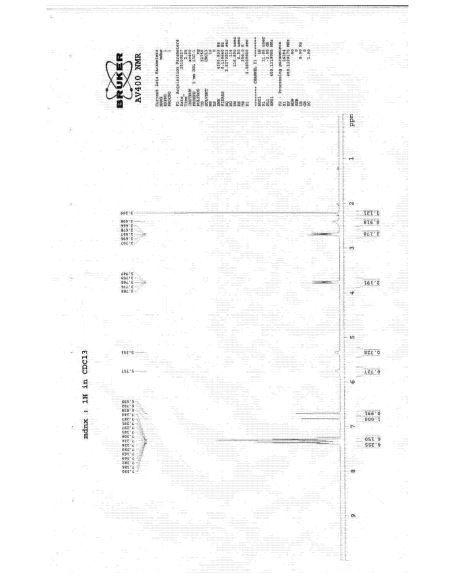
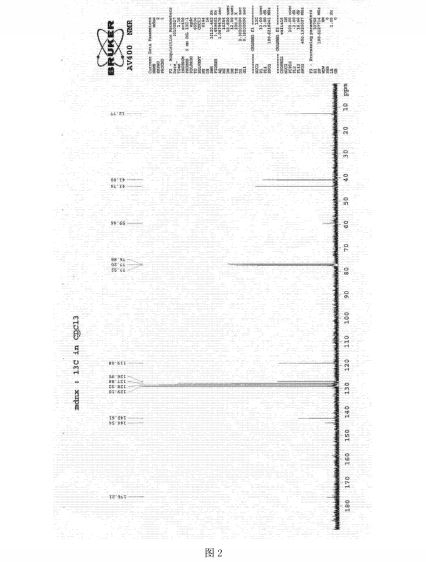
1H and 13C NMR spectra of 2-acyl-substituted cyclohexane-1,3-diones (acyl = formyl, 1; 2-nitrobenzoyl, 2; 2-nitro-4-trifluoromethylbenzoyl, 3) and lithium sodium and potassium salts of 1have been measured. The compound 3, known as NTBC, is a life-saving medicine applied in tyrosinemia type I. The optimum molecular structures of the investigated objects in solutions have been found using the DFT method with B3LYP functional and 6-31G** and/or 6-311G(2d,p) basis set. The theoretical values of the NMR parameters of the investigated compounds have been calculated using GIAO DFT B3LYP/6-311G(2d,p) method. The theoretical data obtained for compounds 1−3 have been exploited to interpret their experimental NMR spectra in terms of the equilibrium between different tautomers. It has been found that for these triketones an endo-tautomer prevails. The differences in NMR spectra of the salts of 1 can be rationalized taking into account the size of the cation and the degree of salt dissociation. It seems that in DMSO solution the lithium salt exists mainly as an ion pair stabilized by the chelation of a lithium cation with two oxygen atoms. The activation free energy the of formyl group rotation for this salt has been estimated to be 51.5 kJ/mol. The obtained results suggest that in all the investigated objects, including the free enolate ions, all atoms directly bonded to the carbonyl carbons lie near the same plane. Some observations concerning the chemical shift changes could indicate strong solvation of the anion of 1 by water molecules. Implications of the results obtained in this work for the inhibition mechanism of (4-hydroxyphenyl) pyruvate dioxygenase by NTBC are commented upon.
Home » Posts tagged 'JAPAN 2015'
Tag Archives: JAPAN 2015
Nitisinone, ニチシノン
Nitisinone
ニチシノン
Orfadin
Launched – 2002, NTBC
SC-0735
SYN-118
2-(alpha,alpha,alpha-Trifluoro-2-nitro-p-tuluoyl)-1,3-cyclohexanedione
2-(2-Nitro-4-trifluoromethylbenzoyl)cyclohexane-1,3-dione
Priority, Orphan
| Formula |
C14H10F3NO5
|
|---|---|
| CAS |
104206-65-7
|
| Mol weight |
329.2281
|
Nitisinone is a synthetic reversible inhibitor of 4-hydroxyphenylpyruvate dioxygenase. It is used in the treatment of hereditary tyrosinemia type 1. It is sold under the brand name Orfadin.
Nitisinone was first approved by the U.S. Food and Drug Administration (FDA) on January 18, 2002, then approved by the European Medicines Agency (EMA) on February 21, 2005. It was developed and marketed as Orfadin® by Swedish Orphan Biovitrum AB (SOBI) in the US .
The mechanism of action of nitisinone involves reversibile inhibition of 4-Hydroxyphenylpyruvate dioxygenase(HPPD). It is indicated for use as an adjunct to dietary restriction of tyrosine and phenylalanine in the treatment of hereditary tyrosinemia type 1 (HT-1).
Orfadin® is available as capsule for oral use, containing 2, 5 or 10 mg of free Nitisinone. The recommended initial dose is 1 mg/kg/day divided into two daily doses. Maximum dose is 2 mg/kg/day.
Nitisinone was launched in 2002 by Swedish Orphan (now Swedish Orphan Biovitrum) in a capsule formulation as an adjunct to dietary restriction of tyrosine and phenylalanine in the treatment of hereditary tyrosinemia type I. In 2015, this product was launched in Japan for the same indication. The same year, an oral suspension formulation for pediatric patients was registered in the E.U., and launch took place in the United Kingdom shortly after. This formulation was approved in 2016 in the U.S. for the same indication. In 2016, nitisinone tablet formulation developed by Cycle Pharmaceuticals was approved in Canada (this formulation is also available also in the U.S.).
Indication
Used as an adjunct to dietary restriction of tyrosine and phenylalanine in the treatment of hereditary tyrosinemia type 1.
Associated Conditions
EU

Nitisinone MendeliKABS
22 June 2017 EMA/CHMP/502860/2017
Product name, strength, pharmaceutical form: Orfadin • Marketing authorisation holder: Swedish Orphan Biovitrum International AB • Date of authorisation: 21/02/2005
Procedure No. EMEA/H/C/004281/0000
During the meeting on 22 June 2017, the CHMP, in the light of the overall data submitted and the scientific discussion within the Committee, issued a positive opinion for granting a Marketing authorisation to Nitisinone MendeliKABS.
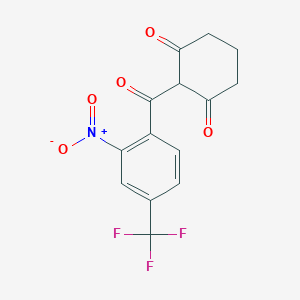
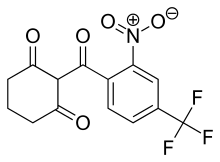
The chemical name of nitisinone is 2-[2-Nitro-4-(trifluoromethyl)benzoyl]-1,3-cyclohexanedione corresponding to the molecular formula C14H10F3NO5. It has a relative molecular mass of 329.23 g/mol and the following structure: Figure 1. Structure of nitisinone.
Nitisinone appears as off-white to yellowish non-hygroscopic fine crystalline powder. It is practically insoluble in unbuffered water. It is freely soluble in dichloromethane, sparingly soluble in ethyl alcohol, slightly soluble in isopropyl alcohol and 70% aqueous isopropyl alcohol and in pH 6.8 phosphate buffer, very slightly soluble in pH 4.5 acetate buffer and practically insoluble at pH 1.1. Solubility in acidified aqueous media depends on the acid counter ion. Solubility increases with increasing pH. Its pKa was found to be around 10. Nitisinone is achiral and does not show polymorphism.
ALSO
2005
Nitisinone is a white to yellowish-white crystalline powder poorly soluble in water. The active substance is a weak acid and it is highly soluble in the pH range 4.5-7.2 in phosphate buffer solutions. Nitisinone has the chemical name 2-(2-nitro-4-trifluoromethylbenzoyl)-cyclohexane-1,3-dione. It does not show polymorphism.
US FDA
https://www.accessdata.fda.gov/drugsatfda_docs/nda/2016/206356Orig1s000ChemR.pdf
Company: Swedish Orphan Biovitrum AB
Application No.: 206356Orig1
Approval Date: April 22, 2016
- Approval Letter(s) (PDF)
- Printed Labeling (PDF)
- Summary Review (PDF)
- Officer/Employee List (PDF)
- Cross Discipline Team Leader Review (PDF)
- Chemistry Review(s) (PDF)
- Pharmacology Review(s) (PDF)
- Clinical Pharmacology Biopharmaceutics Review(s) (PDF)
- Proprietary Name Review(s) (PDF)
- Other Review(s) (PDF)
- Administrative Document(s) & Correspondence (PDF)
Nitisinone (INN), also known as NTBC (an abbreviation of its full chemical name) is a medication used to slow the effects of hereditary tyrosinemia type 1. Since its first use for this indication in 1991, it has replaced liver transplantation as the first-line treatment for this rare condition. It is also being studied in the related condition alkaptonuria. It is marketed under the brand name Orfadin by the company Swedish Orphan Biovitrum (Sobi); it was first brought to market by Swedish Orphan International. It was originally developed as a candidate herbicide.
Uses
Nitisinone is used to treat hereditary tyrosinemia type 1, in combination with restriction of tyrosine in the diet.[1][2][3]
Since its first use for this indication in 1991, it has replaced liver transplantation as the first-line treatment for this rare condition.[4] I It is marketed under the brand name Orfadin.
It has been demonstrated that treatment with nitisinone can reduce urinary levels of homogentisic acid in alkaptonuria patients by 95%.[5] A series of clinical trials run by DevelopAKUre to determine whether nitisinone is effective at treating the ochronosis suffered by patients with alkaptonuria are ongoing.[6] If the trials are successful, DevelopAKUre will try to get nitisinone licensed for use by alkaptonuria patients.[7]
Mechanism of action
The mechanism of action of nitisinone involves reversibile inhibition of 4-Hydroxyphenylpyruvate dioxygenase (HPPD).[8][9] This is a treatment for patients with Tyrosinemia type 1 as it prevents the formation of maleylacetoacetic acid and fumarylacetoacetic acid, which have the potential to be converted to succinyl acetone, a toxin that damages the liver and kidneys.[4] This causes the symptoms of Tyrosinemia type 1 experienced by untreated patients.[10]
Alkaptonuria is caused when an enzyme called homogentisic dioxygenase (HGD) is faulty, leading to a buildup of homogenisate.[11]Alkaptonuria patients treated with nitisinone produce far less HGA than those not treated (95% less in the urine),[5] because nitisinone inhibits HPPD, resulting in less homogenisate accumulation. Clinical trials are ongoing to test whether nitisinone can prevent ochronosisexperienced by older alkaptonuria patients.[6]
Adverse effects
Nitisinone has several negative side effects; these include but are not limited to: bloated abdomen, dark urine, abdominal pain, feeling of tiredness or weakness, headache, light-colored stools, loss of appetite, weight loss, vomiting, and yellow-colored eyes or skin.[12]
Research
Nitisinone is being studied as a treatment for alkaptonuria.[13]
Research at the National Institutes of Health (NIH) has demonstrated that nitisinone can reduce urinary levels of HGA by up to 95% in patients with alkaptonuria. The primary parameter of the NIH trial was range of hip motion, for which the results were inconclusive.[citation needed]
Research done using alkaptonuric mice has shown that mice treated with nitisinone experience no ochronosis in knee joint cartilage. In contrast, all of the mice in the untreated control group developed ochronotic knee joints.[14]
The efficacy of Nitisinone is now being studied in a series international clinical trials called DevelopAKUre.[15] The studies will recruit alkaptonuria patients in Europe.[16] A larger number of patients will be recruited in these trials than in the previous NIH trial.[17] The trials are funded by the European Commission.[18]
Nitisinone has been shown to increase skin and eye pigmentation in mice, and has been suggested as a possible treatment for oculocutaneous albinism.[19][20]
History
Nitisinone was discovered as part of a program to develop a class of herbicides called HPPD inhibitors. It is a member of the benzoylcyclohexane-1,3-dione family of herbicides, which are chemically derived from a natural phytotoxin, leptospermone, obtained from the Australian bottlebrush plant (Callistemon citrinus).[21] HPPD is essential in plants and animals for catabolism, or breaking apart, of tyrosine.[22] In plants, preventing this process leads to destruction of chlorophyll and the death of the plant.[22] In toxicology studies of the herbicide, it was discovered that it had activity against HPPD in rats[23] and humans.[24]
In Type I tyrosinemia, a different enzyme involved in the breakdown of tyrosine, fumarylacetoacetate hydrolase is mutated and doesn’t work, leading to very harmful products building up in the body.[1] Fumarylacetoacetate hydrolase acts on tyrosine after HPPD does, so scientists working on making herbicides in the class of HPPD inhibitors hypothesized that inhibiting HPPD and controlling tyrosine in the diet could treat this disease. A series of small clinical trials attempted with one of their compounds, nitisinone, were conducted and were successful, leading to nitisinone being brought to market as an orphan drug Swedish Orphan International,[8] which was later acquired by Swedish Orphan Biovitrum (Sobi).
Sobi is now a part of the DevelopAKUre consortium. They are responsible for drug supply and regulatory support in the ongoing clinical trials that will test the efficiacy of nitisinone as a treatment for alkaptonuria.[25] It is hoped that if the trials are successful, nitisinone could also be licensed for treatment of alkaptonuria.[7]
Generic versions
There is no generic version of Orfadin in G7 countries. Prior to the market authorization of MDK-Nitisinone in Canada, the only Nitisinone product available globally was Orfadin.[26]Until recently, Nitisinone was not approved in Canada where it was distributed for over 20 years via a Health Canada Special Access Program. In September 2016, MendeliKABS was granted approval of a Priority New Drug Submission (PNDS) by Health Canada for a bioequivalent generic version of Orfadin capsules (MDK-Nitisinone). In November 2016 Cycle Pharma was also granted approval of a PNDS by Health Canada for Nitisinone tablets that are bioequivalent to Orfadin capsules.[27] SOBI was granted approval of a PNDS in December 2016.[28]
PAPER
1H NMR, 13C NMR, and Computational DFT Studies of the Structure of 2-Acylcyclohexane-1,3-diones and Their Alkali Metal Salts in Solution
2-(2-Nitro-4-trifluoromethylbenzoyl)cyclohexane-1,3-dione (NTBC; 3). The compound was prepared in the same manner as 2. The synthesis of an appropriate benzoic acid derivative was started from the transformation of commercially available 2-nitro-4-trifluoromethylaniline into benzonitrile by the classical Sandmeyer method. Then the nitrile was hydrolyzed in 65% sulfuric acid to give 2-nitro-4-trifluoromethylbenzoic acid.13 The obtained triketone 3 had a mp of 140−142 °C (lit.14 141−143 °C). For NMR data, see Supporting Information….. https://pubs.acs.org/doi/suppl/10.1021/jo060583g/suppl_file/jo060583gsi20060420_080852.pdf
NMR data for 2-(2-nitro-4-trifluoromethylbenzoyl)cyclohexane-1,3-dione, 3, in CDCl3

1 H NMR: 16.25 (s, 1H, OH), 8.47 (ddq, 1H, H10, J10,12=1.7 Hz, J10,13=0.4 Hz, J10,F=0.7 Hz), 7.94 (ddq, 1H, H12, J12,13=8.0 Hz, J12,F=0.7 Hz), 7.39 (ddq, 1H, H13, J13,F=0.8 Hz), 2.81 (t-like m, 2H, H4, H4’, JH4,H4’= -18.8 Hz, JH4,H5=5.4 Hz, JH4,H5’=7.3 Hz, JH4,H6=0.7 Hz, JH4,H6’= -0.8 Hz), 2.37 (tlike m, 2H, H6, H6’, JH6,H6’= -16.5 Hz, JH6,H5=4.6 Hz, JH6,H5’=8. 5 Hz), 2.04 (pentet-like m, 2H, H5, H5’, JH5,H5’= -13.6 Hz.

13C NMR: 196.3 (s, C(O)Ph), 195.8 (s, C3), 194.1 (s, C1), 145.5 (s, C9), 139.7 (s, C8), 132.0 (q, C11, J11,F=34.3 Hz), 130.8 (q, C12 J12,F=3.5 Hz), 127.7 (s, C13), 122.6 (q, CF3, JC,F=272.9 Hz), 121.1 (q, C10, J10,F=3.9 Hz), 112.7 (s, C2), 37.3 (s, C6) 31.6 (s, C4), 19.1 (s, C5).
PATENT
EP 186118
US 4780127



