Home » Posts tagged 'Muscle Relaxant'
Tag Archives: Muscle Relaxant
Cyclobenzaprine


Cyclobenzaprine
- Molecular FormulaC20H21N
- Average mass275.387 Da
- MK-130
- TNX-102
1-(3-Dimethylaminopropylidene)-2,3:6,7-dibenzo-4-suberene
1-Propanamine, 3-(5H-dibenzo[a,d]cyclohepten-5-ylidene)-N,N-dimethyl-[ACD/Index Name]
206-145-8[EINECS]
3-(5H-Dibenzo[a,d]cyclohepten-5-ylidene)-N,N-dimethyl-1-propanamine
303-53-7[RN]
5-(3-Dimethylaminopropylidene)dibenzo[a,e]cycloheptatriene
циклобензаприн[Russian][INN]
سيكلوبنزابرين[Arabic][INN]
环苯扎林[Chinese][INN]
Cyclobenzaprine, CAS Registry Number: 303-53-7
CAS Name: 3-(5H-Dibenzo[a,d]cyclohepten-5-ylidene)-N,N-dimethyl-1-propanamine
Additional Names:N,N-dimethyl-5H-dibenzo[a,d]cyclohepten-D5,g-propylamine; 5-(3-dimethylaminopropylidene)dibenzo[a,e]cycloheptatriene; 1-(3-dimethylaminopropylidene)-2,3:6,7-dibenzo-4-suberene; proheptatriene
Manufacturers’ Codes: MK-130; Ro-4-1577; RP-9715
Molecular Formula: C20H21N, Molecular Weight: 275.39
Percent Composition: C 87.23%, H 7.69%, N 5.09%
Literature References: Prepn: GB858187 (1961 to Hoffmann-La Roche); Villani et al.,J. Med. Pharm. Chem.5, 373 (1962); Winthrop et al.,J. Org. Chem.27, 230 (1962). Pharmacology: C. D. Barnes, W. L. Adams, Neuropharmacology17, 445 (1978); N. N. Share, ibid. 721; and toxicology: J. Metysova et al.,Arch. Int. Pharmacodyn. Ther.144, 481 (1963). Metabolism: G. Belvedere et al.,Biomed. Mass Spectrom.1, 329 (1974); H. B. Hucker et al.,Drug Metab. Dispos.6, 184 (1978). Bioavailability: eidem,J. Clin. Pharmacol.17, 719 (1977). Clinical studies: J. V. Basmajian, Arch. Phys. Med. Rehabil.5, 58 (1978); B. R. Brown, J. Womble, J. Am. Med. Assoc.240, 1151 (1978). Comprehensive description: M. L. Cotton, G. R. B. Down, Anal. Profiles Drug Subs.17, 41-72 (1988).
Properties: bp1 175-180°. uv max: 224, 289 nm (log e 4.57, 4.02), (Villani et al.)
Boiling point: bp1 175-180°
Absorption maximum: uv max: 224, 289 nm (log e 4.57, 4.02), (Villani et al.)
Derivative Type: Hydrochloride
CAS Registry Number: 6202-23-9
Trademarks: Flexeril (Merck & Co.); Flexiban (Merck & Co.)
Molecular Formula: C20H21N.HCl, Molecular Weight: 311.85
Percent Composition: C 77.03%, H 7.11%, N 4.49%, Cl 11.37%
Literature References: Use as muscle relaxant: N. N. Share, FR2100873 (1972 to Frosst), C.A.78, 47801n (1973).
Properties: Crystals from isopropanol, mp 216-218°. Soly in water: >20 g/100 ml. Freely sol in water, methanol, ethanol; sparingly sol in isopropanol; slightly sol in chloroform, methylene chloride. Practically insol in hydrocarbons. uv max: 226, 295 nm (e 52300, 12000). LD50 in mice (mg/kg): 35 i.v., 250 orally (Metysova).
Melting point: mp 216-218°
Absorption maximum: uv max: 226, 295 nm (e 52300, 12000)
Toxicity data: LD50 in mice (mg/kg): 35 i.v., 250 orally (Metysova)
Therap-Cat: Muscle relaxant (skeletal).
Keywords: Muscle Relaxant (Skeletal).
Cyclobenzaprine, a centrally-acting muscle relaxant, was first synthesized in 196111 and has been available for human use since 1977.10 It was initially studied for use as antidepressant given its structural similarity to tricyclic antidepressants – it differs from Amitriptyline by only a single double bond.11,10 Since its approval, it has remained relatively popular as an adjunctive, short-term treatment for acute skeletal muscle spasms secondary to musculoskeletal injury.
Cyclobenzaprine (sold under the brand name Flexeril, among others) is a medication used for muscle spasms from musculoskeletal conditions of sudden onset.[6] It is not useful in cerebral palsy.[6] It is taken by mouth.[6] Use is not recommended for more than a few weeks.[6]
Common side effects include headache, feeling tired, dizziness, and dry mouth.[6] Serious side effects may include an irregular heartbeat.[6] There is no evidence of harm in pregnancy, but it has not been well studied in this population.[6] It should not be used with an MAO inhibitor.[6] How it works is unclear.[6]
Cyclobenzaprine was approved for medical use in the United States in 1977.[6] It is available as a generic medication.[6] In 2019, it was the 45th most commonly prescribed medication in the United States, with more than 15 million prescriptions.[7][8] It was not available in the United Kingdom as of 2012.[9]
Synthesis Reference
Villani, F.J.; US. Patent 3,409,640; November 5,1968; assigned to Schering Corporation.
Paper
By: Gowda, Narendra B.; Rao, Gopal Krishna; Ramakrishna, Ramesha A.
Tetrahedron Letters (2010), 51, (43), 5690-5693.
https://www.sciencedirect.com/science/article/abs/pii/S0040403910014668
A simple and convenient protocol for deoxygenation of aliphatic and aromatic N-oxides to the corresponding amines in good to excellent yield using sodium borohydride–Raney nickel in water is reported. Other functional moieties such as alkenes, halides, ethers, and amides are unaffected under the present reaction condition.
Graphical abstract

