Home » Posts tagged 'GM-1020'
Tag Archives: GM-1020
Blixeprodil
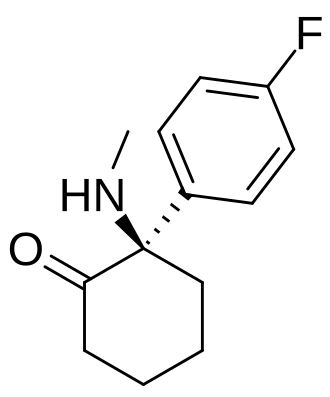
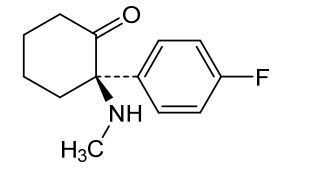
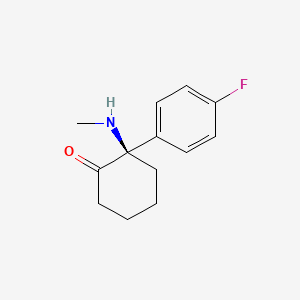
Blixeprodil
CAS 2881017-49-6
MF C13H16FNO MW 221.27 g/mol
Cyclohexanone, 2-(4-fluorophenyl)-2-(methylamino)-, (2R)-
(2R)-2-(4-fluorophenyl)-2-(methylamino)cyclohexan-1-one
N-methyl-D-aspartate (NMDA) receptor antagonist, GM-1020, GM1020, (R)-4-Fluorodeschloroketamine, (R)-4-FDCK, (R)-4FDCK, S2MGG2PC5K
Blixeprodil,[5] also known by its developmental code name GM-1020 or as (R)-4-fluorodeschloroketamine ((R)-4-FDCK), is an NMDA receptor antagonist related to ketamine which is under development for the treatment of major depressive disorder, bipolar depression, and other depressive disorders.[1][6][2][3][7][8] It is taken by mouth.[1][2][3]
The drug is orally active, in contrast to the poor oral bioavailability of ketamine.[3] Its oral bioavailability is >60%.[4][9] The time to peak levels of blixeprodil is 1.5 hours and its elimination half-life is 4.3 hours.[4] In a clinical study comparing it with the serotonergic psychedelic bretisilocin (GM-2505), both blixeprodil and bretisilocin produced hallucinogenic effects.[10]
Blixeprodil shows antidepressant-like effects in rodents.[3][11][4][9] It appears to have a greater separation between antidepressant-like and ataxia-inducing doses than ketamine in rodents and hence might have better tolerability.[3][7][9] Whereas ketamine shows only 3-fold separation between antidepressant-like and ataxic doses, there was 13-fold separation for blixeprodil, and it did not produce hyperlocomotion at doses >20-fold higher than the minimum antidepressant-like dose.[9] In relation to the preceding, blixeprodil is claimed to be non-dissociative at therapeutic doses.[2][4] However, dissociative and other related effects have been observed at low incidences and at higher doses.[4]
The drug is a close analogue of ketamine, with a 4-fluoro group instead of a 2-chloro group on the phenyl ring and in (2R)-enantiopure form.[12] Hence, blixeprodil is related to arketamine ((R)-ketamine); it is said to “bet” on the notion that arketamine is importantly involved in the antidepressant effects of ketamine, in spite of arketamine having less propensity for inducing dissociation.[13]
Blixeprodil is being developed by Gilgamesh Pharmaceuticals.[1][6][2] As of July 2024, it is in phase 2 clinical trials for major depressive disorder and bipolar depression and is in phase 1 trials for other depressive disorders.[1][6][2]
SYN
Example 15: Preparation of Compounds 117rac and 18rac
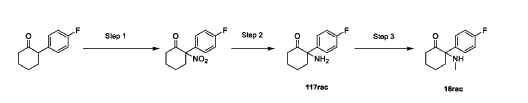
Step 1: Preparation of 2-(4-fluorophenyl)-2-nitrocyclohexan-1-one