The condensation of cyclohexane-1,3-dione (I) with 2-nitro-4-(trifluoromethyl)benzoyl chloride by means of TEA in dichloromethane gives the target Nitisinone.EP 0186118
JP 1986152642, US 4774360, US 4780127
Nitisinone
-
- Synonyms:NTBC, SC 0735
- ATC:A16AX04
- Use:treatment of inherited tyrosinemia type I
- Chemical name:2-[2-nitro-4-(trifluoromethyl)benzoyl]-1,3-cyclohexanedione
- Formula:C14H10F3NO5
- MW:329.23 g/mol
- CAS-RN:104206-65-7
Substance Classes
Synthesis Path
Substances Referenced in Synthesis Path
| CAS-RN | Formula | Chemical Name | CAS Index Name |
|---|---|---|---|
| 504-02-9 | C6H8O2 | cyclohexane-1,3-dione | 1,3-Cyclohexanedione |
| 81108-81-8 | C8H3ClF3NO3 | 2-nitro-4-trifluoromethylbenzoyl chloride |
Trade Names
| Country | Trade Name | Vendor | Annotation |
|---|---|---|---|
| D | Orfadin | Orphan Europe | |
| USA | Orfadin | Swedish Orphan ,2002 |
Formulations
- cps. 2 mg
References
-
- WO 9 300 080 (ICI; 7.1.1993; appl. 18.6.1992; GB-prior. 24.6.1991).
- US 4 774 360 (Stauffer Chemical; 27.9.1988; appl. 29.6.1987).
-
synergistic herbicidal combination:
- WO 9 105 469 (Hoechst AG; 2.5.1991; appl. 12.10.1990; D-prior. 18.10.1989).
-
preparation of benzoylcyclohexanedione herbicides:
- US 4 780 127 (Stauffer Chemical; 25.10.1988; appl. 30.6.1986; USA-prior. 25.3.1982).
-
certain 2-(2-nitrobenzoyl)-1,3-cyclohexanediones:
- EP 186 118 (Stauffer Chemical; 2.7.1986; appl. 18.12.1985; USA-prior. 20.12.1984).
-
stable herbicidal compositions:
- WO 9 727 748 (Zeneca; 7.8.1997; appl. 3.2.1997; USA-prior. 2.2.1996).
PATENT
https://patents.google.com/patent/US9783485B1/en
NTBC is a drug marketed by Swedish Orphan Biovitrum International AB under the brand name Orfadin® and it is used to slow the effects of hereditary tyrosinemia type 1 (HT-1) in adult and pediatric patients. It has been approved by FDA and EMA in January 2002 and February 2005 respectively.
HT-1 disease is due to a deficiency of the final enzyme of the tyrosine catabolic pathway fumarylacetoacetate hydrolase. NTBC is a competitive inhibitor of 4-hydroxyphenylpyruvate dioxygenase (HPPD), an enzyme which precedes fumarylacetoacetate hydrolase. By inhibiting the normal catabolism of tyrosine in patients with HT-1, NTBC prevents the accumulation of the toxic intermediates maleylacetoacetate and fumarylacetoacetate, that in patients with HT-1 are converted to the toxic metabolites succinylacetone and succinylacetoacetate, the former inhibiting the porphyrin synthesis pathway leading to the accumulation of 5-aminolevulinate.
Usefulness of NTBC in the treatment of further diseases has also been documented. A non-comprehensive list is reported hereinafter.
Effectiveness of Orfadin® in the treatment of diseases where the products of the action of HPPD are involved (e.g., HT-1) has been described notably in EP0591275B1 corresponding to U.S. Pat. No. 5,550,165B1. Synthesis of NTBC is also described in this patent.
WO2011106655 reports a method for increasing tyrosine plasma concentrations in a subject suffering from oculocutaneous/ocular albinism, the method comprising administering to the subject a pharmaceutically acceptable composition comprising NTBC in the range of between about 0.1 mg/kg/day to about 10 mg/kg/day.
U.S. Pat. No. 8,354,451B2 reports new methods of combating microbial infections due to fungi or bacteria by means of administration to a subject of a therapeutically active amount of NTBC.
WO2010054273 discloses NTBC-containing compositions and methods for the treatment and/or prevention of restless leg syndrome (RLS).
EP1853241B1 claims the use of NTBC in the treatment of a neurodegenerative disease, notably Parkinson disease.
Introne W. J., et al., disclosed usefulness of nitisinone in the treatment of alkaptonuria (Introne W. J., et al., Molec. Genet. Metab., 2011, 103, 4, 307). The key step of the synthesis reported in EP0591275B1 (now propriety of Swedish Orphan Biovitrum International AB, SE), involves the reaction of 2-nitro-4-trifluromethylbenzoyl chloride and cyclohexane-1,3-dione in the presence of triethylamine and then use of acetone cyanohydrin in order to promote the rearrangement of the key intermediate enol ester. After washing and extraction from CH2Cl2, the crude product is recrystallized from ethyl acetate to get the desired 2-(2-nitro-4-trifluoromethylbenzoyl)-1,3-cyclohexanedione as a solid having a melting point of 88-94° C.
Another patent (U.S. Pat. No. 4,695,673) filed in name of Stauffer Chemical Company disclosed a process of synthesis of acylated 1,3-dicarbonyl compounds in which the intermediate enol ester is isolated prior to its rearrangement into the final product, said rearrangement making use of a cyanohydrin compound derived from alkali metal, methyl alkyl ketone, benzaldehyde, cyclohexanone, C2-C5aliphatic aldehyde, lower alkyl silyl or directly by using hydrogen cyanide.
Yet another patent (U.S. Pat. No. 5,006,158) filed in name of ICI Americas Inc. disclosed a process similar to the one disclosed in U.S. Pat. No. 4,695,673 wherein the intermediate enol ester was isolated prior to its rearrangement into the final product by use of potassium cyanide. Said reaction can optionally be done by concomitant use of a phase transfer catalyst such as Crown ethers. The preferred solvent for conducting such a reaction is 1,2-dichloroethane.
Still a further patent (EP0805791) filed in name of Zeneca Ltd disclosed an alternative synthesis of nitisinone involving the reaction of 1,3-cyclohexanedione and variously substituted benzoyl chloride in the presence of sodium or potassium carbonate in CH3CN or DMF. Best yields were obtained using CH3CN as solvent and sodium carbonate as the base. Reaction was performed at 55-57° C. in 17 hours.
It is well known that one of the problems of the actual drug formulation (i.e., Orfadin® capsules) is its chemical instability. Indeed, even if Orfadin® has to be stored in a refrigerator at a temperature ranging from 2° C. to 8° C., its shelf life is of only 18 months. After first opening, the in-use stability is a single period of 2 months at a temperature not above 25° C., after which it must be discarded. It will be evident that such storage conditions have an impact in the distribution chain of the medicine, in terms of costs and also in terms of logistics for the patient. Therefore, there is an urgent need of more stable formulations, both from a logistic supply chain point of view, and from the patient compliance point of view. Since the formulation of Orfadin® contains only the active ingredient and starch as excipient, relative instability may be attributed to the active pharmaceutical ingredient itself; in other words it can derive from the way it is synthesized and/or the way it is extracted from the reaction mixture, and/or the way it is finally crystallized. Furthermore, some impurities may contribute to render the final product less stable overtime. Consequently, it is of major importance to identify a process of synthesis and/or a crystallization method that enable the reliable production of a highly pure and stable product.
Impurities as herein-above mentioned can derive either from the final product itself (through chemical degradation) or directly from the starting materials/solvents used in the process of synthesis. Regarding the latter option, it is therefore primordial to ascertain that at each step, impurities are completely removed in order not to get them at the final stage, also considering that some of them could potentially be cyto/genotoxic.
The impurities correlated to nitisinone can be either derived from the starting materials themselves (i.e., impurities 1 and 2) or obtained as side products during the process of synthesis and/or under storage conditions (i.e., impurities 3 to 5) and are the following:
-
- 2-nitro-4-(trifluoromethyl) benzoic acid (Impurity no 1),
- 1,3-cyclohexanedione (CHD) (Impurity no 2),
- 4-(trifluoromethyl)salicylic acid (Impurity no 3),
- 2-[3-nitro-4-(trifluoromethyl)benzoyl]-1,3-cyclohexanedione (Impurity no 4), and
- 6-trifluoromethyl-3,4-dihydro-2H-xanthene-1,9-dione (Impurity no 5).
Impurity-2, impurity-3, and impurity-5 have been previously reported in WO2015101794. Strangely, impurity-4 has never been reported, even if it is an obvious side-product which can easily be formed during the coupling reaction between 1,3-cyclohexanedione and 2-nitro-4-(trifluoromethyl) benzoic acid, the latter being not 100% pure but containing some amount of regioisomer 3-nitro-4-(trifluoromethyl) benzoic acid.
Potential genotoxicity of impurity no 4 which possesses an aromatic nitro moiety was assessed using in-silico techniques and resulted to be a potential genotoxic impurity. According to the FDA ICH M7 guidelines, daily intake of a mutagenic impurity (Threshold of Toxicological Concern, TTC) in an amount not greater than 1.5 μg per person is considered to be associated with a negligible risk to develop cancer over a lifetime of exposure. Consequently, assuming a daily dose of 2 mg/kg, for a person weighing 70 kg, the maximum tolerated impurity content of such a compound would be of about 11 ppm, as calculated according to the equation underneath.
concentration limit ( ppm ) = T T C ( µg / day ) Dose ( g / day )
It is therefore of paramount importance to ensure that the process of synthesis of nitisinone and the purification steps of the same give rise to an API devoid of such impurity no 4, or at least far below the threshold of 11 ppm as indicated above. The skilled person will understand that total absence of said impurity is highly desirable.
It is well known in the pharmaceutical field that investigation of potential polymorphism of a solid API is of crucial importance and is also recommended by major regulatory authorities such as FDA.
Notwithstanding the fact that nitisinone has been used for years to treat HT-1 patients, it appears that no NTBC formulation fully satisfies the requisites of stability and/or compliance standard for the patients. Therefore, there is an unmet medical need of long-term pure and stable formulations.
Example 1
Thionyl chloride (162 g, 1.36 mol) was added dropwise into a suspension of 2-nitro-4-trifluoromethylbenzoic acid (228 g, 0.97 mol) in toluene (630 g) at 80° C. The thus obtained solution was kept under stirring at 80° C. for 20 hours, and then cooled to 50° C. The volatiles were removed under reduced pressure in order to get the expected 2-nitro-4-trifluoromethylbenzoyl chloride as an oil. The latter, cooled to 25° C. was added dropwise to a suspension of 1,3-cyclohexanedione (109 g, 0.97 mol) and potassium carbonate (323 g, 2.33 mol) in CH3CN (607 g). After 18 h the mixture was diluted with water (500 ml) and slowly acidified to about pH=1 with HCl 37%. The mixture was then warmed to about 55° C. and the phases were separated. The organic layer was washed with a 10% aqueous solution of sodium chloride and then, concentrated under reduced pressure at a temperature below 55° C. to reach a volume of 380 ml. The thus obtained mixture was stirred at 55° C. for 1 h and then cooled to 0° C. in 16 to 18 h. The resulting solid was filtered and rinsed several times with pre-cooled (0° C.) toluene. The wet solid was dried at 60° C. under vacuum for 6 h to provide nitisinone (164 g) as a white to yellowish solid with a purity of 98.4% as measured by HPLC and a content of potentially genotoxic impurity no 4 of 6.1 ppm measured by HPLC/MS.
Example 2
Nitisinone as obtained from example 1 (164 g) was added to a 3/1 (w/w) mixture of CH3CN/toluene (volume of solvent: 638 ml). The mixture was warmed gently to about 55° C. under stirring until solids were completely dissolved. The solution was then concentrated under reduced pressure maintaining the internal temperature below 50° C. to reach a volume of 290 ml. Then, more toluene (255 g) was added and the solution was concentrated again under reduced pressure until the residual volume reached 290 ml. The solution was heated to about 55° C. for 1 h and successively cooled slowly in 10 to 12 h to 10° C. The resulting solid was filtered and rinsed several times with pre-cooled (0° C.) toluene. The wet solid was dried at about 60° C. under vacuum for 4 h to provide nitisinone (136 g) as a white to yellowish solid, with a purity of 99.94% and a 99.8% assay measured by HPLC and a d(90) particle size between 310 and 350 μm. The content of potential genotoxic impurity no 4 resulted below 1 ppm.
CLIP

References
- ^ Jump up to:a b National Organization for Rare Disorders. Physician’s Guide to Tyrosinemia Type 1Archived 2014-02-11 at the Wayback Machine.
- Jump up^ “Nitisinone (Oral Route) Description and Brand Names”. Mayoclinic.com. 2015-04-01. Retrieved 2015-06-04.
- Jump up^ Sobi Orfadin® (nitisinone)
- ^ Jump up to:a b McKiernan, Patrick J (2006). “Nitisinone in the Treatment of Hereditary Tyrosinaemia Type 1”. Drugs. 66 (6): 743–50. doi:10.2165/00003495-200666060-00002. PMID 16706549.
- ^ Jump up to:a b Introne, Wendy J.; Perry, Monique B.; Troendle, James; Tsilou, Ekaterini; Kayser, Michael A.; Suwannarat, Pim; O’Brien, Kevin E.; Bryant, Joy; Sachdev, Vandana; Reynolds, James C.; Moylan, Elizabeth; Bernardini, Isa; Gahl, William A. (2011). “A 3-year randomized therapeutic trial of nitisinone in alkaptonuria”. Molecular Genetics and Metabolism. 103(4): 307–14. doi:10.1016/j.ymgme.2011.04.016. PMC 3148330
 . PMID 21620748.
. PMID 21620748. - ^ Jump up to:a b “About DevelopAKUre | DevelopAKUre”. Developakure.eu. 2014-06-20. Archived from the original on 2015-05-12. Retrieved 2015-06-04.
- ^ Jump up to:a b “A Potential Drug – Nitisinone”. Akusociety.org. Archived from the original on 2015-05-05. Retrieved 2015-06-04.
- ^ Jump up to:a b Lock, E. A.; Ellis, M. K.; Gaskin, P.; Robinson, M.; Auton, T. R.; Provan, W. M.; Smith, L. L.; Prisbylla, M. P.; Mutter, L. C.; Lee, D. L. (1998). “From toxicological problem to therapeutic use: The discovery of the mode of action of 2-(2-nitro-4-trifluoromethylbenzoyl)-1,3-cyclohexanedione (NTBC), its toxicology and development as a drug”. Journal of Inherited Metabolic Disease. 21 (5): 498–506. doi:10.1023/A:1005458703363. PMID 9728330.
- Jump up^ Kavana, Michael; Moran, Graham R. (2003). “Interaction of (4-Hydroxyphenyl)pyruvate Dioxygenase with the Specific Inhibitor 2-[2-Nitro-4-(trifluoromethyl)benzoyl]-1,3-cyclohexanedione†”. Biochemistry. 42 (34): 10238–45. doi:10.1021/bi034658b. PMID 12939152.
- Jump up^ “Newborn Screening”. Newbornscreening.info. 2013-05-14. Retrieved 2015-06-04.
- Jump up^ “What is Alkaptonuria?”. Akusociety.org. Archived from the original on 2015-04-05. Retrieved 2015-06-04.
- Jump up^ “Nitisinone (Oral Route) Side Effects”. Mayoclinic.com. 2015-04-01. Retrieved 2015-06-04.
- Jump up^ Phornphutkul, Chanika; Introne, Wendy J.; Perry, Monique B.; Bernardini, Isa; Murphey, Mark D.; Fitzpatrick, Diana L.; Anderson, Paul D.; Huizing, Marjan; Anikster, Yair; Gerber, Lynn H.; Gahl, William A. (2002). “Natural History of Alkaptonuria”. New England Journal of Medicine. 347 (26): 2111–21. doi:10.1056/NEJMoa021736. PMID 12501223.
- Jump up^ Preston, A. J.; Keenan, C. M.; Sutherland, H.; Wilson, P. J.; Wlodarski, B.; Taylor, A. M.; Williams, D. P.; Ranganath, L. R.; Gallagher, J. A.; Jarvis, J. C. (2013). “Ochronotic osteoarthropathy in a mouse model of alkaptonuria, and its inhibition by nitisinone”. Annals of the Rheumatic Diseases. 73 (1): 284–9. doi:10.1136/annrheumdis-2012-202878. PMID 23511227.
- Jump up^ “DevelopAKUre”. Developakure.eu. 2014-06-20. Retrieved 2015-06-04.
- Jump up^ “2012-005340-24”. Clinicaltrialsregister.eu. Retrieved 2015-06-04.
- Jump up^ “The Programme | DevelopAKUre”. Developakure.eu. 2014-06-20. Archived from the original on 2015-05-12. Retrieved 2015-06-04.
- Jump up^ “European Commission : CORDIS : Search : Simple”. Cordis.europa.eu. 2012-05-30. Retrieved 2015-06-04.
- Jump up^ Onojafe, Ighovie F.; Adams, David R.; Simeonov, Dimitre R.; Zhang, Jun; Chan, Chi-Chao; Bernardini, Isa M.; Sergeev, Yuri V.; Dolinska, Monika B.; Alur, Ramakrishna P.; Brilliant, Murray H.; Gahl, William A.; Brooks, Brian P. (2011). “Nitisinone improves eye and skin pigmentation defects in a mouse model of oculocutaneous albinism”. Journal of Clinical Investigation. 121 (10): 3914–23. doi:10.1172/JCI59372. PMC 3223618
 . PMID 21968110. Lay summary – ScienceDaily (September 26, 2011).
. PMID 21968110. Lay summary – ScienceDaily (September 26, 2011). - Jump up^ “Nitisinone for Type 1B Oculocutaneous Albinism – Full Text View”. ClinicalTrials.gov. Retrieved 2015-06-04.
- Jump up^ G. Mitchell, D.W. Bartlett, T.E. Fraser, T.R. Hawkes, D.C. Holt, J.K. Townson, R.A. Wichert Mesotrione: a new selective herbicide for use in maize Pest Management Science, 57 (2) (2001), pp. 120–128
- ^ Jump up to:a b Moran, Graham R. (2005). “4-Hydroxyphenylpyruvate dioxygenase”. Archives of Biochemistry and Biophysics. 433 (1): 117–28. doi:10.1016/j.abb.2004.08.015. PMID 15581571.
- Jump up^ Ellis, M.K.; Whitfield, A.C.; Gowans, L.A.; Auton, T.R.; Provan, W.M.; Lock, E.A.; Smith, L.L. (1995). “Inhibition of 4-Hydroxyphenylpyruvate Dioxygenase by 2-(2-Nitro-4-trifluoromethylbenzoyl)-cyclohexane-1,3-dione and 2-(2-Chloro-4-methanesulfonylbenzoyl)-cyclohexane-1,3-dione”. Toxicology and Applied Pharmacology. 133 (1): 12–9. doi:10.1006/taap.1995.1121. PMID 7597701.
- Jump up^ Lindstedt, Sven; Odelhög, Birgit (1987). “4-Hydroxyphenylpyruvate dioxygenase from human liver”. In Kaufman, Seymour. Metabolism of Aromatic Amino Acids and Amines. Methods in Enzymology. 142. pp. 139–42. doi:10.1016/S0076-6879(87)42021-1. ISBN 978-0-12-182042-8. PMID 3037254.
- Jump up^ “Others | DevelopAKUre”. Developakure.eu. 2014-06-20. Retrieved 2015-06-04.
- Jump up^ Pr MDK-Nitisinone Summary Basis of Decisions, Health Canada 2016. http://www.hc-sc.gc.ca/dhp-mps/prodpharma/sbd-smd/drug-med/sbd-smd-2016-mdk-nitisinone-190564-eng.php
- Jump up^ Pr Nitisinone Tablets Regulatory Decision Summary Health Canada, 2016. http://www.hc-sc.gc.ca/dhp-mps/prodpharma/rds-sdr/drug-med/rds-sdr-nitisinone-tab-193770-eng.php
- Jump up^ PrOrfadin Regulatory Decision Summary Health Canada, 2016. http://www.hc-sc.gc.ca/dhp-mps/prodpharma/rds-sdr/drug-med/rds-sdr-orfadin-193226-eng.php
External links
 |
|
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Consumer Drug Information |
| License data |
|
| Routes of administration |
Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | Approximately 54 h |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| ECHA InfoCard | 100.218.521 |
| Chemical and physical data | |
| Formula | C14H10F3NO5 |
| Molar mass | 329.228 g/mol |
| 3D model (JSmol) | |
////////////////Nitisinone, ニチシノン , Orfadin, FDA 2002, NTBC , SC-0735 , SYN-118 , JAPAN 2015, JAP 2015, EU 2005, Priority, Orphan
[O-][N+](=O)C1=C(C=CC(=C1)C(F)(F)F)C(=O)C1C(=O)CCCC1=O
Imidafenacin, イミダフェナシン

Imidafenacin
イミダフェナシン
Cas 170105-16-5
C20H21N3O, 319.408
APPROVED JAPAN 2015-07-29
|
4-(2-methyl-1H-imidazol-1-yl)-2,2-diphenylbutanamide
|

Imidafenacin (INN) is a urinary antispasmodic of the anticholinergic class. It’s molecular weight is 319.40 g/mol
| Imidafenacin (INN) is a urinary antispasmodic of the anticholinergic class. |
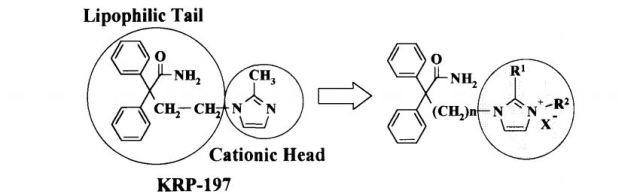
Kyorin and Ono have developed and launched imidafenacin, an oral M1 and M3 muscarinic receptor antagonist. Family members of the product case, WO9515951, expire in the US in 2019
Imidafenacin was approved by Pharmaceuticals Medical Devices Agency of Japan (PMDA) on Apr 18, 2007. It was marketed as Uritos® by Kyorin, and marketed as Staybla® by Ono.
Imidafenacin is a potent M1 and M3-subtype antagonist indicated for the treatment of urinary urgency, frequent urination and urgency urinary incontinence due to overactive bladder.
Uritos® is available as tablet for oral use, containing 0.1 mg of free Imidafenacin. The recommended dose is 0.1 mg twice daily, and it can be increased to 0.2 mg twice daily, if the efficacy was not enough.
Uritos® / Staybla®


MOA:Muscarinic acetylcholine receptor antagonist
Indication:Urinary incontinence; Urinary urgency and frequency

PAPER
Imidafenacin, the compound of formula (I), is an antimuscarinic agent marketed in Japan under the brand name Uritos® used to treat overactive bladder, a disease defined by the presence of urinary urgency, usually accompanied by frequency and nocturia, with or without urge incontinence. Overactive bladder dysfunction has a considerable impact on patient quality of life, although it does not affect survival.

(I)
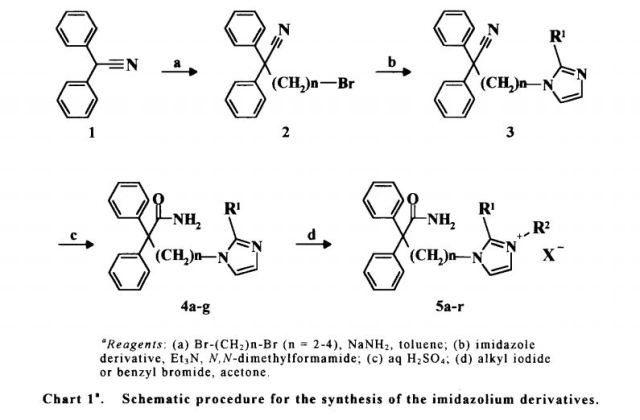
Synthesis of 4-(2-methyl-1 -imidazolyl)-2,2-diphenylbutanamide is first disclosed in Japanese patent JP3294961 B2 as shown in Scheme 1 . 4-bromo-2,2-diphenylbutanenitrile (II) is reacted with three equivalents of 2-methylimidazol, in dimethylformamide and in the presence of triethylamine as a base, to afford 4-(2-methyl-1 H-imidazol-1 -yl)-2,2-diphenylbutanenitrile, compound of formula (III), which is purified by column chromatography and, further, converted into its hydrochloride salt and recrystallized. Then, compound (III) is hydrolyzed with an excess of 70% sulfuric acid at 140-150 °C, followed by basification and recrystallization to provide imidafenacin (I), in an overall yield of only 25% (as calculated by data provided in docume

(III) (I)
Scheme 1
This route of document JP3294961 B2 implies several drawbacks. Firstly, purification of intermediate (III) is carried out by means of chromatographic methods, which are generally expensive, environmentally unfriendly and time consuming. Secondly, the hydrolysis of the nitrile group is carried out under strong acidic conditions and high temperature not convenient for industrial application.
Japanese document JP2003-201281 discloses a process for preparing imidafenacin as shown in Scheme 2. 4-bromo-2,2-diphenylbutanenitrile (II) is reacted, with five equivalents of 2-methylimidazol, which acts also as a base, in dimethylsufoxide to provide intermediate (III), which after an isolation step is further reacted with phosphoric acid in ethanol to provide the phosphate salt of 4-(2-methyl-1 H-imidazol-1-yl)-2,2-diphenylbutanenitrile. Hydrolysis with potassium hydroxide, followed by purification with a synthetic adsorbent provides imidafenacin (I) in a moderate overall yield

(II) (I)
Scheme 2
The use of a synthetic adsorbent is associated with problems with operativities and purification efficiencies from the viewpoint of industrial production, therefore, the process disclosed in document JP2003-201281 is not suitable for industrial application.
EP1845091 A1 discloses a process for preparing imidafenacin, according to previous document JP2003-201281 , however the purification step is carried out by either preparing the hydrochloride or the phosphate salt of imidafenacin followed by neutralization as shown in Scheme 3. Purified imidafenacin is provided in low yield, overall yield of about 31 % (as calculated by data provided in document EP1845091 A1 ). This process has several disadvantages. Firstly, EP1845091 A1 states that the penultimate intermediate, the 4-(2-methyl-1 H-imidazol-1 -yl)-2,2-diphenylbutanenitrile phosphate is hygroscopic, which implies handling problems. Secondly, the additional steps carried out for purification increases the cost of the final imidafenacin process and the pharmaceutical compositions containing it, which already resulted in expensive medications.