Cyclobenzaprine N-oxide, CAS RN: 6682-26-4
Dissolve (1 mmol) of cyclobenzaprine N-oxide in 2.5 mL of water at 60 °C. 2. Add Raney nickel (0.10 g, W6 grade) to the solution. 3. Stir the reaction mixture for 10 minutes. 4. Add (2 mmol) of sodium borohydride slowly in portions over 15-20 minutes to the reaction mixture. 5. Stir the reaction mixture at the same temperature for 2.5 hours (the completion of the reaction as monitored by TLC). 6. Once the reaction is completed, add chloroform (50 mL) to the reaction mixture. 7. Filter the resulted mixture to remove Raney nickel. 8. Dry the chloroform layer over anhydrous magnesium sulfate. 9. Filter the reaction mixture. 10. Evaporate the solvent under vacuum. 11. Purify the obtained residue through short path flash chromatography with silica gel and chloroform.
1H NMR (400 MHz, CDCl3) δ: 1.12 (s, 6H, N-CH3), 1.23- 1.34 (m, 4H, CH2), 4.58 (t, J= 4.0 Hz, 1H, CH), 5.82(d, J= 4.0 Hz, 2H, CH), 6.21- 6.33 (m, 8H, ArH).
13C NMR (100 MHz, CDCl3) δ: 27.89, 45.93, 60.12, 127.40, 127.55, 128.30, 128.59, 128.92, 129.33, 129.45, 129.67, 131.74, 131.96, 132.40, 134.63, 135.39, 137.97, 142.95, 143.30.
SYN
PATENT
https://patents.google.com/patent/WO2012098563A2/en
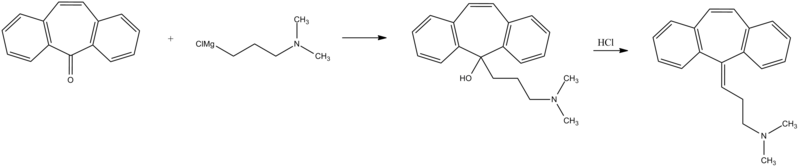
Cyclobenzaprine hydrochloride, chemically known as 5-(3-dimethylaminopropylidene)- dibenzo (a,e) cycloheptatriene hydrochloride (Formula I),

Formula I is a commonly prescribed tricyclic amine having muscle relaxant pharmaceutical activity. After sustaining an injury, muscle spasms may occur to stabilize the affected body part and prevent further damage. Cyclobenzaprine hydrochloride is used to treat such muscle spasm associated with acute, painful musculoskeletal conditions.
Few multistep processes for the preparation of this tricyclic amine are already available in the literature which involves isolation and purification of intermediate compounds. The conventional route of synthesis as reported in US3454643, ES8201950 includes preparation of Grignard reagent (GR) of 3-dimethylaminopropyl chloride in a first step, reacting with 5-dibenzosuberenone (Formulall) in a second step. The reaction mass was extracted with benzene, solid obtained was recrystallized from alcohol to produce 5- hydroxy intermediate (Formula III) and further dehydrated in third step using acetyl chloride or acetic anhydride in presence of chloroform as a solvent medium followed by purging HC1 gas to produce hydrochloride salt (Formula I). CH,
CI-(CH2)3 NS
CH,
Dimeth laminopropyl chloide

Di methy lam i nopropy I 5-dibenzosubrenone – y roxy compoun magnesium chloide
(Formula II) (Formula III)

Cyclobenzaprine base Cyclobenzaprine hydrochloride
(Formula IV) (Formula I)
The multistep synthesis is cumbersome and use of hazardous solvents and reagents like chloroform, benzene and acetyl chloride etc are not recommended for the preparation of pharmaceutical substances.
J. Org. Chem. Vol. 27, 230-240 (1961) also portrayed similar procedure for the synthesis of cyclobenzaprine hydrochloride, wherein 5-hydroxy compound of formula III was isolated and recrystallized before dehydration reaction.
Synthetic Comm. 11 (3), 241-246 (1981) described a process which involves isolation and purification of the intermediate at magnesium -complex stage. Hydrolysis of the isolated complex afforded desired tricyclic amine. GB858186 and GB858187 jointly described a process which comprises preparation of 5- hydroxy compound (Formula III) and subsequent conversion of the same to cyclobenzaprine hydrochloride. However the overall yield reported is significantly low.
In a different approach, a high temperature dehydrogenation of amitriptyline base resulting in formation of cyclobenzaprine hydrochloride is reported in Indian patent application 387/CHE/2005.

. EXAMPLE:
In a reaction vessel, THF (1 10ml), magnesium turnings 20gm (0.823mole) were charged and the mixture was warmed to 45-55°C for 20 min. A solution of l OOgm (0.823mole) of 3-dimethylaminopropyl chloride prepared in 1 10ml THF was added dropwise to the reaction mixture by controlling the reflux generated due to reaction initiation and maintained for 2hrs. The formed Grignard reagent was then cooled to 0-5°C and a solution of lOOgm (0.485mole) 5-dibenzosuberenone prepared in 220ml THF was charged to the reaction mass at temperature below 10°C. The reaction mass was stirred for 45 min at temperature 10-15°C. The absence of 5-dibenzosuberenone was checked by TLC and 770ml of 20% aq. HC1 was charged to the reaction mass at a temperature below 10°C. The reaction mass was then heated to 70-80°C for 3 hrs. The acidic mass was neutralized by using aqueous Na2C03 solution and extracted with 900ml methylene dichloride. The solvent was removed completely under reduced pressure and oil thus formed was dissolved in 450ml IPA and acidified with 240 ml of 20% IPA .HC1 solution and stirred for 2 hrs at 0-5°C for complete precipitation. The precipitate is filtered, recrystallized from IPA (800 ml) and dried to obtain 1 18 gm (78%) white crystalline cyclobenzaprine hydrochloride with purity 99.93% by HPLC.

PATENT
PATENT
CN 111393305
CLIP
Muscle Relaxants
R.S. Vardanyan, V.J. Hruby, in Synthesis of Essential Drugs, 2006
Cyclobenzaprine
Cyclobenzaprine, N,N-dimethyl-3-(dibenzo[a,d]cyclohepten-5-ylidene) propylamine (15.3.9), is synthesized by reacting 5H-dibenzo[a,d]cyclohepten-5-one with 3-dimethylaminopropylmagnesium chloride and subsequent dehydration of the resulting carbinol (15.3.8) in acidic conditions into cyclobenzaprine (15.3.9) [30–32].

