Home » Posts tagged 'veonetinib'
Tag Archives: veonetinib
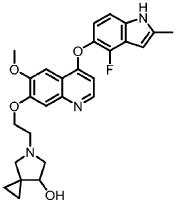
Veonetinib
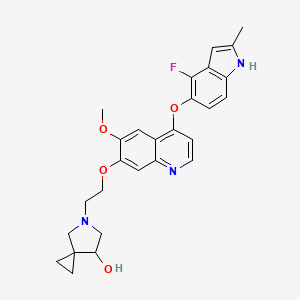
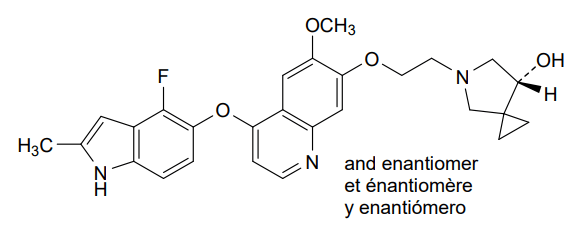
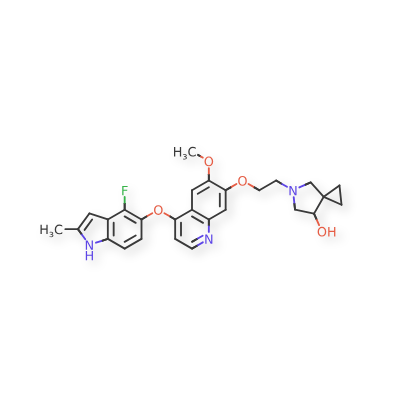
Veonetinib
👉CAS 1210828-09-3
MF C27H28FN3O4 MW 477.5 g/mol
5-[2-[4-[(4-fluoro-2-methyl-1H-indol-5-yl)oxy]-6-methoxyquinolin-7-yl]oxyethyl]-5-azaspiro[2.4]heptan-7-ol
5-AZASPIRO(2.4)HEPTAN-7-OL, 5-(2-((4-((4-FLUORO-2-METHYL-1H-INDOL-5-YL)OXY)-6-METHOXY-7-QUINOLINYL)OXY)ETHYL)-
5-(2-(4-(4-fluoro-2-methyl-1H-indol-5-yloxy)-6-methoxyquinolin-7-yloxy)ethyl)-5-azaspiro[2.4]-heptan-7-ol
(7RS)-5-[2-({4-[(4-fluoro-2-methyl-1H-indol-5-yl)oxy]-6-methoxyquinolin7-yl}oxy)ethyl]-5-azaspiro[2.4]heptan-7-ol
tyrosine kinase inhibitor, antineoplastic, U7PA8S6XGJ
👉SYN
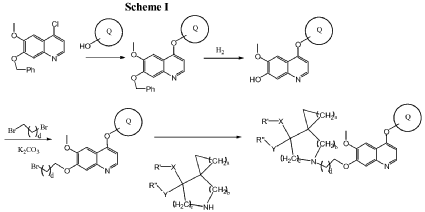
Example 3
5-(2-(4-(4-fluoro-2-methyl-lH-indol-5-yloxy)-6-methoxyquinolin-7-yloxy)ethyl)-5-azaspiro[2.4]-heptan-7-ol
The above product from Example 2 (75 mg) was dissolved into MeOH (8 ml) and stirred at RT.