(II) (I)
HCI or
H3PO4

purified (I) HCI or Ή3ΡΟ4
Scheme 3
The intermediate phosphate salt of 4-(2-methyl-1 H-imidazol-1 -yl)-2,2-diphenylbutanenitrile obtained and used in prior art processes is a solid form having needle-shaped crystals, which are difficult to filtrate. Moreover, said needle-shaped crystals are very hygroscopic and unstable and transform over time to other solid forms. In addition, the water absorbed by this solid form described in the prior art may react with the intermediate to generate further impurities.
Therefore, there is still a need to develop an improved industrially feasible process for the manufacture of imidafenacin in good purity and good yield, involving the use of stable intermediates having also improved handling characteristics.
Example 1 :
Preparation of 4-(2-methyl-1 H-imidazol-1 -yl)-2,2-diphenylbutanenitrile phosphate in solid Form I
4-bromo-2,2-diphenylbutanenitrile (II, 1.000 Kg, 3.33 mol) and 2-methylimidazol (1 .368 Kg, 16.66 mol) were heated in DMSO (0.8 L) at 100-105 °C for 7 hours. The solution was then cooled to 20-25 °C and toluene (2 L) and water (4 L) were added and stirred for 30 minutes. After phase separation, the aqueous layer was extracted with toluene (1 L). Organic layers were combined and washed twice with water (2 x 1 L). Distillation of toluene provided 4-(2-methyl-1 H-imidazol-1-yl)-2,2-diphenylbutanenitrile as a brown oil (0.915 Kg), which was, then, dissolved in dry acetone (3 L) and water (0.1 L), heated to 40-45°C and seeded with 4-(2-methyl-1 H-imidazol-1 -yl)-2,2-diphenylbutanenitrile phosphate. A solution of orthophosphoric acid (0.391 Kg, 3.39 mol) in acetone (2 L) was then added dropwise, maintaining temperature at 40-45 °C. Once the addition was finished, the reaction mixture was maintained 1 hour at 40-45 °C, cooled to 20-25 °C and stirred for 1 hour. The solid was filtered, washed with acetone (1 L), suspended in 2-propanol (10 L), heated at 80 °C and 2 L of solvent were distilled. The obtained suspension was then seeded with 4-(2-methyl-1 H-imidazol-1 -yl)-2,2-diphenylbutanenitrile phosphate solid Form I and maintained at 80 °C for 5 hours. The suspension was cooled down to 20-25°C, filtered off, washed with 2-propanol (1 L) and, finally, dried (45 °C, 0.5 torr, 12 hours).
Yield: 0.967 Kg (73%)
HPLC: 99.5 %
KF: 0.2 %
Optical microscopy: plate-shaped crystal habit as substantially in accordance to Figure 2.
PSD: D90 of 105 m
PXRD: Crystalline solid form as substantially in accordance to Figure 3.
DSC (10 °C/min): Endothermic peak with onset at 177 °C (-1 18 J/g), as substantially in accordance to Figure 4.
TGA (10 °C/min): Decomposition starting at 180 °C.
DVS: No significant weight gain up to 90% of relative humidity. At this humidity, a total increase of only 0.45% in weight was observed.
SCXRD: Crystal structure substantially in accordance to Figure 5. There are not water or solvent molecules in the crystal structure.
PATENT
https://www.google.com/patents/CN103351344A?cl=en
Overactive Bladder (symptomatic overactive bladder, 0AB) is a common chronic lower urinary tract dysfunction. Its incidence, United States and Europe over 75 year-old male incidence up to 42%, slightly lower incidence of women 31%; the incidence of domestic in Beijing 50 years of age for men was 16.4% for women over the age of 18 mixed The overall incidence of urinary incontinence and urge incontinence was 40.4 percent, seriously affecting the physical and mental health of the patient, reduced quality of life. Common antimuscarinic drugs in vivo and in vivo M receptor in some or all of binding with different affinities to improve the symptoms of OAB, but will also cause many side effects, such as dry mouth, constipation, cognitive impairment , tachycardia, blurred vision and so on. Imidafenacin have diphenylbutanoic amide structure, is a new high anticholinergic drugs, which selectively acts on the M3 and Ml receptors, blocking the contraction of the detrusor choline, so detrusor relaxation, reduce side effects of drugs. Meanwhile imidafenacin inhibit smooth muscle of the bladder and inhibiting acetylcholine free dual role, and selectivity for the bladder stronger than the salivary glands.
imidafenacin is a new diphenylbutanoic amides from Japan Ono Pharmaceutical Co., Ltd. jointly developed with Kyorin Pharmaceutical anticholinergics, structure (I) as follows:
The goods listed in June 2007 in Japan under the trade name: STAYBLA, chemical name: 4- (2-methyl-1-imidazolyl) _2,2- diphenylbutyric amide.
At present the preparation imidafenacin few reports, can be summed up as the following ways:
China Patent CN10699098 reported to bromoethyl diphenyl acetonitrile and 2-methylimidazole as a raw material, at 150 ° C condition, after the reaction DMF / triethylamine system, sulfuric acid hydrolysis reuse imidafenacin. The reaction equation is as follows:
BACKGROUND OF THE INVENTION This two-step method was 24% overall yield is too low, and the second step of the reaction is difficult to control. And the reaction product was purified by column chromatography required to obtain a purified product, is not conducive to industrial production.
Chinese patent CN101362721A referred to as the hydrolysis conditions for the preparation of sulfuric acid and organic acid mixed use imidafenacin yield have mentioned the smell.
Although this method increases the yield, but still more by-product of the reaction, the product is not easy purification.
Japanese Patent No. JP2005 / 023216 proposes hydrolysis under alkaline environment, and the use of products and solutions of salts hydrochloride salt and then purified product.
This method improves the yield of the second step of the hydrolysis reaction and simplified purification methods. But the need to use this method to purify salt activated carbon, and filtration devices require more stringent; and a need to be re-crystallized salt solution salt after the operation, a total of four steps of unit operations. Process more cumbersome and more stringent requirements for equipment, it is not conducive to industrial scale production. In addition, the product is dried for a long time, still remaining after solvent treatment product obtained, the purity of the product is still low.
DETAILED DESCRIPTION
The following typical examples are intended to illustrate the present invention, simple replacement of skill in the art of the present invention or improvement made in all part of the present invention within the protection of technical solutions.
Example 1
4- (2-methyl-1-imidazolyl) -2,2-diphenyl butanamide hydrobromide. The 16.5 g (52 mmol) 4- (2- methyl-1-imidazolyl) -2,2-diphenyl butyramide crude into 100 mL of isopropanol, stirring was added 8.0 mL hydrobromic acid and isopropyl alcohol mixed solution (volume ratio of 1: 1), the solid gradually dissolved, was nearly colorless and transparent liquid. After maintaining the reaction mixture was stirred for half an hour, the reaction mixture was added to 100 mL of ethyl acetate, stirred for I hour at room temperature, solid precipitated. Filtration, and the cake was rinsed with an appropriate amount of ethyl acetate. The solid was collected, 40 ° C drying oven and dried to constant weight to give 19.5 g white 4- (2-methyl-1-imidazolyl) -2,2-diphenyl butyramide hydrobromide, yield 98.9%. ?] \ 1 .228.4-229.00C0MS (m / z): 320 [M + 1] +. 1H-NMR (DMS0-1 / 6, 400 MHz) δ: 2.25 (3H, s), 2.73-2.74 (2H, m), 3.68-3.91 (2H, m), 6.81 (1H, s), 7.28-7.35 (I OH, m), 7.39 (1H, s), 7.49 (1H, d, /=2.4 Hz), 7.55 (1H, d, J = 2.2 Hz), 14.39 (1¾ br s).
Example 2
4- (2-methyl-1-imidazolyl) -2,2-diphenyl butyramide. -2,2-Diphenyl butyric acid amide acetate was dissolved in 900 mL of water to 19.5 g (0.051mmol) obtained in Example 1 4- (2-methyl-1-imidazolyl) embodiment. Extracted with 900mL diethyl ether solution, collecting the inorganic layer. Was added to an aqueous solution of 200 mL of ethanol, was added to the system with stirring in an aqueous solution of KOH 2mol / L, there is a solid precipitated. The reaction was stirred I h after filtration. Cake was washed with 40% ethanol solution rinse, rinsed with water several times. Collect the cake, put 40 ° C drying oven dried to constant weight to give 14.8 g white 4- (2-methyl-1-imidazolyl) -2,2-diphenyl methylbutanamide, yield 91.0% (total yield 90% two steps). Μ.p.192.3-193.00C (CN101076521A 191-193O). MS (m / z): 320 [M + l] +. 1H-NMR (DMSO-J6, 400MHz) δ: 2.11 (3Η, s), 2.69-2.73 (2H, m), 3.61-3.65 (2H, m), 6.75 (1H, d, J = L OMHz), 7.01 (1H, br s), 7.04 (1H, d, J = L 0 MHz), 7.34-7.49 (11H, m).
Example 3
4- (2-methyl-1-imidazolyl) -2,2-diphenyl butyramide. The 14.5 g (0.045mmol) obtained in Example 4- (2-methyl-1-imidazolyl) -2,2-diphenyl butanamide 2 was added 116 mL of ethyl acetate was slowly heated to reflux reflux for 30 min, cooled to room temperature for crystallization 5 h. Suction filtered, the filter cake was rinsed with a small amount of ethanol, collected cake was put 40 ° C drying oven and dried to constant weight to give 13.4 g white 4- (2-methyl-1-imidazolyl) -2,2- diphenyl methylbutanamide refined products, yield 92.4% (three-step total yield 83.1%). Mp192.5-193 (TC (CN101076521A 191_193 ° C) .MS (m / z):.. 320 [M + 1] + 1H-NMR (DMSO-J6, 400 MHz) δ
2.11 (3H, 7.01 (1H,
s), 2.69-2.73 (2H, br s), 7.04 (1H, d,
m), 3.61-3.65 (2H, m), 6.75 (1H, J = L 0 MHz), 7.34-7.49 (11H, m).

PATENT
CN103772286A.
imidafenacin (Imidafenacin) is a new diphenylbutanoic amides from Japan Ono Pharmaceutical Co., Ltd. jointly developed with Kyorin Pharmaceutical anticholinergic drugs, bladder is highly selective for the treatment of overactive bladder, in 2007 in June in Japan. Its chemical name is 4- (2-methyl -1H- imidazol-1-yl) -2,2-diphenyl butyramide chemical structure shown by the following formula I:
Reported in U.S. Patent No. US5932607 imidafenacin preparation method, the method is based on 4-bromo-2 ‘2 ~ phenyl butyronitrile, 2-methylimidazole, triethylamine as raw materials, with DMF as a solvent at 150 ° C reaction 30h, to give the intermediate 4- (2-methyl-imidazol-1-yl) -2,2-diphenyl-butyronitrile, 77% yield, then body 140 ~ 150 ° C with 70% sulfuric acid The resulting intermediate hydrolyzed to the amide, after completion of the reaction required excess soda and sulfuric acid, the reaction is as follows:
Which preclude the use of the dilute sulfuric acid hydrolysis, although succeeded in getting the product, but the yield is very low, only 32%, greatly increasing the production cost, mainly due to 70% sulfuric acid, the reaction is difficult to control amide phase, the product will continue to acid hydrolysis byproducts, resulting in decreased yield.
European Patent No. EP1845091 reports imidafenacin Another preparation method, the method using potassium hydroxide and isopropyl alcohol 4- (2-methyl-imidazol-1-yl) diphenyl _2,2- Hydrolysis of nitrile to amide phosphates, and the crude product was converted to the hydrochloride or phosphate, and recrystallized to remove impurities and then basified imidafenacin obtained, which reaction is as follows:
This method uses a lot of bases, product purification is too much trouble, and the total yield of 45%.
Chinese Patent Publication No. CN102746235 also disclosed imidafenacin preparation method of 4- (2-methyl-1-yl) -2,2-diphenyl phosphate or nitrile salt in methanol / ethanol, dimethyl sulfoxide, and the presence of a base, with hydrogen peroxide in 40 ~ 60 ° C under through improved Radziszewski the target compound, the reaction is as follows:
The method used in the hydrogen peroxide solution, but a solution of hydrogen peroxide has strong oxidizing, and has a certain corrosive, inhalation of the vapor or mist respiratory irritation strong, direct eye contact with the liquid may cause irreversible damage and even blindness, security It is not high on the human body and environmentally unfriendly. Alkaline environment, easily decomposed hydrogen peroxide, as the temperature increases, the decomposition reaction increased, and therefore reaction requires a large excess of hydrogen peroxide solution.
The method comprises the steps of: (1) 4-Bromo-2,2-diphenyl-butyronitrile is hydrolyzed to the amide under basic conditions; (2) The obtained 4-bromo-2,2-diphenylbutyric amide is reacted with 2-methylimidazole to give the desired product.
Example 1
2L reaction flask was added 400mL of dry tetrahydrofuran, under a nitrogen atmosphere was added 60% sodium hydride (82.8g, 2.06mol), stirred to obtain a gray turbid solution A. With 400mL dry tetrahydrofuran was sufficiently dissolved diphenyl acetonitrile (200g, 1.04mol), I, 2- dibromoethane (204.2g, 1.08mol), to give a colorless clear liquid B; 5 ~ 15 ° C, a solution of turbid solution B dropwise to solution A, 10 ~ 15 ° C the reaction was incubated 6h, TLC until the reaction was complete, to the reaction system a small amount of water was added dropwise until no bubbles. After addition of 800mL water, 400mL ethyl acetate and stirred, liquid separation, the organic layer was washed with water, saturated sodium chloride solution, respectively, and the organic layer was dried over anhydrous sodium sulfate, suction filtered, concentrated under reduced pressure to give a yellow liquid 310g.
[0018] The resulting yellow liquid with 800mL 90% ethanol and stirred to dissolve at 40 ° C, then cooling and crystallization, filtration, 45 ° C and concentrated under reduced pressure to give a white solid 232.8g, 75% yield.
Preparation of bromo-2,2-diphenyl 4_ butanamide: [0019] Example 2
3L reaction flask was added 4-bromo-2,2-diphenyl-butyronitrile (15 (^, 0.511101), 7501 ^ 6mol / L KOH solution, 750mL dimethylsulfoxide and heated to 100 ~ 120 ° C under stirring The reaction, the reaction lh, until the reaction was complete by TLC after cooling to 40 V, add 2000mL water, 2000mL of methylene chloride was stirred, liquid separation, the organic layer was washed with water, washed with saturated sodium bicarbonate and sodium chloride solution, separated, dried over anhydrous The organic layer was dried over sodium sulphate, filtration, concentrated under reduced pressure to give brown oily liquid 161.92g, 96% yield.
Preparation of bromo-2,2-diphenyl 4_ butanamide: [0020] Example 3
3L reaction flask was added 4-bromo-2,2-diphenyl-butyronitrile (150g, 0.5mol), 666mL 6mol / L NaOH solution, 750mL dimethylsulfoxide, the reaction mixture was stirred and heated to 100 ~ 120 ° C under The reaction lh, until the reaction was complete by TLC after cooling to 40 ° C, add water 2000mL, 2000mL of methylene chloride was stirred, liquid separation, the organic layer was washed with water, washed with saturated sodium bicarbonate and sodium chloride solution, separated, dried over anhydrous sulfate sodium organic layer was dried, filtration, concentrated under reduced pressure to give brown oily liquid 146.73g, 87% yield.
Preparation of bromo-2,2-diphenyl 4_ butanamide: [0021] Example 4
The reaction was stirred 3L reaction flask was added 4-bromo-2,2-diphenyl-butyronitrile (15 (^, 0.511101), 8331 ^ 36% Na2CO3 solution, 750mL dimethylsulfoxide and heated to 100 ~ 120 ° C under The reaction lh, until the reaction was complete by TLC after cooling to 40 ° C, add water 2000mL, 2000mL of methylene chloride was stirred, liquid separation, the organic layer was washed with water, washed with saturated sodium bicarbonate and sodium chloride solution, separated, dried over anhydrous The organic layer was dried over sodium sulphate, filtration, concentrated under reduced pressure to give brown oily liquid 153.48g, yield 91%.
`[0022] Example 5: 4- (2-methyl-imidazol _1_ -1H- yl) butyramide _2,2_ diphenyl (imidafenacin) Preparation 5L reaction flask was added 4-bromo-2 2-diphenyl butyric amide (160g, L 5mol), 2- methyl imidazole (123g,
1.5mol), triethylamine (50.6g, 0.5mol), potassium iodide (5g, 0.03mol), fully dissolved with 1000mL DMF solution was heated to 120 ° C at a reaction 5h, until completion of the reaction by TLC, heating was stopped, to be After cooling, water was added 3000mL system stirred 0.5h, filtration, washed with water until the filtrate is neutral, concentrated under reduced pressure and dried to give a brown solid 146.14g, a yield of 91%.
[0023] Example 6: 4- (2-methyl-imidazol _1_ -1H- yl) butyramide _2,2_ diphenyl (imidafenacin) Preparation 5L reaction flask was added 4-bromo-2, 2- diphenyl butyramide (160g, 0.5mol), 2- methyl imidazole (82.1g,
1.011101), triethylamine (50.68,0.5mol), potassium iodide (5g, 0.03mol), fully dissolved with 1000mL DMF solution was heated to 120 ° C at a reaction 5h, until completion of the reaction by TLC, heating was stopped, the system was cooled until After adding 3000mL water, stirring 0.5h, filtration, washed with water until the filtrate is neutral, concentrated under reduced pressure and dried to give a brown solid 120.45g, 80% yield.
[0024] Example 7: 4- (2-methyl-imidazol _1_ -1H- yl) butyramide _2,2_ diphenyl (imidafenacin) Preparation 5L reaction flask was added 4-bromo-2, 2- diphenyl butyramide (160g, 0.5mol), 2_ methylimidazole (164.2g,
2.011101), triethylamine (50.68,0.5mol), potassium iodide (5g, 0.03mol), fully dissolved with 1000mL DMF solution was heated to 120 ° C at a reaction 5h, until completion of the reaction by TLC, heating was stopped, the system was cooled until After adding water, stirring 3000mL
0.5h, suction filtered, washed with water until the filtrate was neutral, and concentrated under reduced pressure, and dried to give a brown solid 141.33g, yield 88%.
[0025] Example 8: 4- (2-methyl imidazole -1H- _1_ group) _2,2_ diphenylbutanoic amide (imidafenacin) refining up to 80g microphone said that new crude added 300mL of absolute ethanol, the system was warmed to reflux, refluxed
0.5h, after cooling the ethanol was distilled off to IOOmL about 500mL of ethyl acetate was added to precipitate a white solid, a small amount of ethyl acetate and wash the filter cake, 45 ° C and dried in vacuo to give 74.6g of white crystals, yield 93%.

CLIP
| EP 0733621; US 5932607; US 6103747; WO 9515951 |


Alkylation of diphenylacetonitrile (I) with dibromoethane provided bromide (II). This was condensed with 2-methylimidazole (III) in the presence of Et3N in DMF to afford the substituted imidazole (IV). Finally, hydrolysis of the cyano group of (IV) with 70% sulfuric acid produced the target amide.