[0364] A mixture of 2-(4-fluorophenyl)cyclohexan-1-one (5 g, 26.01 mmol, 1 eq), ceric ammonium nitrate (CAN, 28.52 g, 52.02 mmol, 2 eq), and Cu(OAc)2 (945 mg, 5.20 mmol, 0.2 eq) in DCE (50 mL) was stirred at 85 °C for 12 hrs. The mixture was cooled, filtered and concentrated. The residue was purified by column chromatography (SiO2, Petroleum ether/Ethyl acetate=100/1 to 0/1) to afford 2-(4-fluorophenyl)-2-nitrocyclohexan-1-one (2.5 g, 10.54 mmol, 40.52% yield) as a yellow oil.1H NMR (400MHz, CHLOROFORM-d) δ = 7.47 – 7.29 (m, 2H), 7.22 – 7.04 (m, 2H), 3.12 (ddd, J = 3.6, 10.0, 14.0 Hz, 1H), 2.86 – 2.76 (m, 1H), 2.75 – 2.62 (m, 1H), 2.61 – 2.47 (m, 1H), 2.08 – 1.86 (m, 3H), 1.80 (dt, J = 3.6, 9.2 Hz, 1H).
Step 2: Preparation of 2-amino-2-(4-fluorophenyl)cyclohexan-1-one (117rac)
[0365] A mixture of 2-(4-fluorophenyl)-2-nitrocyclohexan-1-one (3 g, 12.65 mmol, 1 eq) and Zn (19.85 g, 303.51 mmol, 24 eq) in AcOH (25 mL) was stirred at 20 °C for 12 hrs. The mixture was cooled, filtered, and concentrated. The residue was dissolved in DCM, washed with sat.
NaHCO3, H2O, and brine, dried over Na2SO4, filtered, and concentrated. The residue was
purified by silica gel (PE:EA = 50:1 – 8:1) to afford 2-amino-2-(4-fluorophenyl)cyclohexan-1- one (1.5 g, 7.24 mmol, 57.23% yield) (117rac) as a brown oil. LCMS (RT = 1.336 min, MS calc.: 207.11, [M+H]+ = 208.1) 1H NMR (400MHz, CHLOROFORM-d) δ = 7.26 – 7.19 (m, 2H), 7.11 – 7.01 (m, 2H), 2.87 – 2.73 (m, 1H), 2.50 – 2.42 (m, 1H), 2.41 – 2.29 (m, 1H), 2.04 – 1.96 (m, 1H), 1.93 (s, 2H), 1.83 – 1.63 (m, 4H); 3C NMR (101 MHz, CHLOROFORM-d) δ = 213.28, 163.27, 160.82, 137.67, 137.63, 127.99, 127.91, 116.16, 115.95, 65.93, 39.71, 28.08, 22.61
Step 3: Preparation of 2-(4-fluorophenyl)-2-(methylamino)cyclohexan-1-one (18rac)
[0366] A mixture of 2-amino-2-(4-fluorophenyl)cyclohexan-1-one (1.3 g, 6.27 mmol, 1 eq) and methyl trifluoromethanesulfonate (1.03 g, 6.27 mmol, 1 eq) in hexafluoroisopropanol (HFIP, 130 mL) was stirred at 0 – 25 °C for 12 hrs under N2 atmosphere. The mixture was filtered and concentrated. The residue was adjusted to pH = 7 with sat. Na2CO3 (20 ml). The aqueous phase was extracted with EA (50 mL x 2). The combined organic phase was washed with brine (50 mL x 2), dried with anhydrous Na2SO4, filtered, and concentrated in vacuo. The residue was purified by prep-HPLC (column: Welch Xtimate C18250*70 mm, 10 μm; mobile phase: A: water(0.05% NH3H2O), B: ACN; B%: 18% – 48%, 32 min) to afford 2-(4-fluorophenyl)-2- (methylamino)cyclohexan-1-one (590 mg, 4.02 mmol, 42.45% yield) (18rac) as a white solid. LCMS (RT = 1.415 min, MS calc.: 221.12, [M+H]+ = 222.1); 1H NMR (400 MHz, CHLOROFORM-d) δ = 7.26 – 7.17 (m, 2H), 7.07 (br t, J = 8.4 Hz, 2H), 2.92 – 2.74 (m, 1H), 2.50 – 2.26 (m, 3H), 2.12 – 1.93 (m, 4H), 1.90 – 1.63 (m, 4H); 13C NMR (101 MHz, CHLOROFORM-d) δ = 211.15, 163.20, 160.75, 134.68, 134.65, 128.99, 128.91, 115.79, 115.58, 69.37, 39.70, 35.85, 28.87, 27.70, 22.21.
