Home » Posts tagged 'zeprumetostat'
Tag Archives: zeprumetostat
Zeprumetostat
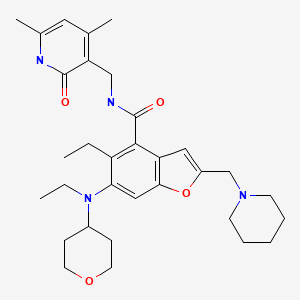
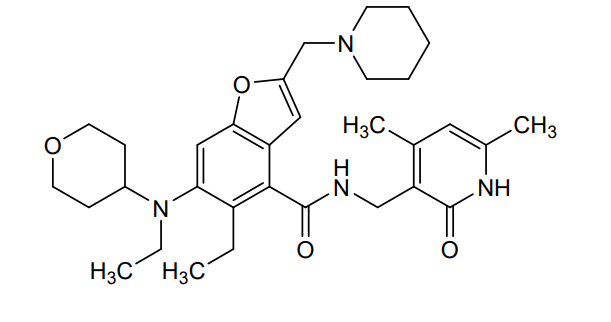
Zeprumetostat
CAS 2098545-98-1
MF C32H44N4O4 MW 548.7 g/mol
CHINA 2025, APPROVALS 2025
- 4-Benzofurancarboxamide, N-[(1,2-dihydro-4,6-dimethyl-2-oxo-3-pyridinyl)methyl]-5-ethyl-6-[ethyl(tetrahydro-2H-pyran-4-yl)amino]-2-(1-piperidinylmethyl)-
- N-[(1,2-Dihydro-4,6-dimethyl-2-oxo-3-pyridinyl)methyl]-5-ethyl-6-[ethyl(tetrahydro-2H-pyran-4-yl)amino]-2-(1-piperidinylmethyl)-4-benzofurancarboxamide
N-[(4,6-dimethyl-2-oxo-1,2-dihydropyridin-3-yl)methyl]-5-ethyl-6-[ethyl(oxan-4-yl)amino]-2-[(piperidin-1-yl)methyl]-1-benzofuran-4-carboxamide
enhancer of zeste homolog 2 (EZH2) inhibitor, antineoplastic, Airijing® (China), EZH2-IN-15, SHR 2554
The chemical structure for zeprumetostat was obtained from WHO proposed INN list 131 (August 2024). The INN record describes the compound as an enhancer of zeste homolog 2 (EZH2) inhibitor and antineoplastic. The chemical structure is claimed in patent WO2017084494A1 [3]. Based on Hengrui’s declared development pipeline, we predicted at that time that zeprumetostat was likely the INN for their EZH2 inhibitor clinical lead SHR2554.
Zeprumetostat is an orally available selective inhibitor of the histone lysine methyltransferase (HMT) enhancer of zeste homolog 2 (EZH2), with potential antineoplastic activity. Upon oral administration, zeprumetostat selectively targets, binds to and inhibits the activity of EZH2. Inhibition of EZH2 specifically prevents the methylation of histone H3 on lysine 27 (H3K27). This decrease in histone methylation alters gene expression patterns associated with cancer pathways and results in decreased proliferation of EZH2-expressing cancer cells. EZH2, an HMT class enzyme and the catalytic subunit of the polycomb repressive complex 2 (PRC2), is overexpressed or mutated in a variety of cancer cells and plays a key role in tumor cell proliferation; its expression is correlated with tumor initiation, progression, stem cell self-renewal, migration and angiogenesis.
Zeprumetostat is a small molecule drug. The usage of the INN stem ‘-metostat’ in the name indicates that Zeprumetostat is a histone N-methyltransferase inhibitor. Zeprumetostat has a monoisotopic molecular weight of 548.34 Da.