NaBH4 (75 mg) was added to the reaction and stirred at RT for 30 min. The reaction was evaporated and purified by column chromatography to give title compound (60 mg). Mass: (M + 1), 478
Patent Scope
- Covers:
- Quinoline–indole kinase inhibitors
- VEGFR / angiogenesis targets
- Broad Markush structures
Fragment A: Indole Phenol
4-fluoro-2-methyl-1H-indol-5-ol
Typical Preparation
- Fischer indole synthesis or substituted aniline cyclization
- Fluorination / directed substitution
- Hydroxyl introduction at C-5
Fragment B: Quinoline Electrophile
4-chloro-6-methoxy-7-(leaving group) quinoline
Typical Route
- Start from 6-methoxyaniline
- Skraup / Doebner–Miller → quinoline core
- Chlorination at C-4
- Functionalization at C-7 (OH or halide)
Fragment C: Chiral Spiro Amine
(R)-5-azaspiro[2.4]heptan-7-ol
- Usually from:
- Chiral pool OR
- Resolution of racemate
- Important: defines final stereochemistry
4. STEP-BY-STEP SYNTHESIS (PATENT-ALIGNED)
Step 1: Indole–Quinoline Ether Formation
Reaction: SNAr / Ullmann-type coupling
Indole phenol + 4-chloroquinoline → aryl ether
Conditions
- Base: K2CO3 / Cs2CO3
- Solvent: DMF / DMSO
- Temp: 80–120°C
Forms:
Indole–O–quinoline core
Step 2: Introduction of Linker (C-7 substitution)
If quinoline has OH:
Quinoline–OH + Br–CH2–CH2–X → O–CH2CH2–X
If halide:Direct alkylation
Conditions
- Base: NaH / K2CO3
- Solvent: DMF
- Temp: 50–90°C
Product:
Quinoline–O–CH2CH2–X
Step 3: Coupling with Spiro Amine
Quinoline–O–CH2CH2–X + spiro amine → final amine linkage
Reaction Type
- SN2 substitution
Conditions
- Base: DIPEA / Et3N
- Solvent: ACN / DMF
- Temp: 50–80°C
Step 4: Final Deprotection / Purification
- Remove protecting groups (if any)
- Chiral purity control
- Crystallization
Step 1: Preparation of Indole–Quinoline Ether
Starting materials:
- 4-fluoro-2-methyl-1H-indol-5-ol
→ 1.00 equiv (e.g., 5.0 g, ~30 mmol) - 4-chloro-6-methoxyquinoline
→ 1.10 equiv (~33 mmol)
Reagents:
- Potassium carbonate (K₂CO₃) → 2.0 equiv (~60 mmol)
- Solvent: DMF (50–60 mL)
Procedure:
- Charge indole phenol and K₂CO₃ in DMF under nitrogen.
- Add 4-chloroquinoline portionwise.
- Heat to 100–110°C.
- Stir for 8–12 h.
Workup:
- Cool to RT
- Pour into water (200 mL)
- Extract with EtOAc (3×)
- Wash with brine, dry (Na₂SO₄)
- Concentrate
Purification:
- Silica gel chromatography (EtOAc/hexane)
Yield: ~70–80%
Product: Indole–quinoline ether intermediate
Step 2: Installation of Ethylene Linker
Starting material: Step 1 product (~25 mmol)
Reagents:
- 1,2-dibromoethane → 1.5–2.0 equiv
- Base: K₂CO₃ → 2 equiv
- Solvent: DMF (40 mL)
Procedure:
- Dissolve intermediate in DMF
- Add K₂CO₃
- Add dibromoethane
- Heat to 80–90°C for 6–8 h
Workup:
- Pour into water
- Extract with EtOAc
- Dry and concentrate
Product: Quinoline–O–CH₂CH₂–Br
Yield: ~65–75%
Step 3: Coupling with Chiral Spiro Amine
Starting materials:
- Bromo intermediate → 1.0 equiv (~15–20 mmol)
- (R)-5-azaspiro[2.4]heptan-7-ol → 1.2 equiv
Reagents:
- DIPEA or Et₃N → 2 equiv
- Solvent: Acetonitrile or DMF (30–40 mL)