SYN
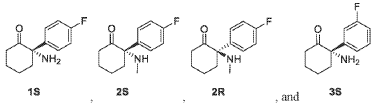
Example 1: Preparation of Compounds 1 and 2 and Their Enantiomers.
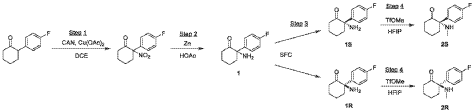
Step 1: Preparation of 2-(4-fluorophenyl)-2-nitrocyclohexan-1-one
[0110] A mixture of 2-(4-fluorophenyl)cyclohexan-1-one (14 g, 72.83 mmol, 1 eq), CAN (79.85 g, 145.66 mmol, 72.59 mL, 2 eq), and Cu(OAc)2 (2.65 g, 14.57 mmol, 0.2 eq) in DCE (140 mL) was stirred at 85 °C for 12 h. On completion, the mixture was filtered and concentrated. The residue was purified by column chromatography (SiO2, Petroleum ether/Ethyl acetate=100/1 to 0/1) to afford 2-(4-fluorophenyl)-2-nitrocyclohexan-1-one (6.1 g, 25.71 mmol, 35.31% yield) as a yellow solid.1H NMR (400 MHz, CHLOROFORM-d) δ = 7.41 – 7.31 (m, 2H), 7.16 (t, J=8.4 Hz, 2H), 3.11 (ddd, J=3.6, 10.4, 14.0 Hz, 1H), 2.87 – 2.76 (m, 1H), 2.73 – 2.64 (m, 1H), 2.60 -2.48 (m, 1H), 2.02 – 1.88 (m, 3H), 1.84 – 1.72 (m, 1H).
Step 2: Preparation of 2-amino-2-(4-fluorophenyl)cyclohexan-1-one (1)
[0111] To a mixture of 2-(4-fluorophenyl)-2-nitrocyclohexan-1-one (5.6 g, 23.61 mmol, 1 eq) in AcOH (10 mL) was added Zn (15.44 g, 236.06 mmol, 10 eq) in several portions and the resulting mixture was stirred at 30 °C for 12 h. On completion, the mixture was filtered and concentrated. The residue was dissolved in DCM (20 mL), washed with sat. aq. NaHCO3 (10 mL), H2O (5 mL), and brine (10 mL), dried over Na2SO4, filtered, and concentrated. The residue was purified by prep-HPLC (column: Agela DuraShell C18 (250 mm*80 mm, 10 μm); mobile phase: A: water (NH4HCO3), B: ACN; B%: 35%, 20 min) to afford 2-amino-2-(4-fluorophenyl)cyclohexan-1-one (2.9 g, 13.99 mmol, 59.28% yield, 1) as a brown oil.1H NMR (400 MHz, CHLOROFORM-d) δ = 7.52 – 7.40 (m, 2H), 7.32 (br s, 1H), 7.34 – 7.20 (m, 2H), 2.93 – 2.92 (m, 1H), 3.08 – 2.92 (m, 1H), 2.74 – 2.63 (m, 1H), 2.63 – 2.50 (m, 1H), 2.28 – 2.16 (m, 1H), 2.10 (br s, 2H), 2.04 – 1.85 (m, 4H).