Treatment of acetonitrile derivative (I) with dibromoethane (II) in toluene in the presence of NaNH2 affords bromo compound (III), which is then condensed with imidazole derivative (IV) by means of Et3N in DMF to provide compound (V). Hydrolysis of the cyano group of (V) with aqueous H2SO4 yields amide derivative (VI), which is finally subjected to alkyl quaternization by reaction with bromobenzyl bromide (VI) in acetone to furnish the desired product.
Paper
Bioorganic & Medicinal Chemistry Letters 9 (1999) 3003-3008
PAPER
Bioorganic & Medicinal Chemistry 7 (1999) 1151±1161
4-(2-methyl-1-imidazolyl)- 2,2-diphenylbutyramide (2.02 g, 24%) as a colorless needle:
mp 189.0±190.0 C (from ethyl acetate:ethanol);
High MS (EI+) m/z calcd for C20H21N3O 319.1685, found 319.1671;
1 H NMR (400 MHz, CDCl3) d 2.23 (3H, s), 2.69±2.74 (2H, m), 3.77±3.82 (2H, m), 5.33 (1H, s), 5.49 (1H, s), 6.73 (1H, s), 6.85 (1H, s), 7.31±7.42 (10H, m).
PATENT
CN103880751A.
imidafenacin chemical name 4- (2-methyl–1H–1-yl) -2,2-diphenyl methylbutanamide (I).
In Patent JP93-341467, JP94-319355 and literature Bioorganic & Medicinal ChemistryLetters, 1999, vol.9,3003 – 3008 reported in the chemical synthesis routes to diphenyl acetonitrile (4) as the starting material,
Condensation and hydrolysis reaction step to give imidafenacin (1).
The new method is simple, mild reaction conditions, easy to control, good high yield and purity of the product, do not pollute the environment, suitable for industrial production.
[0012] The first method from 2-methylimidazole and I, 2- dibromoethane under phase transfer catalyst is tetrabutylammonium bromide (TBAB) and inorganic base catalyzed generate 1- (2-bromoethyl) – methyl -1H- imidazole (5), and diphenyl acetonitrile (4) a phase transfer catalyst and an inorganic base catalyzed condensation of 4- (2-methyl–1H- imidazol-1-yl) -2,2 – diphenylbutyronitrile hydrochloride (2), and then hydrolyzed to imidafenacin (I)
FIG. 1 imidafenacin IH-NMR spectrum
FIG. 2 imidafenacin 13C-NMR spectra
Examples I
1- (2-bromoethyl) -1H- -2_ methyl-imidazole (5) Preparation of
The 1,2_ dibromoethane (50ml), 2- methylimidazole (2.5g, 30.5mmol), tetrabutylammonium bromide (TBAB) (0.5g) and K2C03 (3.6g), K0H ( 4.6g) were added sequentially 100mL three-necked flask and stirred and heated to 50 ° C reaction 7h. Cooling to room temperature, the reaction solution was filtered, and the filtrate was washed with saturated aqueous sodium bicarbonate, dried over anhydrous sodium sulfate. Concentrated, added to a mixed solvent of isopropyl ether and ethyl acetate (3: 1) was stirred resolved crystal dissolved, to give the product 5.lg, yield 88.5%, mp.79_80 ° C.
Preparation of 4- (2-methyl-1-imidazolyl) -2,2-diphenyl-butyronitrile hydrochloride (2)
The diphenyl acetonitrile (5.8g, 30mmol) and 50% aqueous KOH (15ml), dimethyl sulfoxide (DMSO) (100ml), tetrabutylammonium bromide (TBAB) (0.9g) in toluene 50ml was added to the reaction flask and stirred for 0.5h in the 40 ° C. 1- (2-bromoethyl) -2-methyl -1H- imidazole (4) (5.lg, 27mmol), was heated to 20 ° C, the reaction was stirred, TLC tracking and monitoring the reaction was complete, the mixture was poured into 100mL water, extracted three times with ethyl acetate 240ml water phase. Washed three times with 300ml of water The organic phase was dried over anhydrous sodium sulfate, the organic phase was concentrated. Analytical crystal solution with hydrogen chloride ether solution, filtered crystals with a mixed solvent of isopropyl ether and recrystallized from ethyl acetate to give the condensation product of 4- (2-methyl-1-imidazolyl) -2,2-diphenylbutyric carbonitrile hydrochloride (2) of a white solid 7.lg, yield 77.8%, mp: 156.5-158 ° C. 1H-NMR (400MHz, CDCl3), δ (ppm): 7.35-7.42 (IOH, m), 6.90 (1H, s), 6.77 (1H, s), 3.90-3.94 (2H, m), 2.75-2.79 ( 2H, m), 2.25 (3H, s).
The preparation imidafenacin (I),
4- (2-methyl-1-imidazolyl) -2,2_ diphenyl butyronitrile hydrochloride (2) (8.78g, 26mmol) in 70% concentrated sulfuric acid (25ml) was added to the reaction bottle, the reaction was stirred at 90 ° C, the end of the reaction was monitored by TLC tracking. The reaction solution was poured into 120ml of water, solid sodium carbonate was added to adjust PH to weakly alkaline, sufficiently stirred. With 180ml of dichloromethane and 35ml of ethanol mixed solvent was extracted three times, the organic phase was washed with water, dried over anhydrous sodium sulfate, the organic phase was concentrated. The residue was mixed with a solvent of ethyl acetate and recrystallized from ethanol to give 4- (2-methyl-1-imidazolyl) 2,2-diphenyl butyramide 7.0g, yield 84.5%, mp: 188.0-190 (. TC.1H-NMR and 13C-NMR data are as follows (see Figure 1-2 spectra):
1H-NMR (CDC13,400ΜΗζ) δ: 2.209 (s, 3H, -CH3), 2.666-2.707 (t, 2H, -CH2-CH2-),
3.747-3.788 (t, 2H, -CH2-CH2 -), 5.341 (s, 1H, -NH -), 5.757 (s, 1H, -NH -), 6.699 (s, 1H, Ar-H)
, 6.828 (s, 1H, Ar-H), 7.287-7.390 (m, I OH, Ar-H).
[0030] 13C-NMR (CDC13,400MHz) δ: 12.17 (-CH3), 41.00 (-CH2 -), 43.74 (-CH2-), 59.44 (quaternary carbon, coupled with strong electron-withdrawing group), 119.08 (-C = C -), 126.95 (aromatic carbon), 127.88 (aromatic carbon), 128.52 (aromatic carbon), 129.10 (aromatic carbon), 142.61 (= CN), 144.54 (-C = N), 176.21 (carbonyl carbon).
Example 2
[0032] 1- (2-bromoethyl) -1H- -2_ methyl-imidazole (5) Preparation of
[0033] The 1,2_ dibromoethane (50ml), 2- methylimidazole (2.5g, 30.5mmol), tetrabutylammonium chloride (0.43g) and Na2CO3 (2.8g), NaOH (3.3g) followed by adding 100mL three-necked flask, stirred and heated to 40 ° C reaction 5h.Cooling to room temperature, the reaction solution was filtered, and the filtrate was washed with saturated aqueous sodium bicarbonate, dried over anhydrous sodium sulfate. Concentrated, added to a mixed solvent of isopropyl ether and ethyl acetate (3: 1) was dissolved with stirring parsing crystal give the product 4.9g, yield 85.1%, mp.79-80 ° C.
Preparation of [0034] 4- (2-methyl-1-imidazolyl) -2,2-diphenyl-butyronitrile hydrochloride (2)
[0035] A two phenylethyl chest (5.8g, 30mmol) and 50% aqueous NaOH (15ml), dimethylethylene Bitterness (DMSO) (100ml), tetrabutylammonium chloride (0.8g) was added to a toluene 50ml The reaction flask, stirred 0.5h in the 40 ° C. Join
1- (2-bromoethyl) -2-methyl -1H- imidazole (4) (5.lg, 27mmol), was heated to 60 ° C, the reaction was stirred, TLC tracking and monitoring the reaction was complete, the mixture was poured into 100mL of water and extracted three times with ethyl acetate 240ml water phase. Washed three times with 300ml of water The organic phase was dried over anhydrous sodium sulfate, the organic phase was concentrated. Solution of hydrogen chloride in ether solution with analytical crystal, crystals were filtered with a mixed solvent of isopropyl ether and recrystallized from ethyl acetate to give the condensation product of 4- (2-methyl-1-imidazolyl) -2,
2-phenyl-butyronitrile hydrochloride (2) as a white solid 7.0g, yield 76.8%, mp: 156.5-158 ° C. 1H-NMR (400MHz, CDCl3), δ (ppm): 7.35-7.42 (IOH, m), 6.90 (1H, s), 6.77 (1H, s), 3.90-3.94 (2H, m), 2.75-2.79 ( 2H, m), 2.25 (3H, s).
Preparation imidafenacin (I),
[0037] 4- (2-methyl-1-imidazolyl) -2,2_ diphenyl butyronitrile hydrochloride (2) (8.78g, 26mmol) in 70% concentrated sulfuric acid (25ml) was added to the reaction bottle, the reaction was stirred at 110 ° C, the end of the reaction was monitored by TLC tracking. The reaction solution was poured into 120ml of water, solid sodium carbonate was added to adjust PH to weakly alkaline, sufficiently stirred. With 180ml of dichloromethane and 35ml of ethanol mixed solvent was extracted three times, the organic phase was washed with water, dried over anhydrous sodium sulfate, the organic phase was concentrated. The residue was mixed with a solvent of ethyl acetate and recrystallized from ethanol to give 4- (2-methyl-1-imidazolyl) 2,2-diphenyl butyramide 7.2g, yield 86.8%, mp: 188.0-190 (. TC.1H-NMR and 13C-NMR data are as follows (see Figure 1-2 spectra):
1H-NMR (CDC13,400ΜΗζ) δ: 2.209 (s, 3H, -CH3), 2.666-2.707 (t, 2H, -CH2-CH2-),
3.747-3.788 (t, 2H, -CH2-CH2 -), 5.341 (s, 1H, -NH -), 5.757 (s, 1H, -NH -), 6.699 (s, 1H, Ar-H), 6.828 ( s, 1H, Ar-H), 7.287-7.390 (m, I OH, Ar-H).
13C-NMR (CDC13,400MHz) δ: 12.17 (-CH3), 41.00 (-CH2 -), 43.74 (-CH2-), 59.44 (quaternary carbon, coupled with strong electron-withdrawing group), 119.08 (-C = C -), 126.95 (aromatic carbon), 127.88 (aromatic carbon), 128.52 (aromatic carbon), 129.10 (aromatic carbon), 142.61 (= CN), 144.54 (-C = N), 176.21 (carbonyl carbon).
Example 3
[0041] 1- (2-bromoethyl) -1H- -2_ methyl-imidazole (5) Preparation of
[0042] The 1,2_ dibromoethane (50ml), 2- methylimidazole (2.5g, 30.5mmol), benzyltriethylammonium chloride (TEBA) (0.35g) and Na2CO3 (2.8g), Na0H (3.3g) were added sequentially 100mL three-necked flask, stirred and heated to 45 ° C reaction 4h. Cooling to room temperature, the reaction solution was filtered, washed with a saturated aqueous sodium bicarbonate paint filtrate was dried over anhydrous sodium sulfate. Concentrated, added to a mixed solvent of isopropyl ether and ethyl acetate (3: 1) was dissolved with stirring parsing crystal give the product 5.0g, yield 86.8%, mp.79-80. . .
Preparation of [0043] 4- (2-methyl-1-imidazolyl) -2,2-diphenyl-butyronitrile hydrochloride (2)
The diphenyl acetonitrile (5.8g, 30mmol) and 50% aqueous KOH (15ml), dimethyl sulfoxide (DMSO) (100ml), benzyltriethylammonium chloride (TEBA) (0.66g) 50ml Toluene was added to the reaction flask and stirred at 40 ° C under
0.5h0 was added 1- (2-bromoethyl) -2-methyl -1H- imidazole (4) (5.lg, 27mmol), was heated to 60 ° C, the reaction was stirred, TLC tracking and monitoring the reaction was complete, the mixture was poured into 100mL of water and extracted three times with ethyl acetate 240ml water phase. Washed three times with 300ml of water The organic phase was dried over anhydrous sodium sulfate, the organic phase was concentrated. Analytical crystal solution with hydrogen chloride ether solution, filtered crystals with a mixed solvent of isopropyl ether and recrystallized from ethyl acetate to give the condensation product of 4- (2-methyl-1-imidazolyl) -2,2-diphenylbutyric carbonitrile hydrochloride (2) as a white solid 7.0g, yield 76.8%, mp: 156.5-158. . . 1H-NmrgoomHzADCI3), δ (ppm): 7.35-7.42 (10H, m), 6.90 (1H, s), 6.77 (1H, s), 3.90-3.94 (2H, m), 2.75-2.79 (2H, m) , 2.25 (3H, s).
Preparation imidafenacin (I),
[0046] 4- (2-methyl-1-imidazolyl) -2,2_ diphenyl butyronitrile hydrochloride (2) (8.78g, 26mmol) in 70% concentrated sulfuric acid (25ml) was added to the reaction bottle, the reaction was stirred at 100 ° C, the end of the reaction was monitored by TLC tracking. The reaction solution was poured into 120ml of water, solid sodium carbonate was added to adjust PH to weakly alkaline, sufficiently stirred. With 180ml of dichloromethane and 35ml of ethanol mixed solvent was extracted three times, the organic phase was washed with water, dried over anhydrous sodium sulfate, the organic phase was concentrated. The residue was mixed with a solvent of ethyl acetate and recrystallized from ethanol to give 4- (2-methyl-1-imidazolyl) 2,2-diphenyl butyramide 7.lg, yield 85.5%, mp: 188.0-190. (TC.1H-NMR and 13C-NMR data are as follows (see Figure 1-2 spectra):
[0047] 1H-NMR (CDC13,400ΜΗζ) δ: 2.209 (s, 3H, -CH3), 2.666-2.707 (t, 2H, -CH2-CH2-),
3.747-3.788 (t, 2H, -CH2-CH2 -), 5.341 (s, 1H, -NH -), 5.757 (s, 1H, -NH -), 6.699 (s, 1H, Ar-H)
, 6.828 (s, 1H, Ar-H), 7.287-7.390 (m, I OH, Ar-H).
13C-NMR (CDC13,400MHz) δ: 12.17 (-CH3), 41.00 (-CH2 -), 43.74 (-CH2-), 59.44 (quaternary carbon, coupled with strong electron-withdrawing group), 119.08 (-C = C -), 126.95 (aromatic carbon), 127.88 (aromatic carbon), 128.52 (aromatic carbon), 129.10 (aromatic carbon), 142.61 (= CN), 144.54 (-C = N), 176.21 (carbonyl carbon).
Example 41- (2-bromoethyl) -1H- -2_ methyl-imidazole (5) Preparation of
[0051] The 1,2_ dibromoethane (50ml), 2- methylimidazole (2.5g, 30.5mmol), tetrabutylammonium bromide (TBAB) (0.5g) and K2C03 (3.6g), K0H ( 4.6g) were added sequentially 100mL three-necked flask, stirred and heated to 60 ° C reaction 4h.Cooling to room temperature, the reaction solution was filtered, and the filtrate was washed with saturated aqueous sodium bicarbonate, dried over anhydrous sodium sulfate. Concentrated, added to a mixed solvent of isopropyl ether and ethyl acetate (3: 1) was dissolved with stirring parsing crystal give the product 4.5g, yield 78.1%, mp.79_80 ° C.
Preparation of [0052] 4- (2-methyl-1-imidazolyl) -2,2-diphenyl-butyronitrile hydrochloride (2)
[0053] The diphenyl acetonitrile (5.8g, 30mmol) and 50% aqueous KOH (15ml), dimethyl sulfoxide (DMSO) (100ml), tetrabutylammonium bromide (TBAB) (0.9g) in toluene 50ml was added to the reaction flask and stirred for 0.5h in the 40 ° C. Plus Λ 1- (2- bromoethyl) -2-methyl -1H- imidazole (4) (5.lg, 27mmol), was heated to 100 ° C, the reaction was stirred, TLC tracking and monitoring the reaction was complete, the mixture was poured into 100mL of water and extracted three times with ethyl acetate 240ml water phase. Washed three times with 300ml of water The organic phase was dried over anhydrous sodium sulfate, the organic phase was concentrated. Analytical crystal solution with hydrogen chloride ether solution, filtered crystals with a mixed solvent of isopropyl ether and recrystallized from ethyl acetate to give the condensation product of 4- (2-methyl-1-imidazolyl) -2,2-diphenylbutyric carbonitrile hydrochloride (2) as a white solid 6.7g, yield 73.4%, mp: 156.5-158 ° C. 1H-NMR (400MHz, CDCl3), δ (ppm): 7.35-7.42 (IOH, m), 6.90 (1H, s), 6.77 (1H, s), 3.90-3.94 (2H, m), 2.75-2.79 ( 2H, m), 2.25 (3H, s).
The preparation imidafenacin (I),
4- (2-methyl-1-imidazolyl) -2,2_ diphenyl butyronitrile hydrochloride (2) (8.78g, 26mmol) in 70% concentrated sulfuric acid (25ml) was added to the reaction bottle, the reaction was stirred at 150 ° C at the end of the reaction was monitored TLC tracking. The reaction solution was poured into 120ml of water, solid sodium carbonate was added to adjust PH to weakly alkaline, sufficiently stirred. With 180ml of dichloromethane and 35ml of ethanol mixed solvent was extracted three times, the organic phase was washed with water, dried over anhydrous sodium sulfate, the organic phase was concentrated. The residue was mixed with a solvent of ethyl acetate and recrystallized from ethanol to give 4- (2-methyl-1-imidazolyl) 2,2-diphenyl butyramide 6.2g, yield 74.8%, mp: 188.0-190 (. TC.1H-NMR and 13C-NMR data are as follows (see Figure 1-2 spectra):
[0056] 1H-NMR (CDC13,400ΜΗζ) δ: 2.209 (s, 3H, -CH3), 2.666-2.707 (t, 2H, -CH2-CH2 -), 3.747-3.788 (t, 2H, -CH2-CH2 -), 5.341 (s, 1H, -NH -), 5.757 (s, 1H, -NH -), 6.699 (s, 1H, Ar-H)
, 6.828 (s, 1H, Ar-H), 7.287-7.390 (m, I OH, Ar-H).
13C-NMR (CDC13,400MHz) δ: 12.17 (-CH3), 41.00 (-CH2 -), 43.74 (-CH2-), 59.44 (quaternary carbon, coupled with strong electron-withdrawing group), 119.08 (-C = C -), 126.95 (aromatic carbon), 127.88 (aromatic carbon), 128.52 (aromatic carbon), 129.10 (aromatic carbon), 142.61 (= CN), 144.54 (-C = N), 176.21 (carbonyl carbon).
Example 5
1- (2-bromoethyl) -1H- -2_ methyl-imidazole (5) Preparation of
The 1,2_ dibromoethane (50ml), 2- methylimidazole (2.5g, 30.5mmol), tetrabutylammonium bromide (TBAB) (0.5g) and K2CO3 (3.6g), K0H ( 4.6g) were added sequentially 100mL three-necked flask, stirred and heated to 20 ° C reaction 10h. Cooling to room temperature, the reaction solution was filtered, washed with a saturated aqueous sodium bicarbonate paint filtrate was dried over anhydrous sodium sulfate. Concentrated, added to a mixed solvent of isopropyl ether and ethyl acetate (3: 1) was dissolved with stirring parsing crystal give the product 4.1g, yield 71.2%, mp.79-80. . .
Preparation of [0061] 4- (2-methyl-1-imidazolyl) -2,2-diphenyl-butyronitrile hydrochloride (2)
[0062] The diphenyl acetonitrile (5.8g, 30mmol) and 50% aqueous KOH (15ml), dimethyl sulfoxide (DMSO) (100mL), tetrabutylammonium bromide (TBAB) (0.9g) in toluene 50ml was added to the reaction flask and stirred at 20 ° C in Ih. Join
1- (2-bromoethyl) -2-methyl -1H- imidazole (4) (5.lg, 27mmol), was heated to 60 ° C, the reaction was stirred, TLC tracking and monitoring the reaction was complete, the mixture was poured into 100mL of water and extracted three times with ethyl acetate 240ml water phase. Washed three times with 300ml of water The organic phase was dried over anhydrous sodium sulfate, the organic phase was concentrated. Solution of hydrogen chloride in ether solution with analytical crystal, crystals were filtered with a mixed solvent of isopropyl ether and recrystallized from ethyl acetate to give the condensation product of 4- (2-methyl-1-imidazolyl) -2,
2-phenyl-butyronitrile hydrochloride (2) as a white solid 6.5g, yield 71.2%, mp: 156.5-158 ° C. 1H-NMR (400MHz, CDCl3), δ (ppm): 7.35-7.42 (IOH, m), 6.90 (1H, s), 6.77 (1H, s), 3.90-3.94 (2H, m), 2.75-2.79 ( 2H, m), 2.25 (3H, s).
[0063] Preparation of imidafenacin (I), [0064] 4- (2-methyl-1-imidazolyl) -2,2-diphenyl-butyronitrile hydrochloride (2) (8.78g, 26mmol ) and 70% of concentrated sulfuric acid (25ml) was added to the reaction flask, and stirred in at 50 ° C, the end of the reaction was monitored by TLC tracking. The reaction solution was poured into 120ml of water, solid sodium carbonate was added to adjust PH to weakly alkaline, sufficiently stirred. With 180ml of dichloromethane and 35ml of ethanol mixed solvent was extracted three times, the organic phase was washed with water, dried over anhydrous sodium sulfate, the organic phase was concentrated. The residue was mixed with a solvent of ethyl acetate and recrystallized from ethanol to give 4- (2-methyl-1-imidazolyl) 2,2-diphenyl butyramide 6.6g, yield 79.7%, mp: 188.0-190 (. TC.1H-NMR and 13C-NMR data are as follows (see Figure 1-2 spectra):
[0065] 1H-NMR (CDC13,400ΜΗζ) δ: 2.209 (s, 3H, -CH3), 2.666-2.707 (t, 2H, -CH2-CH2-),
3.747-3.788 (t, 2H, -CH2-CH2 -), 5.341 (s, 1H, -NH -), 5.757 (s, 1H, -NH -), 6.699 (s, 1H, Ar-H)
, 6.828 (s, 1H, Ar-H), 7.287-7.390 (m, I OH, Ar-H).
[0066] 13C-NMR (CDC13,400MHz) δ: 12.17 (-CH3), 41.00 (-CH2 -), 43.74 (-CH2-), 59.44 (quaternary carbon, coupled with strong electron-withdrawing group), 119.08 (-C = C -), 126.95 (aromatic carbon), 127.88 (aromatic carbon), 128.52 (aromatic carbon), 129.10 (aromatic carbon), 142.61 (= CN), 144.54 (-C = N), 176.21 (carbonyl carbon).
[0067] Example 6
[0068] 1- (2-bromoethyl) -1H- -2_ methyl-imidazole (5) Preparation of
[0069] The 1,2_ dibromoethane (50ml), 2- methylimidazole (2.5g, 30.5mmol), benzyltriethylammonium chloride (TEBA) (0.35g) and Na2CO3 (2.8g), Na0H (3.3g) were added sequentially 100mL three-necked flask and stirred and heated to 40 ° C reaction 8h. Cooling to room temperature, the reaction solution was filtered, washed with a saturated aqueous sodium bicarbonate paint filtrate was dried over anhydrous sodium sulfate. Concentrated, added to a mixed solvent of isopropyl ether and ethyl acetate (3: 1) was dissolved with stirring parsing crystal give the product 4.4g, yield 76.4%, mp.79-80. . .
Preparation of [0070] 4- (2-methyl-1-imidazolyl) -2,2-diphenyl-butyronitrile hydrochloride (2)
[0071] The diphenyl acetonitrile (5.8g, 30mmol) and 50% aqueous KOH (15ml), dimethyl sulfoxide (DMSO) (100ml), benzyltriethylammonium chloride (TEBA) (0.66g) 50ml Toluene was added to the reaction flask and stirred 0.5h0 1- (2-bromoethyl) -2-methyl -1H- imidazole (4) at 40 ° C (5.lg, 27mmol), was heated to 50 ° C, the reaction mixture was stirred, TLC tracking and monitoring the reaction was complete, the mixture was poured into 100mL of water and extracted three times with ethyl acetate. The aqueous phase was 240ml. Washed three times with 300ml of water The organic phase was dried over anhydrous sodium sulfate, the organic phase was concentrated. Analytical crystal solution with hydrogen chloride ether solution, filtered crystals with a mixed solvent of isopropyl ether and recrystallized from ethyl acetate to give the condensation product of 4- (2-methyl-1-imidazolyl) -2,2-diphenylbutyric carbonitrile hydrochloride (2) as a white solid 6.8g, yield 74.6%, mp: 156.5-158. . . 1H-NmrgoomHzADCI3), δ (ppm): 7.35-7.42 (10H, m), 6.90 (1H, s), 6.77 (1H, s), 3.90-3.94 (2H, m), 2.75-2.79 (2H, m) , 2.25 (3H, s).
[0072] Preparation imidafenacin (I),
[0073] 4- (2-methyl-1-imidazolyl) -2,2_ diphenyl butyronitrile hydrochloride (2) (8.78g, 26mmol) in 70% concentrated sulfuric acid (25ml) was added to the reaction bottle, the reaction was stirred at 80 ° C, the end of the reaction was monitored by TLC tracking. The reaction solution was poured into 120ml of water, solid sodium carbonate was added to adjust PH to weakly alkaline, sufficiently stirred. With 180ml of dichloromethane and 35ml of ethanol mixed solvent was extracted three times, the organic phase was washed with water, dried over anhydrous sodium sulfate, the organic phase was concentrated. The residue was mixed with a solvent of ethyl acetate and recrystallized from ethanol to give 4- (2-methyl-1-imidazolyl) 2,2-diphenyl butyramide 6.Sg, yield 81.8%, mp: 188.0-190. (TC.1H-NMR and 13C-NMR data are as follows (see Figure 1-2 spectra):
[0074] 1H-NMR (CDC13,400ΜΗζ) δ: 2.209 (s, 3H, -CH3), 2.666-2.707 (t, 2H, -CH2-CH2-),
3.747-3.788 (t, 2H, -CH2-CH2 -), 5.341 (s, 1H, -NH -), 5.757 (s, 1H, -NH -), 6.699 (s, 1H, Ar-H)
, 6.828 (s, 1H, Ar-H), 7.287-7.390 (m, I OH, Ar-H). [0075] 13C-NMR (CDC13,400MHz) δ: 12.17 (_CH3), 41.00 (-CH2 -), 43.74 (-CH2-), 59.44 (quaternary carbon, coupled with strong electron-withdrawing group), 119.08 (-C = C -), 126.95 (aromatic carbon), 127.88 (aromatic carbon), 128.52 (aromatic carbon), 129.10 (aromatic carbon), 142.61 (= CN), 144.54 (-C = N), 176.21 (carbonyl carbon).

