonin reuptake, 5-HT3 and 5-HT1a activity for the treatment of cognitive impairment |
| US2015005318 | 2015-01-01 | 1-[2-(2,4-Dimethylphenylsulfanyl)-Phenyl]Piperazine As A Compound With Combined Serotonin Reuptake, 5-HT3 And 5-HT1a Activity For The Treatment Of Cognitive Impairment |
| US2014371453 | 2014-12-18 | 1-[2-(2,4-Dimethylphenylsulfanyl)-Phenyl]Piperazine As A Compound With Combined Serotonin Reuptake, 5-HT3 And 5-HT1a Activity For The Treatment Of Cognitive Impairment |
| US2014343287 | 2014-11-20 | Process for the Manufacture of 1-[2-(2,4-Dimethyl-Phenylsulfanyl)-Phenyl]-Piperazine |
| US2014315921 | 2014-10-23 | 1-[2-(2,4-Dimethylphenylsulfanyl)-Phenyl]Piperazine As A Compound With Combined Serotonin Reuptake, 5-HT3 And 5-HT1a Activity For The Treatment Of Cognitive Impairment |
| US2014256943 | 2014-09-11 | 1-[2-(2,4-Dimethylphenylsulfanyl)-Phenyl]Piperazine As A Compound With Combined Serotonin Reuptake, 5-HT3 And 5-HT1a Activity For The Treatment Of Cognitive Impairment |
| US2014248355 | 2014-09-04 | 1-[2-(2,4-Dimethylphenylsulfanyl)-Phenyl]Piperazine As A Compound With Combined Serotonin Reuptake, 5-HT3 And 5-HT1a Activity For The Treatment Of Cognitive Impairment |
| US2014248356 | 2014-09-04 | 1-[2-(2,4-Dimethylphenylsulfanyl)-Phenyl]Piperazine As A Compound With Combined Serotonin Reuptake, 5-HT3 And 5-HT1a Activity For The Treatment Of Cognitive Impairment |
| US2014163043 | 2014-06-12 | PHENYL-PIPERAZINE DERIVATIVES AS SEROTONIN REUPTAKE INHIBITORS |
| Patent ID | Date | Patent Title |
|---|---|---|
| US2016083359 | 2016-03-24 | 1-[2-(2,4-DIMETHYLPHENYLSULFANYL)-PHENYL]PIPERAZINE AS A COMPOUND With COMBINED SEROTONIN REUPTAKE, 5-HT3 AND 5-HT1A ACTIVITY FOR THE TREATMENT OF COGNITIVE IMPAIRMENT |
| US2016060215 | 2016-03-03 | New Process For The Synthesis Of 1-(2-((2,4-Dimethylphenyl)Thio)Phenyl)Piperazine |
| US2016015706 | 2016-01-21 | CRYSTALLINE FORMS OF AN ANTIDEPRESSANT COMPOUND |
| US2016009670 | 2016-01-14 | VORTIOXETINE MANUFACTURING PROCESS |
| US9211288 | 2015-12-15 | Compositions comprising vortioxetine and donepezil |
| US2015297585 | 2015-10-22 | COMPOSITIONS COMPRISING VORTIOXETINE AND DONEPEZIL |
| US2015266841 | 2015-09-24 | Novel Crystalline Form Of Vortioxetine Hydrobromide |
| US2015150867 | 2015-06-04 | COMPOSITIONS OF 1-[2-(2,4-DIMETHYL-PHENYLSULFANYL)-PHENYL]PIPERAZINE |
| US2015110873 | 2015-04-23 | ENTERIC TABLET |
| US2015094316 | 2015-04-02 | LIQUID FORMULATIONS OF SALTS OF 1-[2-(2,4-DIMETHYLPHENYLSULFANYL)PHENYL]PIPERAZINE |
 |
|
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
1-[2-(2,4-Dimethyl-phenylsulfanyl)-phenyl]piperazine
|
|
| Clinical data | |
| Trade names | Trintellix, Brintellix |
| License data |
|
| Pregnancy category |
|
| Routes of administration |
Oral |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 75% (peak at 7–11 hours) |
| Protein binding | 98% |
| Metabolism | extensive hepatic, primarilyCYP2D6-mediated oxidation |
| Biological half-life | 66 hours |
| Excretion | 59% in urine, 26% in feces |
| Identifiers | |
| CAS Number | 508233-74-7 |
| ATC code | N06AX26 (WHO) |
| PubChem | CID 9966051 |
| IUPHAR/BPS | 7351 |
| ChemSpider | 8141643 |
| KEGG | D10184 |
| ChEBI | CHEBI:76016 |
| Synonyms | Lu AA21004 |
| Chemical data | |
| Formula | C18H22N2S |
| Molar mass | 298.45 g/mol (379.36 as hydrobromide) |
/////////////
-
CC(C=C(C)C=C1)=C1SC2=C(N3CCNCC3)C=CC=C2
FDA Approves Perjeta for Neoadjuvant Breast Cancer Treatment
pertuzumab
Sept. 30, 2013 — The U.S. Food and Drug Administration today granted accelerated approval to Perjeta (pertuzumab) as part of a complete treatment regimen for patients with early stage breast cancer before surgery (neoadjuvant setting). Perjeta is the first FDA-approved drug for the neoadjuvant treatment of breast cancer.
Perjeta was approved in 2012 for the treatment of patients with advanced or late-stage (metastatic) HER2-positive breast cancer. HER2-positive breast cancers have increased amounts of the HER2 protein that contributes to cancer cell growth and survival
cut paste of my old article
he European Medicines Agency (EMA) has approved Roche’s PERJETA (pertuzumab) for patients with previously untreated HER2-positive metastatic breast cancer (mBC)
MARCH 5, 2013 8:59 AM / 4 COMMENTS /

Pertuzumab (also called 2C4, trade name Perjeta) is a monoclonal antibody. The first of its class in a line of agents called “HER dimerization inhibitors”. By binding to HER2, it inhibits the dimerization of HER2 with other HER receptors, which is hypothesized to result in slowed tumor growth.[1] Pertuzumab received US FDA approval for the treatment of HER2-positive metastatic breast cancer on June 8, 2012.[2] Pertuzumab was developed at Genentech and is now owned by Roche which acquired Genentech in 2009.
Clinical trials
Early clinical trials of pertuzumab in prostate, breast, and ovarian cancers have been met with limited success.[3]
The dosage of pertuzumab used in the pivotal phase III CLEOPATRA (Clinical Evaluation of Pertuzumab and Trastuzumab) trial was as follows: IV 840 mg loading dose followed by IV 420 mg every three weeks.[4]
The pharmacokinetics of intravenous pertuzumab appear to be unaffected by age and no drug-drug interaction has been reported with docetaxel. The pharmacokinetics and pharmacodynamics of pertuzumab were summarized in a Feb 2012 review by Gillian Keating.[4]
The combination of pertuzumab plus trastuzumab plus docetaxel, as compared with placebo plus trastuzumab plus docetaxel, when used as first-line treatment for HER2-positive metastatic breast cancer, significantly prolonged progression-free survival, with no increase in cardiac toxic effects in the randomized, double-blind, multinational, phase III CLEOPATRA trial.[5]
Intravenous pertuzumab is currently being evaluated in patients with breast cancer in the following trials: MARIANNE (advanced breast cancer), NEOSPHERE (early breast cancer), TRYPHAENA (HER2-positive stage II/III breast cancer) and APHINITY (HER2-positive nonmetastatic breast cancer).[4]
References
- de Bono, Johann S.; Bellmunt, J; Attard, G; Droz, JP; Miller, K; Flechon, A; Sternberg, C; Parker, C et al. (20 January 2007). “Open-Label Phase II Study Evaluating the Efficacy and Safety of Two Doses of Pertuzumab in Castrate Chemotherapy-Naive Patients With Hormone-Refractory Prostate Cancer”. Journal of Clinical Oncology 25 (3): 257–262.doi:10.1200/JCO.2006.07.0888. PMID 17235043.
- “FDA Approves Perjeta (Pertuzumab) for People With HER2-Positive Metastatic Breast Cancer” (Press release). Genentech. Retrieved 2012-06-09.
- Genentech press release – May 15, 2005
- Keating GM. Pertuzumab: in the first-line treatment of HER2-positive metastatic breast cancer. Drugs 2012 Feb 12; 72 (3): 353-60.Link text
- Baselga J, Cortés J, Kim SB, and the CLEOPATRA Study Group. Pertuzumab plus trastuzumab plus docetaxel for metastatic breast cancer. N Engl J Med 2012 Jan 12; 366 (2): 109-19. Link text
Certolizumab pegol – FDA gave green light to UCB’s Cimzia to treat psoriatic arthritis

