Home » Posts tagged 'Mumbai'
Tag Archives: Mumbai
PharmmaEx Mumbai INDIA 3-4 October 2025

Congratulations Pharmmaexians,
We have signed as our Chief Guest Dr Anthony Melvin Crasto Advisor AfricurePharma Row2Tech Glenmark IPCA AdvectProc Niper-G Dept Pharma Min Chem and Fert Govt of India .
Thanks and Regards
Shivam Sharma
PharmmaEx Mumbai
3rd and 4th October 2025
Bombay Exhibition Centre Nesco Goregaon, .Mumbai India
Anthony Crasto conferred ABPnews award for “Outstanding contribution to Education Sector”
 Conferred prestigious award at event ABP News Presents Healthcare Leadership Awards 26th November, 2018 at Taj Lands End, Mumbai India
Conferred prestigious award at event ABP News Presents Healthcare Leadership Awards 26th November, 2018 at Taj Lands End, Mumbai India
Dedicated to Shobha Crasto Aishal crasto Lionel crasto
Service to education is service to humanity
Society recognises efforts done towards it
/////////////award, event ABP News, Healthcare, Leadership, 26th, November, 2018, Taj Lands End, Mumbai, India, education, anthony crasto
National award to Anthony Melvin Crasto for contribution to Pharma society from Times Network for Excellence in HEALTHCARE) | 5th July, 2018 | Taj Lands End, Mumbai, India
DR ANTHONY MEVIN CRASTO Conferred prestigious individual national award at function for contribution to Pharma society from Times Network, National Awards for Marketing Excellence ( For Excellence in HEALTHCARE) | 5th July, 2018 | Taj Lands End, Mumbai India
////////////National award, contribution to Pharma society, Times Network, Excellence in HEALTHCARE, 5th July, 2018, Taj Lands End, Mumbai, India, ANTHONY CRASTO
#hotpersoninawheelchair
#worlddrugtracker
CHIRAL INDIA 2016, 5th International Conference & Exhibition Nov 8-9 2016, Holiday Inn, Mumbai, India
India's only conference focusing on new chiral technologies for pharmaceutical fine chemicals. The event is a unique platform to learn about recent advances in chiral chemistry, technology and application.
Chiral India series which began in 2012 has now grown into a major must-attend event for the Pharmaceutical industry. This platform is the most popular chiral technology platform bringing together the top experts from China, Canada, USA, Japan, India and other countries to present the latest developments in chiral drug developments and brainstorm with leading R&D personnel from Indian pharmaceutical industry.
The fifth edition of Chiral India to be held on 8-9 November 2016, at Holiday Inn (Mumbai), follows the success of previous four annual editions (2012, 2013, 2014 and 2015) and is now an event awaited by R&D professionals across the industry.
|
||||||||||||
R Rajagopal
+9198211 28341
rraj@chemicalweekly.com
kiran@chemicalweekly.com
| Dr. R. Rajagopal | B-602, Godrej Coliseum | Tel: +91 22 24044477 |
| Editorial Advisor | K.J. Somaiya Hospital Road | Fax: +91 22 24044450 |
| Chemical Weekly | Sion (East) Mumbai 400 022 | www.chemicalweekly.com |
DOWNLOAD BROCHURE…..
Please use http://www.chiralindia.com/Brochure.pdf link to download the Brochure.
Our website URL is www.chiralindia.com
|
Oganised By
|
 |

SCROLL USING MOUSE TO VIEW 5 PAGES
////////CHIRAL INDIA 2016, 5th International Conference, Exhibition, Nov 8-9, 2016, Holiday Inn, Mumbai, India
P7435 from Piramal Enterprises Mumbai, India
P7435
Piramal Enterprises Mumbai, India
P-7435; P7435-DGAT1, P7435, P 7435
- Molecular Weight, 454.47
GDAT1 inhibitor
- Phase IDiabetes mellitus; Lipid metabolism disorders
- ClassAntihyperglycaemics; Antihyperlipidaemics; Small molecules
- Mechanism of ActionDiacylglycerol O acyltransferase inhibitors

| Latest Stage of Development | Phase I |
| Standard Indication | Metabolic (unspecified) |
| Indication Details | Treat metabolic disorders |
https://clinicaltrials.gov/ct2/show/NCT01910571
https://clinicaltrials.gov/ct2/show/NCT01764425
- 24 Nov 2014Piramal Enterprises completes a phase I trial in healthy, overweight or obese subjects in USA (NCT01910571)
- 17 Jun 2014Adverse events and pharmacokinetics data from a phase I trial in healthy male volunteers presented at the 74th Annual Scientific Sessions of the American Diabetes Association (ADA-2014)
- 17 Jun 2014Pharmacodynamics data from preclinical studies in Dyslipidaemia and obesity presented at the 74th Annual Scientific Sessions of the American Diabetes Association (ADA-2014)

