Home » Posts tagged 'pebezertinib'
Tag Archives: pebezertinib
Pebezertinib
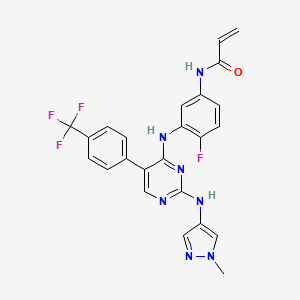
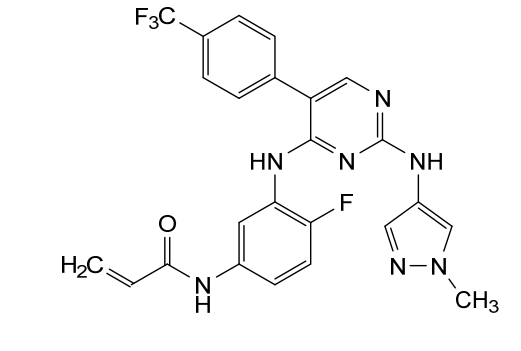
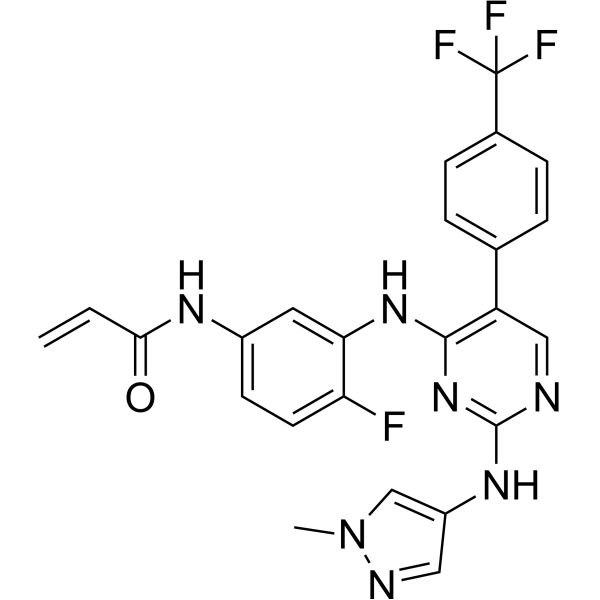
Pebezertinib
CAS 2769954-39-2
MF C24H19F4N7O MW 497.4 g/mol
N-[4-fluoro-3-[[2-[(1-methylpyrazol-4-yl)amino]-5-[4-(trifluoromethyl)phenyl]pyrimidin-4-yl]amino]phenyl]prop-2-enamide
N-[4-fluoro-3-({2-[(1-methyl-1H-pyrazol-4-yl)amino]-5-[4-(trifluoromethyl)phenyl]pyrimidin-4-yl}amino)phenyl]prop-2-enamide
epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor, antineoplastic, BLU 451, BLU 203139, G8G5AU5GLJ, LNG 451
Pebezertinib is a small molecule drug. The usage of the INN stem ‘-ertinib’ in the name indicates that Pebezertinib is a epidermal growth factor receptor (EGFR) inhibitor. Pebezertinib is under investigation in clinical trial NCT05241873 ((Concerto) Study of BLU-451 in Advanced Cancers With EGFR Exon 20 Insertion Mutations). Pebezertinib has a monoisotopic molecular weight of 497.16 Da.
Pebezertinib is an orally bioavailable, central nervous system (CNS) penetrating, mutant-selective covalent inhibitor of epidermal growth factor receptor (EGFR) exon 20 insertion (Ex20ins) activating mutations, with potential antineoplastic activity. Upon oral administration, pebezertinib selectively targets, irreversibly binds to and inhibits the activity of EGFR Ex20ins and some other oncogenic point mutations. This prevents EGFR Ex20ins-mediated signaling. This may induce cell death and inhibit tumor growth in EGFR Ex20ins-overexpressing tumor cells. EGFR, a receptor tyrosine kinase mutated in many tumors, plays a key role in tumor cell proliferation and tumor vascularization. Pebezertinib is able to penetrate the blood-brain-barrier (BBB) and may therefore exert its activity against EGFR Ex20ins-driven CNS primary tumors and CNS metastases. Pebezertinib does not inhibit the activity of wild-type (WT) EGFR. EGFR Ex20ins are oncogenic driver mutations that constitutively upregulate kinase activity.