Certolizumab pegol
The US Food and Drug Administration has approved UCB’s Cimzia for the treatment of adults with psoriatic arthritis, the third indication approved by the agency.
The UCB’s biologic drug Cimzia is already on the market for rheumatoid arthritis and Crohn’s disease in both US and Europe. Cimzia, also known as Certolizumab pegol, is a monoclonal antibody directed against tumor necrosis factor alpha. It is a PEGylated Fab’ fragment of a humanized TNF inhibitor monoclonal antibody
read all at http://www.pharmatopics.com/2013/09/fda-gave-green-light-to-ucbs-cimzia-to-treat-psoriatic-arthritis/
Certolizumab pegol (CDP870, tradename Cimzia) is a therapeutic monoclonal antibody to tumor necrosis factor alpha (TNF-α), for the treatment of Crohn’s disease and rheumatoid arthritis, manufactured by UCB.
certolizumab pegol is a monoclonal antibody directed against tumor necrosis factor alpha. More precisely, it is a PEGylated Fab’fragment of a humanized TNF inhibitor monoclonal antibody.
Polyethylene glycol does not cross the placenta, so it should be safe in pregnancy.
Positive results have been demonstrated in two phase III trials (PRECiSE 1 and 2) of certolizumab pegol versus placebo in moderate to severe active Crohn’s disease. In addition, data from both trials suggest it is well tolerated. As yet its efficacy has not been directly compared to other anti-TNF-α agents.
Preliminary results of the RAPID 1 and 2 phase III studies were also reportedly positive.
In 2013, a phase 3 double blind randomized placebo-controlled study found significantly positive result in patient self-reported questionnaires, with rapid improvement of function and pain reduction.
On April 22, 2008, the U.S. Food and Drug Administration (FDA) approved Cimzia for use in the United States for the treatment of Crohn’s disease in people who did not respond sufficiently or adequately to standard therapy.
On June 26, 2009, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMEA) issued a positive opinion recommending that the European Commission grant a marketing authorisation for Cimzia for the treatment of rheumatoid arthritis only – the CHMP refused approval for the treatment of Crohn’s disease. The marketing authorisation was granted to UCB Pharma SA on October 1, 2009.



Daclizumab
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Humanized (from mouse) |
| Target | CD25 |

Daclizumab is a humanized monoclonal antibody indicated in the United States for prophylaxis of acute organ rejection in patients receiving renal transplants.
It was marketed as Zenepax, but discontinued by Roche in 2009 due to diminishing market demand for that indication. Biogen Idec is currently conducting phase III trials for daclizumab in MS. A phase III trial started in March 2010 is being conducted to determine efficacy of preventing MS relapse.
Study dosing of daclizumab is 150 mg subcutaneously once every 4 weeks versus interferon beta-1a (Avonex) 30 mg intramuscularly given once weekly for 96 to 144 weeks.
Daclizumab (Zenapax®) (molecular wt = 144 kd.) is a humanized monoclonal antibody (IgG1) produced by recombinant DNA technology. It gained FDA approval in Dec 1997. It is known by several other names including HAT (Humanized Anti-Tac), SMART anti-Tac, anti-CD25, and humanized anti-IL2-receptor. It was developed and patented by Protein Design Laboratories (Mountain View, CA) and it is marketed by Hoffman LaRoche (Nutley, NJ ).
Daclizumab is a composite of human (90%) and murine (10%) antibody sequences. In the model below, the murine portions are shown in red and dark blue; the rest of the molecule (gray color) represents the human sequence
The study is aiming for enrollment of 1500 patients and is expected to be complete in January 2014.
more info
Daclizumab (trade name Zenapax) is a therapeutic humanized monoclonal antibody. It is used to prevent rejection in organ transplantation, especially in kidney transplants. The drug is also under investigation for the treatment of multiple sclerosis.
Daclizumab works by binding to CD25, the alpha subunit of the IL-2 receptor of T cells. The drug is marketed in the US, but not in Europe.
Uses
Prevention of organ transplants
Daclizumab is given in multiple doses, the first 1 hour before the transplant operation and 5 further doses given at two week intervals after the transplant. These saturate the receptors and prevent T cell activation and thus prevent formation of antibodiesagainst the transplant.
Like the similar drug basiliximab, daclizumab reduces the incidence and severity of acute rejection in kidney transplantation without increasing the incidence of opportunistic infections.
Daclizumab usage may also be indicated in place of a calcineurin-inhibitor (ciclosporin or tacrolimus) during the early phase after kidney transplantation, when the kidney is recovering and vulnerable to calcineurin-inhibitor toxicity. This has been shown to be beneficial in non-heart beating donor kidney transplantation.
In the United Kingdom, the National Institute for Health and Clinical Excellence (NICE) has recommended its use be considered for all kidney transplant recipients.[citation needed]
Multiple sclerosis
In 2006 it began a Phase II clinical trial that finished in 2007 as a possible multiple sclerosis (MS) treatment. Participants were nine patients with multiple sclerosis not controlled with interferon. Daclizumab was effective in reducing lesions and improving clinical scores.[1] As of June 2013, the drug is in Phase III trials for this indication.[2]
Autoimmune diseases
Daclizumab has also been used to slow the progression of autoimmune diseases, particularly that of birdshot chorioretinopathy.[3]
Common side effects with a frequency of at least 10% include sleeplessness, tremor, headache, arterial hypertension, dyspnoea, gastrointestinal side effects and oedema. In rare cases, the drug can cause severe anaphylaxis.[4]
Daclizumab must not be administered to lactating women.[4]
History
Daclizumab was developed by PDL Biopharma, building on research at the National Institutes of Health (NIH).[5] Since December 1997, it is marketed by Hoffmann-La Roche in the US.
In April 2008, Hoffmann-La Roche submitted an application to have its marketing authorisation withdrawn in the EU for commercial reasons. The drug faced diminishing market demand, according to the company. There were no safety concerns with its use. As of January 2009, its marketing authorisation has been withdrawn and the product discontinued completely.[6][7]
- Rose JW, Burns JB, Bjorklund J, Klein J, Watt HE, Carlson NG (2007). “Daclizumab phase II trial in relapsing and remitting multiple sclerosis: MRI and clinical results”.Neurology 69 (8): 785–789. doi:10.1212/01.wnl.0000267662.41734.1f.PMID 17709711.
- ClinicalTrials.gov NCT01462318 An Immunogenicity and Pharmacokinetics (PK) Study of DAC HYP Prefilled Syringe in Relapsing Remitting Multiple Sclerosis (RRMS) (OBSERVE)
- Sobrin L, Huang JJ, Christen W, Kafkala C, Choopong P, Foster CS (2008). “Daclizumab for treatment of birdshot chorioretinopathy”. Arch Ophthalmol. 126 (2): 186–191. doi:10.1001/archophthalmol.2007.49. PMID 18268208.
- “EPAR for Zenapax”. European Medicines Agency. 2007.
- Tsurushita, N.; Hinton, P. R.; Kumar, S. (2005). “Design of humanized antibodies: From anti-Tac to Zenapax”. Methods 36 (1): 69–83.doi:10.1016/j.ymeth.2005.01.007. PMID 15848076. edit
- British National Formulary, Edition 57
- EMEA: Withdrawal of the marketing authorisation in the European Union
Ipilimumab – Yervoy Battles Melanoma, but Can It Become a Blockbuster?

