Home » Posts tagged 'WV7377RGM8'
Tag Archives: WV7377RGM8
Teprosulvose
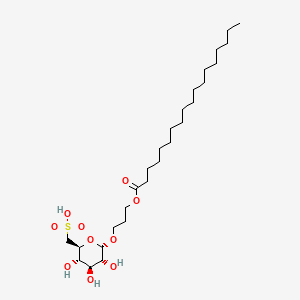
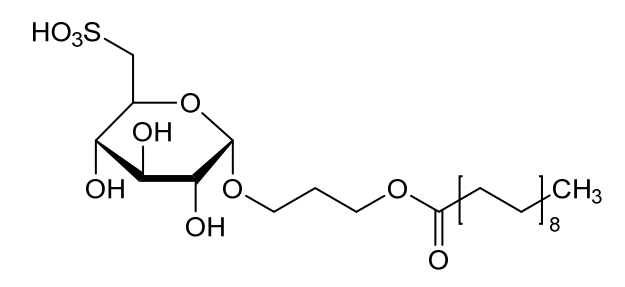

Teprosulvose
CAS 1983131-47-0
MF C27H52O10S MW568.761
| Sulfoquynovosylacylpropanediol [(2S,3S,4S,5R,6S)-3,4,5-trihydroxy-6-(3-octadecanoyloxypropoxy)oxan-2-yl]methanesulfonic acid |
3-(octadecanoyloxy)propyl 6-deoxy-6-sulfo-α-D-glucopyranoside
radiosensitizer (veterinary use), WV7377RGM8, SQAP
Teprosulvose (CAS 1983131-47-0) is a novel synthetic glycolipid, specifically a sulfoquinovosylacylpropanediol (SQAP). It is primarily developed for use in veterinary medicine as a radiosensitizer, intended to enhance the effectiveness of radiation therapy in treating malignant tumors.
1. Chemical Identity and Structure
- USAN/INN Name: Teprosulvose
- Systemic Name: 3-(octadecanoyloxy)propyl 6-deoxy-6-sulfo-$\alpha$-D-glucopyranoside
- Molecular Weight: 568.76 g/mol
- Structure: It consists of a glucose derivative (6-deoxy-6-sulfo-$\alpha$-D-glucopyranoside) linked via a propyl bridge to a long-chain fatty acid (stearic acid/octadecanoic acid).
Regulatory Data
Teprosulvose is currently in the investigational stage, primarily focused on veterinary oncology.
- USAN/INN Status: The name “Teprosulvose” was officially adopted by the USAN Council in 2024 (File LM-156).
- Classification: Radiosensitizer.
- Target Application: Adjuvant therapy for malignant tumors in animals (e.g., canine or feline cancers).
- Current Status: It has not yet received full FDA or EMA approval for human use. In the U.S., it is typically handled under Investigational New Animal Drug (INAD) protocols for clinical trials in veterinary patients.
Note: Because it is a specialized veterinary investigative agent, detailed safety data (LD50, pharmacokinetics) is generally found in specific FDA Freedom of Information (FOI) summaries or peer-reviewed veterinary oncology journals rather than standard human drug databases.
INN List 131 (WHO): Teprosulvose was officially included in the World Health Organization’s International Nonproprietary Names (INN) list in 2024. This confirms its unique status as a distinct drug substance.
USAN Council: The United States Adopted Names Council assigned the name in 2024, classifying it as a radiosensitizer.
FDA Status: It is currently under investigation (INAD) for canine oral melanoma and other solid tumors in veterinary medicine. Human clinical trial data is not yet widely available as the primary focus remains on the “Veterinary First” pathway.
Mechanism of Action: It is a potent inhibitor of DNA polymerase $\alpha$ and $\beta$. By inhibiting the repair of radiation-induced DNA damage, it effectively “locks in” the damage to tumor cells while sparing normal tissue due to differential uptake.
PAT
US Patent 10,206,942 (and related continuations): Covers the use of SQAP compounds in combination with radiation.
WO 2017/023812: International filing regarding the composition and therapeutic application of these glycolipids.
PAT
PAT
- Sulfonated sugar compounds, pharmaceutical compositions which contain the same, and methods of treating tumors with the samePublication Number: US-7973145-B2Priority Date: 2007-07-20Grant Date: 2011-07-05
- Sulfonated sugar compounds, pharmaceutical compositions which contain the same, and methods of treating tumors with the samePublication Number: US-2009209475-A1Priority Date: 2007-07-20
- Novel sulfonated sugar derivative, and use thereof for medicinal agentPublication Number: EP-2130834-A1Priority Date: 2007-07-20
- Sulfonated sugar compounds, pharmaceutical compositions which contain the same, and methods of treating tumors with the samePublication Number: US-2010298246-A1Priority Date: 2007-07-20
- Novel sulfonated sugar derivatives and their use as pharmaceuticalsPublication Number: JP-4435861-B2Priority Date: 2007-07-20Grant Date: 2010-03-24
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

//////////teprosulvose, radiosensitizer (veterinary use), WV7377RGM8, SQAP

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