PATENT
CN 105399678
https://www.google.com/patents/CN105399678A?cl=en
CLIP
http://dmd.aspetjournals.org/content/35/9/1624/T3.expansion.html
TABLE 3
Chemical shifts of protons and carbons in 1H NMR and 13C NMR spectra of major (M-11b) and minor (M-11a) constituents of reference products obtained from imidafenacin
|
Position of Proton |
1H NMR Data (in D2O) |
||
|---|---|---|---|
| Major Constituent (M-11b)
|
Minor Constituent (M-11a)
|
||
| 1 | 2.18a (3Hb, sc) | 2.11a (3Hb, sc) | |
| 2 | 2.82 (2H, m) | 2.79 (2H, m) | |
| 3 | 3.45 (2H, m) | 3.41 (2H, m) | |
| 5 | 5.26 (1H, s) | 5.43-5.47d (1H, d, J = 8.1e) | |
| 6 | 5.33 (1H, s) | 5.43-5.47d (1H, d, J = 8.1e) | |
| 8, 9, and 10 | 7.39-7.49 (10H, m) | 7.40-7.48 (10H, m) | |
| 13
|
8.45 (1.3H, s)
|
8.45 (2H, s)
|
|
| Position of Carbon
|
13C-NMR Data (in D2O)xc
|
||
|---|---|---|---|
| Major Constituent (M-11b)
|
Minor Constituent (M-11a)
|
||
| 1 | 14.61a | 14.48a | |
| 2 | 39.04 | 38.49 | |
| 3 | 43.49 | 42.90 | |
| 4 | 61.95-61.99f | 61.95-61.99f | |
| 5 | 87.61 | 80.22 or 85.78f | |
| 6 | 93.10 | 80.22 or 85.78f | |
| 7 | 144.2-144.4f | 144.2-144.4f | |
| 8, 9, and 10 | 130.7-131.8f | 130.7-131.8f | |
| 11 | 170.8 | 169.5 | |
| 12 | 181.9-182.2f | 181.9-182.2f | |
| 13
|
173.8
|
173.8
|
|
-
↵ a Chemical shifts are reported in parts per million.
-
↵ b Intensities are represented as number of protons.
-
↵ c Multiplicity: s, singlet; d, doublet; m, multiplet.
-
↵ d These proton signals could not be distinguished.
-
↵ e Coupling constants (J) are given in Hertz.
-
↵ f These carbon signals could not be distinguished.

FIG. 1.
Chemical structures of [14C]imidafenacin and postulated metabolites, and their fragment ions. *, 14C labeled position; broken line, precursor and product ions obtained by collision-induced dissociation in LC/MS/MS.
| Cited Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| CN101076521A * | Dec 13, 2005 | Nov 21, 2007 | 杏林制药株式会社 | Process for producing muscarine receptor antagonist and intermediate therefor |
| CN102746235A * | Jul 20, 2012 | Oct 24, 2012 | 北京科莱博医药开发有限责任公司 | Improved method for preparing imidafenacin |
| CN103275007A * | May 27, 2013 | Sep 4, 2013 | 朱雪琦 | Pyrazole derivatives and preparation method thereof |
| US7351429 | 2008-04-01 | Oral solid preparation |
| US2008004247 | 2008-01-03 | Combinations of Statins with Bronchodilators |
| US2007270436 | 2007-11-22 | NOVEL AMINO- AND IMINO-ALKYLPIPERAZINES |
| US2007219237 | 2007-09-20 | Chromane Derivatives |
| US2007185129 | 2007-08-09 | ACID ADDITION SALTS OF THIENOPYRANCARBOXAMIDE DERIVATIVES |
| US2007092566 | 2007-04-26 | Oral sustained-release tablet |
| US2006188554 | 2006-08-24 | Transdermal absorption preparation |
| EP0733621 | 2002-05-15 | NOVEL IMIDAZOLE DERIVATIVE AND PROCESS FOR PRODUCING THE SAME |
| US6103747 | 2000-08-15 | Imidazole derivatives and process for preparing the same |
| US5932607 | 1999-08-03 | Imidazole derivatives and process for preparing the same |
| Patent ID | Date | Patent Title |
|---|---|---|
| US2015064232 | 2015-03-05 | TRANSDERMAL ABSORPTION PREPARATION |
| US8729056 | 2014-05-20 | Preventive and/or therapeutic agent of hand-foot syndrome |
| US8722133 | 2014-05-13 | Method for production of orally rapidly disintegrating tablet comprising imidafenacin as active ingredient |
| US2013211352 | 2013-08-15 | PERCUTANEOUSLY ABSORBED PREPARATION |
| US2013211353 | 2013-08-15 | PERCUTANEOUS ABSORPTION TYPE FORMULATION |
| US8343544 | 2013-01-01 | Oral sustained-release tablet |
| US2012289563 | 2012-11-15 | COMBINATIONS OF IMIDAFENACIN AND SALIVARY STIMULANTS FOR THE TREATMENT OF OVERACTIVE BLADDER |
| US8247415 | 2012-08-21 | Hydroxymethyl pyrrolidines as [beta]3 adrenergic receptor agonists |
| US8158152 | 2012-04-17 | Lyophilization process and products obtained thereby |
| US8124633 | 2012-02-28 | HYDROXYMETHYL ETHER HYDROISOINDOLINE TACHYKININ RECEPTOR ANTAGONISTS |
- Kobayashi F, Yageta Y, Segawa M, Matsuzawa S: Effects of imidafenacin (KRP-197/ONO-8025), a new anti-cholinergic agent, on muscarinic acetylcholine receptors. High affinities for M3 and M1 receptor subtypes and selectivity for urinary bladder over salivary gland. Arzneimittelforschung. 2007;57(2):92-100. [PubMed:17396619 ]
- Miyachi H, Kiyota H, Uchiki H, Segawa M: Synthesis and antimuscarinic activity of a series of 4-(1-Imidazolyl)-2,2-diphenylbutyramides: discovery of potent and subtype-selective antimuscarinic agents. Bioorg Med Chem. 1999 Jun;7(6):1151-61. [PubMed:10428387 ]
Reference
- Kobayashi F, Yageta Y, Segawa M, Matsuzawa S (2007). “Effects of imidafenacin (KRP-197/ONO-8025), a new anti-cholinergic agent, on muscarinic acetylcholine receptors. High affinities for M3 and M1 receptor subtypes and selectivity for urinary bladder over salivary gland”. Arzneimittelforschung. 57 (2): 92–100. doi:10.1055/s-0031-1296589. PMID 17396619.
- Miyachi H, Kiyota H, Uchiki H, Segawa M (June 1999). “Synthesis and antimuscarinic activity of a series of 4-(1-Imidazolyl)-2,2-diphenylbutyramides: discovery of potent and subtype-selective antimuscarinic agents”. Bioorg. Med. Chem. 7 (6): 1151–61.doi:10.1016/S0968-0896(99)00003-6. PMID 10428387.
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
4-(2-methyl-1H-imidazol-1-yl)-2,2-diphenylbutanamide
|
|
| Clinical data | |
| Routes of administration |
Oral |
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number | 170105-16-5 |
| ATC code | none |
| PubChem | CID 6433090 |
| ChemSpider | 4938278 |
| UNII | XJR8Y07LJO |
| ChEMBL | CHEMBL53366 |
| Chemical data | |
| Formula | C20H21N3O |
| Molar mass | 319.40 g/mol |
//////////イミダフェナシン , D06273, KRP-197, KRP 197, ONO-8025, ONO 8025, UNII:XJR8Y07LJO, Kyorin, Ono ,Imidafenacin, 170105-16-5, JAPAN 2015, Uritos® , Staybla®
Olanexidine, オラネキシジングルコン酸塩

Olanexidine Gluconate
OPB-2045G, Gluconate olanexidin, Olanedine, OPB-2045, OPB 2045G,
(Olanedine®)Approved in Japan PMDA 2015-07-03, Olanedine® by Otsuka

A disinfectant uesd to prevent of postoperative bacterial infections.


CAS .146510-36-3(Olanexidine free form),
Imidodicarbonimidic diamide, N-((3,4-dichlorophenyl)methyl)-N’-octyl
| C17H27Cl2N5 | |
| Formula Weight: | 372.341 |
CAS 799787-53-4(Olanexidine Gluconate)
| 568.49 | |
| Formula | C17H27Cl2N5 ● C6H12O7 |
1-(3,4-Dichlorobenzyl)-5-octylbiguanide mono-D-gluconate
| オラネキシジングルコン酸塩 Olanexidine Gluconate  C17H27Cl2N5▪C6H12O7 : 568.49 [799787-53-4] |
Indication:Bacterial infection
Otsuka (Originator)

- Marketed Bacterial infections

Most Recent Events
- 16 Sep 2015 Launched for Bacterial infections (Prevention) in Japan (Topical)
- 03 Jul 2015 Registered for Bacterial infections (Prevention) in Japan (Topical) – First global approval
- 30 Sep 2014 Preregistration for Bacterial infections (Prevention) in Japan (Topical)

SEE ALSO
Olanexidine hydrochloride [USAN]
146509-94-6 HCL
RN: 218282-71-4 HCL HYDRATE
UNII: R296398ALN
Molecular Formula, C17-H27-Cl2-N5.Cl-H.1/2H2-O
Molecular Weight, 835.6192
Imidodicarbonimidic diamide, N-((3,4-dichlorophenyl)methyl)-N’-octyl-, monohydrochloride, hydrate (2:1)
INTRODUCTION
Olanexidine gluconate was approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on Jul 03, 2015. It was developed and marketed as Olanedine® by Otsuka in Japan.
Olanexidine gluconate is an antiseptic/disinfectant compound with potent bactericidal activity against Gram-negative and Gram-positive bacteria, for use in preparing patients for surgery and preventing of postoperative bacterial infections.
Olanedine® is available as topical solution (1.5%), containing 3 g/200 mL, 0.15 g/10 mL and 0.375 g/25 mL, and the recommendation is applying appropriate amount of the drug.
PRODUCT PATENT
Kazuyoshi Miyata, Yasuhide Inoue, Akifumi Hagi, Motoya Kikuchi, Hitoshi Ohno, Kinji Hashimoto, Kinue Ohguro, Tetsuya Sato,Hidetsugu Tsubouchi, Hiroshi Ishikawa,Takashi Okamura, Koushi Iwata,
| Otsuka Pharmaceutical Co., Ltd., Otsuka Pharmaceutical Factory, Inc. |
SYNTHESIS
PATENT
CN1065453A
http://www.google.com.au/patents/CN1065453A?cl=en
WO2008026757A1
https://google.com/patents/WO2008026757A1?cl=en
Example 1: l-cyano-3-n-octylguanidine
A 7.00-kg quantity of Compound (4) (54.16 mol) was dissolved in 105 liters of ethyl acetate, and the resulting mixture was cooled to 5°C or below. A 2.66-kg quantity of concentrated sulfuric acid (27.12 mol) was added thereto dropwise at a temperature of 4O0C or below while stirring. To the thus- obtained suspension of 1/2 sulfate of Compound (4) was added 5.06 kg of sodium dicyanamide (56.83 mol), and the resulting suspension was heated under reflux for 7 hours. The reaction solution was cooled to 400C or below, and 70 liters of water was added thereto. Subsequently, the resulting solution was heated to 80 to 900C (internal temperature) to distill the ethyl acetate off. The remaining liquid was cooled to 400C or below, and 70 liters of toluene was then added thereto, followed by the extraction of 1-cyano — 3-n-octyl guanidine at about 500C. The extracted toluene layer was washed with 35 liters of water at about 500C and cooled to 100C or below, followed by stirring for about 30 minutes. The resulting precipitated crystals were separated and washed with 7 liters of toluene. The resulting crystals were dried at 400C for 7.5 hours, yielding l-cyano-3-n- octylguanidine. 2007/067107
-16-
Yield: 9.11 kg (The yield was 85.7% based on the Compound(4).) White crystals having a melting point of 69 to 740C (no clear melting point was observed)
IR(KBr) spectrum: 3439, 3296, 2916, 2164, 1659, 1556, 1160, 718, and 572 cm“1
Thermogravimetric measurement/differential thermal analysis: 73.5°C (weak), an endothermic peak at 77.50C
1H-NMR(CDCl3) spectrum: 0.88 ppm (t, J = 6.6 Hz, 3H), 1.20-1.38 ppm (m, 10H), 1.43-1.62 ppm (m, 2H), 3.17 ppm (dd, J = 6.9 Hz, J = 6.0 Hz, 2H), 5.60-5.70 ppm (bs, 2H), 5.80-5.95 ppm (bs, IH)
Reference Example 2: Acidolysis of 1- (3,4-dichlorobenzyl) -5- octylbiguanide dihydrochloride
A 1-g quantity of 1- (3, 4-dichlorobenzyl) -5-octyl biguanide dihydrochloride was dissolved in 15 ml of 10% ethanol, followed by refluxing for 5 hours. HPLC analysis was conducted under the conditions described below.
The yield of 1-[N- (3,4-dichlorobenzyl) carbamoyl-3- octyl]guanidine (holding time: 9.84 minutes) was 0.91%, and the yield of 1- (N-octyl-carbamoyl) -3- (3, 4-dichlorobenzyl) guanidine
(holding time: 10.54 minutes) was 0.22%.
HPLC analysis conditions:
Column: YMC AM302 4.6 mm I. D. x 150 mm
Eluate: MeCN/0.05 M aqueous solution of sodium 1- octanesulfonate/acetic acid = 700/300/1
Detector: UV 254 nm
The physical property values of the resulting 1-[N- (3,4- dichlorobenzyl) carbamoyl-3-octyl] guanidine were as follows: NMR (DMSO-de) δ: 0.86 (3H, t, J = 6.0 Hz), 1.07-1.35 (1OH, m) , 1.35-1.49 (2H, m) , 2.95-3.15 (2H, m) , 4.12 (2H, d, J = 6.3 Hz), 6.78-7.40 (4H, m) , 7.23 (IH, dd, J = 2.1 Hz, J = 8.4 Hz), 7.46 (IH, d, J = 2.1 Hz), 7.54 (IH, d, J = 8.4 Hz)
The physical property values of the resulting 1- (N-octyl- carbamoyl) -3- (3, 4-dichlorobenzyl) guanidine were as follows: NMR (DMSO-d6) δ: 0.85 (3H, t, J = 6.6 Hz), 1.02-1.40 (12H, m) , 2.89-2.95 (2H, m) , 4.33 (2H, bs) , 5.76-7.00 (4H, m) , 7.28 (IH, dd, J = 2.1 Hz, J = 8.1 Hz), 7.52 (IH, d, J = 2.1 Hz), 7.58 (IH, d, J = 8.1 Hz)
Example 1: 1- (3, 4-dichlorobenzyl) -5-octylbiguanide monohydrochloride 1/2 hydrate
A 9.82-g quantity of Compound (2) (0.05 mol) and 10.63 g of 3, 4-dichlorobenzylamine (0.05 mol) were added to 49 ml of butyl acetate, followed by refluxing for 6 hours. The reaction solution was concentrated under reduced pressure, and a mixture of 12 ml of water and 47 ml of isopropyl alcohol was added and dissolved into the remainder. To the thus-obtained solution was added, dropwise, 10.13 g of concentrated hydrochloric acid. The resulting mixture was stirred at 28 to 300C for 30 minutes, and the precipitated crystals were then filtered out. The thus- obtained crystals were washed with a small amount of isopropyl alcohol, yielding 23.42 g of (non-dried) 1- (3, 4-dichlorobenzyl) – 5-octylbiguanide dihydrochloride. The resulting crystals were suspended in 167 ml of water without drying, the suspension was then stirred at 25 to 27°C for 2 hours, followed by separation of the crystals by filtration. The thus-obtained crystals were washed with a small amount of water and dried at 400C for 20 hours, yielding 17.05 g of 1- (3, 4-dichlorobenzyl) -5-octyl biguanide monohydrochloride 1/2 hydrate having a purity of 99.9% at a yield of 81.6%.
Example 2 : 1- (3, 4-dichlorobenzyl) -5-octylbiguanide dihydrochloride
A 100-g quantity of Compound (4) (0.774 mol) was dissolved in 1 liter of n-butyl acetate, and 37.6 g of concentrated sulfuric acid (0.383 mol) was added thereto while stirring. To the thus-obtained suspension of 1/2 sulfate of Compound (4) was added 68.9 g of sodium dicyanamide (0.774 mol), 7107
-18- and the resulting suspension was heated under reflux for 3 hours. The reaction solution was cooled to about 200C, and the organic layer thereof was sequentially washed with about 500 ml each of (i) 5% hydrochloric acid, (ii) 5% aqueous caustic soda solution, (iii) 5% aqueous sodium bicarbonate solution, and (iv) water.
To the thus-obtained n-butyl acetate solution of Compound (2) were added 118.5 g of Compound (3) (0.673 mol) and then 58.4 ml of concentrated hydrochloric acid while stirring. The reaction solution was heated, and about 800 ml of n-butyl acetate was distilled off under atmospheric pressure (ordinary pressure) , followed by heating the reaction solution under reflux for 3.5 hours . Subsequently, the reaction solution was cooled to about 400C, and 900 ml of isopropanol, 100 ml of water, and 134 ml of concentrated hydrochloric acid were added thereto. The mixture was stirred at 60 to 70°C for 1 hour and cooled to 100C or below and the precipitated crystals were then separated. The resulting crystals were washed with 200 ml of isopropanol and dried at 6O0C, yielding 1- (3, 4-dichlorobenzyl) -5-octylbiguanide dihydrochloride. Yield: 243.8 g (The yield was 81.3% based on the Compound (3).) Melting point: 228.90C IR(KBr) spectrum: 2920, 1682, 1634, 1337, 1035, 820, and 640 cm“1
PATENT
WO2004105745A1
PATENT
WO2009142715A1
PATENT
https://www.google.com/patents/US8334248
Olanexidine is a compound with high bactericidal activity having the chemical name 1-(3,4-dichlorobenzyl)-5-octylbiguanide. Research has been carried out into bactericides containing, olanexidine hydrochloride as an active ingredient (see Japanese Patent No. 2662343, etc.).
Olanexidine has very poor solubility in water, and hitherto known salts of olanexidine are also poorly soluble in water. For example, the solubility at 0° C. of olanexidine hydrochloride in water has been measured to be less than 0.05% (W/V), and the solubility of free olanexidine is a further order of magnitude less than this. Consequently, sufficient bactericidal activity cannot be expected of an aqueous solution merely having olanexidine dissolved therein, and moreover, depending on the conditions the olanexidine may precipitate out.
In the case of making an aqueous preparation of olanexidine in particular, to make the concentration of the olanexidine sufficient for exhibiting effective bactericidal activity, and to reduce the possibility of the olanexidine precipitating out, it has thus been considered necessary to use a dissolution aid such as a surfactant.
EXAMPLE 1 Preparation of an Aqueous Solution Aqueous Solution 1
20.9 g (50 mmol) of olanexidine hydrochloride hemihydrate was added to 250 mL of a 1 N aqueous sodium hydroxide solution, and the suspension was stirred for 1.5 hours at room temperature (25° C.). The solid was filtered off, and washed with water. The solid obtained was further suspended in 250 mL of purified water, the suspension was stirred for 5 minutes at room temperature, and the solid was filtered off, and washed with water. This operation was carried out once more to remove sodium chloride formed. The solid obtained (free olanexidine) was put into purified water in which 8.9 g (50 mmol) of gluconolactone had been dissolved, and the mixture was stirred at room temperature until the solid dissolved, and then purified water was further added to give a total volume of 300 mL. The concentration of olanexidine in the aqueous solution obtained was measured by using high performance liquid chromatography to be 6% in terms of free olanexidine.
This aqueous solution was still transparent and colorless even after being left for several months at room temperature.
CLIP
http://dmd.aspetjournals.org/content/28/12/1417/F9.expansion.html