- Zeprumetostat, Azacitidine Combined With Lipo-MIT in R/R PTCLCTID: NCT07372352Phase: Phase 2Status: RecruitingDate: 2026-01-28
- EZH2 Inhibitor Zeprumetostat in Combination Therapy for Patients With Relapsed or Refractory Mature T-cell and NK-cell LymphomasCTID: NCT07339527Phase: Phase 1/Phase 2Status: Not yet recruitingDate: 2026-01-14
PAT
WO2017084494
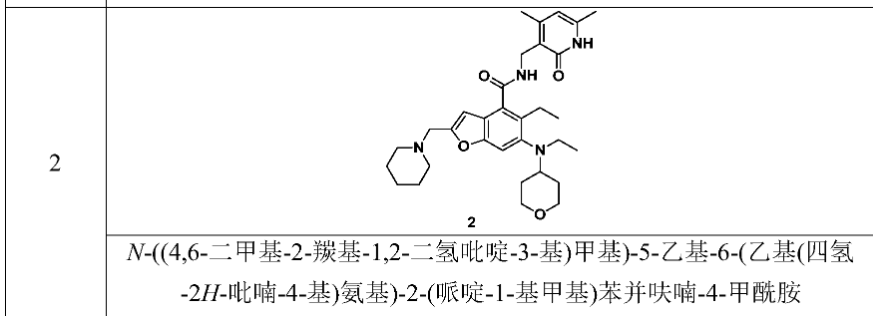
[0234]N-((4,6-dimethyl-2-carbonyl-1,2-dihydropyridin-3-yl)methyl)-5-ethyl-6-(ethyl(tetrahydro-2H-pyran-4-yl)amino)-2-(piperidin-1-ylmethyl)benzofuran-4-carboxamide
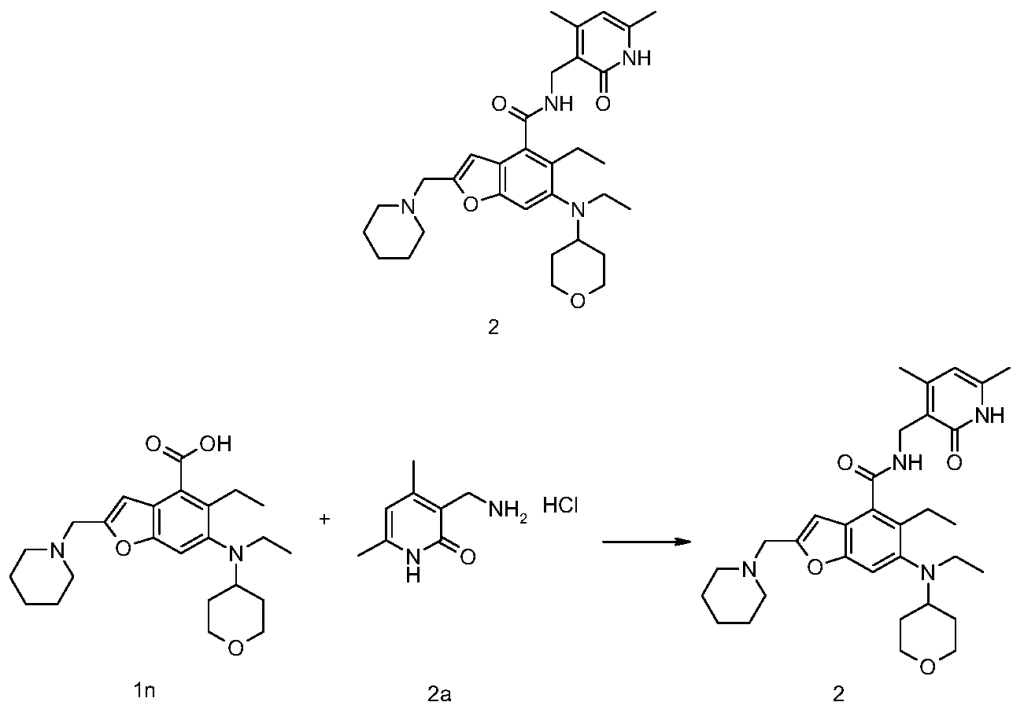
5-Ethyl-6-(ethyl(tetrahydro-2H-pyran-4-yl)amino)-2-(piperidin-1-ylmethyl)benzofuran-4-carboxylic acid 1n (1.0 g, 2.4 mmol) was dissolved in 30 mL of N,N-dimethylformamide, and 1-ethyl-3-(3-dimethylpropylamine)carbodiimide (696 mg, 3.6 mmol), 1-hydroxybenzotriazole (490 mg, 3.6 mmol), and N,N-diisopropylethylamine (1.56 g, 12.1 mmol) were added. The mixture was stirred for 1 hour, and then 3-(aminomethyl)-4,6-dimethylpyridin-2(1H)-one hydrochloride 2a (593 mg, 3.0 mmol, prepared by the method disclosed in patent application “WO2014097041”) was added. The mixture was stirred at room temperature for 12 hours. After the reaction was complete, excess water was added, and the mixture was extracted with a mixed solvent of dichloromethane and methanol (V:V = 8:1). The organic phases were combined, washed with water and saturated sodium chloride solution, dried over anhydrous sodium sulfate, filtered, and the filtrate was concentrated under reduced pressure. The residue was purified by silica gel column chromatography using eluent system A to give the title product N-((4,6-dimethyl-2-carbonyl-1,2-dihydropyridin-3-yl)methyl)-5-ethyl-6-(ethyl(tetrahydro-2H-pyran-4-yl)amino)-2-(piperidin-1-ylmethyl)benzofuran-4-carboxamide 2 (750 mg, white solid), yield: 57%.