Cyclobenzaprine is structurally similar to tricyclic antidepressants. It acts at the brain stem level. It is used as an adjuvant agent for relieving muscle spasms associated with severe diseased conditions of the muscle. A synonym of this drug is flexeril.
///////////////////////////////////////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
Medical use
Cyclobenzaprine is used, in conjunction with physical therapy, to treat muscle spasms that occur because of acute musculoskeletal conditions.[10] After sustaining an injury, muscle spasms to stabilize the affected body part occur, which may increase pain to prevent further damage. Cyclobenzaprine is used to treat such muscle spasms associated with acute, painful musculoskeletal conditions.[11] It decreases pain in the first two weeks,[12][13] peaking in the first few days, but has no proven benefit after two weeks.[12][14] Since no benefit is proven beyond that, therapy should not be continued long-term.[11] It is the best-studied muscle relaxer.[12] It is not useful for spasticity due to neurologic conditions such as cerebral palsy.[11][15]
A 2004 review found benefit for fibromyalgia symptoms, with a reported number needed to treat of 4.8 (meaning that 1 person out of every 4.8 benefits from treatment) for pain reduction, but no change in fatigue or tender points.[16] A 2009 Cochrane review found insufficient evidence to justify its use in myofascial pain syndrome.[17] It may also be used along with other treatments for tetanus.[18]
Side effects
Cyclobenzaprine results in increased rates of drowsiness (38%), dry mouth (24%), and dizziness (10%).[14] Drowsiness and dry mouth appear to intensify with increasing dose.[19] The sedative effects of cyclobenzaprine are likely due to its antagonistic effect on histamine, serotonin, and muscarinic receptors.[medical citation needed]
Agitation is a common side effect observed, especially in the elderly. Some experts[who?] believe that cyclobenzaprine should be avoided in elderly patients because it can cause confusion, delirium, and cognitive impairment.[20][21] In general, the National Committee for Quality Assurance recommends avoiding the use of cyclobenzaprine in the elderly because of the potential for more severe side effects.[22]
Dysphagia, a life-threatening side-effect, may rarely occur.[23] Treatment protocols and support should follow the same as for any structurally related tricyclic, such as tricyclic antidepressants.[24]
Overdose
The most common effects of overdose are drowsiness and tachycardia.[11] Rare but potentially critical complications are cardiac arrest, abnormal heart rhythms, severe low blood pressure, seizures, and neuroleptic malignant syndrome.[11] Life-threatening overdose is rare,[11] however, as the median lethal dose is about 338 milligrams/kilogram in mice and 425 mg/kg in rats.[11] The potential harm is increased when central nervous system depressants and antidepressants are also used; deliberate overdose often includes alcohol among other drugs.[11]
Interactions
Cyclobenzaprine has major contraindications with monoamine oxidase inhibitors (MAOIs). At least one study also found increased risk of serotonin syndrome when cyclobenzaprine was taken with the serotonergic drugs duloxetine or phenelzine.[25]
These substances may interact with cyclobenzaprine:
- Central nervous system depressants (e.g. alcohol, opioids, benzodiazepines, nonbenzodiazepines, phenothiazines, carbamates, barbiturates, major tranquilizers)
- Monoamine oxidase inhibitors taken within two weeks of cyclobenzaprine may result in serious, life-threatening side effects.[11]
Cyclobenzaprine may affect the medications used in surgical sedation and some surgeons request that patients temporarily discontinue its use prior to surgery.[26]
Pharmacology
Cyclobenzaprine is a centrally acting muscle relaxant.[27] Cyclobenzaprine is a 5-HT2 receptor antagonist; it relieves muscle spasm through action on the central nervous system at the brain stem, rather than targeting the peripheral nervous system or muscles themselves.[28]
Pharmacodynamics
| Site | CBP | NCBP | Action | Ref |
|---|---|---|---|---|
| 5-HT1A | 5.3 | 3.2 | Agonist | [29] |
| 5-HT2A | 5.2 | 13 | Antagonist | [29] |
| 5-HT2B | 100 | ??? | Antagonist | [29] |
| 5-HT2C | 5.2 | 43 | Antagonist | [29] |
| α1A | 5.6 | 34 | ND | [29] |
| α2A | 4.3 | 6.4 | Antagonist | [29] |
| α2B | 21 | 150 | ND | [29] |
| α2C | 21 | 48 | ND | [29] |
| H1 | 1.3 | 5.6 | ND | [29] |
| M1 | 7.9 | 30 | ND | [29] |
| Values are Ki (nM), unless otherwise noted. The smaller the value, the more strongly the drug binds to the site. |
Pharmacokinetics
Cyclobenzaprine has an oral bioavailability of about 55% and approximately 93% is bound to proteins in plasma. The half-life of the drug is 18 hours and it has a plasma clearance of 0.7 litres per minute.[27][30][31]
Comparison to other medications
Cyclobenzaprine has been found to be not inferior to tizanidine, orphenadrine, and carisoprodol in the treatment of acute lower back pain, although none have been proven to be effective for long-term use (beyond two weeks of treatment). No differences in pain or spasm scores were noted among these agents, nor when compared to benzodiazepines.[32] However, nonbenzodiazepine (including cyclobenzaprine) treatment was found to have a lower risk of medication abuse and continuation of use against medical advice.[medical citation needed] Side effects such as sedation and ataxia are also less pronounced with nonbenzodiazepine antispasmodics.[medical citation needed]
In a study on the treatment of musculoskeletal pain treatment with cyclobenzaprine alone or in combination with ibuprofen, no significant differences in pain scores were noted among the three treatment groups. Peak benefit was found to occur on day seven of the treatment for all groups.[33]
Formulations
Cyclobenzaprine 10mg tablets
By mouth, cyclobenzaprine is marketed as Apo-Cyclobenzaprin, Fexmid, Flexeril and Novo-Cycloprine. It is available in generic form. A once-a-day, extended-release formulation, Amrix, is available.[34] Cyclobenzaprine is also used by compounding pharmacies in topical creams.[citation needed]
References
- ^ Micromedex® 2010 – DRUGDEX Evaluations (Cyclobenzaprine Hydrochloride)
- ^ “Cyclobenzaprine Hydrochloride Tablets USP Revised: April 2005 Rx only”. nih.gov. Retrieved 1 October 2016.
- ^ Teva Pharmaceuticals USA, Inc (May 2016). “AMR40470 (Amrix) Prescribing Information” (PDF).
- ^ U.S. Food and Drug Administration. “NDA 17-821/S-045 Flexeril (Cyclobenzaprine HCl) Tablets” (PDF).
- ^ Teva Pharmaceuticals USA, Inc (May 2016). “AMR40470 (Amrix) Prescribing Information” (PDF).
