Home » Posts tagged 'Gennova Biopharmaceuticals'
Tag Archives: Gennova Biopharmaceuticals
PTX-COVID19-B
PTX-COVID19-B
| mRNA-based vaccine |
Providence Therapeutics; Canadian government
bioRxiv (2021), 1-50.
https://www.biorxiv.org/content/10.1101/2021.05.11.443286v1
Safe and effective vaccines are needed to end the COVID-19 pandemic caused by SARS-CoV-2. Here we report the preclinical development of a lipid nanoparticle (LNP) formulated SARS-CoV-2 mRNA vaccine, PTX-COVID19-B. PTX-COVID19-B was chosen among three candidates after the initial mouse vaccination results showed that it elicited the strongest neutralizing antibody response against SARS-CoV-2. Further tests in mice and hamsters indicated that PTX-COVID19-B induced robust humoral and cellular immune responses and completely protected the vaccinated animals from SARS-CoV-2 infection in the lung. Studies in hamsters also showed that PTX-COVID19-B protected the upper respiratory tract from SARS-CoV-2 infection. Mouse immune sera elicited by PTX-COVID19-B vaccination were able to neutralize SARS-CoV-2 variants of concern (VOCs), including the B.1.1.7, B.1.351 and P.1 lineages. No adverse effects were induced by PTX-COVID19-B in both mice and hamsters. These preclinical results indicate that PTX-COVID19-B is safe and effective. Based on these results, PTX-COVID19-B was authorized by Health Canada to enter clinical trials in December 2020 with a phase 1 clinical trial ongoing (ClinicalTrials.gov number: NCT04765436).
PTX-COVID19-B is a messenger RNA (mRNA)-based COVID-19 vaccine, a vaccine for the prevention of the COVID-19 disease caused by an infection of the SARS-CoV-2 coronavirus, created by Providence Therapeutics—a private Canadian drug company co-founded by Calgary, Alberta-based businessman Brad T. Sorenson and San Francisco-based Eric Marcusson.[1] in 2013. A team of eighteen working out of Sunnybrook Research Institute in Toronto, Ontario developed PTX-COVID19-B[2] in less than four weeks, according to the Calgary Herald.[3] Human trials with sixty volunteers began on January 26, 2021 in Toronto.[4][5][6]
Providence, which has no manufacturing facilities, partnered with Calgary-based Northern mRNA—the “anchor tenant” in their future manufacturing facilities pending financing.[2]
On 30 April 2021, Sorenson announced that Providence Therapeutics would be leaving Canada and any vaccine that it developed would not be manufactured in Canada.[2]
Overview
Providence Therapeutics Holdings Inc. was co-founded in Toronto, Ontario[7][8] by Calgary, Alberta-based businessman Brad T. Sorenson and San Francisco-based Eric Marcusson Ph.D, who was also the Chief Scientific Officer.[9][3]
PTX-COVID19-B is a messenger RNA (mRNA)-based COVID-19 vaccine. In an interview with CTV news, Sorenson said they were “building some of the important building blocks for the messenger RNA … that provides instructions to cells … to build proteins that may treat or prevent disease”.
As of January 2021, Northern RNA’s Calgary lab was proposed as the site where manufacturing of PTX-COVID19-B would take place.[10] Providence Therapeutics’ partner, Northern RNA, which located at 421 7 Avenue SW in Calgary, has been described as Providence Therapeutics northern division.[7][8]
A February 2021 Manitoba government press release said that the Winnipeg-based Emergent BioSolutions would be manufacturing the vaccine.[11]
Human trials
Phase 1
Human trials began on January 26, 2021 with 60 volunteers between the ages of 18 to 65 in Toronto.[12][13][3] Of these, 15 would receive a placebo and 3 groups of 15 would receive different doses of the vaccine.[10] The volunteers will be monitored for 13 months. The company said that enough data would be available in May which could result in a Phase 2 clinical testing beginning soon after that, pending regulatory approval. If the results of a subsequent larger human trial are positive, the vaccine could enter a commercialization phase in 2022.[14] The Phase 1 clinical trial lead was Piyush Patel. At the 29 April meeting with the House of Commons, Sorenson estimated that PTX-COVID19-B could be approved by Health Canada by “January or February 2022”.[15]:8
Provincial funding
Shortly after the first human trials on PTX-COVID19-B began in late January, on 11 February 2021, Manitoba Premier Brian Pallister announced a “term sheet” between the province and Providence Therapeutics through which Manitoba would receive 2 million doses of PTX-COVID19-B pending its approval by Health Canada.[11] The term sheet includes “best-price guarantee” PTX-COVID19-B.[13] According to a provincial statement released by the Manitoba government, pending approval of the vaccine, the actual manufacturing would take place in Winnipeg by Emergent BioSolutions.[11] Pallister said that, “Building a secure, made-in-Canada vaccine supply will put Canadians at the head of the line to get a COVID vaccine, where we belong.”[11] The down payment would be 20% with a subsequent 40% to be paid when the vaccine was approved by Health Canada; the balance would be paid on delivery of the doses.[13] Specifics about the contract were released in April 2021: the total cost was estimated as CAD $36 million and the agreement included a clause for a non-refundable advance payment of CAD $7.2 million.[2] Sorenson made this comment to Global News: “Under no circumstances is Manitoba going to be on the hook for $7.2 million unless they get real value out of it”.
