
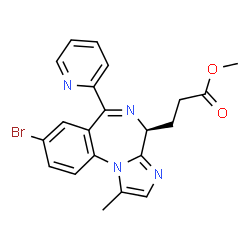
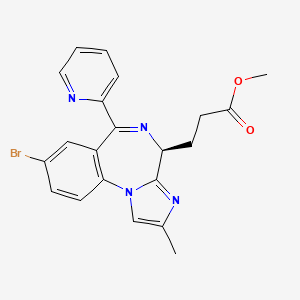

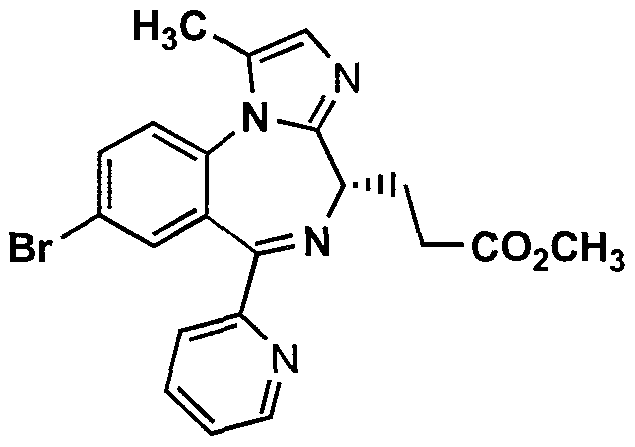
Remimazolam
- Molecular FormulaC21H19BrN4O2
- Average mass439.305 Da
3-[(4S)-8-bromo-1-methyl-6-(2-pyridyl)-4H-imidazo[1,2-a][1,4]benzodiazepin-4-yl]propionic acid methyl ester
CNS-7056
methyl 3-[(4S)-8-bromo-1-methyl-6-(2-pyridyl)-4H-imidazo[1,2-a][1,4]benzodiazepin-4-yl]propanoate
methyl 3-[(4S)-8-bromo-1-methyl-6-pyridin-2-yl-4H-imidazo[1,2-a][1,4]benzodiazepin-4-yl]propanoate
methyl 3-[(7S)-12-bromo-3-methyl-9-(pyridin-2-yl)-2,5,8-triazatricyclo[8.4.0.02,6]tetradeca-1(14),3,5,8,10,12-hexaen-7-yl]propanoate
MFCD18633229
UNII:7V4A8U16MB
CAS 308242-62-8 [RN]
PHASE 3, PAION, Anesthesia
Approved 2021/3/26 eu Byfavo
4H-Imidazo[1,2-a][1,4]benzodiazepine-4-propanoic acid, 8-bromo-1-methyl-6-(2-pyridinyl)-, methyl ester, (4S)-
7V4A8U16MB
9232
Methyl 3-[(4S)-8-bromo-1-methyl-6-(2-pyridinyl)-4H-imidazo[1,2-a][1,4]benzodiazepin-4-yl]propanoate
Methyl 3-[(4S)-8-bromo-l-methyl-6-(2-pyridinyl)-4H-imidazo[l,2- a] [ 1 ,4]benzodiazepin-4-yl]propanoate
methyl 3-[(4S)-8-bromo-2-methyl-6-pyridin-2-yl-4H-imidazo[1,2-a][1,4]benzodiazepin-4-yl]propanoate
D0L5KU; GTPL8442; SCHEMBL13862667; Short-acting sedatives, Therasci; CNS-7056B; CNS-7056X
- CNS 7056
- methyl 3-(8-bromo-1-methyl-6-(2-pyridinyl)-4H-imidazo(1,2-a)(1,4)benzodiazepin-4-yl)propanoate
- ONO 2745
- ONO-2745
- ONO2745
Remimazolam[1] (CNS-7056) is a benzodiazepine derivative drug, developed by PAION, in collaboration with Japanese licensee Ono Pharmaceutical as an alternative to the short-acting imidazobenzodiazepine midazolam, for use in induction of anaesthesia and conscious sedation for minor invasive procedures. Remimazolam was found to be both faster acting and shorter lasting than midazolam, and human clinical trials showed a faster recovery time and predictable, consistent pharmacokinetics, suggesting some advantages over existing drugs for these applications.[2][3]
Remimazolam (CNS-7056) is a water-soluble, rapid and short-acting GABA (A) benzodiazepine (BZ) site receptor agonist in phase III trials at PAION as procedural sedation in patients undergoing colonoscopy or diagnostic endoscopy of the upper gastrointestinal tract, and also with patients undergoing bronchoscopy.
PAION AG and its subsidiary PAION Inc, following its acquisition of CeNeS Pharmaceuticals (following CeNeS’ acquisition of TheraSci ), and licensees Mundipharma , Yichang Humanwell Pharmaceutical , Pendopharm , Cosmo and R-Pharm are developing remimazolam, the lead from a series of short-acting GABA A receptor agonists, as an iv sedative and/or anesthetic for potential use in day case surgical and non-surgical procedures