- ^ Jump up to:a b c d e f g h i j k “Cyclobenzaprine Monograph for Professionals”. Drugs.com. AHFS. Retrieved 22 December 2018.
- ^ “The Top 300 of 2019”. ClinCalc. Retrieved 16 October 2021.
- ^ “Cyclobenzaprine – Drug Usage Statistics”. ClinCalc. Retrieved 16 October 2021.
- ^ “Fibromyalgia, psychiatric comorbidity, and the somatosensory cortex”. British Journal of Medical Practitioners. 5 (2): a522. 2012.
- ^ Yang YW, Macdonald JB, Nelson SA, Sekulic A (December 2017). “Treatment of vismodegib-associated muscle cramps with cyclobenzaprine: A retrospective review”. Journal of the American Academy of Dermatology. 77 (6): 1170–1172. doi:10.1016/j.jaad.2016.12.017. PMID 29132849. S2CID 8265576.
- ^ Jump up to:a b c d e f g h i “Cyclobenzaprine- cyclobenzaprine hydrochloride tablet, film coated”. DailyMed. 30 December 2019. Retrieved 26 September 2020.
- ^ Jump up to:a b c Chou R, Peterson K, Helfand M (August 2004). “Comparative efficacy and safety of skeletal muscle relaxants for spasticity and musculoskeletal conditions: a systematic review”. Journal of Pain and Symptom Management. 28 (2): 140–75. doi:10.1016/j.jpainsymman.2004.05.002. PMID 15276195.
- ^ van Tulder MW, Touray T, Furlan AD, Solway S, Bouter LM (2003). Van Tulder MW (ed.). “Muscle relaxants for non-specific low back pain”. The Cochrane Database of Systematic Reviews. 2 (2): CD004252. doi:10.1002/14651858.CD004252. PMC 6464310. PMID 12804507.
- ^ Jump up to:a b Browning R, Jackson JL, O’Malley PG (July 2001). “Cyclobenzaprine and back pain: a meta-analysis”. Archives of Internal Medicine. 161 (13): 1613–20. doi:10.1001/archinte.161.13.1613. PMID 11434793.
- ^ Ashby P, Burke D, Rao S, Jones RF (October 1972). “Assessment of cyclobenzaprine in the treatment of spasticity”. Journal of Neurology, Neurosurgery, and Psychiatry. 35 (5): 599–605. doi:10.1136/jnnp.35.5.599. PMC 494138. PMID 4563483.
- ^ Tofferi JK, Jackson JL, O’Malley PG (February 2004). “Treatment of fibromyalgia with cyclobenzaprine: A meta-analysis”. Arthritis and Rheumatism. 51 (1): 9–13. doi:10.1002/art.20076. PMID 14872449.
- ^ Leite FM, Atallah AN, El Dib R, Grossmann E, Januzzi E, Andriolo RB, da Silva EM (July 2009). “Cyclobenzaprine for the treatment of myofascial pain in adults”. The Cochrane Database of Systematic Reviews (3): CD006830. doi:10.1002/14651858.CD006830.pub3. PMC 6481902. PMID 19588406.
- ^ Smith BT (2014). Pharmacology for Nurses. Jones & Bartlett Publishers. p. 122. ISBN 9781449689407.
- ^ “Flexeril: Side effects”. RxList.com. Archived from the original on 12 September 2008. Retrieved 22 February 2010.
- ^ “Long-term Use of Cyclobenzaprine for Pain: A Review of the Clinical Effectiveness”. CADTH Rapid Response Reports. Ottawa, Ontario: Canadian Agency for Drugs and Technologies in Health. 23 February 2015. PMID 25763449.
- ^ Potentially inappropriate medications for the elderly according to the revised Beers criteria. 2012. Duke Clinical Research Institute website. [1]
- ^ “High risk medications” (PDF). National Committee for Quality Assurance. Archived from the original (PDF) on 1 February 2010. Retrieved 22 February 2010.
- ^ “MEDICATIONS AND DYSPHAGIA/ SWALLOWING RISKS” (PDF).
- ^ Chabria SB (July 2006). “Rhabdomyolysis: a manifestation of cyclobenzaprine toxicity”. Journal of Occupational Medicine and Toxicology. 1 (1): 16. doi:10.1186/1745-6673-1-16. PMC 1540431. PMID 16846511.
- ^ Keegan MT, Brown DR, Rabinstein AA (December 2006). “Serotonin syndrome from the interaction of cyclobenzaprine with other serotoninergic drugs”. Anesthesia and Analgesia. 103 (6): 1466–8. doi:10.1213/01.ane.0000247699.81580.eb. PMID 17122225.
- ^ Medical Practice of William H. Gorman, M.D. (18 February 2014). “Medications to Avoid, Continue, or Stop – Before & After Surgery”.
- ^ Jump up to:a b “Cyclobenzaprine”. http://www.drugbank.ca.
- ^ Kobayashi H, Hasegawa Y, Ono H (September 1996). “Cyclobenzaprine, a centrally acting muscle relaxant, acts on descending serotonergic systems”. European Journal of Pharmacology. 311 (1): 29–35. doi:10.1016/0014-2999(96)00402-5. PMID 8884233.
- ^ Jump up to:a b c d e f g h i j k “Cyclobenzaprine (CBP) and Its Major Metabolite Norcyclobenzaprine (nCBP) Are Potent Antagonists of Human Serotonin Receptor 2a (5HT2a), Histamine Receptor H-1 and á-Adrenergic Receptors: Mechanistic and Safety Implications for Treating Fibromyalgia Syndrome by Improving Sleep Quality”. ACR Meeting Abstracts. Retrieved 27 January 2022.
- ^ “Cyclobenzaprine”. pubchem.ncbi.nlm.nih.gov.
- ^ Winchell GA, King JD, Chavez-Eng CM, Constanzer ML, Korn SH (January 2002). “Cyclobenzaprine pharmacokinetics, including the effects of age, gender, and hepatic insufficiency”. Journal of Clinical Pharmacology. 42 (1): 61–9. doi:10.1177/0091270002042001007. PMID 11808825. S2CID 7749001.
- ^ “Medscape: Medscape Access”. medscape.com. Retrieved 1 October 2016.
- ^ Childers MK, Petri M, Laudadio C, Harrison D, Silber S, Bowen D (2004). “Comparison of cyclobenzaprine alone versus cyclobenzaprine plus ibuprofen in patients with acute musculoskeletal spasm and pain”. Annals of Emergency Medicine. 44 (4): S87–S88. doi:10.1016/j.annemergmed.2004.07.286.
- ^ “Patient Web site for Amrix (Cyclobenzaprine Hydrochloride Extended‐Release Capsules)”. amrix.com. Retrieved 1 October 2016.
External links
- “Cyclobenzaprine”. Drug Information Portal. U.S. National Library of Medicine.
| Clinical data | |
|---|---|
| Trade names | Flexeril, Amrix, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682514 |
| License data | US DailyMed: Cyclobenzaprine |
| Routes of administration | By mouth |
| ATC code | M03BX08 (WHO) |
| Legal status | |
| Legal status | US: ℞-onlyIn general: ℞ (Prescription only) |
| Pharmacokinetic data | |
| Bioavailability | 33–55%[1][2] |
| Protein binding | 93% |
| Metabolism | major: CYP3A4, CYP1A2; minor: CYP2D6, N-demethylation[5] |
| Metabolites | Norcyclobenzaprine |
| Elimination half-life | 32 hours (extended-release, range 8-37 hours),[3] 18 hours (immediate release, range 8–37 hours)[4] |
| Excretion | Kidney |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 303-53-7 |
| PubChem CID | 2895 |
| IUPHAR/BPS | 7152 |
| DrugBank | DB00924 |
| ChemSpider | 2792 |
| UNII | 69O5WQQ5TI |
| KEGG | D07758 |
| ChEBI | CHEBI:3996 |
| ChEMBL | ChEMBL669 |
| CompTox Dashboard (EPA) | DTXSID0046933 |
| ECHA InfoCard | 100.005.588 |
| Chemical and physical data | |
| Formula | C20H21N |
| Molar mass | 275.395 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
| (verify) |
///////////////cyclobenzaprine, циклобензаприн , سيكلوبنزابرين , 环苯扎林 , MK-130, TNX-102, Muscle Relaxant
CN(C)CCC=C1C2=CC=CC=C2C=CC2=CC=CC=C12