Federal funding
Canada’s National Research Council (NRC) provided Providence Therapeutics with CAD $5 million for the launch of January 2021 first phase of PTX-COVID19-B clinical trials.[2]
As part of the federal government’s “next generation manufacturing supercluster” program, Providence and Northern mRNA had also been “cleared to access up to $5 million” towards the manufacturing start up process, according to a federal government spokesperson.[2]
The CBC report in late April 2021 also stated that “it could be eligible for a slice of $113 million in additional funding from the National Research Council of Canada Industrial Research Assistance Program”. The federal government had provided funding to some other companies in Canada that were also working to develop a COVID-19 vaccine.[2]
Sorenson as Providence Therapeutics CEO posted an open letter to Prime Minister Justin Trudeau, in which he requested $CDN 150 million upfront to be used to pay for clinical trial and material costs.[16][9]
On 29 April 2021, Sorenson appeared before the House of Commons standing committee on international trade, to ask the Minister of Procurement, Anita Anand, to consider PTX-COVID19-B as an alternative to Moderna and Pfizer for the “2022 booster vaccines”.[15] Sorenson said that the NRC had approached Providence Therapeutics in 2020 after the company had announced their Phase I trial PTX-COVID19-B. Sorenson told the Standing Committee that, “We’ve had really good dialogue ever since phase I started. That process has gone on. That started probably [in February], as we geared up to conclude our phase I trial and release data. Although the NRC is capped at $10 million, which is certainly not sufficient to carry out phase II and phase III trials, the NRC has, through the bureaucracy, elevated us back up to the strategic innovation fund. That occurred about three weeks ago. We’re now working with the strategic innovation fund.”[15]:7
He later said that no reply had been received from the government.[17]
In a meeting with the federal COVID-19 vaccine task force and Sorenson, task force members expressed concerns that “Providence might not be able to scale up production fast enough”.[2]
Clinical trials
PTX-COVID19-B, an mRNA Humoral Vaccine, is Intended for Prevention of COVID-19 in a General Population. This Study is Designed to Evaluate Safety, Tolerability, and Immunogenicity of PTX-COVID19-B Vaccine in Healthy Seronegative Adults Aged 18-64… https://clinicaltrials.gov/ct2/show/NCT04765436
Hyderabad Drugmaker To Make Canada Firm’s mRNA Covid Vaccine In India.. https://www.ndtv.com/india-news/hyderabad-drugmaker-biological-e-to-make-canada-firms-mrna-covid-vaccine-in-india-2454000
Biological E., will run a clinical trial of Providence’s vaccine in India and seek emergency use approval for it, the company said in a statement
Hyderabad-based Biological E said on Tuesday it has entered into a licensing agreement with Providence Therapeutics Holdings to manufacture the Canadian company’s mRNA COVID-19 vaccine in India.
Biological E., which also has a separate deal to produce about 600 million doses of Johnson & Johnson’s COVID-19 shot annually, will run a clinical trial of Providence’s vaccine in India and seek emergency use approval for it, the company said in a statement.
Providence will sell up to 30 million doses of its mRNA vaccine, PTX-COVID19-B, to Biological E, and will also provide the necessary technology transfer of the shot, with a minimum production capacity of 600 million doses in 2022 and a target capacity of 1 billion doses.
Financial details of the transaction were not disclosed.