Procedure:
- Combine bromo intermediate and amine in solvent
- Add DIPEA
- Heat to 60–70°C
- Stir 12–16 h
Workup:
- Remove solvent
- Dissolve in EtOAc
- Wash with water + brine
- Dry and concentrate
Yield: ~70–85%
Step 4: Final Purification
Purification options:
- Silica chromatography OR
- Recrystallization (EtOAc/hexane or IPA)
Optional:
- Convert to pharmaceutically acceptable salt
Final Yield (overall): ~35–45%
PAT
EXAMPLE 1
4-(4-Fluoro-2-methyl-1H-indol-5-yloxy)-6-methoxy-7-[2-(5,8-Dioxa-10-azadispiro[2.0.4.3]-undecane)ethoxy]quinoline
Preparation of 4-(4-fluoro-2-methyl-1H-indol-5-yloxy)-6-methoxy-7-benzyloxyquinoline
Method A:
Method B:
Preparation of Title Compound
Method C:
Method D:
EXAMPLE 2
5-(2-(4-(4-Fluoro-2-methyl-1H-indol-5-yloxy)-6-methoxyquinolin-7-yloxy)ethyl)-5-azaspiro[2.4]-heptan-7-one
EXAMPLE 3
5-(2-(4-(4-fluoro-2-methyl-1H-indol-5-yloxy)-6-methoxyquinolin-7-yloxy)ethyl)-5-azaspiro[2.4]-heptan-7-ol
PAT
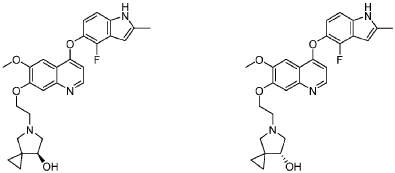
PAT
CN201710900497.6 CN201610649732.2 → leads to US10689361B2
PAT
- Compounds as kinase inhibitorsPublication Number: CA-2733250-CPriority Date: 2008-08-19Grant Date: 2016-06-21
- Compounds that act as kinase inhibitorsPublication Number: ES-2617678-T3Priority Date: 2008-08-19Grant Date: 2017-06-19
- Compounds as kinase inhibitorsPublication Number: WO-2010021918-A1Priority Date: 2008-08-19
- Compounds as kinase inhibitorsPublication Number: EP-2312950-A1Priority Date: 2008-08-19
- Compounds as kinase inhibitorsPublication Number: US-8211911-B2Priority Date: 2008-08-19Grant Date: 2012-07-03
- Compounds as kinase inhibitorsPublication Number: JP-2012500269-APriority Date: 2008-08-19
- Compounds as kinase inhibitorsPublication Number: KR-20110044749-APriority Date: 2008-08-19
- Compounds as kinase inhibitorsPublication Number: EP-2312950-B1Priority Date: 2008-08-19Grant Date: 2016-11-30
- Biological activities of 5-(2-(4-(4-fluoro-2-methyl-1h-indol-5-yloxy)-6-methoxyquinolin-7-yloxy)ethyl)-5-azaspiro[2.4]-heptan-7-ol crystalline, phosphoric acid salt and its enantiomersPublication Number: US-2023399313-A1Priority Date: 2022-06-10
- Btological activities of 5-(2-(4-(4-fluoro-2-methyl-1h-indol-5- yloxy)-6-methoxyquinolin-7-yloxy)ethyl)-5-azaspiro[2.4]-heptan- 7-ol crystalline, phosphoric acid salt and its enantiomersPublication Number: WO-2023239945-A1Priority Date: 2022-06-10
- Btological activities of 5-(2-(4-(4-fluoro-2-methyl-1h-indol-5- yloxy)-6-methoxyquinolin-7-yloxy)ethyl)-5-azaspiro[2.4]-heptan- 7-ol crystalline, phosphoric acid salt and its enantiomersPublication Number: EP-4536652-A1Priority Date: 2022-06-10
- Compounds as kinase inhibitorsPublication Number: CA-2733250-A1Priority Date: 2008-08-19
- Compounds As Kinase InhibitorsPublication Number: US-2010048599-A1Priority Date: 2008-08-19
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

REF
//////////veonetinib, ANAX LAB, tyrosine kinase inhibitor, antineoplastic, U7PA8S6XGJ

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