(Concerto) Study of BLU-451 in Advanced Cancers With EGFR Exon 20 Insertion Mutations
CTID: NCT05241873
Phase: Phase 1
Status: Terminated
Date: 2025-02-10
Conditions: Lung Neoplasm Malignant; Carcinoma, Non-Small-Cell Lung; Respiratory Tract Neoplasms; Neoplasms; Neoplasms by Site; Lung Diseases; Respiratory Tract Disease; Carcinoma, Bronchogenic; Bronchial Neoplasms; Adenocarcinoma; Carcinoma; Neoplasms by Histologic Type; EGFR Exon 20 Mutation; EGFR Exon 20 Insertion Mutation; EGFR Activating Mutation; Antineoplastic Agents; Metastatic Lung Cancer; Brain Metastases; EGFR-mutated NSCLC; EGFR Atypical Mutations, Including G719X and L861Q
Interventions: Pemetrexed
Linked Compound CID: 426756; 135410875; 10339178; 163280903
SYN
Scheme 21: Synthesis of N-(4-fluoro-3-((2-((1-methyl-1H-pyrazol-4-yl)amino)-5-(4-(trifluoromethyl)phenyl)pyrimidin-4-yl)amino)phenyl)acrylamide (Compound 37):
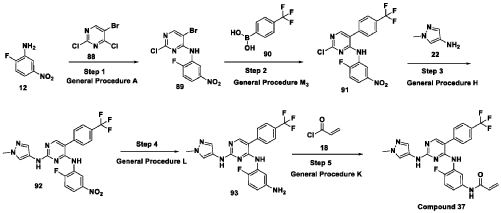
Step 1: Synthesis of 5-bromo-2-chloro-N-(2-fluoro-5-nitrophenyl)pyrimidin-4-amine (89):
[0286] Title compound was prepared in a manner substantially similar to procedure mentioned in General Procedure A. The crude was purified by combiflash eluted with 40% ethyl acetate in hexane to get (89) as pale yellow solid (1.3 g, Yield: 44.24 %). MS: [M+H]+ 346.97.
Step 2: Synthesis of 2-chloro-N-(2-fluoro-5-nitrophenyl)-5-(4-(trifluoromethyl)phenyl)pyrimidin-4-amine (91):
[0287] Title compound was prepared in a manner substantially similar to procedure mentioned in General Procedure M3. The crude was purified by combiflash eluted with 35% ethyl acetate in hexane to get desired product (91) as light yellow solid (700 mg; Yield: 50.12%). MS:
[M+H]+ 413.10
Step 3: Synthesis of N4-(2-fluoro-5-nitrophenyl)-N2-(1-methyl-1H-pyrazol-4-yl)-5-(4-(trifluoromethyl)phenyl)pyrimidine-2,4-diamine (92):
[0288] Title compound was prepared in a manner substantially similar to procedure mentioned in General Procedure H. The crude was purified by combiflash eluted with 1% methanol in dichloromethane to get desired product (92) as pale yellow solid (500 mg; Yield: 70.24%). MS:
[M+H]+ 474.09
Step 4: Synthesis of N4-(5-amino-2-fluorophenyl)-N2-(1-methyl-1H-pyrazol-4-yl)-5-(4-(trifluoromethyl)phenyl)pyrimidine-2,4-diamine (93):
[0289] Title compound was prepared in a manner substantially similar to procedure mentioned in General Procedure L to get (93) as semi solid (350 mg; Yield: 74.78%). MS: [M+H]+ 444.11
Step 5: Synthesis of N-(4-fluoro-3-((2-((1-methyl-1H-pyrazol-4-yl)amino)-5-(4-(trifluoromethyl)phenyl)pyrimidin-4-yl)amino)phenyl)acrylamide (Compound 37):
[0290] Title compound was prepared in a manner substantially similar to procedure mentioned in General Procedure K. The crude was purified by Prep HPLC to get Compound 37 as off white solid (30 mg, Yield: 13.33%).1H NMR (400 MHz, DMSO-d6): δ 10.21 (bs, 1H), 9.24 (bs, 1H), 8.53 (bs, 1H), 7.99 (s, 1H), 7.71-7.81 (m, 5H), 7.57 (s, 1H), 7.08-7.16 (m, 3H), 6.37-6.44 (m, 1H), 6.21-6.26 (m, 1H), 5.74 (d, J = 8.4 Hz, 1H), 3.54 (s, 3H). LCMS: [M+H]+ 498.35.