India has been struggling with a devastating second wave of the pandemic and has managed to fully vaccinate only about 3% of its population. On Monday, the Serum Institute of India said it will increase production of AstraZeneca’s shot by nearly 40% in June, a step towards bridging the shortfall in the country.
“The mRNA platform has emerged as the front runner in delivering the first vaccines for emergency use to combat the COVID-19 pandemic,” said Mahima Datla, Biological E.’s managing director.
Messenger ribonucleic acid (mRNA) vaccines prompt the body to make a protein that is part of the virus, triggering an immune response. US companies Pfizer and Moderna use mRNA technology in their COVID-19 shots.
The drug regulator has approved clinical trials of another mRNA vaccine developed by local firm Gennova Biopharmaceuticals, and the government has said it will fund the studies.
Providence Therapeutics Announces Very Favorable Interim Phase 1 Trial Data for PTX-COVID19-B, its mRNA Vaccine Against COVID-19
CALGARY, AB, May 12, 2021 / – Providence Therapeutics Holdings Inc. (“Providence”) announced today very favorable interim clinical data of PTX-COVID19-B, its vaccine candidate against SARS-CoV-2 (“COVID-19”), from its Phase 1 study entitled “PRO-CL-001, A Phase 1, First-in-Human, Observer-Blinded, Randomized, Placebo Controlled, Ascending Dose Study to Evaluate the Safety, Tolerability, and Immunogenicity of PTX-COVID19-B Vaccine in Healthy Seronegative Adults Aged 18-64” (the “Phase 1 Study”), which found that PTX-COVID19-B met Providence’s target results for safety, tolerability, and immunogenicity in the participants of the Phase 1 Study.
Highlights from Providence Therapeutics’ “Phase 1 Study”:
- PTX-COVID19-B was generally safe and well tolerated
- PTX-COVID19-B exhibited strong virus neutralization capability across the 16µg, 40µg and 100µg dose cohorts
- PTX-COVID19-B 40µg dose was selected for Phase 2 study
- PTX-COVID19-B will be evaluated in additional Phase 1 population cohorts
The Phase 1 Study was designed with dose-escalations and was performed in seronegative adult subjects without evidence of recent exposure to COVID-19. The subjects were randomized to receive either the PTX-COVID19-B vaccine or a placebo in a 3:1 ratio. A total of 60 subjects participated in the Phase 1 Study.
The overall results of the Phase 1 Study are that PTX-COVID19-B was safe and well tolerated at the three dose levels of 16µg, 40µg and 100µg. Adverse events identified in the Phase 1 Study were generally mild to moderate in severity, self-resolving and transient. There were no serious adverse events reported in the Phase 1 Study. The most common adverse event reported in the Phase 1 Study was redness and pain at the injection site. Systemic reactions reported in the Phase 1 Study were generally mild to moderate and well tolerated with headache being the most common reaction reported. The reported adverse events of the Phase 1 Study were in line with the expectations of management of Providence as they compare very favorably to the adverse events data published on other mRNA vaccines for COVID-19 that have been approved for use by various health authorities around the world.
Based on the results of the Phase 1 Study, Providence intends to use a 40µg dose for the Phase 2 study of PTX-COVID19-B that is anticipated to be initiated in June 2021. Additional Phase 1 studies in adolescent and elderly populations are also planned to be undertaken by Providence.
PTX-COVID19-B vaccination induced high anti-S IgG antibodies:
Participants in the Phase 1 Study were vaccinated on day zero and day twenty-eight. Plasma samples were collected on day 1, day 8, day 28 (prior to the participant receiving the second dose), and day 42 to determine levels of IgG anti-S protein using electrochemiluminescence (“ECL”) assays from Meso Scale Discovery (“MSD”). Study participants in all three vaccine dose cohorts of the Phase 1 Study developed a strong IgG antibody response against Spike protein that was detected by day 28 and enhanced by day 42. No antibodies against S protein were detected in participants in the Phase 1 Study injected with placebo. The highest levels of antibodies were found in the 40 and 100 µg doses. By day 42, PTX-COVID19-B vaccinated participants had more than one log higher antibody levels than convalescent subjects-plasma (indicated in the dotted line) which was evaluated in the same assay.