[0238]
1H NMR(400MHz,DMSO-d 6):δ11.48(s,1H),8.15(t,1H),7.39(s,1H),6.46(s,1H),5.86(s,1H),4.32(d,2H),3.83(d,2H),3.54(s,2H),3.21(t,2H),3.01-3.07(m,2H),2.92-2.97(m,1H),2.77-2.82(m,2H),2.39(brs,4H),2.23(s,3H),2.11(s,3H),1.64-1.67(brd,2H),1.47-1.55(m,6H),1.36-1.37(brd,2H),1.02(t,3H),0.82(t,3H).
PAT
WO2019091450]
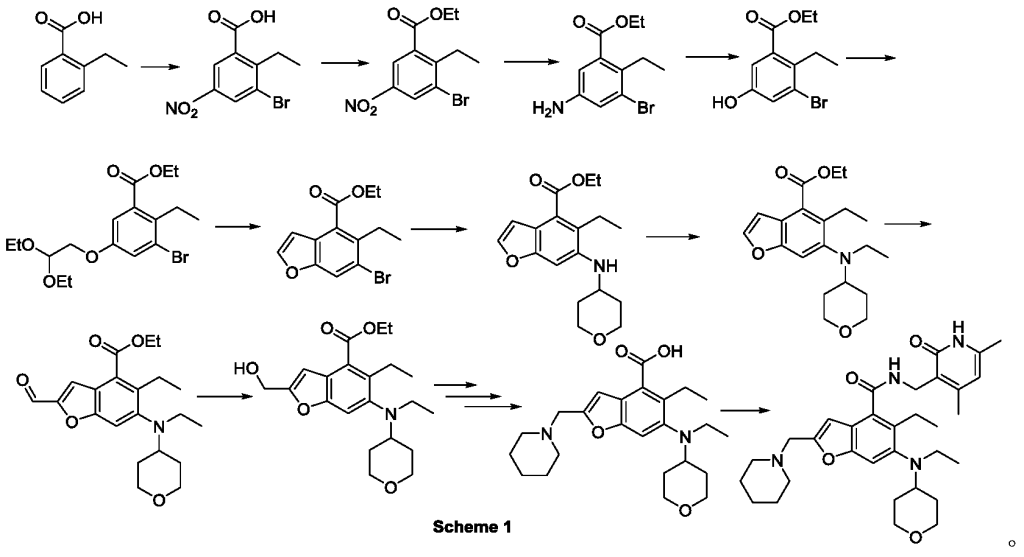
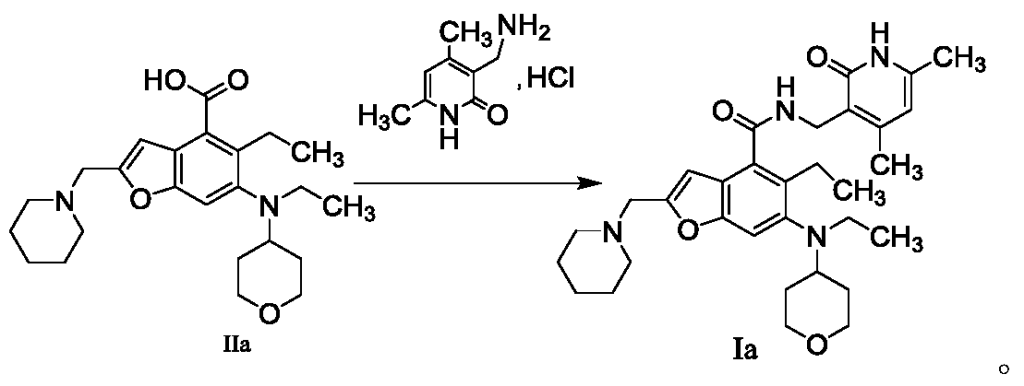
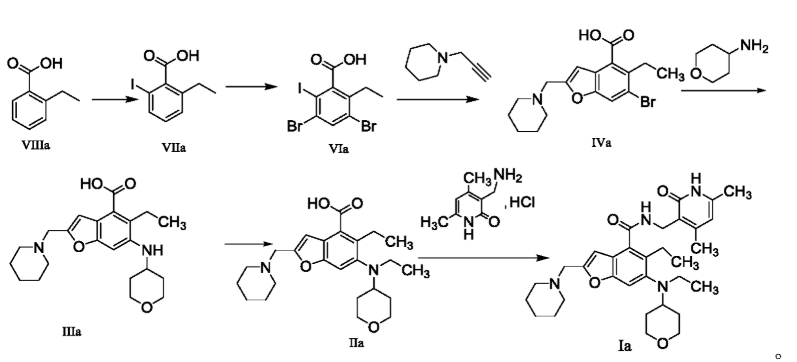
The method for preparing compounds from Formula IIa to Formula Ia provided by this invention can be specifically referred to in the methods for preparing amides disclosed in PCT applications WO2017084494A, WO2012142513, WO2013039988, WO2015-141616, and WO2011140325.