NEW DRUG APPROVALS
ONE TIME
$10.00
METHOCARBAMOL

Methocarbamol
- Molecular FormulaC11H15NO5
- Average mass241.240 Da
- метокарбамол , ميثوكاربامول , 美索巴莫
1,2-Propanediol, 3-(2-methoxyphenoxy)-, 1-carbamate
208-524-3[EINECS]
2-Hydroxy-3-(2-methoxyphenoxy)propyl carbamate
532-03-6[RN]
MethocarbamolCAS Registry Number: 532-03-6
CAS Name: 3-(2-Methoxyphenoxy)-1,2-propanediol 1-carbamate
Additional Names: 3-(o-methoxyphenoxy)-2-hydroxypropyl 1-carbamate; 2-hydroxy-3-(o-methoxyphenoxy)propyl 1-carbamate; guaiacol glyceryl ether carbamate
Manufacturers’ Codes: AHR-85Trademarks: Neuraxin; Miolaxene (Lepetit); Lumirelax; Etroflex; Delaxin (Ferndale); Robamol (Cenci); Traumacut (Brenner-Efeka); Tresortil; Relestrid; Robaxin (Robins)
Molecular Formula: C11H15NO5, Molecular Weight: 241.24Percent Composition: C 54.77%, H 6.27%, N 5.81%, O 33.16%
Literature References: Prepn from 3-(o-methoxyphenoxy)-2-hydroxypropyl chlorocarbonate: Murphey, US2770649 (1956 to A. H. Robins). Comprehensive description: S. Alessi-Severini et al.,Anal. Profiles Drug Subs. Excip.23, 371-399 (1994).
Properties: Crystals from benzene, mp 92-94°. uv max (water): 222, 274 nm (E1%1cm 298, 94). 1og P -0.06. Soly in water at 20°: 2.5 g/100 ml. Sol in alcohol, propylene glycol. Sparingly sol in chloroform. Practically insol in n-hexane.
Melting point: mp 92-94°
Absorption maximum: uv max (water): 222, 274 nm (E1%1cm 298, 94)
Therap-Cat: Muscle relaxant (skeletal).
Therap-Cat-Vet: Muscle relaxant (skeletal).
Keywords: Muscle Relaxant (Skeletal).
Methocarbamol, sold under the brand name Robaxin among others, is a medication used for short-term musculoskeletal pain.[3][4] It may be used together with rest, physical therapy, and pain medication.[3][5][6] It is less preferred in low back pain.[3] It has limited use for rheumatoid arthritis and cerebral palsy.[3][7] Effects generally begin within half an hour.[3] It is taken by mouth or injection into a vein.[3]
Methocarbamol is a CNS depressant indicated with rest, physical therapy and other treatments to control the discomfort associated with various acute musculoskeletal conditions.
Methocarbamol was developed in the early 1950s as a treatment for muscle spasticity and the associated pain.6,7 It is a guaiacol glyceryl ether.7
Methocarbamol tablets and intramuscular injections are prescription medicines indicated in the United States as an adjunct to rest, physical therapy, and other measures for the relief of discomforts associated with acute, painful musculoskeletal conditions.Label,9 In Canada, methocarbamol can be sold as an over the counter oral medicine at a lower dose that may be combined with acetaminophen or ibuprofen.10 A combination product with acetylsalicylic acid and codeine is available in Canada by prescription.10
Methocarbamol was FDA approved on 16 July 1957.8
Common side effect include headaches, sleepiness, and dizziness.[3][8] Serious side effects may include anaphylaxis, liver problems, confusion, and seizures.[4] Use is not recommended in pregnancy and breastfeeding.[3][4] Because of risk of injury, skeletal muscle relaxants should generally be avoided in geriatric patients.[3] Methocarbamol is a centrally acting muscle relaxant.[3] How it works is unclear, but it does not appear to affect muscles directly.[3]
Methocarbamol was approved for medical use in the United States in 1957.[3] It is available as a generic medication.[3][4] It is relatively inexpensive as of 2016.[9] In 2019, it was the 136th most commonly prescribed medication in the United States, with more than 4 million prescriptions.[10][11]
SYN
CN 109970606

SYN
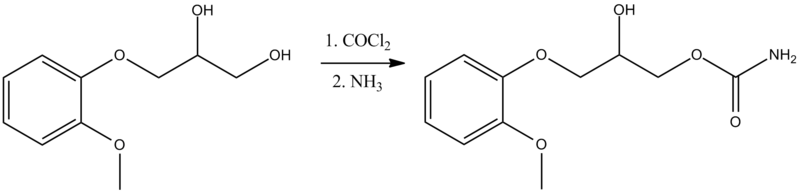
Synthesis of methocarbamol from guaifenesin. (a) methocarbamol and (b) β-isomer of methocarbamol.
SYN
https://www.sciencedirect.com/science/article/abs/pii/S0957416607003801
The muscle relaxant methocarbamol 2 and tranquilizer mephenoxalone 3, as well as intermediate cyclic carbonate 4, have been prepared in enantiopure form by starting from enantiopure guaifenesin 1 easily available by an entrainment resolution procedure. Thermal investigations reveal that 2 is probably a conglomerate forming substance, 3 forms a stable racemic compound, and 4 occupies an intermediate position. The enantiomeric excess of a binary phase eutectic point for these substances comprises 0%, 85%, and 10%, respectively.
Graphical abstract

