Home » Posts tagged 'PRAX-628'
Tag Archives: PRAX-628
Vormatrigine
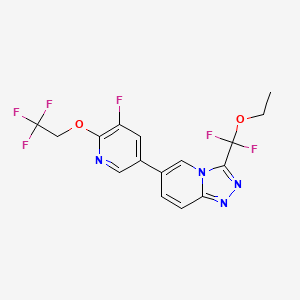
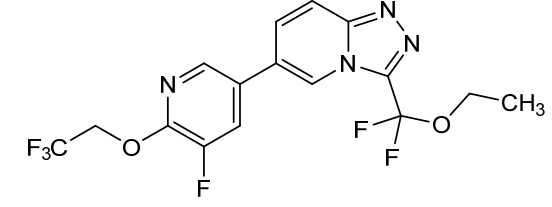
Vormatrigine
CAS 2392951-18-5
MF C16H12F6N4O2 MW406.28 g/mol
3-(ethoxydifluoromethyl)-6-(5-fluoro-6-(2,2,2-trifluoroethoxy)pyridin-3-yl)-[1,2,4]triazolo[4,3-a]pyridine
3-[ethoxydi(fluoro)methyl]-6-[5-fluoro-6-(2,2,2-trifluoroethoxy)pyridin-3-yl][1,2,4]triazolo[4,3-a]pyridine
sodium channel blocker, PRAX-628, PRAX 628, QU3C48T4NV,
Vormatrigine is a small molecule drug. The usage of the INN stem ‘-trigine’ in the name indicates that Vormatrigine is a sodium channel blocker, signal transduction modulator. Vormatrigine has a monoisotopic molecular weight of 406.09 Da.
Vormatrigine (formerly PRAX-628) is an oral, small-molecule, voltage-gated sodium channel inhibitor being developed by Praxis Precision Medicines for focal onset and generalized epilepsy. Phase II RADIANT trial data indicated that 22% of participants achieved complete seizure freedom, with a 56.3% median reduction in seizure frequency. IGMPI +2
Key Aspects of Vormatrigine:
- Mechanism: Targets the hyperexcitable state of NaV channels, acting as a functional state modulator.
- Efficacy: In the Phase II RADIANT trial (NCT06908356) for focal epilepsy, the drug showed significant seizure reduction (56.3% median reduction), with 60% of participants achieving a $\ge$50% reduction in seizures.
- Dosing: Developed for once-daily, oral administration without the need for complex titration.
- Safety Profile: Vormatrigine was generally well-tolerated in clinical studies, with mostly mild, transient adverse events reported, as noted in the Phase 1 PRAX-628-102 study.
- Future Development: Praxis is moving forward with Phase III trials (POWER2) following the successful Phase II results, with plans to potentially redefine treatment for patients with treatment-resistant focal epilepsy.
- Drug-Drug Interactions: Preliminary data suggests a favorable profile with minimal interaction risks, supporting its potential use in polytherapy.
Neurology Live +4
The drug is expected to be a major competitor in the epilepsy market if approved, with potential for high sales due to its efficacy profile compared to existing treatments. IGMPI
SYN
WO2019232209A1
- Assignee: Praxis Precision Medicines
- Title: Pyridin-3-yl substituted triazolopyrazines / triazolopyridines
- Year: 2019
- Covers:
- Vormatrigine structure (explicitly or as a close analog)
- General synthetic routes
- Multiple heterocycle construction strategies
This is the core patent used by all CRO/CDMO reverse synthesis work.Example ~178
Patent Landscape
The primary patent coverage for Vormatrigine and its related analogs is held by Praxis Precision Medicines, Inc. The chemical structure is a 1,2,4-triazolo[4,3-a]pyridine derivative.
- Primary Compound Patent:WO 2020/033839 (and its US equivalent US 11,447,489).
- Title: Preparation of pyridin-3-yl substituted triazolopyrazines, triazolopyridazines, and triazolopyridines as ion channel modulators.
- Scope: This patent covers the specific structure of Vormatrigine (Example 167 in some filings) and its use as a voltage-gated sodium channel (VGSC) inhibitor.
- Other Relevant Filings: * WO 2022/173918: Focuses on specific crystalline forms (polymorphs) and manufacturing improvements.
- CN 116444437 / CN 111087324: While these specific numbers often relate to broader triazolopyridine research (like Darolutamide, which you’ve researched previously), Praxis holds several Chinese counterparts for the PRAX-628 series.