In a 25 mL three-necked flask, starter IIa (50 mg, 0.12 mmol), 1-ethyl-3-(3-dimethylpropylamine)carbodiimide (34.5 mg, 0.18 mmol), 1-hydroxybenzotriazole (23.67 mg, 0.18 mmol), and N,N-diisopropylethylamine (77.89 mg, 0.6 mmol) were mixed and dissolved in 3 mL of N,N-dimethylformamide and stirred until homogeneous. Then, starter 3-(aminomethyl)-4,6-dimethylpyridine-2(1H)-one hydrochloride (24.9 mg, 0.13 mmol) was added and the mixture was stirred at room temperature until the thin-layer chromatography showed that starter IIa had disappeared. The reaction was then terminated. Excess water was added to the reaction solution, and the mixture was extracted with a mixed solvent of dichloromethane and methanol. The organic phases were combined, washed with water, washed with saturated sodium chloride solution, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure, and the residue was purified by silica gel column chromatography with a dichloromethane-methanol eluent system to give 30.1 mg of white solid, yield 47.0%. [0151]m/z[M+H]
1H NMR(400MHz,DMSO-d6)ppm 11.51(s,1H)8.17(t,1H)7.39(s,1H)6.47(s,1H)5.86(s,1H)4.32(d,2H)3.83(d,2H)3.53(s,2H)3.21(t,2H)3.04(d,2H)2.94(br.s .,1H)2.79(d,2H)2.38(br.s.,4H)2.23(s,3H)2.08-2.14(m,3H)1.65(d,2H)1.44-1.56(m,6H)1.36(d,2H)1.02(t,3H)0.81(t,3H).
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Benzofuran derivative, preparation method thereof and use thereof in medicinePublication Number: US-11059811-B2Priority Date: 2015-11-19Grant Date: 2021-07-13
- Derived from benzofuran, method of preparing it and using it in medicinePublication Number: ES-2760510-T3Priority Date: 2015-11-19Grant Date: 2020-05-14
- Benzofuran derivative, a method for production thereof and use thereof in medicinePublication Number: RU-2727198-C2Priority Date: 2015-11-19Grant Date: 2020-07-21
- Benzofuran derivative, preparation method thereof and use thereof in medicinePublication Number: US-2020354349-A1Priority Date: 2015-11-19
- BENZOFURAN DERIVATIVE, ITS USES AND ITS PREPARATION PROCESS, AND PHARMACEUTICAL COMPOSITIONPublication Number: BR-112018007876-B1Priority Date: 2015-11-19
- Benzofuran derivative, preparation method thereof and use thereof in medicinePublication Number: US-2018327394-A1Priority Date: 2015-11-19
- Benzofuran derivative, preparation method thereof and use thereof in medicinePublication Number: EP-3378859-B1Priority Date: 2015-11-19Grant Date: 2019-10-30
- Benzofuran derivative, preparation method thereof and use thereof in medicinePublication Number: US-10759787-B2Priority Date: 2015-11-19Grant Date: 2020-09-01
- Benzofuran derivative, preparation method thereof and use thereof in medicinePublication Number: EP-3378859-A1Priority Date: 2015-11-19
- Crystal of benzofuran derivative free base and preparation methodPublication Number: US-11155537-B2Priority Date: 2017-05-18Grant Date: 2021-10-26
- Use of ezh2 inhibitor combined with btk inhibitor in preparing drug for treating tumorPublication Number: US-2021030736-A1Priority Date: 2017-05-18
- Use of EZH2 inhibitor combined with BTK inhibitor in preparing drug for treating tumorPublication Number: US-11065239-B2Priority Date: 2017-05-18Grant Date: 2021-07-20
- Crystal of benzofuran derivative free base and preparation methodPublication Number: US-2021130333-A1Priority Date: 2017-05-18
- Determination and preparation method of benzofuran derivative free basePublication Number: KR-102612379-B1Priority Date: 2017-05-18Grant Date: 2023-12-12
//////////zeprumetostat, ANAX LAB, CHINA 2025, APPROVALS 2025, antineoplastic, Airijing® (China), EZH2-IN-15, SHR 2554

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