(Salt/Parent)
|
1
|
Remimazolam [INN]
308242-62-8 |

MW: 439.3111 |
|
2
|
Remimazolam besilate
1001415-66-2 |

MW: 597.4875 |
|
3
|
Remimazolam tosylate
1425904-79-5 |

MW: 611.5143 |
Trials
Phase I[4] and Ib[5] dose-finding studies for procedural sedation with patients recovering faster from remimazolam than midazolam. Phase II trials comparing remimazolam to the standard anesthesia protocols for cardiac surgery and colonoscopy were presented at major conferences in October 2014.[6]
A phase IIa trial comparing remimazolam to midazolam for upper endoscopy was published in December 2014, finding a similar safety profile.[7] Remimazolam was originally discovered in the late 1990s at Glaxo Wellcome in their labs in Research Triangle Park, NC.
BY CHENGDU
WO-2018103119
Novel crystalline forms of hydrobromate salt of remimazolam , processes for their preparation and compositions comprising them are claimed.
Remazolam, whose structure is shown in formula (I), has the chemical name 3-[(4S)-8-bromo-1-methyl-6-(2-pyridyl)-4H-imidazole [1,2] -a]

methyl [1,4]benzodiazepin- 4-yl]propanoate.
This compound is currently known as a CNS (Central Nervous System) inhibitor and has sedative, hypnotic, anxiolytic, muscle relaxing, and anticonvulsant effects. It is currently used intravenously in the following clinical treatment programs: preoperative sedation, anxiolysis and forgetfulness during surgery; awake sedation during short-term diagnosis, surgery, or endoscopic procedures; and administration of other anesthetics and analgesia Before and/or at the same time as a component for induction and maintenance of general anesthesia; ICU sedation and the like. It is reported in patent application CN101501019 that the free base stability of the compound is poor, and it is only suitable for storage at a low temperature of 5°C. Under conditions of 40°C/75% relative humidity (open), the sample is deliquescent and discolored, and the content is significantly reduced.
Due to the stability problem of the free base of the compound, researchers from various countries have studied the salts of the compound. For example, patent applications CN101501019B and WO2008/007081A1 respectively report the besylate and ethanesulfonate of the compound of formula (I). And shows that the above salts have good thermal stability, low hygroscopicity, and high water solubility, and that CN104968348A clearly states that the above benzenesulfonates and ethanesulfonates are the most preferred compounds of formula (I). Salts.
Immediately afterwards, CN 103221414B proposes a toxilate salt of a compound of formula (I) and indicates that the toxitic acid salt is less toxic than benzene sulphonate, and the thermal stability, water solubility and the like of certain crystal forms are even higher. For good.
To sort out the existing technology information, you can draw the following related content (Table 1):
From the above table, it can be seen that regardless of whether it is a free base of remazolam or a known salt derivative of remazolam, the water solubility is not higher than 11 mg/ml, and only in the slightly soluble range, which will increase The safety risk of its use in clinical use requires resolving and dissolving for a long time during clinical reconstitution. It may also leave insoluble materials, resulting in inaccurate drug dosage and potential safety risks. In addition, it is used for general anesthesia. Indications with a large demand will increase the amount of diluent and cause extreme inconvenience for clinical use. Therefore, the solubility of the known salt derivatives of remazolam is a big disadvantage and needs to be further improved.
The raw material remazolam of the compound of the formula (I) used in the present invention can be obtained by purchasing a commercially available product or can be prepared according to a known method (for example, patent US200,700,934,75A, etc.).
Example 1 Preparation of Form III Hydrobromide Salt of Compound of Formula (I)
Accurately weigh 1.8 g of the compound of formula (I) into a 100 mL three-necked flask, add 8.2 mL of isopropanol and stir to dissolve it completely, then dissolve 0.83 g of 47% aqueous hydrobromic acid in 6.3 mL of isopropanol and drip To the solution of the compound of formula (I) in isopropanol, the crystals were stirred, filtered, and dried at 55°C under reduced pressure to give the hydrobromide salt of the compound of formula (I).
The X-ray diffraction pattern of this crystal is shown in FIG. 1, the DSC and TGA patterns are shown in FIG. 2, and the melting point is 163 DEG C. It is defined that the crystal form is the hydrobromide III crystal form of the compound of Formula (I).
PATENT
WO0069836
Family members of remimazolam’s product case WO0069836 , have production in most of the EU states until May 2020 and expire in the US in April 2020.
PRODUCT PATENT
WO 2000069836
https://encrypted.google.com/patents/WO2000069836A1?cl=en
Example Ic-8
Methyl 3-[(4S)-8-bromo-l-methyl-6-(2-pyridinyl)-4H-imidazo[l,2- a] [ 1 ,4]benzodiazepin-4-yl]propanoate