Ipilimumab
by Todd Campbell, The Motley Fool Sep 28th 2013 1:00PM
Updated Sep 28th 2013 1:02PM
In early 2011, the Food and Drug Administration approved Bristol-Myers Squibb‘s drug Yervoy as a treatment for skin cancer melanoma. The drug marked the first approved treatment proven to extend the life of a person diagnosed with the disease. It marked a big leap forward in medicine as an early leader in immunotherapy, or the unleashing of the body’s immune system on cancer.
read all at
http://www.dailyfinance.com/2013/09/28/yervoy-battles-melanoma-but-can-it-become-a-blockb/

Ipilimumab’s molecular target is CTLA-4 (Uniprot: P16410; canSAR ; PFAM: P16410), a negative regulator of T-cell activation. Ipilimumab augments T-cell activation and proliferation by binding to CTLA-4 and preventing its interaction with its ligands (CD80 and CD86). CTLA-4 is a membrane-bound, 223 amino acid long, T-cell protein. It contains an immunoglobulin V-type domain (PFAM:PF07686). The structure of CTLA-4 is determined (see e.g. PDBe:3osk)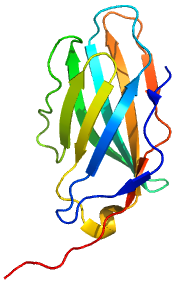
Ipilimumab (i pi lim′ ue mab; also known as MDX-010 and MDX-101), marketed asYervoy, is a drug used for the treatment of melanoma, a type of skin cancer. It is a U.S. Food and Drug Administration (FDA) approved human monoclonal antibody developed byBristol-Myers Squibb, and works by activating the immune system by targeting CTLA-4.
Cytotoxic T lymphocytes (CTLs) can recognize and destroy cancer cells. However, there is also an inhibitory mechanism that interrupts this destruction. Ipilimumab turns off this inhibitory mechanism and allows CTLs to continue to destroy cancer cells.
In addition to melanoma, ipilimumab is undergoing clinical trials for the treatment of non-small cell lung carcinoma (NSCLC), small cell lung cancer (SCLC) and metastatic hormone-refractory prostate cancer.
Yervoy is a monoclonal antibody drug indicated for treating metastatic melanoma. The drug was developed by Bristol-Myers Squibb.
In March 2011, The US Food and Drug Administration (FDA) approved Yervoy to treat patients with newly diagnosed or previously-treated unresectable or metastatic melanoma. Yervoy is the first drug approved vor the treatment of metastatic melanoma in the US.
Bristol-Myers Squibb submitted a marketing authorisation application to the European Medicines Agency in May 2010. The drug received approval from the European Commission in July 2011.
Approval from Australia’s Therapeutic Goods Association was received in July 2011. The drug is currently being reviewed by Health Canada.
Metastatic melanoma
Melanoma responsible for majority of skin cancer deaths in the US. In metastatic melanoma the cancer spreads to other parts of the body from its starting point. It becomes difficult to treat the disease once it spreads beyond the skin to other parts of the body. The disease is also known as stage IV melanoma.
If the melanoma spreads to the lungs then the patient faces breathing problems. The patients with metastatic melanoma may feel symptoms of fatigue, loss of weight, and appetite and bowel problems.
The incidence of the disease has increased steadily in the US after 1970s. The American Cancer Society (ACS) estimated that more than 68,000 new cases of melanoma were registered in the US in 2009. The ACS estimated that the number of deaths occurred due to melanoma in 2010 was more than 8,700.
Yervoy mechanism
Yervoy treats metastatic melanoma by activating the immune system. The drug works by binding or inhibiting cytotoxic T lymphocyte-associated antigen 4 (CTLA-4), a molecule that plays vital role in relating natural immune responses. The presence or absence of CTLA-4 can curb or increase the immune system’s T-cell response in fighting disease.
The drug also works by blocking a complex set of interactions in the immune system. It is designed to inhibit the activity of CTLA-4, thereby sustaining an active immune response in its attack on cancer cells.
Approvals and indications
Ipilimumab was approved by the FDA in March 2011 to treat patients with late-stage melanoma that has spread or cannot be removed by surgery. On February 1, 2012, Health Canada approved ipilimumab for “treatment of unresectable or metastatic melanoma in patients who have failed or do not tolerate other systemic therapy for advanced disease.” Additionally Ipilimumab was approved in the European Union (EU), for second line treatment of metastatic melanoma, November 2012

Vitamin A
Vitamin A is actually a group of compounds that include retinol and beta-carotene among others. The science behind it dates back to 1816 when scientists noticed nutrition deprived dogs developed corneal ulcers. It was later found to be a fat soluble vitamin (as opposed to water-soluble) meaning it is stored in the fat (along with vitamins D, E, and K).
Vitamin A is indeed important in maintaining good vision. It is also important for cell growth and differentiation, skin health, and the immune system. For women the recommended daily allowance (RDA) is 700 mcg and 900 for men. The widely considered upper safe limit is considered to be 3000 mcg.
Animal sources of vitamin A include cheddar cheese, eggs, butter, and liver. Plant source include carrots, broccoli, kale and spinach. Dandelion greens have a particularly high amount of vitamin A and are used in natural medicine.
Worldwide Vitamin A deficiency is a huge problem. Approximately…
View original post 1,116 more words
ONE LAKH VIEWS ON ALL BLOGS—DR ANTHONY CRASTO
DR ANTHONY MELVIN CRASTO Ph.D
WORLDDRUGTRACKER
ANNOUNCING ONE LAKH PLUS VIEWS ON ALL BLOGS- DR ANTHONY CRASTO
- Eurekamoments in Organic Chemistry
- NEW DRUG APPROVALS
- WORLD DRUG TRACKER
- Green Chemistry International
- drug regulatory affairs international
- ORGANIC SPECTROSCOPY INTERNATIONAL
- ORGANIC SYNTHESIS INTERNATIONAL
- ALL ABOUT DRUGS
- ORGANIC CHEMISTRY INTERNATIONAL
- Drug Scaleup and Manufacturing International
SEE ALSO
- Organic Chemistry by Dr Anthony
- Technology Transfer
- Stereochemistry
- Spectroscopy
- Polymorphism
- Reactions in Org Chem
DR ANTHONY MELVIN CRASTO, Worlddrugtracker, Born in Mumbai in 1964 and graduated from Mumbai University, Completed his PhD from ICT ,1991, Mumbai, India, in Organic chemistry, The thesis topic was Synthesis of Novel Pyrethroid Analogues, Currently he is working with GLENMARK- GENERICS LTD, Research centre as Principal Scientist, Process Research (bulk actives) at Mahape, Navi Mumbai, India. Prior to joining Glenmark, he worked with major multinationals like Hoechst Marion Roussel, now sSanofi, Searle India ltd, now Rpg lifesciences, etc. he is now helping millions, has million hits on google on all organic chemistry websites. His New Drug Approvals, Green Chemistry International, Eurekamoments in organic chemistry are some most read blogs He has hands on experience in initiation and developing novel routes for drug molecules and implementation them on commercial scale over a 25 year tenure, good knowledge of IPM, GMP, Regulatory aspects, he has several international drug patents published worldwide . He gas good proficiency in Technology transfer, Spectroscopy, Stereochemistry, Synthesis, polymorphism etc He suffered a paralytic stroke in dec 2007 and is bound to a wheelchair, this seems to have injected feul in him to help chemists around the world, he is more active than before and is pushing boundaries, he has one lakh connections on all networking sites, He makes himself available to all, contact him on +91 9323115463, amcrasto@gmail.com
Personal Links
- my sites on the net
- DR ANTHONY MELVIN CRASTO
- GOOGLE GROUP ORGANIC PROCESS DEVELOPMENT
- mixxt
- epernicus
- scipeople
- jimdo
- yolasite
- my cv
- slidestaxx
- wordpress blog
- ABOUT ME
- BRANDSITE
- SKILLPAGES
- Academia.edu
- RESEARCHGATE
- DIIGO
- SLIDESHATE
- WIX
- WIX BLOG
- ISSUU
- SCRIBD
- BIZ
- GOOGLE BLOG
- APNACIRCLE
- Eurekamoments in Organic Chemistry
- Organic Chemistry by Dr Anthony
- Green Chemistry International
- Technology Transfer
- Stereochemistry
- Spectroscopy
- Polymorphism
- Reactions in Org Chem
- DR ANTHONY MELVIN CRASTO Ph.D
- Pharmaceuticals
- Medicinal chemistry
- Organic chemistry literature
- Patent related site
- Green chemistry
- Reagents
- R & D
- Molecules
- Heterocyclic chem
- Sourcing
- NEW DRUG APPROVALS
- WORLD DRUG TRACKER
- Green Chemistry International
- drug regulatory affairs international
- ORGANIC SPECTROSCOPY INTERNATIONAL
- ORGANIC SYNTHESIS INTERNATIONAL
- ALL ABOUT DRUGS
- ORGANIC CHEMISTRY INTERNATIONAL
- GOOGLE PLUS
- Drug Scaleup and Manufacturing International
amcrasto@gmail.com
email me if u like my posts
VINCRISTINE……..Chemistry, Isolation