Chairman Ajay Piramal

Swati Piramal-The Vice Chairperson of Piramal Enterprises Ltd

Nandini Piramal, Executive Director, Piramal Enterprises
Piramal Enterprises gets US FDA approval for P7435 IND
http://www.pharmabiz.com/NewsDetails.aspx?aid=76992&sid=2
Our Bureau, Mumbai
Tuesday, August 06, 2013, 12:25 Hrs [IST]
Piramal Enterprises Ltd has received US Food and Drug Administration (FDA) approval for its Investigational New Drug (IND) P7435. This is a novel, potent and highly selective, oral diacylglycerolacyltransferase 1 (DGAT1) inhibitor.
P7435 has been developed by the NCE Research Division of PEL for the management of metabolic disorders such as lipid abnormalities and diabetes. It is well-established that increased lipid levels’ (including triglycerides) is one of the major risk factors for cardiovascular disease (CVD). It has been reported by the World Health Organisation, that CVD, is the number one cause of deaths globally, representing approximately 30 per cent of all deaths. Currently, there is a significant medical need for effective and safe drugs for the management of lipid abnormalities and metabolic disorders.
P7435 has demonstrated its lipid lowering potential in various preclinical studies by showing significant reduction in triglyceride levels, glucose and insulin levels,and decrease in food intake and body weight gain -factors which are associated with lipid abnormalities and metabolic disorders.
PEL has established the safety and tolerability of P7435 in a phase I trial recently completed in India. This extension trial in the US will further evaluate the safety and efficacy of P7435 in a larger population.
Dr Swati Piramal, vice chairperson, Piramal Enterprises, said, “The NCE Research division of PEL continues its ambitious diabetes/metabolic disorders programme to discover and develop NCEs to fight against diseases like diabetes and lipid disorders. With P7435 we are looking at addressing a serious need for effective and well-tolerated drugs that treat lipid disorders, which are commonly associated with diabetes and CVDs. Expansion of this trial will allow testing this NCE in a wider population,which is critical to the development of this drug and will provide therapeutic solutions not just to India but also to the rest of the world.”
The NCE Research division of Piramal Enterprises focuses on the discovery and development of innovative small molecule medicines to improve the lives of patients suffering from cancer, metabolic disorders and inflammatory conditions. The key elements of its strategy include capitalizing on Piramal’s strengths, in particular the India advantage, and leveraging external partnerships to achieve high levels of R&D productivity. Piramal’s state-of-the-art Research Centre in Mumbai has comprehensive capabilities spanning target identification all the way through clinical development. Its robust pipeline, including 8 compounds in clinical development, bears testimony to its innovative and rigorous drug discovery process.
PAPER
European Journal of Medicinal Chemistry (2012), 54, 324-342
http://www.sciencedirect.com/science/article/pii/S0223523412003133
PATENT
WO 2010023609
http://www.google.co.in/patents/WO2010023609A1?cl=en
/////////Piramal Enterprises, Mumbai, India, P-7435, P7435-DGAT1, P7435, P 7435, GDAT1 inhibitor
O=C(O)[C@@H](NC(=O)c1cc(no1)c2ccc(cc2)Nc3nc4ccc(F)cc4s3)C(C)C
Flow Chemistry India 2016, 21-22 January 2016, Mumbai, India
Flow Chemistry India 2016Date: Thursday, 21 January 2016 – Friday, 22 January 2016
|
SELECTBIO INDIA
http://selectbiosciences.com/conferences/index.aspx?conf=FCINDIA16&se=india
Register…………..http://selectbiosciences.com/conferences/registration.aspx?conf=FCINDIA16&se=india