SYN
International Patent Application No. PCT/US2021/057472, the entire teachings of which are incorporated herein by reference, discloses selective inhibitors of EGFR, including exon 20 mutant proteins, which can be used to treat various cancers. The structure of one of the inhibitors disclosed in PCT Patent Application No. PCT/US2021/057472, referred to
herein as “Compound (I)” is shown below:
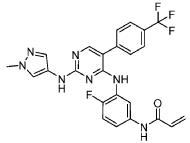
Example 1 : Preparation of Compound (I)
Synthesis of N-(4-fluoro-3-((2-((l-methyl-lH-pyrazol-4-yl)amino)-5-(4-(trifluoro methyl)phenyl)pyrimidin-4-yl)amino)phenyl)acrylamide (Compound I):
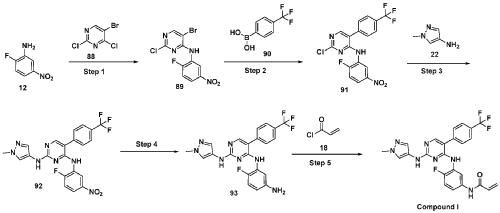
Step 1 : Synthesis of 5-bromo-2-chloro-N-(2-fluoro-5-nitrophenyl)pyrimidin-4-amine (89):
To an ice cold solution of 2-fluoro-5-nitroaniline (12) (1.0 eq) in tetrahydrofuran was added sodium hydride (60% dispersion in mineral oil, 3.0 eq) portion-wise. The resulting reaction mixture was stirred at room temperature for 30 minutes and followed by the addition of 2, 4-di chi oro-5 -bromopyrimidine (88) (1.0 eq). The resulting reaction mixture was heated at 60 °C for 16 hours. After completion (TLC monitoring), quenched with ice, extracted with ethyl acetate (3 times). The combined organic layers were washed with water, brine, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The crude was purified by combiflash eluted with 40% ethyl acetate in hexane to get (89) as pale yellow solid (1.3 g, Yield: 44.24 %). MS: [M+H]+ 346.97.
Step 2: Synthesis of 2-chloro-N-(2-fluoro-5-nitrophenyl)-5-(4-(trifluoromethyl)phenyl) pyrimidin-4-amine (91):
To a solution of halo derivative (89) (1.0 eq) and respective boronate acid/ester derivative (90) (1.1 eq) in A A i methyl form am ide: water (4: 1) was added sodium carbonate or sodium bicarbonate (2.0 eq). The resulting reaction mixture was degassed under argon atmosphere for 15 minutes, followed by addition of tetrakis(triphenylphosphine)palladium(0) (0.1 eq). The resulting reaction mixture was heated at 90 °C for 16 hours. After completion of reaction (TLC monitoring), the reaction mixture was cooled to room temperature, water was added and extracted with ethyl acetate (3 times). The combined organic layers were washed with brine, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The crude was purified by combiflash eluted with 35% ethyl acetate in hexane to get desired product (91) as light yellow solid (700 mg; Yield: 50.12%). MS: [M+H]+413.10.
Step 3 : Synthesis of N4-(2-fluoro-5-nitrophenyl)-N2-(l-methyl-lH-pyrazol-4-yl)-5-(4-(trifluoromethyl)phenyl)pyrimidine-2,4-diamine (92):
To an ice-cold solution of chloro compound (91) (1.0 eq) in isopropanol was added amine (22) (1.2 eq) and trifluoroacetic acid (2.0 eq). The reaction mixture was heated at 110 °C for 16 hours. After completion of the reaction (TLC monitoring), the reaction mixture was concentrated under reduced pressure, added saturated solution of sodium bicarbonate and extracted with dichloromethane (3 times). The combined organic layers were washed with brine solution, dried over anhydrous sodium sulfate and evaporated under reduced pressure. The crude was purified by combiflash eluted with 1% methanol in di chloromethane to get desired product (92) as pale yellow solid (500 mg; Yield: 70.24%). MS: [M+H]+ 474.09.
Step 4: Synthesis of N4-(5-amino-2-fluorophenyl)-N2-(l-methyl-lH-pyrazol-4-yl)-5-(4-(trifluoromethyl)phenyl)pyrimidine-2,4-diamine (93):
To an ice cold solution of nitro derivative (92) (1.0 eq) in methanol: tetrahydrofuran: water (2:2: 1) were added zinc-dust or iron powder (5 eq) and ammonium chloride (5 eq). The resultant reaction mixture was stirred at room temperature for 2 hours. After completion of reaction (TLC monitoring), reaction mixture passed through celite bed washed with 5% methanol in dichloromethane. The filtrate was washed with water, brine, dried over anhydrous sodium sulfate, filtered and concentrated to dryness to get the desired product (93) as semi solid (350 mg; Yield: 74.78%). MS: [M+H]+ 444.11.