VINCRISTINE
(3aR,3a1R,4R,5S,5aR,10bR)-methyl 4-acetoxy-3a-ethyl-9-((5S,7S,9S)-5-ethyl-5-hydroxy-9-(methoxycarbonyl)-2,4,5,6,7,8,9,10-octahydro-1H-3,7-methano[1]azacycloundecino[5,4-b]indol-9-yl)-6-formyl-5-hydroxy-8-methoxy-3a,3a1,4,5,5a,6,11,12-octahydro-1H-indolizino[8,1-cd]carbazole-5-carboxylate
…………………………………………………………..
………………………………………………………………….


Vincristine (brand name, Oncovin), formally known as leurocristine, sometimes abbreviated “VCR”, is a vinca alkaloid from the Catharanthus roseus (Madagascar periwinkle), formerly Vinca rosea and hence its name. It is amitotic inhibitor, and is used in cancer chemotherapy. Vincristine is created by the coupling of indole alkaloids vindoline and catharanthine in the vinca plant.[1]
Mechanism
Tubulin is a structural protein that polymerizes to microtubules. The cell cytoskeleton and mitotic spindle, among other things, are made of microtubules. Vincristine binds to tubulin dimers, inhibiting assembly of microtubule structures. Disruption of the microtubules arrests mitosis in metaphase. Therefore, the vinca alkaloids affect all rapidly dividing cell types including cancer cells, but also those of intestinal epithelium and bone marrow.

