Home » Posts tagged 'PLB 1004'
Tag Archives: PLB 1004
Andamertinib
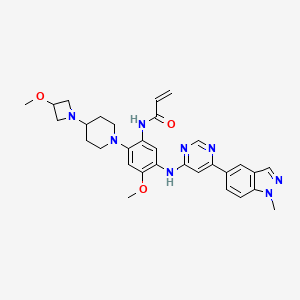
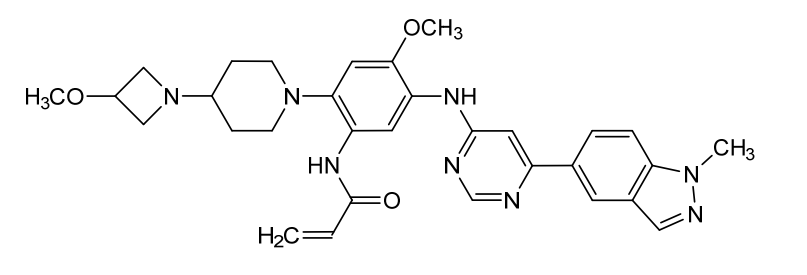
Andamertinib
CAS 2254145-43-0
MF C31H36N8O3 MW568.7 g/mol
N-[4-methoxy-2-[4-(3-methoxyazetidin-1-yl)piperidin-1-yl]-5-[[6-(1-methylindazol-5-yl)pyrimidin-4-yl]amino]phenyl]prop-2-enamide
N-(4-methoxy-2-[4-(3-methoxyazetidin-1-yl)piperidin-1-yl]-5-{[6-(1-methyl-1H-indazol-5-yl)pyrimidin-4-yl]amino}phenyl)prop-2-enamide
epidermal growth factor receptor tyrosine kinase inhibitor, antineoplastic, PLB 1004, 5X3KAG7ZBW
Andamertinib (also known as PLB1004) is an investigational, orally bioavailable, and irreversible small-molecule inhibitor of the epidermal growth factor receptor (EGFR). It is primarily being developed to treat non-small cell lung cancer (NSCLC) with specific genetic mutations.
Key Clinical & Therapeutic Features
- Target Mutations: It specifically targets EGFR exon 20 insertion (ex20ins) mutations, which are often resistant to standard first- and second-generation EGFR inhibitors.
- Broad Selectivity: Beyond ex20ins, it also shows activity against classical mutations like Del19, L858R, and the resistance mutation T790M.
- Brain Penetration: Andamertinib is designed to cross the blood-brain barrier, making it potentially effective for patients with brain metastases.
- Clinical Performance: In phase 2 studies (e.g., the KANNON study), it demonstrated a confirmed objective response rate (ORR) of 42.7% and a median progression-free survival of 6.2 months in pretreated patients.
Regulatory Status (as of Early 2026)
- China: A New Drug Application (NDA) was accepted by the National Medical Products Administration (NMPA) in May 2025 and granted priority review for treating NSCLC with EGFR ex20ins mutations.
- Global: It remains in various stages of clinical trials globally, including studies for first-line treatment and combination therapies with other agents like vebreltinib.
Andamertinib is an orally bioavailable, mono-anilino-pyrimidine, mutant-selective epidermal growth factor receptor (EGFR) inhibitor, with potential antineoplastic activity. Upon oral administration, andamertinib targets, binds to and irreversibly inhibits the activity of various EGFR mutations, including exon 20 insertion (Ex20ins) activating mutations, the gatekeeper mutation T790M, ExDel19, and L858R. This prevents EGFR-mediated signaling, induces cell death and inhibits tumor growth in tumor cells expressing these EGFR mutations. EGFR, a receptor tyrosine kinase mutated in many tumor cell types, plays a key role in tumor cell proliferation and tumor vascularization.
SYN

SYN
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Aminopyrimidine compound, preparation method therefor and use thereofPublication Number: US-11352352-B2Priority Date: 2017-06-13Grant Date: 2022-06-07
- Aminopyrimidine compound, preparation method therefor and use thereofPublication Number: EP-3640248-A1Priority Date: 2017-06-13
- Aminopyrimidine compound, preparation method therefor and use thereofPublication Number: US-2020087296-A1Priority Date: 2017-06-13
- Aminopyrimidine derivatives, preparation method therefor and use thereofPublication Number: EP-3640248-B1Priority Date: 2017-06-13Grant Date: 2023-08-23
//////////andamertinib, FLAX LAB, antineoplastic, PLB 1004, 5X3KAG7ZBW

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