Step 5 : Synthesis of N-(4-fluoro-3-((2-((l-methyl-lH-pyrazol-4-yl)amino)-5-(4-(trifluoromethyl)phenyl)pyrimidin-4-yl)amino)phenyl)acrylamide (Compound I):
To a solution of amino compound (93) (1.0 eq) in dichloromethane: tetrahydrofuran (1 :1) was cooled to -40 °C followed by triethylamine (3-5 eq) and acryloyl chloride (1.0 eq) were added. The mixture was stirred at the same temperature for 2 hours. After completion of reaction (monitored by TLC), added water and extracted with dichloromethane (3 times). The combined organic layers washed with brine, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The crudes were purified by Prep-HPLC purification to to obtain Compound I as off white solid (30 mg, Yield: 13.33%). ‘H NMR (400 MHz, DMSO-de): 8 10.21 (bs, 1H), 9.24 (bs, 1H), 8.53 (bs, 1H), 7.99 (s, 1H), 7.71-7.81 (m, 5H), 7.57 (s, 1H), 7.08-7.16 (m, 3H), 6.37-6.44 (m, 1H), 6.21-6.26 (m, 1H), 5.74 (d, J= 8.4 Hz, 1H), 3.54 (s, 3H). LCMS: [M+H]+ 498.35.
PAT
- Pyrimidine compounds, compositions, and medicinal applications thereofPublication Number: WO-2022094354-A1Priority Date: 2020-10-30Linked Compounds: 1,056Linked Substances: 1,365
- Pyrimidine compounds, compositions and pharmaceutical uses thereofPublication Number: CN-116685583-APriority Date: 2020-10-30Linked Compounds: 921Linked Substances: 1,108
- Pyrimidine compounds, compositions, and medicinal applications thereofPublication Number: TW-202233603-APriority Date: 2020-10-30Linked Compounds: 531Linked Substances: 575
- Pyrimidine compounds, compositions, and their medicinal applicationsPublication Number: KR-20230116795-APriority Date: 2020-10-30Linked Compounds: 699Linked Substances: 744
- Egfr inhibitors for treatment of cancerPublication Number: WO-2024097270-A1Patent Family: TW-202432143-A; WO-2024097270-A1Priority Date: 2022-11-01Inventor(s): ANKROM WENDY; MAR BRENTON; PANDEY ANJALI; PEARSON PAUL; ZALUTSKAYA ALENAAssignee(s): BLUEPRINT MEDICINES CORPClassification: A61K31/506; A61K31/519; A61K31/555; A61P35/00; A61P35/04Abstract: The present disclosure provides improved methods of treating non-small cell lung cancer characterized by EGFR mutation using Compound (I): or a pharmaceutically acceptable salt thereof.Linked Compounds: 27Linked Substances: 28
- Salt and crystal forms of an epidermal growth factor receptor inhibitorPublication Number: US-2025282761-A1Patent Family: AU-2023265064-A1; CN-119923392-A; EP-4519254-A1; IL-316663-A; JP-2025517634-A; KR-20250012078-A; MX-2024013485-A; TW-202409016-A; US-2025282761-A1; WO-2023215431-A1Priority Date: 2022-05-04Inventor(s): GRUFF ERIC; Kuang Shanming; PANDEY ANJALI; SHAH HARSH; XIE TIANAssignee(s): BLUEPRINT MEDICINES CORPClassification: A61K31/506; C07D403/12Abstract: Various salt forms and free base solid forms of Compound (I) represented by the following formula are disclosed. Pharmaceutical compositions comprising the same, methods of treating a disease associated with an epidermal growth factor receptor (EGFR) family kinase using the same, and methods for making the salt forms of Compound (I) and crystalline forms thereof are also disclosed.Linked Compounds: 11Linked Substances: 13
- Salt and crystal forms of an epidermal growth factor receptor inhibitorPublication Number: WO-2023215431-A1Priority Date: 2022-05-04Linked Compounds: 18Linked Substances: 22
- Salt and crystal forms of an epidermal growth factor receptor inhibitorPublication Number: EP-4519254-A1Priority Date: 2022-05-04Linked Compounds: 14Linked Substances: 16
- Pyrimidine compounds, compositions, and medicinal applications thereofPublication Number: EP-4237418-A1Priority Date: 2020-10-30Linked Compounds: 821Linked Substances: 918



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

[1].
Zhou Y, et al., Anti-PD-1/L1 antibody plus anti-VEGF antibody vs. more VEGFR-targeted TKI as first-line therapy for unresectable hepatocellular carcinoma: a network meta-analysis. Explor Target Antitumor Ther. 2024;5(3):568-580. [Content Brief]
//////////pebezertinib, antineoplastic, BLU 451, BLU 203139, G8G5AU5GLJ, LNG 451

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