venue
Hotel Ramada Powai and Convention Centre, Mumbai, India


Professor & Research Chair, Nelson Mandela Metropolitan University |

Professor, Synthetic Organic Chemistry , The University of Tokyo |

Deputy Director and Chair, National Chemical Laboratory |

Professor, Eindhoven University of Technology |

Scientific Director, University of Lyon |

Chairman, Flow Chemistry Society
Professor, University of Warsaw |


Maninderjit Singh Ahluwalia
Overview
SELECTBIO INDIA is delighted to welcome you all at the 4th International Conference Flow Chemistry India 2016 to be held in Mumbai on January 21-22, 2016 under the auspices of the Flow Chemistry Society. The society aims to unite and represent those who are actively working on this rapidly developing field. This meeting is dedicated to the integration of flow chemistry into everyday practice throughout the world by delivering the latest knowledge and making it available for the entire chemistry community.
Society members save 25% on the registration fee and non-members will receive their first year’s membership included in the fee.
Running alongside the conference will be an exhibition covering the latest technological advances in the area of flow chemistry.
Who Should Attend
• Scientists, Chemists, Chemical Engineers and Researchers working in Pharmaceutical and Fine Chemicals Research and Development including Drug Discovery, Medicinal Chemistry and Chemical Process Development
• Scientists, Chemists and Chemical Engineers working in Pharmaceutical and Fine Chemical Bulk Manufacturing Units
• Corporate Management, Scientists, Managers responsible for development of Pharmaceutical and Fine Chemicals R & D and Manufacturing activities
• Scientists, Chemists & Engineers belonging to the fields of Inorganic, Organic, Medicinal, Natural Products, Analytical, High-throughput and Process Chemistry in the Academic research as well as in Applied research and development in the area of Agrochemical, Petrochemical and Fragrance industry
• Scientists working in or interested in applications of Flow Chemistry in Material science, Green chemistry, Nanotechnology, Biotechnology, Theoretical Chemistry, Information technology and Flow synthesis instruments including Engineering & Automation
Conference Package – Includes Registration, 2 Nights Accommodation, Dinner & Airport Transfers (Valid up to January 5, 2016 only)
Call for Posters
You can also present your research on a poster while attending the meeting. Submit an abstract for consideration now!
Poster Submission Deadline: 30 November 2015
Agenda Topics
- Advances in Micro & Continuous Flow Reactors, Systems & Processes
- Applications in Pharmaceutical Industry & API Synthesis
- Engineering Aspects of Flow Chemistry
- Flow Reactor – Choosing the Right One
- Photochemistry & Multistep Synthesis in Flow
- Quality Issue and QbD in Flow Chemistry
- Scale up – From Micro to Commercial Scale
- Yield Improvement, Cost Cutting and Waste Reduction in Flow Chemistry
Sponsorship and Exhibition Opportunities
http://selectbiosciences.com/conferences/index.aspx?conf=FCINDIA16&se=india

Workshop Tutor
Charlotte Wiles
CEO CHEMTRIX
A Workshop on “Flow Chemistry Demonstrations (Lab & Plant Scale) for Chemical and Pharmaceutical Industry-” will be held one day prior to the training course i.e. on 20th January, 2016 from 10:00 am – 05:00 pm in Mumbai. This workshop is supported by Process Intensification will be jointly conducted by :
Dr. Dinesh Kudav (Mumbai University); Dr. Charlotte Wiles (Chemtrix BV-Neth); Mr. Wouter Stam (Flowid, NV-Neth); Mr. Manjinder Singh (CIPLA & VP-FCS-India Chapter); Dr. Viktor Gyollai, (AM Technology-UK); Dr. Prashant Kini (UPL Ltd.); Mr. Kumar Oza (Pi & TCPL); Mr. Madhav Sapre (Pi & Sharon Bio); et al .
This workshop is specially designed to demonstrate application/capabilities of Flow Chemistry running “live” reactions in Continuous Flow Reactors. The reactions likely to be demonstrated using Flow Chemistry includes :• Nitration • Organometallic reaction• Oxidation • Bi-phasic reaction• Nano-Particle preparation in Flow• Biocatalytic Reaction with enhanced enzyme life.
This workshop is free for the registered delegates of Flow Chemistry India 2016 Conference and Continuous Flow Reactors Training Course.
You can visit Mumbai city
Taj hotel, mumbai
Gateway of india
 Food in mumbai
Food in mumbai
 mumbai skyline
mumbai skyline