PATENT
US 2770649
https://patents.google.com/patent/US2770649A/en
PAPER
Journal of pharmaceutical sciences (1970), 59(7), 1043-4

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
Medical use
Methocarbamol is a muscle relaxant used to treat acute, painful musculoskeletal spasms in a variety of musculoskeletal conditions.[12] However, there is limited and inconsistent published research on the medication’s efficacy and safety in treating musculoskeletal conditions, primarily neck and back pain.[12]
Methocarbamol injection may have a beneficial effect in the control of the neuromuscular spasms of tetanus.[6] It does not, however, replace the current treatment regimen.[6]
It is not useful in chronic neurological disorders, such as cerebral palsy or other dyskinesias.[3]
Currently, there is some suggestion that muscle relaxants may improve the symptoms of rheumatoid arthritis; however, there is insufficient data to prove its effectiveness as well as answer concerns regarding optimal dosing, choice of muscle relaxant, adverse effects, and functional status.[7]
Comparison to similar agents
The clinical effectiveness of methocarbamol compared to other muscle relaxants is not well-known.[12] One trial of methocarbamol versus cyclobenzaprine, a well-studied muscle relaxant, in those with localized muscle spasm found there was no significant differences in their effects on improved muscle spasm, limitation of motion, or limitation of daily activities.[12]
Contraindications
There are few contraindications to methocarbamol. They include:
- Hypersensitivity to methocarbamol or to any of the injection components.[6]
- For the injectable form, suspected kidney failure or renal pathology, due to large content of polyethylene glycol 300 that can increase pre-existing acidosis and urea retention.[6]
Side effects
Methocarbamol is a centrally acting skeletal muscle relaxant that has significant adverse effects, especially on the central nervous system.[3]
Potential side effects of methocarbamol include:
- Most commonly drowsiness, blurred vision, headache, nausea, and skin rash.[8]
- Possible clumsiness (ataxia), upset stomach, flushing, mood changes, trouble urinating, itchiness, and fever.[13][14]
- Both tachycardia (fast heart rate) and bradycardia (slow heart rate) have been reported.[14]
- Hypersensitivity reactions and anaphylatic reactions are also reported.[5][6]
- May cause respiratory depression when combined with benzodiazepines, barbiturates, codeine, or other muscle relaxants.[15]
- May cause urine to turn black, blue or green.[13]
While the product label states that methocarbamol can cause jaundice, there is minimal evidence to suggest that methocarbamol causes liver damage.[8] During clinical trials of methocarbamol, there were no laboratory measurements of liver damage indicators, such as serum aminotransferase (AST/ALT) levels, to confirm hepatotoxicity.[8] Although unlikely, it is impossible to rule out that methocarbamol may cause mild liver injury with use.[8]
Elderly
Skeletal muscle relaxants are associated with an increased risk of injury among older adults.[16] Methocarbamol appeared to be less sedating than other muscle relaxants, most notably cyclobenzaprine, but had similar increased risk of injury.[15][16] Methocarbamol is cited along with “most muscle relaxants” in the 2012 Beers Criteria as being “poorly tolerated by older adults, because of anticholinergic adverse effects, sedation, increased risk of fractures,” noting that “effectiveness dosages tolerated by older adults is questionable.”[17]
Pregnancy
Methocarbamol is labeled by the FDA as a pregnancy category C medication.[6] The teratogenic effects of the medication are not known and should be given to pregnant women only when clearly indicated.[6]
Overdose
There is limited information available on the acute toxicity of methocarbamol.[5][6] Overdose is used frequently in conjunction with CNS depressants such as alcohol or benzodiazepines and will have symptoms of nausea, drowsiness, blurred vision, hypotension, seizures, and coma.[6] There are reported deaths with an overdose of methocarbamol alone or in the presence of other CNS depressants.[5][6]
Abuse
Unlike other carbamates such as meprobamate and its prodrug carisoprodol, methocarbamol has greatly reduced abuse potential.[18] Studies comparing it to the benzodiazepine lorazepam and the antihistamine diphenhydramine, along with placebo, find that methocarbamol produces increased “liking” responses and some sedative-like effects; however, at higher doses dysphoria is reported.[18] It is considered to have an abuse profile similar to, but weaker than, lorazepam.[18]
Interactions
Methocarbamol may inhibit the effects of pyridostigmine bromide.[5][6] Therefore, methocarbamol should be used with caution in those with myasthenia gravis taking anticholinesterase medications.[6]
Methocarbamol may disrupt certain screening tests as it can cause color interference in laboratory tests for 5-hydroxy-indoleacetic acid (5-HIAA) and in urinary testing for vanillylmandelic acid (VMA) using the Gitlow method.[6]
Pharmacology
Mechanism of action
The mechanism of action of methocarbamol has not currently been established.[3] Its effect is thought to be localized to the central nervous system rather than a direct effect on skeletal muscles.[3] It has no effect on the motor end plate or the peripheral nerve fiber.[6] The efficacy of the medication is likely related to its sedative effect.[3] Alternatively, methocarbamol may act via inhibition of acetylcholinesterase, similarly to carbamate.[19]
Pharmacokinetics
In healthy individuals, the plasma clearance of methocarbamol ranges between 0.20 and 0.80 L/h/kg.[6] The mean plasma elimination half-life ranges between 1 and 2 hours, and the plasma protein binding ranges between 46% and 50%.[6] The elimination half-life was longer in the elderly, those with kidney problems, and those with liver problems.[6]
Metabolism
Methocarbamol is the carbamate derivative of guaifenesin, but does not produce guaifenesin as a metabolite, because the carbamate bond is not hydrolyzed metabolically;[8][6] its metabolism is by Phase I ring hydroxylation and O-demethylation, followed by Phase II conjugation.[6] All the major metabolites are unhydrolyzed carbamates.[20][21] Small amounts of unchanged methocarbamol are also excreted in the urine.[5][6]
Society and culture
Methocarbamol was approved as a muscle relaxant for acute, painful musculoskeletal conditions in the United States in 1957.[8] Muscle relaxants are widely used to treat low back pain, one of the most frequent health problems in industrialized countries. Currently, there are more than 3 million prescriptions filled yearly.[8] Methocarbamol and orphenadrine are each used in more than 250,000 U.S. emergency department visits for lower back pain each year.[22] In the United States, low back pain is the fifth most common reason for all physician visits and the second most common symptomatic reason.[23] In 80% of primary care visits for low back pain, at least one medication was prescribed at the initial office visit and more than one third were prescribed two or more medications.[24] The most commonly prescribed drugs for low back pain included skeletal muscle relaxants.[25] Cyclobenzaprine and methocarbamol are on the U.S. Medicare formulary, which may account for the higher use of these products.[16]
Economics
The generic formulation of the medication is relatively inexpensive, costing less than the alternative metaxalone in 2016.[26][9]
Marketing