Note: The free base of this compound is unstable and dimerizes over time. It should be stored frozen or quickly converted to the HCl salt to prevent this.
Step 3: Preparation of (S)-2-amino-2-(4-fluorophenyl)cyclohexan-1-one (1S) and (R)-2-amino-2-(4-fluorophenyl)cyclohexan-1-one (1R)
[0112] The racemate 1 (2.9 g) was separated by SFC (column: DAICEL CHIRALPAK AD (250 mm*30 mm, 10 μm); mobile phase: A: CO2, B: 0.1% NH3H2O in ETOH; B%: 27%, multi-injection process with 6-min spacing between injections) to afford ENT-1 free base (RT = 2.266 min, 1.1 g, 1.62 mmol, 1S_FB) as a yellow oil and ENT-2 free base (RT = 2.945 min, 1.1 g, 1.28 mmol, 1R_FB) as a yellow oil.
[0113] A portion of each free base was further purified by prep-HPLC (column: Welch Xtimate C18 (100 mm*25 mm, 3 μm); mobile phase: A: water (0.04% HCl), B: ACN; B%: 1% – 20%, 8 min) to afford ENT-1 HCl (RT = 2.266 min, 272 mg, HCl salt, 1S) as a white solid and ENT-2 HCl (RT = 2.945 min, 283 mg, HCl salt, 1R) as a white solid.
[0114] ENT-1 HCl, RT = 2.266 min (assigned here as the S isomer, 1S); LCMS (RT = 1.449 min, MS calc.: 207.1, [M+H]+ = 208.1); 1H NMR (400MHz, DMSO-d6) δ = 8.83 (br s, 3H), 7.50 – 7.42 (m, 2H), 7.41 – 7.32 (m, 2H), 3.03 (br dd, J=2.4, 14.0 Hz, 1H), 2.45 – 2.27 (m, 2H), 2.21 -2.05 (m, 1H), 1.97 (td, J=2.8, 9.6 Hz, 1H), 1.81 (br d, J=11.6 Hz, 1H), 1.71 – 1.47 (m, 2H); 13C NMR (101 MHz, DMSO-d6) δ = 206.52, 164.22, 161.76, 130.78, 130.69, 130.08, 130.05, 116.90, 116.68, 66.26, 34.75, 27.52, 21.53; ENT-2 HCl, RT = 2.945 min (assigned here as the R isomer, 1R); LCMS (RT = 1.449 min, MS calc.: 207.1, [M+H]+ = 208.0); 1H NMR (400MHz, DMSO-d6) δ = 8.84 (br s, 3H), 7.49 – 7.42 (m, 2H), 7.40 – 7.33 (m, 2H), 3.03 (br dd, J=1.6, 14.0 Hz, 1H), 2.45 – 2.27 (m, 2H), 2.23 – 2.06 (m, 1H), 1.97 (dt, J=2.8, 6.1 Hz, 1H), 1.81 (br d, J=11.6 Hz, 1H), 1.70 – 1.46 (m, 2H); 13C NMR (101 MHz, DMSO-d6) δ = 206.50, 164.22, 161.76, 130.78, 130.70, 130.08, 130.05, 116.89, 116.68, 66.26, 34.75, 27.51, 21.52.
[0115] The retention times above, which identify the enantiomers, were determined using the free bases using the following chiral analytical method: column: Chiralpak AD-3 (150 mm×4.6 mm I.D., 3 μm); mobile phase: A: CO2 B: EtOH (0.1% IPAm, v/v); gradient (Time (min)/A%/B%): 0.0/90/10, 0.5/90/10, 3.5/50/50, 4.5/50/50, 5.0/90/10; flow rate: 2.5 mL/min; column temp.: 35 °C; ABPR: 2,000 psi.
