Home » Posts tagged 'RP10584'
Tag Archives: RP10584
Methiomeprazine
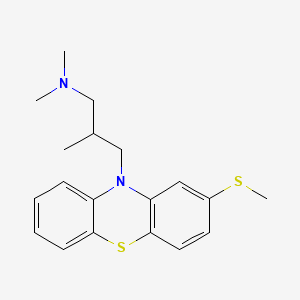
Methiomeprazine
N,N,2-trimethyl-3-(2-methylsulfanylphenothiazin-10-yl)propan-1-amine
CAS 7009-43-0
Molecular Formula, C19-H24-N2-S2, Molecular Weight, 344.5446,
- 10H-Phenothiazine-10-propanamine, N,N,β-trimethyl-2-(methylthio)-, (±)-
- Phenothiazine, 10-[3-(dimethylamino)-2-methylpropyl]-2-(methylthio)-, (±)- (8CI)
- N,N,β-Trimethyl-2-(methylthio)-10H-phenothiazine-10-propanamine
- (±)-10-(3-Dimethylamino-2-methylpropyl)-2-(methylthio)phenothiazine
- 10584-RP
- 2-Methylthio-10-(2-methyl-3-dimethylaminopropyl)phenothiazine
- Methiomeprazine
- SKF 6270
- (+-)-10-(3-Dimethylamino-2-methylpropyl)-2-(methylthio)phenothiazine
- Phenothiazine, 10-(3-(dimethylamino)-2-methylpropyl)-2-(methylthio)-, (+-)-
- 10584 RP
- EINECS 230-285-9
- Methiomeprazinum
- Methiomeprazinum [INN-Latin]
- Metiomeprazina
- Metiomeprazina [INN-Spanish]
- RP 10584
- SKF 6270
- UNII-X2R9QTF0OL
Methiomeprazine hydrochloride
14056-64-5

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
///////////////////////////////////////////////////////////////////////////////////////////////////// Methiomeprazine is an antiemetic drug.
PATENTFR 2705 M 19640831.The title compd. and its derivs. are prepd. and can be used in the prepn. of antiemetic compns. A soln. of 2.280 g. 3-methylthio-10-(3-dimethylamino-2-methylpropyl)phenothiazine (I) in 12 l. EtOH is heated to 70° and added to a soln. (60°) of 969 g. d-tartaric acid in 27 l. EtOH, the soln. kept overnight and filtered, and the mother liquors from the 1st and 2nd crystns. combined and evapd. The residue (2.352 g.) is dissolved in H2O, the soln. made alk. with 700 ml. NaOH (d. 1.33) and extd. with 4 l. CH2Cl2, the org. phase sepd., the aq. phase extd. with 1 l. CH2Cl2, and the exts. combined and evapd. at ∼20 mm. The residue (1.183 g.) is taken up in 7 l. EtOH at 60°, the soln. added to 370 g. maleic acid in 1.7 l. EtOH (60°), and the mixt. kept overnight to give 1.192 g. I acid maleate (II), m. 176-7° (EtOH), [α]24D -21.2° ± 1.5° (c 2, CHCl3). II (300 g.) is added to a mixt. of 1 l. H2O and 2 l. CH2Cl2, 150 ml. NaOH (d. 1.33) added, and the org. phase sepd. and distd. to give 185 g. (-)-3-methylthio-10-(3-dimethylamino-2-methylpropyl)phenothiazine (III), m. 84-5° (iso-PrOH), [α]23D -45° ± 3° (c 2.5, C6H6).