Generic methocarbamol 750mg tablet.
Methocarbamol without other ingredients is sold under the brand name Robaxin in the U.K., U.S., Canada[27] and South Africa; it is marketed as Lumirelax in France, Ortoton in Germany and many other names worldwide.[28] In combination with other active ingredients it is sold under other names: with acetaminophen (paracetamol), under trade names Robaxacet and Tylenol Body Pain Night; with ibuprofen as Robax Platinum; with acetylsalicylic acid as Robaxisal in the U.S. and Canada.[29][30] However, in Spain the tradename Robaxisal is used for the paracetamol combination instead of Robaxacet.[citation needed] These combinations are also available from independent manufacturers under generic names.[citation needed]
Research
Although opioids are a typically first line in treatment of severe pain, several trials suggest that methocarbamol may improve recovery and decrease hospital length of stay in those with muscles spasms associated with rib fractures.[31][32][33] However, methocarbamol was less useful in the treatment of acute traumatic pain in general.[34]
Long-term studies evaluating the risk of development of cancer in using methocarbamol have not been performed.[5][6] There are currently no studies evaluating the effect of methocarbamol on mutagenesis or fertility.[5][6]
The safety and efficacy of methocarbamol has not been established in pediatric individuals below the age of 16 except in tetanus.[5][6]
References
- ^ “Robaxin-750 – Summary of Product Characteristics (SmPC)”. (emc). 8 August 2017. Retrieved 19 April 2020.
- ^ Sica DA, Comstock TJ, Davis J, Manning L, Powell R, Melikian A, Wright G (1990). “Pharmacokinetics and protein binding of methocarbamol in renal insufficiency and normals”. European Journal of Clinical Pharmacology. 39 (2): 193–4. doi:10.1007/BF00280060. PMID 2253675. S2CID 626920.
- ^ Jump up to:a b c d e f g h i j k l m n o p q r “Methocarbamol Monograph for Professionals”. Drugs.com. American Society of Health-System Pharmacists.
- ^ Jump up to:a b c d British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 1093. ISBN 9780857113382.
- ^ Jump up to:a b c d e f g h i “Robaxin- methocarbamol tablet, film coated”. DailyMed. 18 July 2019. Retrieved 19 April 2020.
- ^ Jump up to:a b c d e f g h i j k l m n o p q r s t u v w x “Robaxin- methocarbamol injection”. DailyMed. 10 December 2018. Retrieved 19 April 2020.
- ^ Jump up to:a b Richards, Bethan L.; Whittle, Samuel L.; Buchbinder, Rachelle (18 January 2012). “Muscle relaxants for pain management in rheumatoid arthritis”. The Cochrane Database of Systematic Reviews. 1: CD008922. doi:10.1002/14651858.CD008922.pub2. ISSN 1469-493X. PMID 22258993.
- ^ Jump up to:a b c d e f g h “Methocarbamol”. LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. National Institute of Diabetes and Digestive and Kidney Diseases. 30 January 2017. PMID 31643609.
- ^ Jump up to:a b Fine, Perry G. (2016). The Hospice Companion: Best Practices for Interdisciplinary Care of Advanced Illness. Oxford University Press. p. PT146. ISBN 978-0-19-045692-4.
- ^ “The Top 300 of 2019”. ClinCalc. Retrieved 16 October 2021.
- ^ “Methocarbamol – Drug Usage Statistics”. ClinCalc. Retrieved 16 October 2021.
- ^ Jump up to:a b c d Chou, Roger; Peterson, Kim; Helfand, Mark (August 2004). “Comparative efficacy and safety of skeletal muscle relaxants for spasticity and musculoskeletal conditions: a systematic review”. Journal of Pain and Symptom Management. 28 (2): 140–175. doi:10.1016/j.jpainsymman.2004.05.002. ISSN 0885-3924. PMID 15276195.
- ^ Jump up to:a b “Methocarbamol”. MedlinePlus. Retrieved 18 April 2020.
- ^ Jump up to:a b “Methocarbamol Side Effects: Common, Severe, Long Term”. Drugs.com. Retrieved 18 April 2020.
- ^ Jump up to:a b See, Sharon; Ginzburg, Regina (1 August 2008). “Choosing a skeletal muscle relaxant”. American Family Physician. 78 (3): 365–70. ISSN 0002-838X. PMID 18711953.
- ^ Jump up to:a b c Spence, Michele M.; Shin, Patrick J.; Lee, Eric A.; Gibbs, Nancy E. (July 2013). “Risk of injury associated with skeletal muscle relaxant use in older adults”. The Annals of Pharmacotherapy. 47 (7–8): 993–8. doi:10.1345/aph.1R735. ISSN 1542-6270. PMID 23821610. S2CID 9037478.
- ^ “Beers Criteria Medication List”. DCRI. Retrieved 18 October 2020.
- ^ Jump up to:a b c Preston KL, Wolf B, Guarino JJ, Griffiths RR (1992). “Subjective and behavioral effects of diphenhydramine, lorazepam and methocarbamol: evaluation of abuse liability”. Journal of Pharmacology and Experimental Therapeutics. 262 (2): 707–20. PMID 1501118.
- ^ PubChem. “Methocarbamol”. pubchem.ncbi.nlm.nih.gov. Retrieved 6 July 2020.
- ^ Methocarbamol. In: DRUGDEX System [intranet database]. Greenwood Village, Colorado: Thomson Healthcare; c1974–2009 [cited 2009 Feb 10].
- ^ Bruce RB, Turnbull LB, Newman JH (January 1971). “Metabolism of methocarbamol in the rat, dog, and human”. J Pharm Sci. 60 (1): 104–6. doi:10.1002/jps.2600600120. PMID 5548215.
- ^ Friedman BW, Cisewski D, Irizarry E, Davitt M, Solorzano C, Nassery A, et al. (March 2018). “A Randomized, Double-Blind, Placebo-Controlled Trial of Naproxen With or Without Orphenadrine or Methocarbamol for Acute Low Back Pain”. Annals of Emergency Medicine. 71 (3): 348–356.e5. doi:10.1016/j.annemergmed.2017.09.031. ISSN 1097-6760. PMC 5820149. PMID 29089169.