Step 4: Preparation of (S)-2-(4-fluorophenyl)-2-(methylamino)cyclohexan-1-one (2S) and (R)-2-(4-fluorophenyl)-2-(methylamino)cyclohexan-1-one (2R)
[0116] Compound 1S_FB (540 mg, 2.61 mmol, 1 eq) and methyl trifluoromethanesulfonate (427.59 mg, 2.61 mmol, 285.06 μL, 1 eq) were combined in hexafluoroisopropanol (40 mL) at 0
°C under N2 atmosphere and then the mixture was allowed to warm to 25 °C and stirred for 12 h. On completion, the residue was adjusted to pH 7 with sat. aq. Na2CO3 (10 mL) and the combined organic phase was washed with brine (100 mL * 2), dried over Na2SO4, filtered, and concentrated in vacuum. The residue was purified by prep-HPLC (column: Waters Xbridge C18 (150 mm*50 mm, 10μm); mobile phase: A: water (10 mM NH4HCO3), B: ACN; B%: 30% – 50%, 10 min) to afford 2S (260 mg, 1.18 mmol, 45.10% yield) as a white solid. Compound 2R was prepared by the same procedure starting from 1R_FB (590 mg, 2.85 mmol) in hexafluoroisopropanol (60 mL) (other quantities scaled based on molar equivalents) and obtained as an off-white solid (260 mg, 1.18 mmol, 41.27% yield).
[0117] 2S (assigned here as the S isomer) (free base); LCMS (RT = 1.427 min, MS calc.: 221.1, [M+H]+ = 222.1); 1H NMR (400MHz, CHLOROFORM-d) δ = 7.21 (dd, J = 5.4, 8.8 Hz, 2H), 7.10 – 7.02 (m, 2H), 2.85 – 2.74 (m, 1H), 2.49 – 2.37 (m, 1H), 2.36 – 2.25 (m, 1H), 2.22 (br s, 1H), 2.03 (s, 3H), 1.96 (dt, J = 3.2, 5.8 Hz, 1H), 1.88 – 1.64 (m, 4H); 13C NMR (101 MHz, CHLOROFORM-d) δ = 211.25, 163.22, 160.76, 134.80, 134.77, 128.98, 128.90, 115.80, 115.59, 69.38, 39.73, 35.92, 28.92, 27.72, 22.24; 2R (assigned here as the R isomer) (free base); LCMS (RT = 1.415 min, MS calc.: 221.1, [M+H]+ = 222.1); 1H NMR (400MHz, CHLOROFORM-d) δ = 7.25 – 7.17 (m, 2H), 7.11 – 7.02 (m, 2H), 2.85 – 2.75 (m, 1H), 2.48 – 2.38 (m, 1H), 2.35 – 2.19 (m, 2H), 2.04 (s, 3H), 1.97 (br dd, J = 2.8, 6.1 Hz, 1H), 1.89 – 1.66 (m, 4H); 13C NMR (101 MHz, CHLOROFORM-d) δ = 211.24, 163.22, 160.77, 134.78, 134.74, 128.99, 128.91, 115.81, 115.60, 69.38, 39.73, 35.91, 28.91, 27.72, 22.24.