The Bandra-Worli Sea Link is a cable-stayed bridge that connects central Mumbai with its western suburbs

get in if you can
The Mumbai Suburban Railway system carries more than 6.99 million commuters on a daily basis. It has the highest passenger densities of any urban railway …

Chhatrapati shivaji in mumbai india
British-victoria terminus


VADA PAV


SELECTBIO CONFERENCES PICS










/////////
Indian pharma’s struggle to tighten standards paves way for M&A deals
MUMBAI – India’s smaller generic drugmakers, struggling to cope with a bruised reputation and tougher regulation in the United States, are under pressure to consider branching out to new, less-profitable markets or sell out to larger rivals.
Two years after its most high-profile regulatory setback to date in the United States – Ranbaxy’s $500 million U.S. fine for drug safety violations – India’s $15 billion a year generic drug industry is still rebuilding its image in its biggest market.
Many of its top firms are facing sanctions at some of their factories, as the U.S. Food and Drug Administration (FDA) tightens checks and its approvals process.
Combined with government-mandated price controls on drugs at home, that is piling pressure on smaller players.
“If they want to have a presence globally, they have to make investments. If they can’t, then they’ll have to focus on other markets or scale back their ambition outside of India, and that’s probably what will happen,” said Subhanu Saxena, CEO of Cipla , India’s fourth-largest drugmaker by revenue.
Ashok Anand, president of Hikal Ltd , a Mumbai-based drugmaker with a market value of $167 million, said some peers were putting themselves on the block.
“If they cannot deal with the stricter regulations, they might just prefer to sell out,” he said.
Pressure on U.S. sales has been felt across the Indian industry, with all drugmakers hit by delays in FDA approvals as the U.S. safety body overhauls its review process. Growth in U.S. revenue for drugmakers slowed to 14 percent in the year to March 2015, less than half what it was in the year to March 2012, according to brokerage Edelweiss.
But for larger players who want to plug gaps or, for the likes of Glenmark and Aurobindo who aim to grow in the United States, this pressure has lowered prices and could pave the way for attractive deals, bankers said.
“Now that some of the smaller companies are reeling under intensive regulatory scrutiny and want to cash out on their investments, valuations would be much more realistic,” said the head of India M&A at a large European bank in Mumbai.
SPENDING SPREE
Indian manufacturers say they have spent millions in high-end testing equipment, improved training and have hired larger teams in quality control since Ranbaxy was fined for manipulating clinical data.
Some consultants estimate spending on compliance has more than doubled to reach about 6 to 7 percent of sales for the larger companies.
But while the number of U.S. export bans issued to Indian companies fell to eight in 2014 from 21 in 2013, according to FDA data, the agency continues to find manufacturing violations at the plants of some of the biggest drugmakers in the country, an indication of the pervasiveness of the problem.
Sun Pharmaceutical Industries , Wockhardt , Dr Reddy’s Laboratories and Cadila Healthcarehave all faced FDA rebukes over the past year.
Smaller firms Ipca and Aarti Drugs faced FDA bans on their plants this year.
These failures – which executives blame on India’s “quick fix” culture and consultants blame on a failure to prioritize compliance – have clouded short-term growth prospects and added to pressure on smaller players, pushing some to look elsewhere.
“They can choose to be in lesser-regulated markets, such as Latin America, where there is a lot of demand. But they will have to live with much thinner margins,” said the finance director of a small Indian drugmaker, who did not want to be named. “It’s survival of the fittest.” REUTERS
http://m.todayonline.com/business/indian-pharmas-struggle-tighten-standards-paves-way-ma-deals
///////
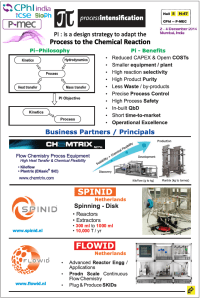
Pi-Process Intensification Experts LLP at CPhI Mumbai India 3rd Dec 2014…My visit