Uses
Vincristine is delivered via intravenous infusion for use in various types of chemotherapy regimens. Its main uses are in non-Hodgkin’s lymphoma as part of the chemotherapy regimen CHOP, Hodgkin’s lymphoma as part of MOPP, COPP, BEACOPP, or the less popular Stanford V chemotherapy regimen, in acute lymphoblastic leukemia, and in treatment for nephroblastoma (Wilms tumor, a kidney tumor most common in young children). It is also used to induce remission in ALL with Dexamethasone and L-Asparaginase. Vincristine is occasionally used as an immunosuppressant, for example, in treating thrombotic thrombocytopenic purpura (TTP) or chronic idiopathic thrombocytopenic purpura (ITP). It is used in combination with prednisone to treat childhood leukemia.
The main side-effects of vincristine are peripheral neuropathy, hyponatremia, constipation, and hair loss.
Peripheral neuropathy can be severe, and hence a reason to avoid, reduce, or stop the use of vincristine. One of the first symptoms of peripheral neuropathy is foot drop: A person with a family history of foot drop and/or Charcot-Marie-Tooth disease (CMT) should avoid the taking of vincristine.[2]
Accidental injection of vinca alkaloids into the spinal canal (intrathecal administration) is highly dangerous, with a mortality rate approaching 100 percent. The medical literature documents cases of ascending paralysis due to massive encephalopathy and spinal nerve demyelination, accompanied by intractable pain, almost uniformly leading to death; a handful of survivors were left with devastating neurological damage with no hope of recovery. Rescue treatments consist of washout of the cerebrospinal fluid and administration of protective medications.[3] A significant series of inadvertent intrathecal vincristine administration occurred in China in 2007 when batches of cytarabine andmethotrexate (both often used intrathecally) manufactured by the company Shanghai Hualian were found to be contaminated with vincristine.[4]
Having been used as a folk remedy for centuries, studies in the 1950s revealed that C. roseus contained 70 alkaloids, many of which are biologically active. While initial studies for its use in diabetes mellitus were disappointing, the discovery that it caused myelosuppression (decreased activity of the bone marrow) led to its study in mice withleukemia, whose lifespan was prolonged by the use of a vinca preparation. Treatment of the ground plant with Skelly-B defatting agent and an acid benzene extract led to a fraction termed “fraction A”. This fraction was further treated withaluminium oxide, chromatography, trichloromethane, benz-dichloromethane, and separation by pH to yield vincristine.[5]
Vincristine was approved by the United States Food and Drug Administration (FDA) in July 1963 as Oncovin. The drug was initially discovered by a team led by Dr. J.G. Armstrong, then marketed by Eli Lilly and Company.
Like LSD, the microtubule toxin vincristine allegedly causes not-unpleasant visual hallucinations in humans. Other side-effects of vincristine include depression, agitation, and insomnia. Very small doses are needed for the effects of LSD or vincristine, for example, these drugs are active at concentrations of 4.3E-7 M-1 vincristine and 1.0E-8 M-1 LSD.
Many researchers have favored the drug-receptor theory to explain drug-induced hallucinations, usually at the 5-HT2A receptor. In the drug-receptor theory, signal amplification takes place when one molecule of drug binds to a receptor, which activates G-proteins, which affects more proteins, thus signaling cascades explain how a small amount of LSD can lead to widespread changes in the cell.
Van Woerkom suggests instead that LSD binds an element of the cytoskeleton, in a fashion similar to colchicine or vinblastine, which directly bind tubulin. The amount of LSD needed to produce hallucinations is so vanishly small, that it seems hard to believe that a submicromolar dosage of LSD could act on a substrate as vast as the cytoskeleton. However, some microtubule inhibitors such as vincristine are effective at very low dosages. The potency of vincristine may partly explain the success of this drug as a chemotherapeutic drug.
Three generic drug makers supply vincristine in the United States – APP, Mayne, and Sicor (Teva).
- ^ “Pharmacognosy of Vinca Alkaloids”.
- Graf, W. D.; Chance, P. F.; Lensch, M. W.; Eng, L. J.; Lipe, H. P.; Bird, T. D. (1996). “Severe Vincristine Neuropathy in Charcot-Marie-Tooth Disease Type 1A”. Cancer 77 (7): 1356–1362. doi:10.1002/(SICI)1097-0142(19960401)77:7<1356::AID-CNCR20>3.0.CO;2-#. PMID 8608515.
- Qweider, M.; Gilsbach, J. M.; Rohde, V. (2007). “Inadvertent Intrathecal Vincristine Administration: A Neurosurgical Emergency. Case Report”. Journal of Neurosurgery: Spine 6 (3): 280–283. doi:10.3171/spi.2007.6.3.280. PMID 17355029.
- Jake Hooker and Walt Bogdanich (January 31, 2008). “Tainted Drugs Tied to Maker of Abortion Pill”. New York Times.
- Johnson, I. S.; Armstrong, J. G.; Gorman, M.; Burnett, J. P. (1963). “The Vinca Alkaloids: A New Class of Oncolytic Agents” (pdf). Cancer Research 23 (8 Part 1): 1390–1427.PMID 14070392.
External links
- Vincristine chemotherapy
- Vincristine and vinblastine
- Description and Natural History of the Periwinkle
- The Boger Route to (-)-Vindoline
- U.S. National Library of Medicine: Drug Information Portal – Vincristine
-
Cytostatic Vinca alkaloids rosea L. Catharanthus roseus G.Don) are now well known anticancer and particularly useful. Given the small amount of vincristine in Catharanthus present, quite a number of ways of preparation have been proposed by chemists. Thus FR-A-2296418 describes the synthesis of vincristine by coupling Catha-ranthine and vindoline. Other laboratories have achieved the transformation of vinblastine vincristine oxidation under controlled conditions, very strict.
-
FR-A-2210393 and US-A-3899493 perform the oxidation by chromic acid at -30, -90 ° C in a mixture of acetic acid-acetone or chloroform-acetic acid at -55 ° C.
-
In U.S. 4,375,432, chromic compound is also used in acid medium at -65 ° C, -50 ° C in a medium based solvent THF. In addition, EP-A-37289 boasts an oxidation mixture ferrous salt, hydrogen peroxide, perchlorate in acetonitrile. ZA-A-82 08939 discloses a method with chromic acid and an ether-chloroform.
-
HU-A-23638 offers diterbutylchromate in pelargonic acid, and finally EP-A-117861 gets vinblastinel transformation vincristine oxidant potassium permanganate in acetic acid medium. It is clear that these dimeric alkaloids are a valuable material because of their low levels in vegetable raw materials, and therefore the processes of synthesis or semi-synthesis performance are of extreme interest.
-
Vincristine is used in cancer chemotherapy, particularly for the treatment of certain acute leukemias.
-
This alkaloid is obtained mainly by extraction from leaves of Catharanthus Ro-seus (U.S. Patent No. 3,205,220) where it is accompanied by other alkaloids bis-Indo-holic, especially vinblastine.Vinblastine (I, R = CH 3), however, is present at a concentration much higher than that of vincristine and is therefore a precursor of choice for the semisynthesis of the latter.
-
Several processes of vincristine from vinblastine were disclosed. We note in particular patents or patent applications include:
- a) Belgian Patent 739,337 (Gedeon Richter) which describes a method for the oxidation of vinblastine vincristine in a mixture chromic acid, acetic acid and acetone.
- b) Belgian Patent 823560 (Gedeon Richter) the oxidation is performed with oxygen in the presence of formic acid and of a catalyst based on platinum at room temperature.
- c) European Patent Application 18231 (Gedeon Richter): is carried out by oxidation with chromic acid or an alkali metal dichromate in the presence of acetic anhydride and, optionally, of ethanol and an organic solvent immis target with water.
- d) European Patent Application 37289 (Eli Lil-ly): the oxidation is effected by the perchlorate of iron (II) in the presence of hydrogen peroxide and acetonitrile.
-
In addition, the European patent application 37. 290 discloses a process for the oxidation of vinblastine base with Na 2 Cr 2 O 7 in the presence of sulfuric acid in tetrahydrofuran. This reaction led to -50 ° C, is achieved with a yield of 80-92% calculated for each estimation.
-
Observed yields or purity of the products obtained characterizing the processes described above are, however, significant disadvantages.
-
Frequently a secondary product formed is N-demethyl vinblastine need then reformulate for vincristine.
Thus Potier and Kutney obtained products with the C18’S-C2’R absolute configuration, which is critical for anti-tumor activity, by a coupling reaction of the N.sup.b -oxide of catharanthine, or its derivatives, with vindoline, in the presence of trifluoroacetic anhydride, followed by a reduction reaction. [See Potier et. al. J. Am. Chem. Soc. 98. 7017 (1976) and Kutney et. al. Helv. Chim. Acta, 59, 2858 (1976)].
The Potier and Kutney coupling process has disadvantages. The yields are not satisfactory except for the coupling of catharanthine N-oxide with vindoline and even there the preparative yield is low. While vindoline is the most abundant alkaloid of Vinca rosea and is thus readily available, the other possible components of the Potier-Kutney coupling process (catharanthine, allocatharanthine, voacangine,) are relatively inaccessible, costly, and they do not allow a wide range of structural variation of that component of the coupling process.
- …………………………………………………………………………………………………………………………………………………………………………………………………………..
-
EP 0117861 B1
-
clips
-
The process of the present invention produces a simple vincristine, in quantity and purity requiring little or no additional purification by recrystallization or chromatography.
-
[0009]The reagent used is oxidation permanganate ion dissolved in toluene or dichloromethane as solvent. An alternative consists in immobilizing the resin on a permanganate anion, for example a polymer such as polystyrene comprising ammonium groups. Solubilization can be achieved by the action of a complexing agent crown ether (“crown-ether”) of potassium permanganate.
-
[0010]The permanganate anion can also be solubilized by preparing an ammonium salt or quaternary phosphonium corresponding which is soluble in methylene chloride or toluene. For this purpose, it is preferable to use potassium permanganate benzyltriethylammonium.
-
[0011]Obtaining from vincristine vinblastine using a permanganate salt is unexpected since the potassium permanganate used in some acetone oxide derivatives of vinblastine at the portion of the molecule velbanamine (Kutney, Balsevich and Worth, Heterocycles, 11, 69, 1978). The N-methyl group of the vindoline part intact.
-
[0012]The formation of N-CHO indoline skeleton on a bis-indole group vinblastine using a permanganate salt has never been reported.
-
[0013]According to one embodiment of the method of the present invention, vinblastine, preferably in the form of sulphate, is treated in the presence of an organic acid such as acetic acid, with an excess of potassium permanganate dissolved in dichloromethane or toluene in the presence of “18-crown-6” or ether derivatives dibenzo-or di-cyclohexylcorrespondants. The reaction is conducted at a temperature between -40 ° C and -75 ° C and is preferably followed by thin layer chromatography. The reaction time generally ranges from 5 minutes to 3 hours.
-
[0014]Potassium permanganate is preferably dissolved in dichloromethane and the oxidation reaction is then carried out at -70 ° C.
-
[0015]The solubility of potassium permanganate is indeed substantially increased in the presence of a macrocyclic polyether as the “18-crown-6” ether (1, 4, 7, 10, 13, 16-hexaoxacy-clooctadécane) or derivative dibenzo – or corresponding dicyclohexyl-hexyl.
-
[0016]The reaction mixture is then treated simultaneously by a mild reducing and alkaline. For this purpose, use is preferably an aqueous solution of bisulfite, disulfite or sodium metabisulfite and ammonia.
-
[0017]The organic phase was separated and the aqueous phase is extracted several times with methylene chloride. The combined organic phases were concentrated in vacuo to give a residue containing 80-85% of base vincristine, a 90-95% yield.
-
[0018]Alternatively, you can proceed with the extraction of the reaction mixture after reduction without conducting a simultaneous alkalinization. The acidic aqueous solution was then extracted with dichloromethane. This route is a novel process for purification of vincristine formed in the reaction medium.
-
[0019]According to another embodiment of the present invention, vincristine is obtained by oxidation of vinblastine by reacting a quaternary ammonium permanganate. The ammonium cation is preferably benzyltriethylammonium group or benzyl trimethyl ammonium (see eg Angew. Chem., Intern. Ed. 13, 170, 1974). The reaction is carried out in 2 to 6 hours at -60 ° C in an inert solvent wherein the ammonium salt is soluble, and an acid, preferably an organic acid of low molecular weight. A mixture of dichloromethane and glacial acetic acid can be used. After treatment with a mild reducing agent in aqueous medium, the resulting acidic solution is extracted with dichloromethane, and the organic phase is made alkaline by washing with a basic aqueous solution and concentrated. Vincristine solvate is isolated with a yield higher than 90%.
-
[0020]The latest variant of the method of the invention is particularly advantageous in terms of economic and technical.
-
[0021]Purification or separation may be effected by crystallization and chromatography using techniques well known this from the crude product of the reaction. The product can also be lyophilized.
-
[0022]In most cases, vincristine thus obtained can be converted directly into an addition salt with an organic or inorganic acid, preferably pharmaceutically acceptable. This salt is preferably a sulfate that may arise in a more or less solvated or hydrated.
-
[0023]We can also prepare vincristine dissolved in a physiologically acceptable solvent and ready to be injected.
-
[0024]In particular, vincristine sulfate is obtained by addition of H 2 S0 4 to a solution of vincristine gross or recrystallized from ethanol, dissolved in a mixture of methylene chloride and anhydrous ethanol, partial removal in vacuo chloride methylene and crystallization.
-
[0025]Vincristine sulfate thus obtained has a purity sufficient for use as a medicament, particularly in the form of injectable solutions.