A solution of the C7-bromo-benzodiazepine Ex 1-10 (7.31 g, 18.2 mmol) in THF (21 mL) was added to a suspension of NaH (870 mg of 60% oil dispersion, 21.8 mmol) in THF (70 mL) at 0 °C. The reaction mixture was stirred at 0 °C for 30 min, warmed to room temperature and stirred for 30 min, then cooled to 0 °C. Bis- morpholinophosphorochloridate (6.48 g, 25.5 mmol) was added, the mixture was allowed to warm to room temperature over 4.5 h, and the mixture was filtered with additional THF (ca. 10 mL). A mixture of the filtrate and DL-l-amino-2-propanol (2.80 mL, 36.4 mmol) was stirred at room temperature for 18 h and concentrated under reduced pressure. The residue was diluted with EtOAc (ca. 250 mL), washed with saturated aqueous NaHCO3 (1 x 75 mL), H2O (2 x 75 mL), saturated aqueous NaCl (1 x 75 mL), dried (Na SO ), and concentrated under reduced pressure. Purification by flash chromatography, elution with 19:1 EtOAc-MeOH, gave 3.06 g
(37%) of the adduct as a foam; ESIMS 459 (M+H, base).
A mixture of DMSO (1.88 mL, 26.6 mmol) and oxalyl chloride (1.16 mL, 13.3 mmol) in CH2C12 (40 mL) was stirred at -78 °C for 30 min. A solution of the alcohol prepared above (3.05 g, 6.64 mmol) in CH2C1 (26 mL) was added. The reaction mixture was warmed to -15 °C and stirred 1 h, cooled to -78 °C, treated with
E-3N (5.55 mL, 39.9 mmol), and allowed to warm to room temperature over 3 h. The mixture was diluted with EtOAc (ca. 500 mL), washed with saturated aqueous NaHCO3 (1 x 100 mL), H2O (1 x 100 mL), saturated aqueous NaCl (1 x 100 mL), dried (Na SO ), and concentrated under reduced pressure to give a foam. A mixture of this foam and a catalytic amount ofp-toluenesulfonic acid was stirred at room temperature for 18h, neutralized by the addition of saturated aqueous NaHCO3 and diluted with EtOAc (ca. 500 mL). The layers were separated and the organic phase was washed with saturated aqueous NaHCO3 (1 x 100 mL), H2O (2 x 100 mL), saturated aqueous NaCl (1 x 100 mL), dried (Na SO ), and concentrated under reduced pressure. Purification by flash chromatography, elution with 19: 1 EtOAc-
MeOH, gave 2.56 g (88%) of Ic-8 as a foam; 1H NMR (400 MHz, CDC13) δ 8.57 (d, J = 4.6 Hz, lH), 8.17 (d J = 7.8 Hz, IH), 7.79 (dd, J = 7.7, 6.2 Hz, IH), 7.71 (dd, J = 8.6, 2.2 Hz, IH), 7.64 (d, J – 2.2 Hz, IH), 7.34 (dd, J = 7.5, 5.0 Hz, IH), 7.30 (d, J = 8.6 Hz, IH), 6.86 (s, IH), 4.05 (m, 1 H), 3.67 (s, 3H), 2.80 (comp, 4H), 2.34 (s, 3H); ESIMS 461 (M+Na, base), 439 (M+H); Anal, calcd. for C2]H19BrN4O2-0.25 H2O: C,
58.63; H, 4.43; N, 12.62. Found: C, 56.88; H, 4.43; N, 12.23.
Example lc-8 was formulated in an aqueous vehicle at a concentration of 10 mg/ml. Accordingly, 10 mg of compound (and 9 mg NaCl) was dissolved in 0.63 ml of 0.1 N HCl. Slowly and while stirring, 0.37 ml of 0.1 N NaOH was added. Adjustments are made to the dose volume depending on the dose being administered.

