Home » Posts tagged 'LAZUVAPAGON'
Tag Archives: LAZUVAPAGON
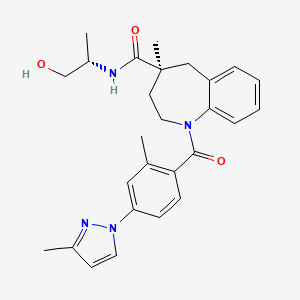
LAZUVAPAGON

LAZUVAPAGON
KRPN-118
CAS 2379889-71-9
Chemical Formula: C27H32N4O3
Molecular Weight: 460.58
(4S)-N-[(2S)-1-hydroxypropan-2-yl]-methyl-1-[2-methyl-4-(3- methyl-1H-pyrazol-1-yl)benzoyl]-2,3,4,5-tetrahydro-1H-1-benzazepine-4-carboxamide
Vasopressin V2 receptor agonist
Kyorin Pharmaceutical under license from Sanwa Kagaku Kenkyusho , is developing SK-1404 ([14C]-SK-1404, presumed to be lazuvapagon), for the iv treatment of nocturia, and as an oral formulation, as KRPN-118
PATENT
WO2020171055
PATENT
WO2014104209
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2014104209
PATENT
WO-2020171073
Process for preparing benzazepine derivatives, particularly lazuvapagon a V2 receptor agonist, and their intermediates, useful for treating diabetes insipidus, hemophilia and overactive bladder.
[Chemical
In the fifth step, compound (IX) is treated with a base (eg, sodium hydroxide, potassium hydroxide, etc.) in a suitable solvent (eg, alcohol solvent such as methanol, ethanol, etc., water), usually at room temperature to an organic solvent. A carboxylic acid compound of the compound (X) can be obtained by reacting at a temperature of the boiling point of the solvent for 30 minutes to 1 day. Next, in the sixth step, the obtained carboxylic acid compound is subjected to amidation with L-alaninol to obtain the compound (V). For the amidation, a method using a condensing agent, a method of reacting L-alaninol with a mixed acid anhydride or acid chloride of carboxylic acid, and the like can be used. In the method using a condensing agent, for example, the carboxylic acid compound and L-alaninol are condensed in a suitable organic solvent (chloroform, dimethylformamide, etc.) in the presence of a base (eg, diisopropylethylamine, triethylamine, etc.) (for example, 1 , 3-dicyclohexylcarbodiimide (DCC), 1-ethyl-3-[3-(dimethylamino)propyl]carbodiimide (EDC), etc.) alone or in combination with 1-hydroxybenztriazole (HOBt). (V) can be obtained. Further, in the method using a mixed acid anhydride, for example, a carboxylic acid derivative in an appropriate organic solvent (eg, dichloromethane, toluene, etc.) in the presence of a base (eg, pyridine, triethylamine, etc.), an acid chloride (eg, pivaloyl chloride, Tosyl chloride, etc.) or an acid derivative (eg, ethyl chloroformate, isobutyl chloroformate, etc.), and the resulting mixed acid anhydride is reacted with L-alaninol usually at 0° C. to room temperature to give compound (V). Can be obtained. Further, in the method of passing through an acid chloride, for example, an acid chloride is obtained by using a chlorinating agent (eg, thionyl chloride, oxalyl chloride, etc.) in a suitable organic solvent (eg, toluene, xylene, etc.) Acid chloride in the presence of a base (eg sodium carbonate, triethylamine etc.) in a suitable organic solvent (eg ethyl acetate, toluene etc.) with L-alaninol,
PATENT
WO-2020171055
The present inventors have investigated the method described in Patent Document 1 by using N-[(S)-1-hydroxypropan-2-yl]-4-methyl-1-[2-methyl-4-(3-methyl-1H). -Pyrazol-1-yl)benzoyl]-2,3,4,5-tetrahydro-1H-benzo[b]azepine-4-carboxamide chiral compound was prepared and analyzed. As a result, the compound was amorphous (amorphous). Solid). Amorphous is known to be a thermodynamically non-equilibrium metastable state and generally has high solubility and dissolution rate, but is low in stability and is often unfavorable in terms of drug development. Therefore, an object of the present invention is to increase the applicability as a drug substance to (S)-N-[(S)-1-hydroxypropan-2-yl]-4 represented by the formula (I). -Methyl-1-[2-methyl-4-(3-methyl-1H-pyrazol-1-yl)benzoyl]-2,3,4,5-tetrahydro-1H-benzo[b]azepine-4-carboxamide It is to provide an alcohol solvate or a crystal thereof.