Madagascar Periwinkle: Public Domain Illustration by Sydenham Edwards
The Madagascar periwinkle, an attractive flowering plant, contains the powerful anti-cancer chemicals vinblastine and vincristine. Velvet beans, which are named from the covering of soft hairs on the young plant, contain L-dopa, a very helpful chemical in the treatment of Parkinson’s disease. The Madagascar periwinkle and the velvet bean are just two of the large number of plants that have been found to contain medicinal chemicals. There are almost certainly many more plants that have undiscovered health benefits.
The Madagascar Periwinkle
The Madagascar periwinkle is native to Madagascar and India, but is now grown in many countries as a garden plant. It has also escaped from gardens and grows as a weed. The red, purple, pink or white flowers often have a center which is a different color from the rest of the flower. Madagascar periwinkles may grow up to one meter tall and have glossy green leaves.
The sap of the Madagascar periwinkle, which has a milky appearance and is poisonous, contains vinblastine, vincristine and many other alkaloids. Researchers are discovering that many of these alkaloids are biologically active inside the human body.
Vinblastine and Vincristine
Vinblastine and vincristine have very similar chemical structures, but their effects on the body are not the same. Vinblastine is used to treat specific types of cancer, such as Hodgkin’s disease, breast cancer, testicular cancer and non-small cell lung cancer. Vincristine is used in the treatment of acute lymphoblastic leukemia (ALL) and has provided a great breakthrough in successful treatment of this disease in children. When vincristine is added to the treatment regimen for children suffering from ALL, the survival rate reaches eighty percent. Vincristine is not so impressive in the treatment of ALL in adults.
Cells contain a supporting network of protein tubules, which are known as microtubules. Microtubules also play a vital role in the process of cell division. Before a cell divides, each chromosome in the cell is replicated. The replicated chromosomes are separated from their partners and pulled to opposite ends of the cell by microtubules during a process called mitosis. The cell then divides down the middle.
Vinblastine and vincristine stop microtubule formation during mitosis and therefore prevent cells from reproducing. This effect is strongest in cells that have a high rate of division, such as cancer cells. However, vinblastine and vincristine also affect cells lining the intestine, the cells in the bone marrow that produce blood cells, and the cells in the hair follicles, since these too have a high rate of cell division.
Possible vinblastine or vincristine side effects include constipation, hair loss, a low platelet count, which can cause increased bleeding, a low white blood cell count, which can lead to increased infections, or a low red blood cell count, resulting in anemia. There may occasionally be nerve damage, possibly due to the effect of the medicines on the microctubules in the nerve cells. Vincristine is more likely to cause nerve damage than vinblastine.

,,,,,,,,,,,,,


Total synthesis of (+)-vincristine (2). TFA, trifluoroacetic acid or trifluoroacetyl; DBU, 1,8-diazabicyclo[5.4.0]undec-7-ene.
Stereocontrolled total synthesis of (+)-vincristine