PAT
- Methods of treating neuropsychiatric disordersPublication Number: US-2024216339-A1Priority Date: 2021-06-08
- Arylcyclohexylamine derivatives and their use in the treatment of psychiatric disordersPublication Number: US-11344510-B2Priority Date: 2019-12-26Grant Date: 2022-05-31
- Arylcyclohexylamine derivatives and their use in the treatment of psychiatric disordersPublication Number: US-2022409555-A1Priority Date: 2019-12-26
- Arylcyclohexylamine derivatives and their use in the treatment of psychiatric disordersPublication Number: WO-2021134086-A1Priority Date: 2019-12-26
- A treatment for patients with mood disorders using n-ethyl-2-(5-fluoro-1h-indol-3-yl)- n-methylethan-1-amine or a pharmaceutically acceptable salt thereofPublication Number: WO-2025111597-A1Priority Date: 2023-11-22
- Methods of treating psychiatric disorders or pain using (r)-2-(4-fluorophenyl)-2-(methylamino)cyclohexan-1-one or pharmaceutically acceptable salts thereofPublication Number: WO-2025106879-A1Priority Date: 2023-11-15
- (ampa-pam)-nmda receptor antagonist combination therapy for treatment of mental conditions and disordersPublication Number: WO-2023154450-A2Priority Date: 2022-02-11
- Arylcyclohexylamine derivatives and their use in the treatment of psychiatric disordersPublication Number: US-2024300886-A1Priority Date: 2021-06-25
- Arylcyclohexylamine derivatives and their use in the treatment of psychiatric disordersPublication Number: EP-4358946-A1Priority Date: 2021-06-25



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
References
- “GM 1020”. AdisInsight. 12 July 2024. Retrieved 20 February 2025.
- Peplow M (June 2024). “Next-generation psychedelics: should new agents skip the trip?”. Nature Biotechnology. 42 (6): 827–830. doi:10.1038/s41587-024-02285-1. PMID 38831049.
Other companies are confident that they can further reduce or even erase those effects without losing therapeutic efficacy. Gilgamesh, for example, is taking that approach with ketamine, DMT and psilocybin. In the case of ketamine, says Kruegel, the dissociative side effects require that the subjects remain under supervision. So Gilgamesh designed a ketamine analog called GM-1020 that has no dissociative effects (distortions in sight, sound and feelings of detachment) and that also has better oral bioavailability than ketamine itself. After completing a phase 1 trial last year, the company began dosing patients with GM-1020 in a phase 2 trial for major depressive disorder in March. “The hope is that the psychoactive effects will be limited enough that this can eventually be taken at home,” says Kruegel.
- Klein AK, Austin EW, Cunningham MJ, Dvorak D, Gatti S, Hulls SK, et al. (May 2024). “GM-1020: a novel, orally bioavailable NMDA receptor antagonist with rapid and robust antidepressant-like effects at well-tolerated doses in rodents”. Neuropsychopharmacology. 49 (6): 905–914. doi:10.1038/s41386-023-01783-1. PMC 11039472. PMID 38177696.
- Marek G, Umbricht D, Christian E, Winters J, Raines S, Kiss L, et al. (December 2023). “ACNP 62nd Annual Meeting: Poster Abstracts P251 – P500: P352. GM-1020: An Oral NMDA Receptor Antagonist for Depression Demonstrates Target Engagement at Doses That Do Not Cause Dissociation, Ataxia or Sedation in a Phase 1 Single Ascending Dose Study”. Neuropsychopharmacology. 48 (Suppl 1): 211–354 (269–269). doi:10.1038/s41386-023-01756-4. PMC 10729596. PMID 38040810.
- https://iris.who.int/bitstream/handle/10665/380497/9789240107038-eng.pdf “blixeprodilum blixeprodil (2R)-2-(4-fluorophenyl)-2-(methylamino)cyclohexan-1-one N-methyl-D-aspartate (NMDA) receptor antagonist”
- “Delving into the Latest Updates on GM-1020 with Synapse”. Synapse. 15 February 2025. Retrieved 20 February 2025.
- Klein A, Dvorak D, Austin E, Marek G, Sporn J, Hughes Z, et al. (2023). “531. GM-1020 is a Novel, Orally Bioavailable NMDA Antagonist With Improved Separation Between Antidepressant and Ataxic Doses Compared to Ketamine”. Biological Psychiatry. 93 (9): S308–S309. doi:10.1016/j.biopsych.2023.02.771.