PAPERJournal of Organic Chemistry (1960), 25, 944-7.https://pubs.acs.org/doi/abs/10.1021/jo01076a019cf. CA 54, 15391b. The prepn. of various 10-aminoalkyl derivs. of the following phenothiazines was described: 2-hydroxyphenothiazine (I), 2-methylthiophenothiazine (II), 2-methylsulfonylphenothiazine (III), 2-trifluoromethylsulfonylphenothiazine (IV), 2-trifluoromethylthiophenothiazine (V), 2-azaphenothiazine (VI), and 8-chloro-2-azaphenothiazine (VII). The direct alkylation of I was not attempted. Instead, 2-benzoyloxyphenothiazine was alkylated with NaNH2 in xylene and the ester group removed by basic hydrolysis during the workup. The alkylation of IV with 3-(4-methylpiperazinyl)propyl chloride required 48 hrs. VI (15 g.), 6.8 g. NaNH2, and 500 ml. PhMe refluxed 45 min. under N, treated with 21 g. 3-chloro-1-(1-formyl-4-piperazinyl)propane-HCl and 300 ml. PhMe, the mixt. cooled, 150 ml. H2O added, the PhMe layer extd. with dil. HCl, the acid exts. made alk., extd. with C6H6, and the solvent evapd. gave 21 g. oil. The oil dissolved in 250 ml. alc., 60 ml. H2O and 7 ml. 40% NaOH, the mixt. refluxed 2 hrs., the solvents removed, the residual oil dissolved in C6H6, the soln. extd. with HCl, made alk., extd. with C6H6, and the whole distd. gave 11 g. 10-[3-(1-piperazinyl)propyl]-2-azaphenothiazine. The distd. material was dissolved in 250 ml. MeOH and refluxed 1.5 hrs. with 1.8 g. ethylene oxide, the solvent evapd., the residue dissolved in 250 ml. C6H6, the soln. azeotropically distd. during 1 hr., cooled, and refluxed 1 hr. with 6.5 g. AcCl, the solvents evapd., the gum treated with 10% NaOH, and the C6H6 evapd. gave 4.3 g. 4-[3-(2-azaphenothiazin-10-yl)propyl]-1-piperazineëthanol; acetate dimaleate m. 147-8° (decompn.) (EtOAc). 1-Piperazinepropanol (57.6 g.) refluxed 1 hr. with 48 g. HCO2Me, the excess HCO2Me removed, and the residue distd. gave 65.3 g. oil, b1.1 174.5-7.0°, n24D 1.5072. This oil (42.8 g.) in 300 cc. CHCl3 treated with excess HCl, then 19 g. SOCl2, the mixt. refluxed 0.5 hr., 3 g. SOCl2 added, refluxing continued 2.5 hrs., and the solvents removed gave a cryst. HCl salt. Conversion of this to the free base gave 60% 1-formyl-4-(3-chloropropyl)piperazine, yellow oil, b0.4 144.5-8.5°, n25D 1.5053. By starting with I-VII the following 2,10-disubstituted phenothiazines were obtained (substituents at 2, 10, b.p./mm., and % yield given); SMe, (CH2)3NMe2, 220-3°/0.7 (HCl salt m. 149-50°), 88; SMe, CH2CHMeCH2NMe2, 218-21°/0.1 (HCl salt m. 173-4°), 93; SMe, (CH2)3N.(CH2)2.NMe.CH2.CH2, 239-42°/0.1 (di-HCl salt m. 224-5°), 92; SMe, CH2CHMeCH2N.(CH2)2. NMe.CH2.CH2, 200-20°/0.03 (dimaleate m. 174-5°), 44; SMe, (CH2)3N.(CH2)2.N[(CH2)2OAc].CH2.CH2 – (dimaleate m. 165-6°), 33; SO2Me, (CH2)3NMe2, 115-16° (HCl salt m. 112-15°), 62; SO2Me, CH2CHMeCH2NMe2, 255-60°/0.2 (HCl salt m. 234-5°), 60; SCF3, (CH2)3NMe2, 153-7°/0.1, 64; SCF3, CH2CHMeCH2NMe2, 153-7°/0.1 (picrate m. 158.5-9.5°), 54; SCF3, I (CH2)3N.(CH2)2.NMe.CH2.CH2, 220-3°/0.3 (dimaleate m. 182-3°), 63; SO2CF3, (CH2)3NMe2, 235-40°/0.04 (HCl salt m. 174-5°), 15; SO2CF3, CH2CHMeCH2NMe2, 182-4°/0.2 (picrate m. 203-4°), 19; SO2CF3, (CH2)3N.(CH2)2.NMe.CH2.CH2, – [di-HCl salt m. 249.5° (decompn.)], 16; OH, (CH2)3NMe2, 220-5°/0.05, m. 90-1° (dimaleate m. 132-3°), 49. The following 8,10-substituted 2-azaphenothiazines were similarly prepd. (8,10 substituents, m.p. or b.p., % yield given): H, (CH2)3NMe2, 165-70°/0.007 [di-HCl salt m. 240.5-4.5° (decompn.)], 63; H, CH2CHMeCH2NMe2, 190-5°/0.6 (di-HCl salt m. 234-5°), 82; H, (CH2)3N.(CH2)2.N[(CH2)2OAc].CH2.CH2, – (dimaleate m. 147-8° (decompn.), 9; Cl, (CH2)3NMe2, 215-20°/1 (di-HCl salt m. 249-50°), 66.