PATENT
CN 103232454
https://patents.google.com/patent/CN103232454A/en
The compounds of the following formula I:
[0003]
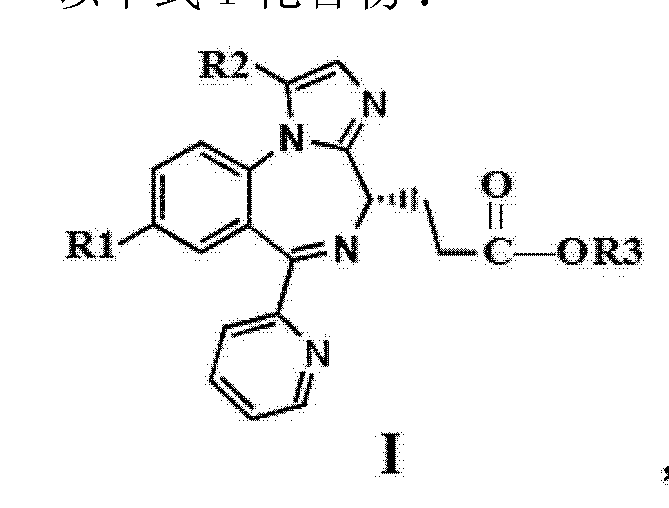
Wherein R1 is bromine, R2 and R3 is methyl, [0004] because it contains the specific configuration, W000 / 69836 reported in the compound (60 specification Example Ic-8) is a short-acting central nervous system (CNS) to suppress agents, including having a sedative-hypnotic, anxiolytic, muscle relaxant and anticonvulsant effect.They can be used for intravenous administration in the clinical treatment: preoperative sedation, such as during surgery, and forgetting anxiolytic purposes; in short diagnostic, operative or endoscopic conscious sedation during the procedure; administration of other anesthetics and analgesics before and / or simultaneously, as a component for the induction and maintenance of general anesthesia in; the ICU sedation, according CN101501019A (PA10N, application No. CN200780028964.5) reports, free base of the compound is not very stable, only suitable stored at low temperatures 5 ° C, at 40 ° C / 75% relative humidity (open) condition, the sample storage deliquescence, to the orange color turned yellow, with respect to the initial content and significantly reduced the content of the display. Thus the synthesis of salts of compounds of formula It (the I), hoping to increase the chemical stability thereof, for use in the preparation of medicaments.
[0005] existing CN101501019A and US20100075955A1 (TILBR00K) reported the benzenesulfonate salt of a compound of formula I, ethanesulfonate.CN102964349A (Henry, Application No. 201110456864.0) reported for compounds of formula ITosylate.
[0006] have reported the presence of a compound of formula I or a salt thereof concerns stability, which is disadvantageous for these compounds used in the clinical treatment of related diseases.
HPLC method [A]:
[0022] According to Chinese Pharmacopoeia 2010 Appendix VD High Performance Liquid Chromatography;
[0023] using Daicel Chrialcel OJ-H (5 μ m) 4.6 X 250mm using chiral chromatographic columns (guard column, if necessary Daicel Chrialcel OJ-H column analysis protected 5 μ m4.0 X IOmm, which is Japan Series Cat (Daicel ) brand), hexane: ethanol = 93: 7 (v / v) as the mobile phase, a flow rate of 1.0ml / min, column temperature 40 ° C, detection wavelength 225nm;
Bulk drug preparation of the present invention: [0204] Example 1