………………….
see docstoc presentation
click below
Vincristine
var docstoc_docid=”51697405″;var docstoc_title=”Vincristine”;var docstoc_urltitle=”Vincristine”;
…………………….
isolation
Kumar A, Patil D, Rajamohanan PR, Ahmad A (2013)
Isolation, Purification and Characterization of Vinblastine and Vincristine from Endophytic Fungus Fusarium oxysporumIsolated from Catharanthus roseus. PLoS ONE 8(9): e71805. doi:10.1371/journal.pone.0071805
http://www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0071805
Isolation, purification and characterization of vinblastine and vincristine from the endophytic fungus Fusarium oxysporum
A two stage fermentation procedure was employed for the isolation of vinblastine and vincristine by Fusarium oxysporum. In the first stage, 500 ml Erlenmeyer flasks containing 100 ml medium (MGYP, (0.3%) malt extract, (1.0%) glucose, (0.3%) yeast extract and (0.5%) peptone) were inoculated with 7 days old culture and incubated at 28°C on a rotary shaker (240 rpm) for 4–5 days, which was used as seed culture (I stage). Later, 10 ml seed culture was transferred to 500 ml Erlenmeyer flask containing 100 ml production medium called as vinca medium-1 (Glucose: 3%, Succinic acid: 1%, Sodium benzoate: 100 mg, Peptone: 1%, Magnesium sulphate: 3.6 mg, Biotin: 1 mg, Thiamine: 1 mg, Pyridoxal: 1 mg, Calcium pentothenate: 1 mg, Phosphate buffer: 1 ml (pH 6.8), L-Tryptophan: 0.1%, Geranium oil: 0.05%.) which were incubated at 28°C for 20 days as shake culture (II stage), after which it was harvested and used for further study. Culture filtrates and mycelia were separated with the help of muslin cloth and then lyophilized. Lyophilized culture filtrate was extracted using ethyl acetate as a solvent system. The organic layer was separated from the aqueous layer using separating funnel. The extraction was repeated thrice and the solvent was dried using anhydrous sodium sulphate and concentrated under vacuum using rotavapour at 40°C in order to get crude extract. A small amount of crude extract was dissolved in ethyl acetate and subjected to thin layer chromatography (TLC) on silica gel-G (0.5 mm thickness) using chloroform:methanol (8:2) as a solvent system. The TLC plates were sprayed with ceric ammonium sulphate reagent. Vinca alkaloids spots produced brilliant violet color as well as purple color with above spraying reagent. Purification of fungal vinblastine and vincristine were done by silica gel column chromatography. The crude extract was loaded on silica gel column (60–120 mesh size, 40 cm×2 cm length width) pre-equilibrated with chloroform and eluted with a gradient of chloroform:methanol (100% chloroform, 9:1, 8:2, 7:3, 1:1 and 3:7 and 100% methanol). Fractions containing compounds with Rf values similar to that of the standard vinblastine and vincristine were pooled and subjected to preparative TLC on a 0.5 mm thick (20 cm×20 cm) silica plate and developed in chloroform:methanol (8:2) solvent system. The putative bands of fungal vinblastine and vincristine were scraped and eluted out with methanol. Purity of the isolated compounds was checked on TLC in the solvent systems such as (a) chloroform:methanol (8:2) (b) chloroform:methanol (9:1) and (c) ethyl acetate: acetonitrile (8:2).
http://www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0071805
see also
http://www.ncbi.nlm.nih.gov/pubmed/20209002
ALSO
large-scale isolation of native catharantine, vindoline and 3′,4′-anhydrovinblastine whereby the isolation of vincristine, vinblastine, leurosine and the corresponding desacetoxy, desacetyl and N-desmethyl derivatives in a manner known per se can also be accomplished.
For the isolation of the two monoindole alkaloids: vindoline and catharantine from the dried plant Vinca rosea L. Svoboda [J. Am. Pharm. Assoc. 48, (11), 659 (1959)] described a method, which can be accomplished only with a very modest yield. From 1 kg. of the dried plant–subjecting the whole plant to a suitable treatment–approximately 0.6 g. of vindoline and 0.05 g. of catharantine were obtained.
3′,4′-ANHYDROVINBLASTINE UNTIL NOW HAS NEITHER BEEN ISOLATED FROM THE PLANT Vinca rosea L. nor identified in it.
For the preparation of the diindole alkaloid components starting from the leaves of Vinca rosea L. there are more methods known in the art (U.S. Pat. nos. 3,097,137; 3,205,220; 3,225,030 and Hungarian Pat. Nos. 153,200; 154,715; 160,967 and 164,958 as well as Austrian Pat. Nos. 313,435, 313,485, Australian pat. No. 458,629 and Swiss Pat. No. 572,488 and British Pat Nos. 1,412,932, 1,382,460 corresponding to the preceding two patents). According to these known processes from 1 kg. of the dried leaves of Vinca rosea L. about 0.1 to 0.2 g. of leurosine can be obtained and vinblastine, vincristine and optionally the corresponding N-desmethyl, desacetyl and desacetoxy derivatives are also simultaneously isolated.
Further on it is well known that the synthetic catharantine and vindoline may be coupled by the Polonovszky reaction to give 3′,4′-anhydrovinblastine which can thereafter be epoxidized to leurosine [Potier et al. Tetrahedron Letters 3945 (1976); DT-OS 25 58,124; Helv. Chim. Acta 59, 2858 (1976); Heterocycles 4, 997 (1976), Belgian patent specification No. 842,200 equivalent to U.S. patent application Ser. No. 582,372]. Leurosine itself has a valuable tumour growth inhibiting activity and the N-desmethyl-N-formyl derivative thereof is the most promising substance against leukemia (Hungarian Pat. No. 165,986 equivalent to U.S. patent application Ser. No. 422,100, and Austrian Pat. No. 332,566 which has issued as British Pat. No. 1,412,932).
Simeprevir has been approved in Japan for the treatment of genotype 1 chronic hepatitis C infection
simeprevir
| CAS number | 923604-59-5 | ||
| Formula | C38H47N5O7S2 | ||
| Weight | 749.93908 |
Stockholm, Sweden — Medivir AB (OMX: MVIR) today reports that Janssen Pharmaceutical R&D Ireland (Janssen) has been informed by the Japanese Ministry of Health, Labour and Welfare (MHLW) that simeprevir has been approved for the treatment of genotype 1 chronic hepatitis C virus (HCV) infection.
read all at
http://www.pharmalive.com/japan-approves-simeprevir
Hepatitis C virus (HCV) infections affect approximately 3 percent of the worldwide population and often lead to cirrhosis and hepatocellular carcinoma. The standard therapy of pegylated- interferon and ribavirin induces serious side effects and provides viral eradication in less than 50% of patients. Combination therapy of HCV including ribavirin and interferonare currently is the approved therapy for HCV. Unfortunately, such combination therapy also produces side effects and is often poorly tolerated, resulting in major clinical challenges in a significant proportion of patients. Numerous direct acting agents (DAAs) have been or are being developed for treatment of HCV, such as telaprevir and boceprevir (both received MA approved in 2011 for use with interferon and ribavirin based therapy), however direct acting agents are linked to increased toxicity of treatment, the emergence of resistance, and to date do not provide a standard of care which is interferon free. The combination of direct acting agents can also result in drug-drug interactions. To date, no HCV therapy has been approved which is interferon free. There is therefore a need for new combination therapies which have reduced side effects, and interferon free, have a reduced emergence of resistance, reduced treatment periods and/or and enhanced cure rates.
Simeprevir (formerly TMC435) is an experimental drug candidate for the treatment of hepatitis C. It is being developed byMedivir and Johnson & Johnson‘s pharmaceutical division Janssen Pharmaceutica and is currently in Phase III clinical trials.[1]
Simeprevir is a hepatitis C virus protease inhibitor.[2]
Simeprevir is being tested in combination regimens with pegylated interferon alfa-2a and ribavirin,[3] and in interferon-free regimens with other direct-acting antiviral agents including daclatasvir[4] and sofosbuvir [5]
Food and Drug Administration (FDA) has granted Priority Review to the New Drug Application (NDA) for simeprevir (TMC435). Simeprevir is an investigational NS3/4A protease inhibitor taken orally (150 mg capsule) once a day along with pegylated interferon and ribavirin for genotype 1 chronic hepatitis C virus (HCV) infection in adult patients with compensated liver disease (meaning the liver is heavily scarred but still functional).
“Hepatitis C is a complex disease and Janssen is committed to working with the HCV community, caregivers, and health care systems to address this global epidemic,” said Gaston Picchio, Hepatitis Disease Area Leader, Janssen Research & Development. “We are pleased that the FDA has granted simeprevir Priority Review, as it is a significant step forward in making this therapy available to physicians and their hepatitis C patients.”
The FDA grants Priority Review to medicines that may offer major advances in care or provide a treatment option where no adequate therapy exists. Under the Prescription Drug User Fee Act, FDA review will begin approximately 60 days after receipt of the application and will aim to be completed within six months from when the review period begins.
The regulatory submission for simeprevir is supported in part by data from three pivotal Phase 3 studies: QUEST-1 and QUEST-2 in treatment-naïve patients and PROMISE in patients who have relapsed after prior interferon-based treatment. Janssen also recently submitted simeprevir for marketing authorization to regulatory authorities in Japan and Europe.
- “Medivir Announces That Simeprevir (TMC435) Data Will Be Presented at the Upcoming AASLD Meeting”. Yahoo News. October 1, 2012. Retrieved November 6, 2012.
- Lin, TI; Lenz, O; Fanning, G; Verbinnen, T; Delouvroy, F; Scholliers, A; Vermeiren, K; Rosenquist, A et al. (2009). “In vitro activity and preclinical profile of TMC435350, a potent hepatitis C virus protease inhibitor”. Antimicrobial agents and chemotherapy 53 (4): 1377–85. doi:10.1128/AAC.01058-08. PMC 2663092. PMID 19171797.
|displayauthors=suggested (help) - “Phase 3 Studies Show Simeprevir plus Interferon/Ribavirin Cures Most Patients in 24 Weeks”. hivandhepatitis.com. December 27, 2012.
- Medivir announces TMC435 in an expanded clinical collaboration. Medivir. 18 April 2012.
- Results from a phase IIa study evaluating Simeprevir and Sofosbuvir in prior null responder Hepatitis C patients have been presented at CROI. 6 March 2013.
IUPAC standard name
(1R, 4R, 6S, 15R, 17R)-N-(cyclopropanesulfonyl) -17 – ({7-methoxy-8-methyl-2-[4 – (propan-2-yl) -1,3-thiazol-2 -yl] quinolin-4-yl} oxy)-13-methyl-2 ,14-dioxo-3 ,13-diazatricyclo [13.3.0.0 4 , 6 ] octadec-7-ene-4-carboxamide
IUPAC traditional name
(1R, 4R, 6S, 15R, 17R)-N-(cyclopropanesulfonyl) -17 – {[2 – (4-isopropyl-1 ,3-thiazol-2-yl)-7-methoxy-8-methylquinolin-4- yl] oxy}-13-methyl-2 ,14-dioxo-3 ,13-diazatricyclo [13.3.0.0 4 , 6 ] octadec-7-ene-4-carboxamide
Aliases
TMC435
TMC435350