PATENTGB 802725N-Aminoalkyl derivs. of I, where the alkyl is a straight or branched 2-5 C atom chain and the amino may be mono- or dialkylated or may be substituted by a pyrrolidino, piperidino, morpholino, or 4-alkyl-1-piperazinyl group, are prepd. by condensing I with the appropriate halo amine or by decompg. a phenothiazine-10-carboxylate of the appropriate amino alcohol. I (4.9 g.) was heated in 50 cc. boiling anhyd. xylene with 0.88 g. sodamide 1 hr., 2.71 g. 3-dimethylamino-1-chloropropane added, the soln. boiled 6 hrs., treated with H2O, then with dil. HCl, made alk. with NaOH, extd. with ether, and the solvent was evapd. in vacuo to give 4.5 g. 3-methylthio-10-(3-dimethylaminopropyl)phenothiazine (III), b0.2 206-18°; III.2HCl m. 160° (acetone-ether); picrate m. 135° (acetone). 3-Methylthio-10-(3-dimethylamino-2-methylpropyl)phenothiazine, m. 88-9°, was prepd. from I and 3-dimethylamino-2-methyl-1-chloropropane; picrate m. 145° (EtOH). The following were similarly prepd.: 3-methylthio-10-[3-(4-methyl-1-piperazinyl)propyl]phenothiazine, b0.1 250-6° [dihydrochloride m. 220° (decompn.) (acetone-ether); dipicrate m. 252-3° (acetone-iso-PrOH); 3-methylthio-10 – (2 – dimethylaminopropyl)phenothiazine, b0.2 202-6° (hydrochloride m. 205-6°; picrate m. 190°); 3-methylthio-10- (3-pyrrolidinopropyl)phenothiazine, b0.9 261° (hydrochloride m. 161°). I was phosgenated in toluene in the presence of pyridine to the 3-methylthiophenothiazine-10-carbonyl chloride (IV), m. 125°; IV heated in toluene with 3-(4- methyl-1-piperazinyl)-2-methylpropanol gave 3-(4-methyl-1- piperazinyl)-2-methylpropyl 3-methylthiophenothiazine-10- carboxylate (V) (dihydrochloride m. 225°). A soln. of 13 7 g. V in 60 cc. ο-Cl2C6H4 was boiled for 5 hrs. till CO2 evolution ceased, the soln. cooled, 60 cc. ether added and the mixt. H2O-washed, extd. with 10% HCl, made alk. with NaOH, and extd. with ether. The ether soln. was dried over anhyd. Na2SO4 and distd. in vacuo to yield 11.25 g. crude base which gave, with an EtOH soln. of maleic acid, 12.7 g. 3-methylthio-10-[3-(4-methyl-1-piperazinyl)-2-methyl-propyl]phenothiazinecarboxylic acid dimaleate, m. 199°. 3-Methylthio-10- [2,3-bis(dimethylamino)propyl] phenothiazine neutral fumarate, m. 198°, was similarly obtained by decarboxylating 1,3-bis(dimethylamino)-2-propyl 3-methylthiophenothiazine-10-carboxylate and treating with fumaric acid. 3-Methylthio-10-(3-diethylaminopropyl)phenothiazine-HCl, m. 172°, was prepd. from 3-methylthio-10-[3-(p-toluenesulfonyloxy)propyl]phenothiazine (VI) and Et2NH; 3-methylthio-10-(3-methylaminopropyl)phenothiazine (H oxalate m. 186°), from VI and MeNH2. VI heated with excess NH3 in toluene gave 3-methylthio-10-(3-aminopropyl)phenothiazine (VII) (oxalate m. 198°). VII in dioxane was neutralized with N HCl and treated with 30% aq. HCHO and PtO2 to give III. These compds. are antiemetics and potentiators of general anasthetics or neuroleptics.
SYN

///////////Methiomeprazine , antiemetic, Metiomeprazina, RP 10584, RP-10584, RP10584, RP 10584, SKF 6270
Systematic name (3):
- 10-[3-(ジメチルアミノ)-2-メチルプロピル]-2-(メチルチオ)-10H-フェノチアジン
- N,N,β-トリメチル-2-(メチルチオ)-10H-フェノチアジン-10-プロパン-1-アミン
- N,N,β-トリメチル-2-メチルチオ-10H-フェノチアジン-10-プロパン-1-アミン
Other name (6):
- メチオメプラジン
- Methiomeprazine
- 10-[3-(Dimethylamino)-2-methylpropyl]-2-(methylthio)-10H-phenothiazine
- SKF-6270
- N,N,β-Trimethyl-2-(methylthio)-10H-phenothiazine-10-propan-1-amine
CSc1ccc2Sc3ccccc3N(CC(C)CN(C)C)c2c1

NEW DRUG APPROVALS
ONE TIME
$10.00

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