REFERENCES
http://www.otsukakj.jp/en/news/photo/photo-14423716650.pdf
| Patent ID | Date | Patent Title |
|---|---|---|
| US8979785 | 2015-03-17 | Fluid application device and method |
| US8911771 | 2014-12-16 | Fluid application device and method |
| US8858484 | 2014-10-14 | Fluid application device and method |
| US2013330114 | 2013-12-12 | FLUID APPLICATION DEVICE AND METHOD |
| US2012095254 | 2012-04-19 | METHOD AND APPARATUS FOR PREPARING A SOLUTION OF A SHEAR SENSITIVE MATERIAL |
| US7868207 | 2011-01-11 | PROCESS FOR PRODUCING 1-(3, 4-DICHLOROBENZYL)-5-OCTYLBIGUANIDE OR A SALT THEREOF |
| US2010331421 | 2010-12-30 | DISINFECTANT AND/OR BACTERICIDAL AQUEOUS COMPOSITIONS |
| US2010331423 | 2010-12-30 | AQUEOUS SOLUTION OF OLANEXIDINE, METHOD OF PREPARING THE AQUEOUS SOLUTION, AND DISINFECTANT |
| US7829518 | 2010-11-09 | Aqueous solution of olanexidine, method of preparing the aqueous solution, and disinfectant |
| US7825080 | 2010-11-02 | Aqueous solution of olanexidine, method of preparing the aqueous solution, and disinfectant |
| Patent ID | Date | Patent Title |
|---|---|---|
| US7622469 | 2009-11-24 | 2, 4-diamino-1, 3, 5-triazine derivatives |
| US2009287021 | 2009-11-19 | METHOD AND APPARATUS FOR PREPARING A SOLUTION OF A SHEAR SENSITIVE MATERIAL |
| US2007053942 | 2007-03-08 | Disinfectant and/or bactericidal aqueous compositions |
| EP0507317 | 1997-01-15 | BIGUANIDE DERIVATIVES, MANUFACTURING METHOD THEREOF, AND DISINFECTANTS CONTAINING THE DERIVATIVES |
| EP0507317A2 * | Apr 3, 1992 | Oct 7, 1992 | Otsuka Pharmaceutical Co., Ltd. | Biguanide derivatives, manufacturing method thereof, and disinfectants containing the derivatives |
| EP1634589A1 * | May 25, 2004 | Mar 15, 2006 | Otsuka Pharmaceutical Co., Ltd. | Aqueous olanexidine solution, method of preparing the same, and disinfectant |
| Reference | ||
|---|---|---|
| 1 | * | TSUBOUCHI H ET AL: “Synthesis and Structure-Activity Relationships of Novel Antiseptics” BIOORGANIC & MEDICINAL CHEMISTRY LETTERS, OXFORD, GB, vol. 7, no. 13, 8 July 1997 (1997-07-08), pages 1721-1724, XP004136287 ISSN: 0960-894X |
//////////Olanexidine Gluconate, OPB-2045G, (Olanedine®, Approved, japan 2015-07-03, Olanedine, Otsuka, PMDA, Olanexidine, オラネキシジングルコン酸塩 , Gluconate olanexidin, Olanedine, OPB-2045, OPB 2045G, JAPAN 2015
CCCCCCCCN=C(N)NC(=NCC1=CC(=C(C=C1)Cl)Cl)N
Clc1ccc(CNC(=N)NC(=N)NCCCCCCCC)cc1Cl.O=C(O)[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO
Trelagliptin

Trelagliptin
865759-25-7; UNII-Q836OWG55H
| Molecular Formula: | C18H20FN5O2 |
|---|---|
| Molecular Weight: | 357.382103 g/mol |
2-[[6-[(3R)-3-aminopiperidin-1-yl]-3-methyl-2,4-dioxopyrimidin-1-yl]methyl]-4-fluorobenzonitrile
(R) -2 – ((6 (3-amino-piperidin-1-yl) -3-methyl-2,4-dioxo-3,4-dihydropyrimidine -1 (2H) – yl) methyl) synthesis of 4-fluoro-benzonitrile
(R)-2-((6-(3-amino-3-methylpiperidin-l-yl)-3-methyl-2,4-dioxo-3,4-dihydropyrimidin-l(2H)-yl)methyl)-4-fluorobenzonitrile
A dipeptidyl peptidase-4 (DPP-4) inhibitor used to treat type 2 diabetes.
Research Code SYR-472
CAS No. 865759-25-7 (Trelagliptin)
1029877-94-8 (Trelagliptin Succinate)

Dipeptidyl Peptidase IV (IUBMB Enzyme Nomenclature EC.3.4.14.5) is a type π membrane protein that has been referred to in the literature by a wide a variety of names including DPP4, DP4, DAP-IV, FAPβ, adenosine deaminase complexing protein 2, adenosine deaminase binding protein (AD Abp), dipeptidyl aminopeptidase IV; Xaa-Pro-dipeptidyl-aminopeptidase; Gly-Pro naphthylamidase; postproline dipeptidyl aminopeptidase IV; lymphocyte antigen CD26; glycoprotein GPI lO; dipeptidyl peptidase IV; glycylproline aminopeptidase; glycylproline aminopeptidase; X-prolyl dipeptidyl aminopeptidase; pep X; leukocyte antigen CD26; glycylprolyl dipeptidylaminopeptidase; dipeptidyl-peptide hydrolase; glycylprolyl aminopeptidase; dipeptidyl-aminopeptidase IV; DPP ΓV/CD26; amino acyl-prolyl dipeptidyl aminopeptidase; T cell triggering molecule TρlO3; X-PDAP. Dipeptidyl Peptidase IV is referred to herein as “DPP-IV.” [0003] DPP-W is a non-classical serine aminodipeptidase that removes Xaa-Pro dipeptides from the amino terminus (N-terminus) of polypeptides and proteins. DPP-IV dependent slow release of dipeptides of the type X-GIy or X-Ser has also been reported for some naturally occurring peptides.
DPP-IV is constitutively expressed on epithelial and endothelial cells of a variety of different tissues (intestine, liver, lung, kidney and placenta), and is also found in body fluids. DPP-IV is also expressed on circulating T-lymphocytes and has been shown to be synonymous with the cell-surface antigen, CD-26. DPP-IV has been implicated in a number of disease states, some of which are discussed below.
[0005] DPP-IV is responsible for the metabolic cleavage of certain endogenous peptides (GLP-I (7-36), glucagon) in vivo and has demonstrated proteolytic activity against a variety of other peptides (GHRH, NPY, GLP-2, VIP) in vitro.
GLP-I (7-36) is a 29 amino-acid peptide derived by post-translational processing of proglucagon in the small intestine. GLP-I (7-36) has multiple actions in vivo including the stimulation of insulin secretion, inhibition of glucagon secretion, the promotion of satiety, and the slowing of gastric emptying. Based on its physiological profile, the actions of GLP-I (7-36) are believed to be beneficial in the prevention and treatment of type II diabetes and potentially obesity. For example, exogenous administration of GLP-I (7-36) (continuous infusion) in diabetic patients has been found to be efficacious in this patient population. Unfortunately, GLP-I (7-36) is degraded rapidly in vivo and has been shown to have a short half -life in vivo (t1/2=1.5 minutes).
Based on a study of genetically bred DPP-IV knock out mice and on in vivo I in vitro studies with selective DPP-IV inhibitors, DPP-IV has been shown to be the primary degrading enzyme of GLP-I (7-36) in vivo. GLP-I (7-36) is degraded by DPP-IV efficiently to GLP-I (9-36), which has been speculated to act as a physiological antagonist to GLP-I (7-36). Inhibiting DPP-TV in vivo is therefore believed to be useful for potentiating endogenous levels of GLP-I (7-36) and attenuating the formation of its antagonist GLP-I (9-36). Thus, DPP-IV inhibitors are believed to be useful agents for the prevention, delay of progression, and/or treatment of conditions mediated by DPP-IV, in particular diabetes and more particularly, type 2 diabetes mellitus, diabetic dislipidemia, conditions of impaired glucose tolerance (IGT), conditions of impaired fasting plasma glucose (WG), metabolic acidosis, ketosis, appetite regulation and obesity.
DPP-IV expression is increased in T-cells upon mitogenic or antigenic stimulation (Mattem, T., et al., Scand. J. Immunol, 1991, 33, 737). It has been reported that inhibitors of DPP-IV and antibodies to DPP-IV suppress the proliferation of mitogen-stimulated and antigen-stimulated T-cells in a dose-dependant manner (Schon, E., et al., Biol. Chem., 1991, 372, 305). Various other functions of T-lymphocytes such as cytokine production, IL-2 mediated cell proliferation and B-cell helper activity have been shown to be dependent on DPP-IV activity (Schon, E., et al., Scand. J. Immunol, 1989, 29, 127). DPP-IV inhibitors, based on boroProline, (Flentke, G. R., et al., Proc. Nat. Acad. Set USA, 1991, 88, 1556) although unstable, were effective at inhibiting antigen-induced lymphocyte proliferation and IL-2 production in murine CD4+ T-helper cells. Such boronic acid inhibitors have been shown to have an effect in vivo in mice causing suppression of antibody production induced by immune challenge (Kubota, T. et al, Clin. Exp. Immun., 1992, 89, 192). The role of DPP-IV in regulating T lymphocyte activation may also be attributed, in part, to its cell-surface association with the transmembrane phosphatase, CD45. DPP-IV inhibitors or non-active site ligands may possibly disrupt the CD45-DPP-TV association. CD45 is known to be an integral component of the T-cell signaling apparatus. It has been reported that DPP-IV is essential for the penetration and infectivity of HTV-I and HTV-2 viruses in CD4+ T-cells (Wakselman, M., Nguyen, C, Mazaleyrat, J.-P., Callebaut, C, Krust, B., Hovanessian, A. G., Inhibition of HIV-I infection of CD 26+ but not CD 26-cells by a potent cyclopeptidic inhibitor of the DPP-IV activity of CD 26. Abstract P.44 of the 24.sup.th European Peptide Symposium 1996). Additionally, DPP-IV has been shown to associate with the enzyme adenosine deaminase (ADA) on the surface of T-cells (Kameoka, J., et al., Science, 193, 26 466). ADA deficiency causes severe combined immunodeficiency disease (SCID) in humans. This ADA-CD26 interaction may provide clues to the pathophysiology of SCID. It follows that inhibitors of DPP-TV may be useful immunosuppressants (or cytokine release suppressant drugs) for the treatment of among other things: organ transplant rejection; autoimmune diseases such as inflammatory bowel disease, multiple sclerosis and rheumatoid arthritis; and the treatment of AIDS.
It has been shown that lung endothelial cell DPP-IV is an adhesion molecule for lung-metastatic rat breast and prostate carcinoma cells (Johnson, R. C, et al., J. Cell. Biol, 1993, 121, 1423). DPP-IV is known to bind to fibronectin and some metastatic tumor cells are known to carry large amounts of fibronectin on their surface. Potent DPP-IV inhibitors may be useful as drugs to prevent metastases of, for example, breast and prostrate tumors to the lungs.
High levels of DPP-PV expression have also been found in human skin fibroblast cells from patients with psoriasis, rheumatoid arthritis (RA) and lichen planus (Raynaud, F., et al., J. Cell. Physiol, 1992, 151, 378). Therefore, DPP-TV inhibitors may be useful as agents to treat dermatological diseases such as psoriasis and lichen planus. [0011] High DPP-TV activity has been found in tissue homogenates from patients with benign prostate hypertrophy and in prostatosomes. These are prostate derived organelles important for the enhancement of sperm forward motility (Vanhoof, G., et al., EMr. /.
Clin. Chem. Clin. Biochem., 1992, 30, 333). DPP-IV inhibitors may also act to suppress sperm motility and therefore act as a male contraceptive agent. Conversely, DPP-IV inhibitors have been implicated as novel for treatment of infertility, and particularly human female infertility due to Polycystic ovary syndrome (PCOS, Stein-Leventhal syndrome) which is a condition characterized by thickening of the ovarian capsule and . formation of multiple follicular cysts. It results in infertility and amenorrhea.
DPP-IV is thought to play a role in the cleavage of various cytokines
(stimulating hematopoietic cells), growth factors and neuropeptides.
[0013] Stimulated hematopoietic cells are useful for the treatment of disorders that are characterized by a reduced number of hematopoietic cells or their precursors in vivo. Such conditions occur frequently in patients who are immunosuppressed, for example, as a consequence of chemotherapy and/or radiation therapy for cancer. It was discovered that inhibitors of dipeptidyl peptidase type PV are useful for stimulating the growth and differentiation of hematopoietic cells in the absence of exogenously added cytokines or other growth factors or stromal cells. This discovery contradicts the dogma in the field of hematopoietic cell stimulation, which provides that the addition of cytokines or cells that produce cytokines (stromal cells) is an essential element for maintaining and stimulating the growth and differentiation of hematopoietic cells in culture. (See, e.g., PCT Intl. Application No. PCT/US93/017173 published as WO 94/03055).
[0014] DPP-IV in human plasma has been shown to cleave N-terminal Tyr-Ala from growth hormone-releasing factor and cause inactivation of this hormone. Therefore, inhibitors of DPP-IV may be useful in the treatment of short stature due to growth hormone deficiency (Dwarfism) and for promoting GH-dependent tissue growth or re-growth.
DPP-IV can also cleave neuropeptides and has been shown to modulate the activity of neuroactive peptides substance P, neuropeptide Y and CLIP (Mentlein, R., Dahms, P., Grandt, D., Kruger, R., Proteolytic processing of neuropeptide Y and peptide YY by dipeptidyl peptidase IV, Regul. Pept., 49, 133, 1993; Wetzel, W., Wagner, T., Vogel, D., Demuth, H.-U., Balschun, D., Effects of the CLIP fragment ACTH 20-24 on the duration of REM sleep episodes, Neuropeptides, 31, 41, 1997). Thus DPP-IV inhibitors may also be useful agents for the regulation or normalization of neurological disorders.
Several compounds have been shown to inhibit DPP-IV. Nonetheless, a need still exists for new DPP-IV inhibitors that have advantageous potency, stability, selectivity, toxicity and/or pharmacodynamics properties. In this regard, synthetic methods are provided that can be used to make a novel class of DPP-IV inhibitors.
Trelagliptin (Zafatek) is a pharmaceutical drug used for the treatment of type 2 diabetes (diabetes mellitus).[1]
Indications for Medical Use
It is a highly selective dipeptidyl peptidase (DPP-4) inhibitor that is typically used as an add on treatment when the first line treatment of metformin is not achieving the expected glycemic goals; though it has been approved for use as a first line treatment when metformin cannot be used.[1]
Biochemistry
DPP-4 inhibitors activate T-cells and are more commonly known as T-cell activation antigens (specifically CD26).[1][2] Chemically, it is a fluorinated derivative of alogliptin.
Development
Formulated as the salt trelagliptin succinate, it was approved for use in Japan in March 2015.[3] Takeda, the company that developed trelagliptin, chose to not get approval for the drug in the USA and EU.[1] The licensing rights that Takeda purchased from Furiex Pharmaceuticals for DPP-4 inhibitors included a clause specific to development of this drug in the USA and EU.[1] The clause required that all services done for phase II and phase III clinical studies in the USA and EU be purchased through Furiex.[1] Takeda chose to cease development of this drug in the USA and EU because of the high costs quoted by Furiex for these services.[1] Gliptins have been on the market since 2006 and there are 8 gliptins currently registered as drugs (worldwide).[4] Gliptins are an emerging market and are thus being developed at an increasing rate; there are currently two gliptins in advanced stages of development that are expected to be on the market in the coming year.[4]
Gliptins are thought to have cardiovascular protective abilities though the extent of these effects is still being studied.[4] They are also being studied for the ability that this class of drugs has at promoting B-cell survival.[4]
Administration and Dosing
Similar drugs in the same class as trelagliptin are administered once daily while trelagliptin is administered once weekly.[1][5] Alogliptin (Nesina) is the other major DPP-4 inhibitor on the market. It is also owned by Takeda and is administered once daily. A dosing of once per week is advantageous as a reduction in the frequency of required dosing is known to increase patient compliance.[1][2]
Zafatek is administered in the form trelagliptin succinate in a 1:1 mixture of trelagliptin and succinic acid.[6] The drug is marketed with the IUPAC name Succinic acid – 2-({6-[(3R)-3-amino-1-piperidinyl]-3-methyl-2,4-dioxo-3,4-dihydro-1(2H)-pyrimidinyl}methyl)-4-fluorobenzonitrile (1:1), has a molecular mass of 475.470143 grams/mol, and has the molecular formula | C=22 | H=26 | F=1 | N=5 | O=6 .[6][7]
SYNTHESIS …………….
PAPER

The discovery of two classes of heterocyclic dipeptidyl peptidase IV (DPP-4) inhibitors, pyrimidinones and pyrimidinediones, is described. After a single oral dose, these potent, selective, and noncovalent inhibitors provide sustained reduction of plasma DPP-4 activity and lowering of blood glucose in animal models of diabetes. Compounds 13a, 27b, and 27j were selected for development.
2-[6-(3-Aminopiperidin-1-yl)-3-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-ylmethyl]-4-fluorobenzonitrile, TFA salt (27j)
PATENT
WO 2007035629
http://www.google.com/patents/WO2007035629A3?cl=en
(R)-2-((6-(3-amino-3-methylpiperidin-l-yl)-3-methyl-2,4-dioxo-3,4-dihydropyrimidin-l(2H)-yl)methyl)-4-fluorobenzonitrile (30). 2-(6-Chloro-3-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-l-ylmethyl)-4-fluoro-benzonitrile (300 mg, 1.0 mmol), (R)-3-amino-3-methyl-piperidine dihydrochloride (266 mg, 1.4 mmol) and sodium bicarbonate (500 mg, 5.4 mmol) were stirred in a sealed tube in EtOH (3 mL) at 1000C for 2 hrs. The final compound was obtained as TFA salt after HPLC purification. 1H-NMR (400 MHz, CD3OD): δ. 7.78-7.83 (m, IH), 7.14-7.26 (m, 2H), 5.47 (s, IH), 5.12-5.36 (ABq, 2H, J = 105.2, 15.6 Hz), 3.21 (s, IH), 2.72-3.15 (m, 4H), 1.75-1.95 (m, 4H), 1.39 (s, 3H). MS (ES) [m+H] calc’d for C19H22FN5O2, 372.41; found, 372.41.
Compound 34