Process Intensification
Creating competitive advantage through Improved and consistent quality, high efficiencies and maximum flexibility.
Safer, Cleaner, Smaller, Cheaper and Smarter processes , The basic principle of Process Intensification is to fit the equipment to the process and not process to the equipment, as is the case now.
Process Intensification can achieve drastic improvement in the time cycle and yields as well as converting batch processes to continuous process using specialized set of equipment. The design philosophy in process intensification is to design a process which has Chemical Kinetics as its only limitation. See the illustration below
“Process Intensification by Kinetics alone controlling the reaction, using specialized equipments; modification / telescoping of process steps achieves drastic reduction in time cycles and converts batch processes to continuous ; Reduced energy consumption, Reduced by-product formation; sustainability , hazard-containment, compliance to QbD and PAT and importantly a much faster time-to-market”
Illustrative examples are as follows:
- Watt’s aldol reaction: Time needed to reach 100 % conversion 20 minutes against 24 hours in batch process
- Fisher Esterification:
 gives 83% yield against 15% in batch process
gives 83% yield against 15% in batch process - Grignard Reaction:
 gives 78% yield against 49% in batch process
gives 78% yield against 49% in batch process - Nitration Reaction: Product purity increase from 56% to 78% and yield of mononitrate increases 55% to 75%.
- Other Reactions: Acetylation, Amine Protection, Carbonylation, Claisen Schmidt Reaction, Esterification, Hydrogenation, Hydrolysis, Methylation, Oxidation, Phosgenation, Sulphonation, Suzuki Coupling Ring Expansion
Sponsored Projects
Scale-up for Retrofitting in existing plant as well as greenfield projects based on flow chemistry data generated in our laboratory. A well-equipped Laboratory and Pilot Plant set-up is available at our “Pi-Lab” for carrying out “FLOW Chemistry” based Reactions and utilizing numerous Process Intensification techniquesfor Unit-Processes & Unit-Operations for the industry to reap the benefits of Process Intensification.
The laboratory and pilot plant data will be utilized to provide the plant scale design using specialized equipments like micro-reactors, micro-plate-reactors in SiC, monolithic loop reactors, spinning disk reactors-cum-heat exchangers, FUMI reactors, dynamic mixing reactors, oscillatory baffled reactors (OBR), Bio-catalytic impregnated membrane Reactors, and other modern state-of-the-art equipments enabling conversion of batch to continuous flow processes.
We handle hazardous chemistries with very high exotherms (upto 1300 J/gm) safely in the range of -70oC to + 250oC with pressures upto 200 bar, and with reaction times from 0.03 sec to 1 hour and reactor volumes from 0.2 ml to 100 ml (Lab) and 1 L (Pilot) — yielding from 20 gms to 8 Kgs/hour (Lab) and 500 gms to 200 Kgs/hour (Pilot).
Scale Up – Flexibility & Adaptability
 …… will provide all the services for scale up to the sizes desired by clients by utilizing data from Laboratory trials.
…… will provide all the services for scale up to the sizes desired by clients by utilizing data from Laboratory trials.


Rental
A range of Flow Chemistry and Process Intensification equipments can be offered on rent. This enables the users to get the hands-on experience so as to select the apt equipments for their needs.
CEO
Pi-Process Intensification Experts LLP
Plot-W-33, M.I.D.C. Industrial Area
TALOJA – 410208, Navi Mumbai, INDIA
email : vk@pi-inc.co
www.pi-inc.co
Tel: +91-9321342022 // +91-9821342022

some pics from hall 5 -H-47 at cphi mumbai india dec 3 2014


Flow Chemistry test facility in India
Flow Chemistry test facility in India
BOOK YOUR TRIAL
on Plantrix® Industrial system.
Contacts:

India
Vijay Kirpalani
vk@pi-inc.co
0091 9821 3420 22
More information
Stan Hoeijmakers
info@chemtrix.com
0031 (0)46 70 22 600
EVENTS
Chemtrix at CPhI India
2 – 4 December 2014
Mumbai, India
Booth H47, Hall 5
Pi Process Intensification
read at
http://hosted.verticalresponse.com/721499/f9f4fc970b/285875213/4942751fec/
|
Plantrix® Industrial Flow Chemistry Test Facility in India
|
||||||||

|
||||||||
|
|
|||||||
|
|
||||||||
Chemtrix Bv.
Urmonderbaan 22
Geleen, 6167RD
NL

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....