[0205] Preparation Example 4 taking the resulting compound of formula I lg, were added to 8ml of ethanol at 50 ° C – lactic acid – water (volume ratio of the three 45: 2: 53) mixed solution was stirred to dissolve; filtration, the filtrate was 5 ° C was allowed to stand at a temperature of 10~12 hours recrystallized, crystals were filtered off, 40 ° C and dried in vacuo; the above operation was repeated once, to give a compound of formula I may be formulated bulk drug used as a pharmaceutical formulation, was recrystallized twice yield rate of 86.1%.Chromatographic purity of product by HPLC 99.22% [B]; R & lt isomer impurity content of 0.39% relative peak area ratio (I / Ix) = 255 HPLC [Method A].
PATENT
EP 2305647
PATENT
WO 2011032692,
See also
References
- Jump up^ EP Patent 1183243
- Jump up^ Rogers WK, McDowell TS (December 2010). “Remimazolam, a short-acting GABA(A) receptor agonist for intravenous sedation and/or anesthesia in day-case surgical and non-surgical procedures”. IDrugs : the Investigational Drugs Journal. 13 (12): 929–37. PMID 21154153.
- Jump up^ Saari TI, Uusi-Oukari M, Ahonen J, Olkkola KT (March 2011). “Enhancement of GABAergic activity: neuropharmacological effects of benzodiazepines and therapeutic use in anesthesiology”. Pharmacological Reviews. 63 (1): 243–67. doi:10.1124/pr.110.002717. PMID 21245208.
- Jump up^ “A placebo- and midazolam-controlled phase I single ascending-dose study evaluating the safety, pharmacokinetics, and pharmacodynamics of remimazolam (CNS 7056): Part I. Safety, efficacy, and basic pharmacokinetics”. Anesth. Analg.accessdate =. 115: 274–83. Aug 2012. doi:10.1213/ANE.0b013e31823f0c28. PMID 22190555.
- Jump up^ “A phase Ib, dose-finding study of multiple doses of remimazolam (CNS 7056) in volunteers undergoing colonoscopy”. Anesth. Analg. 117: 1093–100. Nov 2013. doi:10.1213/ANE.0b013e3182a705ae. PMID 24108261.
- Jump up^ “Two Scientific Remimazolam Presentations Are Accepted for ASA and ACG Meeting in October 2014”. MarketWired. Oct 1, 2014. Retrieved 2014-10-24.
- Jump up^ “A Phase IIa, Randomized, Double-Blind Study of Remimazolam (CNS 7056) Versus Midazolam for Sedation in Upper Gastrointestinal Endoscopy”. Anesthesia. 120: 771–80. Dec 11, 2014. doi:10.1213/ANE.0000000000000548. PMID 25502841
PATENTS
Remimazolam
 |
| Identifiers |
|
|
| CAS Number |
|
| PubChem CID |
|
| ChemSpider |
|
| UNII |
|
| Chemical and physical data |
| Formula |
C21H19BrN4O2 |
| Molar mass |
439.304 g/mol |
| 3D model (JSmol) |
|
-
c4cccnc4C1=NC(CCC(=O)OC)c(ncc2C)n2-c(cc3)c1cc3Br
|
-
InChI=1S/C21H19BrN4O2/c1-13-12-24-21-17(7-9-19(27)28-2)25-20(16-5-3-4-10-23-16)15-11-14(22)6-8-18(15)26(13)21/h3-6,8,10-12,17H,7,9H2,1-2H3/t17-/m0/s1 
-
Key:CYHWMBVXXDIZNZ-KRWDZBQOSA-N 
|
//////////////CNS-7056 , CNS-7056X , ONO-2745 , CNS 7056 , CNS 7056X , ONO 2745, REMIMAZOLAM, PHASE 3, PHASE 3, PAION, Anesthesia, 308242-62-8
CC1=CN2C3=C(C=C(C=C3)Br)C(=NC(C2=N1)CCC(=O)OC)C4=CC=CC=N4

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
