SYN
SYN
Example 3: 3-[ethoxy(difluoro)methyl]-6-[5-fluoro-6-(2,2,2-trifluoroethoxy)-3-pyridyl]-[1,2,4]triazolo[4,3-a]pyridine
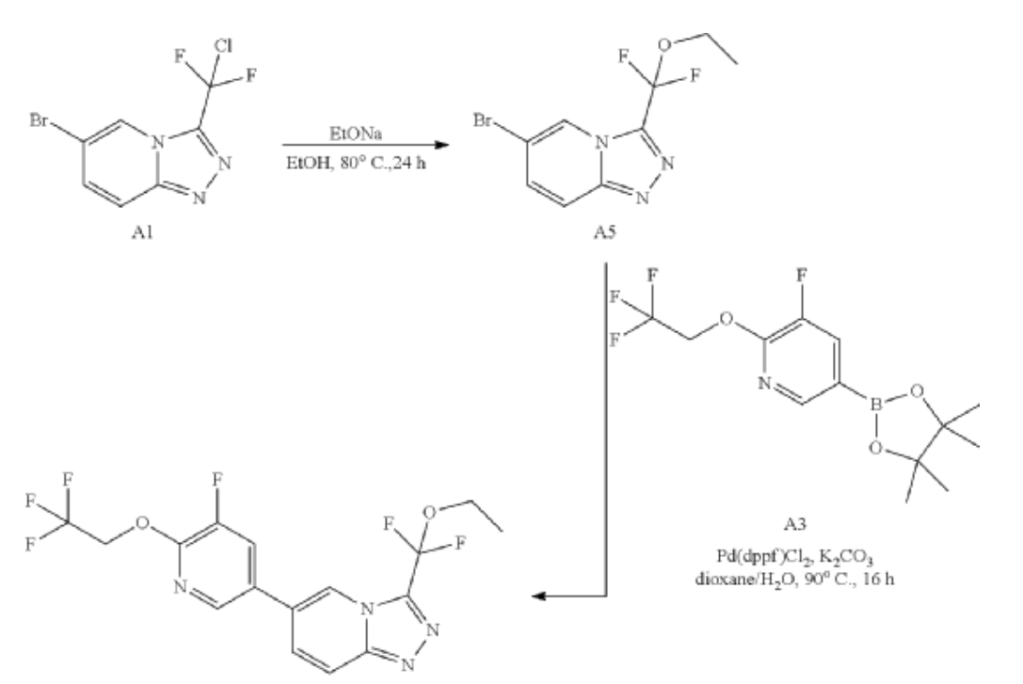
| Synthesis of A5: A mixture of 6-bromo-3-[chloro(difluoro)methyl]-[1,2,4]triazolo[4,3-a]pyridine (300 mg, 1.06 mmol) and EtONa (361.37 mg, 5.31 mmol) in Ethanol (10 mL) was stirred at 80° C. for 24 hours. After cooling to room temperature, the reaction was quenched with sat.NH 4Cl (10 mL), and the mixture was extracted with EtOAc (20 mL×2). The combined organic phase was washed with brine (10 mL), dried over Na 2SO 4, filtered and concentrated to give the crude product. The crude product was purified by flash chromatography on silica gel (EtOAc in PE=10% to 40%) to give the product (70 mg, 0.17 mmol) as a solid. LCMS R t=1.97 min in 4 min chromatography, MS ESI calcd. C 9H 9BrF 2N 3O [M+H+2] + 294.0, found 293.8. |
SYN
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

REF
PAT
- Ion channel modulatorsPublication Number: TW-202012403-APriority Date: 2018-05-30
- 3-(ethoxydifluoromethyl)-6-(5-fluoro-6-(2,2,2-trifluoroethoxy)pyridin-3-yl)-[1,2,4]triazolo[4,3-a]pyridine as an ion channel modulatorPublication Number: US-11731978-B2Priority Date: 2018-05-30Grant Date: 2023-08-22
- Ion channel modulatorsPublication Number: EP-3801535-A1Priority Date: 2018-05-30
- Ion channel modulatorsPublication Number: EP-4487916-A2Priority Date: 2018-05-30
- Ion channel modulatorsPublication Number: US-2022024930-A1Priority Date: 2018-05-30
- 3-(ethoxydifluoromethyl)-6-(5-fluoro-6-(2,2,2-trifluoroethoxy)pyridin-3-yl)-[1,2,4]triazolo[4,3-a]pyrazine as an ion channel modulatorPublication Number: US-11014931-B2Priority Date: 2018-05-30Grant Date: 2021-05-25
- Ion channel modulatorPublication Number: KR-20210024500-APriority Date: 2018-05-30
- Ion channel modulatorsPublication Number: US-2021163488-A1Priority Date: 2018-05-30
- Ion channel modulatorsPublication Number: US-2022220118-A1Priority Date: 2018-05-30
- 3-(ethoxydifluoromethyl)-6-(5-fluoro-6-(2,2,2-trifluoroethoxy)pyridin-3-yl)-[1,2,4]triazolo[4,3-a]pyridine as an ion channel modulatorPublication Number: US-11731976-B2Priority Date: 2018-05-30Grant Date: 2023-08-22
- Treatment of neurological disordersPublication Number: WO-2023211859-A1Priority Date: 2022-04-26
- Methods for the treatment of neurological disordersPublication Number: WO-2023211856-A1Priority Date: 2022-04-26
- Ion channel modulatorsPublication Number: US-11866439-B2Priority Date: 2018-05-30Grant Date: 2024-01-09
- Ion channel modulatorsPublication Number: US-2024132501-A1Priority Date: 2018-05-30
- Ion channel modulatorsPublication Number: US-2021087197-A1Priority Date: 2018-05-30
////////////vormatrigine, ANAX LAB, sodium channel blocker, PRAX-628, PRAX 628, QU3C48T4NV,

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