- Hughes Z (December 2024). “ACNP 63rd Annual Meeting: Panels, Mini-Panels and Study Groups: 19.4 Translational Profile of GM-1020, a Novel Orally Bioavailable NMDA Receptor Antagonist That Achieves Robust Target Engagement Without Dissociation or Sedation”. Neuropsychopharmacology. 49 (Suppl 1): 1–64 (25–25). doi:10.1038/s41386-024-02010-1. PMC 11627185. PMID 39643632.
- Kiss L, Klein A, Austin E, Dvorak D, Gatti S, Papp M, et al. (December 2022). “ACNP 61st Annual Meeting: Poster Abstracts P1 – P270: P215. GM-1020: A Novel, Orally Bioavailable NMDA Receptor Antagonist With Rapid and Robust Antidepressant Effects and Reduced Ataxia in Rodents”. Neuropsychopharmacology. 47 (Suppl 1): 63–219 (185–186). doi:10.1038/s41386-022-01484-1. PMC 9714397. PMID 36456693.
- Dvorak D, Christian E, Hughes Z, Klein A, Austin E, Kiss L, et al. (2024). “ACNP 63rd Annual Meeting: Poster Abstracts P1-P304: P87. GM-1020 (NMDA Antagonist) Vs GM-2505 (5-HT2A Agonist) – Distinct Mechanisms, Same Outcome?”. Neuropsychopharmacology. 49 (S1): 65–235. doi:10.1038/s41386-024-02011-0. ISSN 0893-133X. PMC 11627186. Retrieved 19 January 2026.
- Trunnell ER, Baines J, Farghali S, Jackson T, Jayne K, Smith R, et al. (August 2024). “The need for guidance in antidepressant drug development: Revisiting the role of the forced swim test and tail suspension test”. Regulatory Toxicology and Pharmacology. 151 105666. doi:10.1016/j.yrtph.2024.105666. PMID 38942190.
- Sá VL, de Jesus Santos G, da Fonseca Fraga I, da Silva JM, Santos, MG, et al. (2015). Avaliação farmacológica de um análogo a um antagonista do receptor N-Metil-D-Aspartato [Pharmacological evaluation of an analogue of an N-Methyl-D-Aspartate receptor antagonist] (PDF). I Congresso de Ciências Farmacêuticas do Interior Baiano.
[Translated:] […] ketamine has low oral availability and a narrow therapeutic index, generating adverse effects such as dissociation, cognitive impairment, sedation, and ataxia, which limits the acceptance of the drug in the treatment of depression. The preclinical characterization through in vitro and in vivo studies of GM-1020 ((R)-2-(4-fluorophenyl)-2-(methylamino)cyclohexan-1-one) may indicate a new therapy that presents bioavailability when administered orally and absence of undesirable motor effects.
- Gunther M (31 January 2023). “Gilgamesh Tweaks Known Psychedelics To Improve Therapies”. Lucid News – Psychedelics, Consciousness Technology, and the Future of Wellness. Retrieved 20 February 2025.
| Clinical data | |
|---|---|
| Other names | GM-1020; GM1020; (R)-4-Fluorodeschloroketamine; (R)-4-FDCK; (R)-4FDCK |
| Routes of administration | Oral[1][2][3] |
| Drug class | NMDA receptor antagonist[1][2][3] |
| Pharmacokinetic data | |
| Bioavailability | >60%[4] |
| Elimination half-life | 4.3 hours[4] |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2881017-49-6 |
| PubChem CID | 156552274 |
| Chemical and physical data | |
| Formula | C13H16FNO |
| Molar mass | 221.275 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
///////////blixeprodil, N-methyl-D-aspartate (NMDA) receptor antagonist, GM-1020, GM1020, (R)-4-Fluorodeschloroketamine, (R)-4-FDCK, (R)-4FDCK, S2MGG2PC5K

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