4-Fluoro-2-methylbenzonitrile (31). A mixture of 2-bromo-5-fluorotoluene (3.5 g, 18.5 mmol) and CuCN (2 g, 22 mmol) in DMF (100 mL) was refluxed for 24 hours. The reaction was diluted with water and extracted with hexane. The organics were dried over MgSO4 and the solvent removed to give product 31 (yield 60%). 1H-NMR (400 MHz, CDCl3): δ 7.60 (dd, J=5.6, 8.8 Hz, IH), 6.93-7.06 (m, 2H), 2.55 (s, 3H).
2-Bromomethyl-4-fluorobenzonitrile (32). A mixture of 4-fluoro-2-methylbenzonitrile (2 g, 14.8 mmol), NBS (2.64 g, 15 mmol) and AIBN (100 mg) in CCl4 was refluxed under nitrogen for 2 hours. The reaction was cooled to room temperature. The solid was removed by filtration. The organic solution was concentrated to give crude product as an oil, which was used in the next step without further purification. 1H-NMR (400 MHz, CDCl3): δ 7.68 (dd, J= 5.2, 8.4 Hz, IH), 7.28 (dd, J= 2.4, 8.8 Hz, IH), 7.12 (m, IH), 4.6 (s, 2H).
Alternatively, 32 was made as follows. 4-Fluoro-2-methylbenzonitrile (1 kg) in DCE (2 L) was treated with AJJBN (122 g) and heated to 750C. A suspension of DBH (353 g) in DCE (500 mL) was added at 750C portionwise over 20 minutes. This operation was repeated 5 more times over 2.5 hours. The mixture was then stirred for one additional hour and optionally monitored for completion by, for example, measuring the amount of residual benzonitrile using HPLC. Additional AJ-BN (e.g., 12.5 g) was optionally added to move the reaction toward completion. Heating was stopped and the mixture was allowed to cool overnight. N,N-diisopropylethylamine (1.3 L) was added (at <10°C over 1.5 hours) and then diethyl phosphite (1.9 L) was added (at <20°C over 30 min). The mixture was then stirred for 30 minutes or until completion. The mixture was then washed with 1% sodium metabisulfite solution (5 L) and purified with water (5 L). The organic phase was concentrated under vacuum to afford 32 as a dark brown oil (3328 g), which was used without further purification (purity was 97% (AUC)).
2-(6-Chloro-3-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-l-ylmethyl)-4-fluoro-benzonitrile (33). A mixture of crude 3-methyl-6-chlorouracil (0.6 g, 3.8 mmol), 2-bromomethyl-4-fluorobenzonitrile (0.86 g, 4 mmol) and K2CO3 (0.5 g, 4 mmol) in DMSO (10 mL) was stirred at 6O0C for 2 hours. The reaction was diluted with water and extracted with EtOAc. The organics were dried over MgSO4 and the solvent removed. The residue was purified by column chromatography. 0.66 g of the product was obtained (yield: 60%). 1H-NMR (400 MHz, CDCl3): δ 7.73 (dd, 1=1.2, 8.4Hz, IH), 7.26 (d, J-4.0Hz, IH), 7.11-7.17 (m, IH), 6.94 (dd, J=2.0, 9.0 Hz, IH), 6.034 (s, 2H), 3.39 (s, 3H). MS (ES) [m+H] calc’d for C13H9ClFN3O2, 293.68; found 293.68.
Alternatively, 33 was made as follows. To a solution of 6-chloro-3-methyluracil (750 g) and W,iV-diisopropylethylarnine (998 mL) in NMP (3 L) was added (at <30°C over 25 min) a solution of 32 (2963 g crude material containing 1300 g of 32 in 3 L of toluene). The mixture was then heated at 6O0C for 2 hours or until completion (as determined, for example, by HPLC). Heating was then stopped and the mixture was allowed to cool overnight. Purified water (3.8 L) was added, and the resultant slurry was stirred at ambient temperature for 1 hour and at <5°C for one hour. The mixture was then filtered under vacuum and the wet cake was washed with IPA (2 X 2.25 L). The material was then dried in a vacuum oven at 40±5°C for 16 or more hours to afford 33 as a tan solid (>85% yield; purity was >99% (AUC)).
2-[6-(3-Amino-piperidin-l-yl)-3-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-l-ylmethyl]-4-fluoro-benzonitrile (34). 2-(6-Chloro-3-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-l-ylmethyl)-4-fluoro-benzonitrile (300 mg, 1.0 mmol), (R)-3-amino-piperidine dihydrochloride (266 mg, 1.5 mmol) and sodium bicarbonate (500 mg, 5.4 mmol) were stirred in a sealed tube in EtOH (3 mL) at 1000C for 2 hrs. The final compound was obtained as TFA salt after HPLC purification. 1H-NMR (400 MHz, CD3OD): δ. 7.77-7.84 (m, IH), 7.16-7.27 (m, 2H), 5.46 (s, IH), 5.17-5.34 (ABq, 2H, J = 35.2, 15.6 Hz), 3.33-3.47 (m, 2H), 3.22 (s, 3H), 2.98-3.08 (m, IH), 2.67-2.92 (m, 2H), 2.07-2.17 (m, IH), 1.82-1.92 (m, IH), 1.51-1.79 (m, 2H). MS (ES) [m+H] calc’d for C18H20FN5O2, 357.38; found, 357.38.
Alternatively, the free base of 34 was prepared as follows. A mixture of 33 (1212 g), IPA (10.8 L), (R)-3-amino-piperidine dihydrochloride (785 g), purified water (78 mL) and potassium carbonate (2.5 kg, powder, 325 mesh) was heated at 6O0C until completion (e.g., for >20 hours) as determined, for example, by HPLC. Acetonitrile (3.6 L) was then added at 6O0C and the mixture was allowed to cool to <25°C. The resultant slurry was filtered under vacuum and the filter cake was washed with acetonitrile (2 X 3.6 L). The filtrate was concentrated at 450C under vacuum (for >3 hours) to afford 2.6 kg of the free base of 34.
The HCl salt of 34 was prepared from the TFA salt as follows. The TFA salt (34) was suspended in DCM, and then washed with saturated Na2CO3. The organic layer was dried and removed in vacuo. The residue was dissolved in acetonitrile and HCl in dioxane (1.5 eq.) was added at 00C. The HCl salt was obtained after removing the solvent. 1H-NMR (400 MHz, CD3OD): δ. 7.77-7.84 (m, IH), 7.12-7.26 (m, 2H), 5.47 (s, IH), 5.21-5.32 (ABq, 2H, J = 32.0, 16.0 Hz), 3.35-3.5 (m, 2H), 3.22 (s, 3H), 3.01-3.1 (m, IH), 2.69-2.93 (m, 2H), 2.07-2.17 (m, IH), 1.83-1.93 (m, IH), 1.55-1.80 (m, 2H). MS (ES) [m+H] calc’d for C18H20FN5O2, 357.38; found, 357.38.
Alternatively, the HCl salt was prepared from the free base as follows. To a solution of free base in CH2Cl2 (12 L) was added (at <35°C over 18 minutes) 2 M hydrochloric acid (3.1 L). The slurry was stirred for 1 hour and then filtered. The wet cake was washed with CH2Cl2 (3.6 L) and then THF (4.8 L). The wet cake was then slurried in THF (4.8 L) for one hour and then filtered. The filter cake was again washed with THF (4.8 L). The material was then dried in a vacuum oven at 5O0C (with a nitrogen bleed) until a constant weight (e.g., >26 hours) to afford 34 as the HCl salt as a white solid (1423 g, >85% yield).
The succinate salt of 34 was prepared from the HCl salt as follows. To a mixture of the HCl salt of 34 (1414 g), CH2Cl2 (7 L) and purifed water (14 L) was added 50% NaOH solution (212 mL) until the pH of the mixture was >12. The biphasic mixture was stirred for 30 min and the organic layer was separated. The aqueous layer was extracted with CH2Cl2 (5.7 L) and the combined organic layers were washed with purified water (6 L). The organic layer was then passed through an in-line filter and concentrated under vacuum at 3O0C over three hours to afford the free base as an off-white solid. The free base was slurried in prefiltered THF (15 L) and prefiltered IPA (5.5 L). The mixture was then heated at 6O0C until complete dissolution of the free base was observed. A prefiltered solution of succinic acid (446 g) in THF (7 L) was added (over 23 min) while maintaining the mixture temperature at >57°C. After stirring at 6O0C for 15 min, the heat was turned off, the material was allowed to cool, and the slurry was stirred for 12 hours at 25±5°C. The material was filtered under vacuum and the wet cake was washed with prefiltered IPA (2 X 4.2 L). The material was then dried in a vacuum oven at 70±5°C (with a nitrogen bleed) for >80 hours to afford the succinate salt of 34 as a white solid (1546 g, >90% yield).
The product was also converted to a variety of corresponding acid addition salts. Specifically, the benzonitrile product (approximately 10 mg) in a solution of MeOH (1 mL) was treated with various acids (1.05 equivalents). The solutions were allowed to stand for three days open to the air. If a precipitate formed, the mixture was filtered and the salt dried. If no solid formed, the mixture was concentrated in vacuo and the residue isolated. In this way, salts of 34 were prepared from the following acids: benzoic, p-toluenesulfonic, succinic, R-(-)-Mandelic and benzenesulfonic. The succinate was found to be crystalline as determined by x-ray powder diffraction analysis.
In addition, the methanesulfonate salt was prepared as follows. A 10.5 g aliquot of the benzonitrile product was mixed with 400 mL of isopropylacetate. The slurry was heated to 75°C and filtered through #3 Whatman filter paper. The solution was heated back to 750C and a IM solution of methanesulfonic acid (30.84 mL) was added slowly over 10 minutes while stirring. The suspension was cooled to room temperature at a rate of about 20°C/hr. After 1 hr at room temperature, the solid was filtered and dried in an oven overnight to obtain the methanesulfonate salt.
PATENT
US 2008227798
http://www.google.com/patents/US20080227798
- EXAMPLES
- Example 1Preparation of 2-[6-(3-amino-piperidin-1-yl)-3-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-ylmethyl]-4-fluoro-benzonitrile succinate (Compound I)
A. Preparation of 4-fluoro-2-methylbenzonitrile (Compound B)
-
Compound B was prepared by refluxing a mixture of 2-bromo-5-fluoro-toluene (Compound A) (3.5 g, 18.5 mmol) and CuCN (2 g, 22 mmol) in DMF (100 mL) for 24 hours. The reaction was diluted with water and extracted with hexane. The organics were dried over MgSO4 and the solvent removed to give product B (yield 60%). 1H-NMR (400 MHz, CDCl3): δ 7.60 (dd, J=5.6, 8.8 Hz, 1H), 6.93-7.06 (m, 2H), 2.55 (s, 3H).
B. Preparation of 2-bromomethyl-4-fluorobenzonitrile (Compound C)
-
Compound C was prepared by refluxing a mixture of 4-fluoro-2-methylbenzonitrile (Compound B) (2 g, 14.8 mmol), N-bromosuccinimide (NBS) (2.64 g, 15 mmol) and azo-bis-isobutyronitrile (AIBN) (100 mg) in CCl4 under nitrogen for 2 hours. The reaction was cooled to room temperature. The solid was removed by filtration. The organic solution was concentrated to give the crude product the form of an oil, which was used in the next step without further purification. 1H-NMR (400 MHz, CDCl3): δ 7.68 (dd, J=5.2, 8.4 Hz, 1H), 7.28 (dd, J=2.4, 8.8 Hz, 1H), 7.12 (m, 1H), 4.6 (s, 2H).
C. Preparation of 2-(6-chloro-3-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-ylmethyl)-4-fluoro-benzonitrile (Compound D)
-
Compound E was prepared by stirring a mixture of crude 3-methyl-6-chlorouracil D (0.6 g, 3.8 mmol), 2-bromomethyl-4-fluorobenzonitrile (0.86 g, 4 mmol) and K2CO3 (0.5 g, 4 mmol) in DMSO (10 mL) at 60° C. for 2 hours. The reaction was diluted with water and extracted with EtOAc. The organics were dried over MgSO4 and the solvent removed. The residue was purified by column chromatography. 0.66 g of the product was obtained (yield: 60%). 1H-NMR (400 MHz, CDCl3): δ 7.73 (dd, J=7.2, 8.4 Hz, 1H), 7.26 (d, J=4.0 Hz, 1H), 7.11-7.17 (m, 1H), 6.94 (dd, J=2.0, 9.0 Hz, 1H), 6.034 (s, 2H), 3.39 (s, 3H). MS (ES) [m+H] calc’d for C13H9ClFN3O2, 293.68; found 293.68.
D. Preparation of 2-(6-chloro-3-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-ylmethyl)-4-fluoro-benzonitrile (Compound F)
-
Compound F was prepared by mixing and stirring 2-(6-chloro-3-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-ylmethyl)-4-fluoro-benzonitrile (Compound E) (300 mg, 1.0 mmol), (R)-3-amino-piperidine dihydrochloride (266 mg, 1.5 mmol) and sodium bicarbonate (500 mg, 5.4 mmol) in a sealed tube in EtOH (3 mL) at 100° C. for 2 hrs. The final compound was obtained as trifluoroacetate (TFA) salt after HPLC purification. 1H-NMR (400 MHz, CD3OD): δ. 7.77-7.84 (m, 1H), 7.16-7.27 (m, 2H), 5.46 (s, 1H), 5.17-5.34 (ABq, 2H, J=35.2, 15.6 Hz), 3.33-3.47 (m, 2H), 3.22 (s, 3H), 2.98-3.08 (m, 1H), 2.67-2.92 (m, 2H), 2.07-2.17 (m, 1H), 1.82-1.92 (m, 1H), 1.51-1.79 (m, 2H). MS (ES) [m+H] calc’d for C18H20FN5O2, 357.38; found, 357.38.
E. Preparation of Compound I: the succinic acid salt of 2-(6-Chloro-3-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-ylmethyl)-4-fluoro-benzonitrile
-
The TFA salt prepared in the above step (Example 1, Step D) was suspended in DCM, and then washed with saturated Na2CO3. The organic layer was dried and removed in vacuo. The benzonitrile product (approximately 10 mg) was dissolved in MeOH (1 mL) and to which succinic acid in THF (1.05 equivalents) was added. The solutions were allowed to stand for three days open to the air. If a precipitate formed, the solid was collected by filtration. If no solid formed, the mixture was concentrated in vacuo, and the succinate salt was obtained after removing the solvent. 1H-NMR (400 MHz, CD3OD): δ. 7.77-7.84 (m, 1H), 7.12-7.26 (m, 2H), 5.47 (s, 1H), 5.21-5.32 (ABq, 2H, J=32.0, 16.0 Hz), 3.35-3.5 (m, 2H), 3.22 (s, 3H), 3.01-3.1 (m, 1H), 2.69-2.93 (m, 2H), 2.07-2.17 (m, 1H), 1.83-1.93 (m, 1H), 1.55-1.80 (m, 2H). MS (ES) [m+H] calc’d for C18H20FN5O2, 357.38; found, 357.38.
-
Compound I such prepared was found to be crystalline as determined by x-ray powder diffraction analysis (FIG. 1). The crystal material was designated Form A.
| TABLE A | |||
| Approximate Solubilities of Compound I | |||
| Solubility | |||
| Solvent | (mg/mL)a | ||
| Acetone | 2 | ||
| Acetonitrile (ACN) | <1 | ||
| Dichloromethane (DCM) | <1 | ||
| Dimethyl Formamide (DMF) | 68 | ||
| 1,4-Dioxane | <1 | ||
| Ethanol (EtOH) | 2 | ||
| Ethyl Acetate (EtOAc) | <1 | ||
| di-Ethyl ether | <1 | ||
| Hexanes | <1 | ||
| 2-Propanol (IPA) | <1 | ||
| Methanol (MeOH) | 20 | ||
| Tetrahydrofuran (THF) | <1 | ||
| Toluene | <1 | ||
| Trifluoroethanol (TFE) | >200 | ||
| Water (H2O) | 51 | ||
| ACN:H2O (85:15) | 101 | ||
| EtOH:H2O (95:5) | 5 | ||
| IPA:H2O (88:12) | 11 | ||
| aApproximate solubilities are calculated based on the total solvent used to give a solution; actual solubilities may be greater because of the volume of the solvent portions utilized or a slow rate of dissolution. Solubilities are reported to the nearest mg/mL. | |||
PATENT
Reference Example 2
in the following formula 2, 2 – ((6 – ((3R) -3- amino-piperidin-1-yl) -3-methyl-2,4-dioxo-3,4-dihydropyrimidine -1 (2H ) – yl) shown in the following example of a production process of a methyl) -4-fluoro-benzonitrile succinate (4b).
[Formula 2]
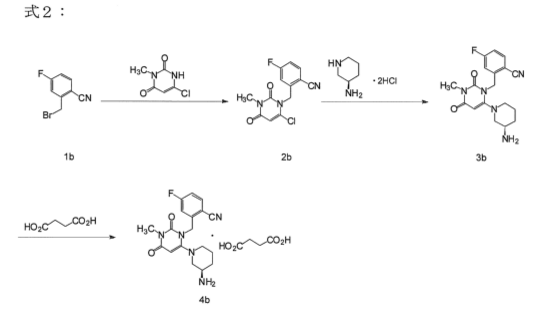
[In the formula 2, 2 – ((6-chloro-3-methyl-2,4-dioxo-3,4-dihydropyrimidine -1 (2H) – yl) methyl) -4-fluorobenzonitrile (2b) manufacturing process]
ethyl acetate (3.5 vol), 2- (bromomethyl) -4-fluorobenzonitrile (1b) (1 equiv, 1wt.), 6- chloro-3-methyl uracil (1.05 eq, 0.79wt), N- methylpyrrolidone (NMP;.. 3.5 times the amount), diisopropylethylamine (Hunig’s base, 2.1 eq, 1.27wt) was heated to an internal temperature of 60 ~ 70 ℃ a.
The mixture was stirred until 2-4 hours or the completion of the reaction at 60 ~ 70 ℃.
Then cooling the solution to 40 ~ 50 ℃, after stirring at least 30 minutes, 40 ~ 50 ℃ isopropanol (1.5 times) while maintaining, water (3.5 times the amount) was added, then at least one hour stirring did. The solution was cooled to 20 ~ 30 ℃, was then stirred for at least 1 hour. The solution was cooled to 0 ~ 10 ℃, was then stirred for at least 1 hour. The resulting slurry was filtered, washed with 0 ~ 10 ℃ in cold isopropanol (4.0 vol), and vacuum dried at 45 ~ 55 ℃, to give the above compound (2b).
[In the formula 2, 2 – ((6 – ((3R) -3- amino-piperidin-1-yl) -3-methyl-2,4-dioxo-3,4-dihydropyrimidine -1 (2H) – yl) methyl) -4-manufacturing process of the fluorobenzonitrile (3b)]
the above compound (2b) (1 eq, 1wt.), (R) -3- aminopiperidine dihydrochloride (1.1 eq, 0.65wt .), potassium carbonate (2.5 equivalents, 1.18wt.), isopropanol (5.0 vol), water (1.5 times) until the completion of the reaction with 65 ~ 75 ℃ (eg, 3 to 7 hours ) was allowed to react. Potassium carbonate in 65 ~ 75 ℃ (7.05 eq, 3.32wt.), Water (5.5 vol) was added, and after stirring for about 30 minutes, the phases were separated at 50 ℃ ~ 70 ℃. The organic solvent was concentrated under reduced pressure to approximately 5 times. And water (5 vol) was added to the solution and concentrated under reduced pressure to approximately 5 times. The solution was stirred for about 40 minutes at 55 ℃ ~ 75 ℃. The solution was cooled to 20 ℃ ~ 30 ℃, was then stirred for at least 1 hour. The solution was cooled to 0 ~ 10 ℃, subsequently stirred for at least 1 hour, the resulting slurry was filtered, washed with 0 ~ 10 ℃ in cold water (2.0 times the amount), 45 ~ 55 ℃ was vacuum dried to give the above compound (3b).
[In the above formula 2, the compound production step of succinate (4b) of (3b)]
Compound (3b), tetrahydrofuran (6.0 vol), isopropanol (3.0 vol), water (0. a 6-fold amount) was heated to 55 ~ 65 ℃. Tetrahydrofuran solution of succinic acid (20 ℃ ~ 30 ℃) was added and the solution was stirred for about 15 minutes and maintained at 55 ~ 65 ℃.
The solution was cooled to 20 ~ 30 ℃, the mixture was stirred for at least 1 hour. The solution was cooled to 0 ~ 10 ℃, was then stirred for at least 1 hour. After the resulting slurry filtered and washed with isopropanol (6.0 vol). The resulting wet crystals were dried at 65 ~ 75 ℃, was obtained succinate of the compound (3b) and (4b) as a white crystalline solid.
PATENT
http://www.google.com/patents/CN103030631A?cl=en
2 – ({6 -! [(3R) -3- amino-piperidin-1-yl] -3-methyl-dihydro-pyrimidin _3,4_ _2,4_ dioxo-1 (2 1) – yl} methyl) benzonitrile is an effective DPP-1V inhibitors class of drugs in recent years in Japan, the structural formula
As shown below.
Chinese Patent Application CN1926128 discloses a process for preparing 2_ ({6_ [(3R) -3- amino-piperidin-1-yl] -3-methyl-2,4-dioxo-3,4- dihydropyrimidine-1 (2 1!) – yl} methyl) benzonitrile method, as shown in Scheme I:
Scheme I
In the above reaction scheme, 6-chloro-uracil and 2-bromomethyl-benzene cyanide in a mixed solvent of DMF-DMSO, in the presence of NaH and LiBr alkylation reaction to give compound 2 in a yield of 54%. Compound 2 is further alkylation reaction of compound yield 3 is 72%. The total yield of the compound 4 prepared in 20% yield is low, and the preparation of compound 4 obtained purity is not high, but also the need for further purification, such as recrystallization, column chromatography and other means in order to obtain high-purity suitable Pharmaceutically acceptable 2 – ({6 – [(3R) -3- amino-piperidin-1-yl] -3-methyl-2,4-dioxo-3,4-dihydro-pyrimidin _1 (2! 1) – yl} methyl) benzonitrile compound. Preparation still find more suitable for industrial production, a higher yield of the 2- ({6- [(3R) -3- amino-piperidin-1-yl] -3-methyl-2,4-dioxo -3, (2Η) 4- dihydropyrimidine-1 – yl} methyl) benzonitrile or a salt or the like.
PATENT
Example 15
(R) -2 – ((6 (3-amino-piperidin-1-yl) -3-methyl-2,4-dioxo-3,4-dihydropyrimidine -1 (2H) – yl) methyl) synthesis of 4-fluoro-benzonitrile
100mL four-necked flask of water and isopropanol 1/1 (v / v) mixture 60mL was added, pyridine 21.4μL [d = 0.98, mw.79.10, 0.26mmol], (R) -1- (3- (2 – cyano-5-fluoro-benzyl) -1-methyl-2,6-dioxo-1,2,3,6-tetra-hydro-4-yl) piperidin-3-carboxamide 2.00g [mw.385.39, 5.19mmol] of It was added to the order. Then, iodobenzene diacetate 1.84g [mw.322.10, 5.71mmol] was added, and the mixture was stirred for 3 h at 20 ℃. After volatile components were distilled off under reduced pressure by an evaporator, and the aqueous solution was washed twice with ethyl acetate 20mL. After cooling to near 0 ℃, potassium carbonate 16g added stepwise at 15 ℃ or less, was extracted by the addition of toluene 6mL and isopropanol 6mL. After separation, the organic layer was washed with saturated brine 10mL, adding toluene 6mL after concentration under reduced pressure by an evaporator, and further subjected to vacuum concentration. It was suspended by the addition of toluene 6mL to concentrate, by the addition of n-heptane 6mL, after 1 hour and aged at 0 ℃, reduced pressure filtration, to obtain the desired compound after drying under reduced pressure at 50 ℃. White crystalline powder, 1.6g, 86% yield.
1 H-NMR (500 MHz, CDCl 3 ) delta (ppm) 1.23 (D, J = 11.03 Hz, 1H) 1.30 (BRS, 2H) 1.56-1.67 (M, 1H) 1.72-1.83 (M, 1H) 1.95 (dd , J = 12.77 Hz, 3.94 Hz, 1H) 2.41 (m, 1H) 2.61 (m, 1H) 2.87-2.98 (m, 2H) 2.99-3.05 (m, 1H) 3.32 (s, 3H) 5.23-5.32 (m , 2H) 5.39 (s, 1H) 6.86 (dd, J = 8.99 Hz, 2.36 Hz, 1H) 7.09 (td, J = 8.04 Hz, 2.52 Hz, 1H) 7.69 (dd, J = 8.51 Hz, 5.36 Hz, 1H ).
13 C NMR (126 MHz, CDCl 3 ) ppm 28.0, 33.4, 46.1, 51.9, 59.7, 90.8, 114.6,114.7, 115.6, 115.8, 116.4, 135.4, 135.5, 144.6, 152.7, 159.5, 162.9.
Reference Example 4
(R) -2 – ((6 (3-amino-piperidin-1-yl) -3-methyl-2,4-dioxo-3,4-dihydropyrimidine -1 (2H) – yl) methyl) synthesis of 4-fluoro-benzonitrile succinate