- ^ Chou, Roger; Huffman, Laurie Hoyt (2 October 2007). “Medications for Acute and Chronic Low Back Pain: A Review of the Evidence for an American Pain Society/American College of Physicians Clinical Practice Guideline”. Annals of Internal Medicine. 147 (7): 505–14. doi:10.7326/0003-4819-147-7-200710020-00008. ISSN 0003-4819. PMID 17909211. S2CID 32719708.
- ^ Cherkin, D. C.; Wheeler, K. J.; Barlow, W.; Deyo, R. A. (1 March 1998). “Medication use for low back pain in primary care”. Spine. 23 (5): 607–14. doi:10.1097/00007632-199803010-00015. ISSN 0362-2436. PMID 9530793. S2CID 23664539.
- ^ Luo, Xuemei; Pietrobon, Ricardo; Curtis, Lesley H.; Hey, Lloyd A. (1 December 2004). “Prescription of nonsteroidal anti-inflammatory drugs and muscle relaxants for back pain in the United States”. Spine. 29 (23): E531–7. doi:10.1097/01.brs.0000146453.76528.7c. ISSN 1528-1159. PMID 15564901. S2CID 72742439.
- ^ Robbins, Lawrence D. (2013). Management of Headache and Headache Medications. Springer Science & Business Media. p. PT147. ISBN 978-1-4612-2124-1.
- ^ “ROBAXIN product appearance in Canada”. ctchealth.ca. Retrieved 13 December 2021.
- ^ “Methocarbamol”. Drugs.com. Retrieved 12 May 2018.
- ^ “New Drugs and Indications Reviewed at the May 2003 DEC Meeting” (PDF). ESI Canada. Archived from the original (PDF) on 10 July 2011. Retrieved 14 November 2008.
- ^ “Tylenol Body Pain Night Overview and Dosage”. Tylenol Canada. Archived from the original (website) on 31 March 2012. Retrieved 23 April 2012.
- ^ Patanwala, Asad E.; Aljuhani, Ohoud; Kopp, Brian J.; Erstad, Brian L. (October 2017). “Methocarbamol use is associated with decreased hospital length of stay in trauma patients with closed rib fractures”. The American Journal of Surgery. 214 (4): 738–42. doi:10.1016/j.amjsurg.2017.01.003. ISSN 0002-9610. PMID 28088301.
- ^ Deloney, Lindsay; Smith, Melanie; Carter, Cassandra; Privette, Alicia; Leon, Stuart; Eriksson, Evert (January 2020). “946: Methocarbamol reduces opioid use and length of stay in young adults with traumatic rib fractures”. Critical Care Medicine. 48 (1): 452. doi:10.1097/01.ccm.0000633320.62811.06. ISSN 0090-3493.
- ^ Smith, Melanie; Deloney, Lindsay; Carter, Cassandra; Leon, Stuart; Privette, Alicia; Eriksson, Evert (January 2020). “1759: Use of methocarbamol in geriatric patients with rib fractures is associated with improved outcomes”. Critical Care Medicine. 48 (1): 854. doi:10.1097/01.ccm.0000649332.10326.98. ISSN 0090-3493.
- ^ Aljuhani, Ohoud; Kopp, Brian J.; Patanwala, Asad E. (2017). “Effect of Methocarbamol on Acute Pain After Traumatic Injury”. American Journal of Therapeutics. 24 (2): e202–6. doi:10.1097/mjt.0000000000000364. ISSN 1075-2765. PMID 26469684. S2CID 24284482.
External links
- “Methocarbamol”. Drug Information Portal. U.S. National Library of Medicine.
| Clinical data | |
|---|---|
| Trade names | Robaxin, Marbaxin, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682579 |
| License data | US DailyMed: Methocarbamol |
| Pregnancy category | AU: B2 |
| Routes of administration | By mouth, intravenous |
| ATC code | M03BA03 (WHO) M03BA53 (WHO) M03BA73 (WHO) |
| Legal status | |
| Legal status | CA: OTCUK: POM (Prescription only) [1]US: ℞-only |
| Pharmacokinetic data | |
| Metabolism | Liver |
| Elimination half-life | 1.14–1.24 hours[2] |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 532-03-6 |
| PubChem CID | 4107 |
| IUPHAR/BPS | 6829 |
| DrugBank | DB00423 |
| ChemSpider | 3964 |
| UNII | 125OD7737X |
| KEGG | D00402 |
| ChEBI | CHEBI:6832 |
| ChEMBL | ChEMBL1201117 |
| CompTox Dashboard (EPA) | DTXSID6023286 |
| ECHA InfoCard | 100.007.751 |
| Chemical and physical data | |
| Formula | C11H15NO5 |
| Molar mass | 241.243 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
| (what is this?) (verify) |
- Sica DA, Comstock TJ, Davis J, Manning L, Powell R, Melikian A, Wright G: Pharmacokinetics and protein binding of methocarbamol in renal insufficiency and normals. Eur J Clin Pharmacol. 1990;39(2):193-4. [Article]
- Bruce RB, Turnbull LB, Newman JH: Metabolism of methocarbamol in the rat, dog, and human. J Pharm Sci. 1971 Jan;60(1):104-6. [Article]
- Witenko C, Moorman-Li R, Motycka C, Duane K, Hincapie-Castillo J, Leonard P, Valaer C: Considerations for the appropriate use of skeletal muscle relaxants for the management of acute low back pain. P T. 2014 Jun;39(6):427-35. [Article]
- Crankshaw DP, Raper C: Mephenesin, methocarbamol, chlordiazepoxide and diazepam: actions on spinal reflexes and ventral root potentials. Br J Pharmacol. 1970 Jan;38(1):148-56. doi: 10.1111/j.1476-5381.1970.tb10343.x. [Article]
- Muir WW 3rd, Sams RA, Ashcraft S: The pharmacology and pharmacokinetics of high-dose methocarbamol in horses. Equine Vet J Suppl. 1992 Feb;(11):41-4. [Article]
- Authors unspecified: Methocarbamol-A New Lissive Agent. Can Med Assoc J. 1958 Dec 15;79(12):1008-9. [Article]
- O’DOHERTY DS, SHIELDS CD: Methocarbamol; new agent in treatment of neurological and neuromuscular diseases. J Am Med Assoc. 1958 May 10;167(2):160-3. [Article]
- FDA Approved Drug Products: Robaxin [Link]
- FDA Approved Drug Products: Robaxin Intramuscular Injection [Link]
- Pfizer Canada: Robax [Link]
////////////////Methocarbamol, метокарбамол , ميثوكاربامول , 美索巴莫, AHS 85, Muscle Relaxant
COC1=C(OCC(O)COC(N)=O)C=CC=C1

NEW DRUG APPROVALS
ONE TIME
$10.00

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