[Chemical 1]
Compound (I) was produced by the following method.
[Chemical 5]
1-(2-Methyl-4-(3-methyl-1H-pyrazol-1-yl)benzoyl)-5-oxo-2,3,4,5-tetrahydro-1H-benzo[b] Azepine-4-carboxylic acid ethyl ester was treated with methyl bromide in the presence of (R,R)-3,5-bistrifluoromethylphenyl-NAS bromide, cesium carbonate and cesium fluoride in a mixed solvent of benzene bromide and water. By carrying out methylation using (R)-4-methyl-1-(2-methyl-4-(3-methyl-1H-pyrazol-1-yl)benzoyl)-5-oxo-2,3,4 ,5-Tetrahydro-1H-benzo[b]azepine-4-carboxylic acid ethyl ester was obtained.
(R)-4-Methyl-1-(2-methyl-4-(3-methyl-1H-pyrazol-1-yl)benzoyl)-5-oxo-2,3,4,5- Reduction of the ketone portion of tetrahydro-1H-benzo[b]azepine-4-carboxylic acid ethyl ester with a borane-ammonia complex prepared from sodium borohydride and ammonium sulfate in a toluene solvent gave (4R)-5. -Hydroxy-4-methyl-1-(2-methyl-4-(3-methyl-1H-pyrazol-1-yl)benzoyl)-2,3,4,5-tetrahydro-1H-benzo[b]azepine- 4-Carboxylic acid ethyl ester was obtained.
(4R)-5-hydroxy-4-methyl-1-(2-methyl-4-(3-methyl-1H-pyrazol-1-yl)benzoyl)-2,3,4,5- By chlorinating the hydroxyl group of tetrahydro-1H-benzo[b]azepine-4-carboxylic acid ethyl ester with phosphorus oxychloride in the presence of pyridine in a toluene solvent, (4S)-5-chloro-4-methyl-1 -(2-Methyl-4-(3-methyl-1H-pyrazol-1-yl)benzoyl)-2,3,4,5-tetrahydro-1H-benzo[b]azepine-4-carboxylic acid ethyl ester was obtained. It was
(4S)-5-chloro-4-methyl-1-(2-methyl-4-(3-methyl-1H-pyrazol-1-yl)benzoyl)-2,3,4,5- By stirring tetrahydro-1H-benzo[b]azepine-4-carboxylic acid ethyl ester in a methanol solvent in the presence of 10% palladium-carbon under slightly pressurized conditions of hydrogen gas, (S)-4-methyl- 1-(2-methyl-4-(3-methyl-1H-pyrazol-1-yl)benzoyl)-2,3,4,5-tetrahydro-1H-benzo[b]azepine-4-carboxylic acid ethyl ester Obtained.
(S)-4-Methyl-1-(2-methyl-4-(3-methyl-1H-pyrazol-1-yl)benzoyl)-2,3,4,5-tetrahydro-1H- Benzo[b]azepine-4-carboxylic acid ethyl ester is hydrolyzed with 30% sodium hydroxide in a solvent of water and methanol to give (S)-4-methyl-1-(2-methyl-4-( 3-Methyl-1H-pyrazol-1-yl)benzoyl)-2,3,4,5-tetrahydro-1H-benzo[b]azepine-4-carboxylic acid was obtained.
(S)-4-Methyl-1-(2-methyl-4-(3-methyl-1H-pyrazol-1-yl)benzoyl)-2,3,4,5-tetrahydro-1H- Benzo[b]azepine-4-carboxylic acid was converted to an acid chloride form using thionyl chloride in a toluene solvent. This acid chloride and L-alaninol are reacted in a mixed solvent of ethyl acetate and water in the presence of sodium carbonate to give (S)-N-((S)-1-hydroxypropan-2-yl)-4-methyl. -1-(2-methyl-4-(3-methyl-1H-pyrazol-1-yl)benzoyl)-2,3,4,5-tetrahydro-1H-benzo[b]azepine-4-carboxamide (compound ( I)) was obtained.
of compound (I) To 5.0 g of amorphous compound (I) of Reference Example 1, 65 mL of isopropanol was added, and the mixture was stirred at room temperature for 30 minutes. After the precipitated suspension was dissolved by heating, it was allowed to cool to room temperature and stirred overnight at 5°C. The suspension was filtered, washed with chilled isopropanol and dried at 40° C. overnight to give 4.9 g of a white solid.
/////////////LAZUVAPAGON, KRPN-118
CC1=NN(C=C1)C2=CC(=C(C=C2)C(=O)N3CCC(CC4=CC=CC=C43)(C)C(=O)NC(C)CO)C

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....