50mL eggplant-shaped flask (R) -2 – ((6- (3- amino-1-yl) -3-methyl-2,4-dioxo-3,4-dihydro-pyrimidine -1 (2H) – yl) methyl) -4-fluorobenzonitrile 1.0g [mw.357.38, 2.8mmol], it was added tetrahydrofuran 4.5mL and water 2 drops. After heated and dissolved at 65 ℃, was dropped to the solution was dissolved at the same temperature 0.331g succinic acid [mw.118.09, 2.8mmol] with tetrahydrofuran 4mL and isopropanol 2.5mL. Aged for 16 hours at room temperature after stirring for 30 min at 65 ℃, and stirred for a further 2 hours at 0 ℃. The crystallization product was collected by terrorism to vacuum filtration. To obtain the desired compound after drying under reduced pressure at 45 ℃. White crystalline powder, 1.2g, 93% yield.
1 H-NMR (500 MHz, DMSO) delta (ppm) 1.35 (D, J = 8.83 Hz, 1H) 1.42-1.57 (M, 1H) 1.66-1.97 (M, 2H) 2.54-2.77 (M, 2H) 2.91 ( d, J = 11.35 Hz, 1H) 3.00-3.07 (m, 1H) 3.08 (m, 1H) 3.09 (s, 3H) 3.14 (m, 1H) 5.12 (d, J = 16.08 Hz, 1H) 5.20 (d, J = 16.39 Hz, 1H) 5.38 (s, 1H) 7.17 (dd, J = 9.62 Hz, 2.36 Hz, 1H) 7.35 (td, J = 8.51 Hz, 2.52 Hz, 1H) 7.95 (dd, J = 8.67 Hz, 5.52 Hz, 1H).
13 C NMR (126 MHz, DMSO) delta ppm 27.9, 31.6, 46.3, 47.0, 51.7, 55.8, 90.3, 106.9, 115.7, 117.1, 136.45, 136.53, 145.8, 152.3, 159.7, 162.7, 164.1 , 166.1, 175.2.
PATENT
http://www.google.com/patents/CN102964196A?cl=en
PATENT
New Patent, Trelagliptin, SUN PHARMA
SUN PHARMACEUTICAL INDUSTRIES LIMITED [IN/IN]; Sun House, Plot No. 201 B/1 Western Express Highway Goregaon (E) Mumbai, Maharashtra 400 063 (IN)
BARMAN, Dhiren, Chandra; (IN).
NATH, Asok; (IN).
PRASAD, Mohan; (IN)
The present invention provides a process for the preparation of 4-fluoro-2- methylbenzonitrile of Formula (II), and its use for the preparation of trelagliptin or its salts. The present invention provides an efficient, simple, and commercially friendly process for the preparation of 4-fluoro-2-methylbenzonitrile, which is used as an intermediate for the preparation of trelagliptin or its salts. The present invention avoids the use of toxic and hazardous reagents, high boiling solvents, and bromo intermediates such as 2-bromo-5-fluorotoluene, which is lachrymatory in nature and thus difficult to handle at a commercial scale.
Trelagliptin is a dipeptidyl peptidase IV (DPP-IV) inhibitor, chemically designated as 2- [[6-[(3i?)-3 -aminopiperidin- 1 -yl] -3 -methyl -2,4-dioxopyrimidin- 1 -yljmethyl] -4-fluorobenzonitrile, represented by Formula I.

Formula I
Trelagliptin is administered as a succinate salt of Formula la, chemically designated as 2-[[6-[(3i?)-3-aminopiperidin-l-yl]-3-methyl-2,4-dioxopyrimidin-l-yl]methyl]-4-fluorobenzonitrile butanedioic acid (1 : 1).

Formula la
U.S. Patent Nos. 7,795,428, 8,288,539, and 8,222,411 provide a process for the preparation of 4-fluoro-2-methylbenzonitrile by reacting 2-bromo-5-fluorotoluene with copper (I) cyanide in N,N-dimethylformamide.
Chinese Patent No. CN 102964196 provides a process for the preparation of 4-fluoro-2-methylbenzonitrile by reacting 4-fluoro-2-methylbenzyl alcohol with cuprous iodide in the presence of 2,2′-bipyridine and 2,2,6,6-tetramethylpiperidine oxide (TEMPO) in an anhydrous ethanol.
Copper (I) cyanide is toxic to humans, and therefore its use in the manufacture of a drug substance is not advisable. In addition, 2-bromo-5-fluorotoluene is converted to 4-fluoro-2-methylbenzonitrile by refluxing in N,N-dimethylformamide at 152°C to 155°C for 24 hours. This leads to some charring, resulting in a tedious work-up process and low yield. Furthermore, the use of reagents like cuprous iodide, 2,2′-bipyridine, and 2,2,6,6-tetramethylpiperidine oxide (TEMPO) is hazardous and/or environmentally-unfriendly, and therefore their use in the manufacture of a drug substance is not desirable.
The present invention provides an efficient, simple, and commercially friendly process for the preparation of 4-fluoro-2-methylbenzonitrile, which is used as an intermediate for the preparation of trelagliptin or its salts. The present invention avoids the use of toxic and hazardous reagents, high boiling solvents, and bromo intermediates such as 2-bromo-5-fluorotoluene, which is lachrymatory in nature and thus difficult to handle at a commercial scale.
EXAMPLES
Example 1 : Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methylbenzaldehyde (1.38 g) was added to ethanol (10 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (2.76 g) and pyridine (1 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 3 hours. The solvent was recovered up to maximum extent from the reaction mixture under reduced pressure to afford the title compound. Yield: 3.1 g
Example 2: Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methylbenzaldehyde (5 g) was added to ethanol (37 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (10 g) and N,N-diisopropylethylamine (3.6 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 2 hours. The solvent was recovered up to maximum extent from the reaction mixture under reduced pressure to afford the title compound. Yield: 3.1 g
Example 3 : Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methylbenzaldehyde (10 g) was added to ethanol (40 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (20 g) and N,N-diisopropylethylamine (7.5 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 4 hours. The solvent was recovered from the reaction mixture under reduced pressure to afford the title compound. Yield: 11.0 g
Example 4: Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methylbenzaldehyde (50 g) was added to ethanol (500 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (70 g) and N,N-diisopropylethylamine (36 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 6 hours. The solvent was recovered from the reaction mixture under reduced pressure to afford the title compound. Yield: 51.0 g
Example 5 : Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methylbenzaldehyde (20 g) was added to ethanol (200 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (20 g) and N,N-diisopropylethylamine (18 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 4 hours. The solvent was recovered from the reaction mixture under reduced pressure to obtain a residue. Deionized water (60 mL) was charged into the residue, and then the slurry was stirred at 0°C to 5°C for 1 hour. The solid obtained was filtered, then washed with deionized water (2 x 20 mL). The wet solid was dried in an air oven at 40°C to 45 °C for 4 hours to 5 hours. The crude product obtained was recrystallized in ethanol (50 mL) to afford the pure title compound. Yield: 21.0 g
Example 6: Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methyl benzaldehyde (50 g) was added to ethanol (500 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (50 g) and N,N-diisopropylethylamine (46.4 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 4 hours. The solvent was recovered from the reaction mixture under reduced pressure to obtain a residue. Deionized water (150 mL) was charged to the residue, and then the slurry was stirred at 0°C to 5°C for 1 hour. The solid obtained was filtered, then washed with deionized water (2 x 50 mL). The wet solid was dried in an air oven at 40°C to 45 °C for 4 hours to 5 hours. The crude product obtained was recrystallized in ethanol (200 mL) to afford the pure title compound. Yield: 53.5 g
Example 7: Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methylbenzaldoxime (3.1 g) and phosphorous pentoxide (1 g) were added to toluene (30 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C for 24 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25°C to 30°C. Deionized water (30 mL) was added to the mixture and then the layers were separated. The organic layer was concentrated under reduced pressure to afford the title compound. Yield: 1.1 g
Example 8: Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methylbenzaldoxime (3 g) and phosphorous pentoxide (2 g) were added to toluene (30 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C for 24 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25°C to 30°C. Deionized water (30 mL) was added to the mixture and then the layers were separated. The organic layer was concentrated under reduced pressure to afford the title compound. Yield: 1.0 g
Example 9: Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methylbenzaldoxime (5 g) and concentrated sulphuric acid (2 mL) were added to toluene (100 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C for 5 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25°C to 30°C. Deionized water (50 mL) was added to the mixture and then the layers were separated. The organic layer was concentrated under reduced pressure to afford the title compound. Yield: 3.24 g
Example 10: Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methylbenzaldoxime (25 g) and concentrated sulphuric acid (35 g) were added to toluene (500 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C for 6 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25°C to 30°C. Deionized water (250 mL) was added to the mixture and then the layers were separated. The organic layer was concentrated under reduced pressure to afford the title compound. Yield: 20.5 g
Example 11 : Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methyl benzaldoxime (5 g) and sodium bisulphate monohydrate (3.1 g) were added to toluene (50 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C for 12 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25°C to 30°C, then filtered, and then washed with toluene (10 mL). The filtrate was concentrated under reduced pressure to afford the title compound. Yield: 3.0 g
Example 12: Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methyl benzaldoxime (50 g) and sodium bisulphate monohydrate (31.6 g) were added to toluene (500 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C using a Dean-Stark apparatus for 12 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25 °C to 30°C, then filtered, and then washed with toluene (100 mL). The filtrate was concentrated under reduced pressure to afford a crude product. The crude product obtained was recrystallized in a mixture of toluene (200 mL) and hexane (500 mL) to afford the title compound.
Yield: 38.0 g

Sun Pharma managing director Dilip Shanghvi.
References
- 1 McKeage, Kate (2015-06-27). “Trelagliptin: First Global Approval”. Drugs 75 (10): 1161–1164. doi:10.1007/s40265-015-0431-9. ISSN 0012-6667.
- 2 Inagaki, Nobuya; Onouchi, Hitoshi; Sano, Hiroki; Funao, Nobuo; Kuroda, Shingo; Kaku, Kohei. “SYR-472, a novel once-weekly dipeptidyl peptidase-4 (DPP-4) inhibitor, in type 2 diabetes mellitus: a phase 2, randomised, double-blind, placebo-controlled trial”. The Lancet Diabetes & Endocrinology 2 (2): 125–132. doi:10.1016/s2213-8587(13)70149-9.
- 3″New Drug Application Approval of Zafatek® Tablets for the treatment of Type 2 Diabetes in Japan | Takeda Pharmaceutical Company Limited”. http://www.takeda.com. Retrieved 2015-11-13.
- 4 Cahn, Avivit; Raz, Itamar (2013-06-01). “Emerging gliptins for type 2 diabetes”. Expert Opinion on Emerging Drugs 18 (2): 245–258. doi:10.1517/14728214.2013.807796. ISSN 1472-8214. PMID 23725569.
- 5Inagaki, Nobuya; Onouchi, Hitoshi; Maezawa, Hideaki; Kuroda, Shingo; Kaku, Kohei. “Once-weekly trelagliptin versus daily alogliptin in Japanese patients with type 2 diabetes: a randomised, double-blind, phase 3, non-inferiority study”. The Lancet Diabetes & Endocrinology 3 (3): 191–197. doi:10.1016/s2213-8587(14)70251-7.
- 6″TRELAGLIPTIN SUCCINATE | C22H26FN5O6 – PubChem”. pubchem.ncbi.nlm.nih.gov. Retrieved 2015-11-13.
- 7″Trelagliptin succinate | C22H26FN5O6 | ChemSpider”. http://www.chemspider.com. Retrieved 2015-11-13.
http://www.cbijournal.com/paper-archive/may-june-2014-vol-3/Review-Paper-1.pdf
| Patent | Submitted | Granted |
|---|---|---|
| TABLET [US2012129878] | 2010-07-27 | 2012-05-24 |
| AROMATIC RING COMPOUND [US2015045378] | 2013-02-12 | 2015-02-12 |
| Patent | Submitted | Granted |
|---|---|---|
| Combination therapy for the treatment of diabetes and related conditions [US2011263617] | 2011-10-27 | |
| Treatment for diabetes in patients with insufficient glycemic control despite therapy with an oral or non-oral antidiabetic drug [US2011275561] | 2011-11-10 | |
| Treatment for diabetes in patients with inadequate glycemic control despite metformin therapy comprising a DPP-IV inhibitor [US2011301182] | 2011-12-08 | |
| COATED PREPARATION [US2010166853] | 2008-07-10 | 2010-07-01 |
| Solid preparation comprising 2-[[6-[(3R)-3-amino-1-piperidinyl]-3,4-dihydro-3-methyl-2,4-dioxo-1(2H)-pyrimidinyl]methyl]-4-fluorobenzonitrile [US7994183] | 2008-03-12 | 2011-08-09 |
| Diabetes therapy [US2012165251] | 2011-06-23 | 2012-06-28 |
| MEDICAL USE OF A DPP-4 INHIBITOR [US2014371243] | 2014-06-13 | 2014-12-18 |
| TREATMENT OF GENOTYPED DIABETIC PATIENTS WITH DPP-IV INHIBITORS SUCH AS LINAGLIPTIN [US2013196898] | 2010-11-26 | 2013-08-01 |
| ANTIDIABETIC MEDICATIONS COMPRISING A DPP-4 INHIBITOR (LINAGLIPTIN) OPTIONALLY IN COMBINATION WITH OTHER ANTIDIABETICS [US2012094894] | 2010-02-12 | 2012-04-19 |
| DPP-IV INHIBITORS FOR TREATMENT OF DIABETES IN PEDIATRIC PATIENTS [US2012122776] | 2010-01-29 | 2012-05-17 |
| Patent | Submitted | Granted |
|---|---|---|
| LAMINATED TABLET AND MANUFACTURING METHOD THEREFOR [US2014023708] | 2012-03-02 | 2014-01-23 |
| Combination therapy for the treatment of diabetes and related conditions [US2013310398] | 2013-07-24 | 2013-11-21 |
| USE OF KERATINOCYTES AS A BIOLOGICALLY ACTIVE SUBSTANCE IN THE TREATMENT OF WOUNDS, SUCH AS DIABETIC WOUNDS, OPTIONALLY IN COMBINATION WITH A DPP-4 INHIBITOR [US2013315975] | 2013-05-23 | 2013-11-28 |
| USE OF A DPP-4 INHIBITOR IN AUTOIMMUNE DIABETES, PARTICULARLY LADA [US2013317046] | 2013-05-21 | 2013-11-28 |
| USE OF A DPP-4 INHIBITOR FOR MODIFYING FOOD INTAKE AND REGULATING FOOD PREFERENCE [US2013324463] | 2013-05-21 | 2013-12-05 |
| COMBINATION THERAPY [US2013281373] | 2011-05-05 | 2013-10-24 |
| USE OF A DPP-4 INHIBITOR IN PODOCYTES RELATED DISORDERS AND/OR NEPHROTIC SYNDROME [US2013303462] | 2013-05-13 | 2013-11-14 |
| USE OF A DPP-4 INHIBITOR IN SIRS AND/OR SEPSIS [US2013303554] | 2013-05-13 | 2013-11-14 |
| Combination of a GPR119 Agonist and the DPP-IV Inhibitor Linagliptin for Use in the Treatment of Diabetes and Related Conditions [US2013109703] | 2011-03-18 | 2013-05-02 |
| Treatment for diabetes in patients inappropriate for metformin therapy [US2011263493] | 2011-10-27 |
| Patent | Submitted | Granted |
|---|---|---|
| DIPEPTIDYL PEPTIDASE INHIBITORS [US7781584] | 2008-07-03 | 2010-08-24 |
| POLYMORPHS OF SUCCINATE SALT OF 2-[6-(3-AMINO-PIPERIDIN-1-YL)-3-METHYL-2,4-DIOXO-3,4-DIHYDRO-2H-PYRIMIDIN-1-YLMETHY]-4-FLUOR-BENZONITRILE AND METHODS OF USE THEREFOR [US2008227798] | 2008-09-18 | |
| GPR119 receptor agonists in methods of increasing bone mass and of treating osteoporosis and other conditions characterized by low bone mass, and combination therapy relating thereto [US7816364] | 2009-10-29 | 2010-10-19 |
| DIPEPTIDYL PEPTIDASE INHIBITORS [US8222411] | 2009-11-05 | 2012-07-17 |
| ADMINISTRATION OF DIPEPTIDYL PEPTIDASE INHIBITORS [US2008287476] | 2008-11-20 | |
| POLYMORPHS OF SUCCINATE SALT OF 2-[6-(3-AMINO-PIPERIDIN-1-YL)-3-METHYL-2,4-DIOXO-3,4-DIHYDRO-2H-PYRIMIDIN-1-YLMETHY]-4-FLUOR-BENZONITRILE AND METHODS OF USE THEREFOR [US8084605] | 2008-11-13 | 2011-12-27 |
| WEEKLY ADMINISTRATION OF DIPEPTIDYL PEPTIDASE INHIBITORS [US8093236] | 2008-11-06 | 2012-01-10 |
| Therapeutic Agent for Diabetes [US2009042863] | 2009-02-12 | |
| ADMINISTRATION OF DIPEPTIDYL PEPTIDASE INHIBITORS [US2007060530] | 2007-03-15 | |
| DIPEPTIDYL PEPTIDASE INHIBITORS [US7795428] | 2008-01-03 | 2010-09-14 |
| Patent | Submitted | Granted |
|---|---|---|
| Dipeptidyl peptidase inhibitors [US7807689] | 2005-11-24 | 2010-10-05 |
| DIPEPTIDYL PEPTIDASE INHIBITORS [US2008108807] | 2008-05-08 | |
| DIPEPTIDYL PEPTIDASE INHIBITORS [US2008108808] | 2008-05-08 | |
| FUSED CYCLIC COMPOUNDS [US7732626] | 2010-01-07 | 2010-06-08 |
| DIPEPTIDYL PEPTIDASE INHIBITORS [US7906523] | 2008-08-07 | 2011-03-15 |
| DIPEPTIDYL PEPTIDASE INHIBITORS [US8188275] | 2008-07-24 | 2012-05-29 |
| DIPEPTIDYL PEPTIDASE INHIBITORS [US8173663] | 2009-01-08 | 2012-05-08 |
| ADMINISTRATION OF DIPEPTIDYL PEPTIDASE INHIBITORS [US2011077402] | 2011-03-31 | |
| DPP-IV INHIBITORS FOR USE IN THE TREATMENT OF NAFLD [US2011092510] | 2011-04-21 | |
| PURIN DERIVATIVES FOR USE IN THE TREATMENT OF FAB-RELATED DISEASES [US2011190322] | 2011-08-04 |
| Patent | Submitted | Granted |
|---|---|---|
| Administration of Dipeptidyl Peptidase Inhibitors [US2011192748] | 2011-08-11 | |
| PHARMACEUTICAL COMPOSITION COMPRISING A GLUCOPYRANOSYL-SUBSTITUTED BENZENE DERIVATE [US2011195917] | 2011-08-11 | |
| DPP-IV INHIBITOR COMBINED WITH A FURTHER ANTIDIABETIC AGENT, TABLETS COMPRISING SUCH FORMULATIONS, THEIR USE AND PROCESS FOR THEIR PREPARATION [US2011206766] | 2011-08-25 | |
| COMBINATION OF A CERTAIN DPP-4 INHIBITOR AND VOGLIBOSE [US2014343014] | 2014-05-16 | 2014-11-20 |
| CARDIO- AND RENOPROTECTIVE ANTIDIABETIC THERAPY [US2014274889] | 2014-03-14 | 2014-09-18 |
| TREATMENT FOR DIABETES IN PATIENTS INAPPROPRIATE FOR METFORMIN THERAPY [US2014274890] | 2014-06-03 | 2014-09-18 |
| Fused ring compound and use thereof [US2010190747] | 2010-07-29 | |
| FUSED RING COMPOUND AND USE THEREOF [US2010197683] | 2010-08-05 | |
| Fused cyclic compounds [US8088821] | 2010-08-05 | 2012-01-03 |
| GPR119 Receptor Agonists in Methods of Increasing Bone Mass and of Treating Osteoporosis and Other Conditions Characterized by Low Bone Mass, and Combination Therapy Relating Thereto [US8101626] | 2010-07-29 | 2012-01-24 |
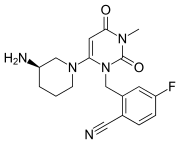 |
|
| Systematic (IUPAC) name | |
|---|---|
|
Succinic acid – 2-({6-[(3R)-3-amino-1-piperidinyl]-3-methyl-2,4-dioxo-3,4-dihydro-1(2H)-pyrimidinyl}methyl)-4-fluorobenzonitrile (1:1)
|
|
| Clinical data | |
| Trade names | Zafatek |
| Chemical data | |
| Formula | C22H26FN5O6 |
| Molar mass | 475.470143 g/mol |
/////////Trelagliptin, PMDA, JAPAN 2015
Cn1c(=O)cc(n(c1=O)Cc2cc(ccc2C#N)F)N3CCC[C@H](C3)N

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....