,,,,,,,,,,,,,
NS3/4A protease inhibitors
Ciluprevir (BILN 2061) Boehringer Ingelheim
Boceprevir (SCH503034) Merck
Telaprevir (VX-950) Vertex
Danoprevir (RG7227) Roche
simeprevir /TMC435 Tibotec / Medivir
Vaniprevir (MK-7009) Merck
Bl 201335 Boehringer Ingelheim
BMS-650032 Bristol-Myers Squibb
GS-9256 Gilead
ABT-450 Abbott / Enanta
Narlaprevir (SCH900518) Merck
PHX1766 Phenomix
ACH-1625 Achillion
IDX320 Idenix
MK-5172 Merck
VX-985 Vertex Drug name Company
GS-9451 Gilead
Telaprevir
Accordin to http://en.wikipedia.Org/wiki/File:Telaprevir.svg, Teaprevir has the structure
Systematic lUPAC Name: (1 S,3aR,6aS)-2-[(2S)-2-[[(2S)-2-Cyclohexyl-2-(pyrazine-2- carbonylamino)acetyl]amino]-3,3-dimethylbutanoyl]-/\/-[(3S)-1-(cyclopropylamino)-1 ,2- dioxohexan-3-yl]-3,3a,4,5,6,6a-hexahydro-1/-/-cyclopenta[c]pyrrole-1-carboxamide
Telaprevir may be administered in a unit dose of, for example between about 250 and about l OOOmg, such as about 750mg/kg. Typically once, twice, three or four times daily, such as three times daily for the duration of the pre-treatment period and/or combination treatment period.
Boceprevir
Accordin to http://en.wikipedia.0rg/wiki/File:B0ceprevir.svg, Boceprevir has the structure:
Systematic lUPAC Name: (1 R,2S,5S)-N-[(2≡)-4-amino-1-cyclobutyl-3,4-dioxobutan-2-yl)]- 3-{(2S)-2-[(tert-butylcarbamoyl)amino]-3,3-dimethylbutanoyl}- 6,6-dimethyl-3- azabicyclo[3.1.0]hexane-2-carboxamide
Boceprevir may be administered in a unit dose of, for example between about 250 and about 1000mg, such as about 800mg/kg. Typically once, twice, three or four times daily, such as three times daily for the duration of the pre-treatment period and/or combination treatment period.
Compound 1: miR-122 inhibitor
As reported in Young et al., JACS 2010, 132, 7976-7981) (hereby incorporated by reference), it is possible to assay for small molecule inhibitors of miR122 and small molecule are known, such as those illustrated below:
» {7.02 ± 1.40) If (4. S3 * 0.45)
The numerical values refer to luciferase expression due to miR-122 deprepression, and values greater than 1 indicate miR-122 inhibition.
EMA Accepts AstraZeneca’s Naloxegol Application

naloxegol
http://www.ama-assn.org/resources/doc/usan/naloxegol.pdf
Morphinan-3,14-diol, 4,5-epoxy-6-(3,6,9,12,15,18,21-heptaoxadocos-1-yloxy)-17-(2-
propen-1-yl)-, (5α,6α)-
4,5α-epoxy-6α-[(3,6,9,12,15,18,21-heptaoxadocosan-1-yl)oxy]-17-(prop-2-en-1-
yl)morphinan-3,14-diol
MOLECULAR FORMULA C34H53NO11
MOLECULAR WEIGHT 651.8
SPONSOR AstraZeneca
CODE DESIGNATION NKTR-118
CAS REGISTRY NUMBER 854601-70-0
WHO NUMBER 9434
Marketing Authorisation Application for naloxegol accepted by European Medicines Agency
Friday, 27 September 2013
AstraZeneca today announced that the European Medicines Agency (EMA) has accepted the Marketing Authorisation Application (MAA) for naloxegol, an investigational peripherally-acting mu-opioid receptor antagonist, which has been specifically designed for the treatment of opioid-induced constipation (OIC) for adult patients 18 years and older, including patients with inadequate response to laxatives.
read more
http://www.pharmalive.com/ema-accepts-astrazeneca-s-naloxegol-application
Naloxegol (INN; NKTR-118), or PEGylated naloxol,[1] is a peripherally–selective opioid antagonist under development byAstraZeneca, licensed from Nektar, for the treatment of opioid-induced constipation.[2]
- ^ Roland Seifert; Thomas Wieland; Raimund Mannhold; Hugo Kubinyi, Gerd Folkers (17 July 2006). G Protein-Coupled Receptors as Drug Targets: Analysis of Activation and Constitutive Activity. John Wiley & Sons. p. 227. ISBN 978-3-527-60695-5. Retrieved 14 May 2012.
- ^ “Nektar | R&D Pipeline | Products in Development | CNS/Pain | Oral Naloxegol (NKTR-118) and Oral NKTR-119”. Retrieved 2012-05-14.
Naloxegol (NKTR-118) is an investigational drug candidate in Phase 3 studies being developed as a once-daily oral tablet for the treatment of opioid-induced constipation. Naloxegol (NKTR-118) was designed using Nektar’s proprietary small molecule polymer conjugate technology. Results of the Phase 2 study of naloxegol (NKTR-118) were presented in October 2009 at the American College of Gastroenterology Annual Clinical Meeting and the American Academy of Pain Management. NKTR-119 is an early stage drug development program that is intended to combine oral naloxegol (NKTR-118) with selected opioids, with the goal of treating pain without the side effect of constipation traditionally associated with opioid therapy.
Nektar and AstraZeneca have a global agreement for both naloxegol (NKTR-118) and NKTR-119. Under the agreement, AstraZeneca has responsibility for the development, global manufacturing and marketing of both naloxegol (NKTR-118) and NKTR-119. For naloxegol (NKTR-118), Nektar is eligible to receive up to $235 million in aggregate payments upon the achievement of certain regulatory milestones, as well as additional tiered sales milestone payments of up to $375 million if the product achieves considerable levels of commercial success. Nektar will also be eligible to receive significant double-digit royalty payments on net sales of naloxegol (NKTR-118) worldwide. For NKTR-119, Nektar would receive development milestone payments as well as tiered sales milestone payments. Nektar will also receive significant double-digit royalty payments on NKTR-119 net sales worldwide.
oxalate derivative
http://www.ama-assn.org/resources/doc/usan/naloxegol-oxalate.pdf
Morphinan-3,14-diol, 4,5-epoxy-6-(3,6,9,12,15,18,21-heptaoxadocos-1-yloxy)-
17-(2-propen-1-yl)-, (5α,6α)-, ethanedioate (1:1)
4,5α-epoxy-6α-[(3,6,7,12,15,18,21-heptaoxadocosyl)oxy]-17-(prop-2-
enyl)morphinan-3,14-diol hydrogen ethanedioate
MOLECULAR FORMULA C34H53NO11 . C2H2O4
MOLECULAR WEIGHT 741.8
TRADEMARK None as yet
SPONSOR AstraZeneca
CODE DESIGNATIONS NKTR-118 oxalate, AZ13337019 oxalate
CAS REGISTRY NUMBER 1354744-91-4

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
















 DACLIZUMAB,
DACLIZUMAB,