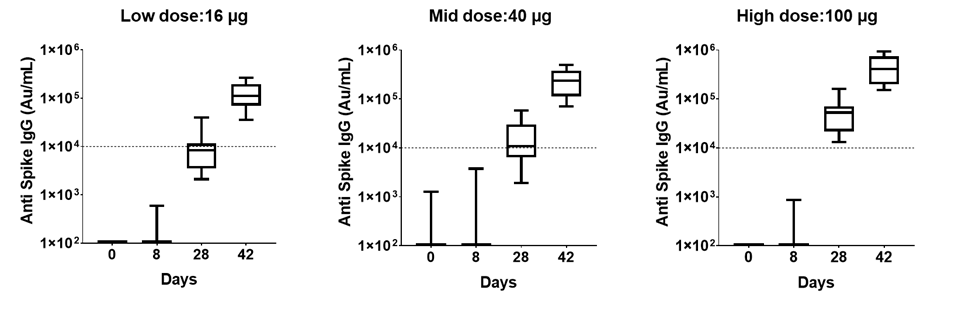
Based on the interim data of the Phase 1 Study, the level of antibodies produced in participants by PTX-COVID19-B compare favorably to the levels of antibodies produced by other mRNA vaccines that have been approved for use against COVID-19 based on the recently published report from Stanford University, where IgG responses in individuals vaccinated with the COVID-19 mRNA vaccine compared to COVID-19 infected patients were evaluated[1].
PTX-COVID19-B vaccination induced high neutralizing antibody levels:
Neutralizing activity from the Phase 1 Study participants’ plasma was evaluated by S-ACE2 MSD assay. The results indicate that the antibodies block the interaction between S protein with the ACE2 receptor and the decrease in ECL signal is used to calculate percentage inhibition of the plasma at the same dilution. All participants in the Phase 1 Study from the 16, 40 and 100 µg dose levels showed blocking activity by day 28 and all of them reached 100% blocking activity by day 42 with samples diluted 1:100 or greater. Moreover, the quantification of the antibody levels in ng/mL with a reference standard showed that all participants in the Phase 1 Study produced neutralizing antibodies by day 28 with the first immunization and increase ten-fold by day 42, two weeks after the administration of the second dose. These results indicate that PTX-COVID19-B induced a strong neutralizing antibody response which compares very favorably to the published results of other mRNA vaccines. Further studies are being conducted by Providence to determine neutralization activity using a pseudo-virus assay.
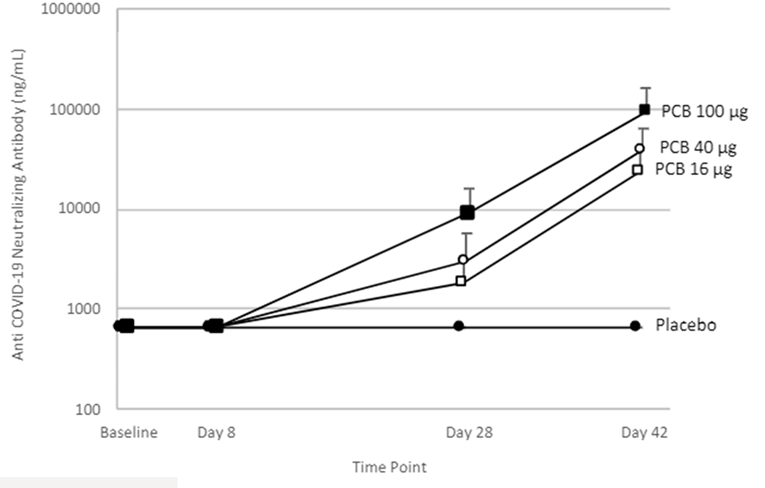
Providence intends to advance a Phase 2 clinical trial in early June 2021, with multiple trial sites in Canada. The Phase 2 clinical trial is anticipated to be structured as a comparator trial using Pfizer/BioNTech vaccine as the positive control.
About Providence Therapeutics
Providence is a leading Canadian clinical stage biotechnology company pioneering mRNA therapeutics and vaccines with operations in Calgary, Alberta and Toronto, Ontario. In response to a worldwide need for a COVID-19 vaccine, Providence expanded its focus beyond oncology therapies and devoted its energy and resources to develop a world-class mRNA vaccine for COVID-19. Providence is focused on serving the needs of Canada, and other countries that may be underserved by large pharmaceutical programs. For more information, please visit providencetherapeutics.com.
References
- ^ “Canadian company urges human trials after COVID-19 vaccine results in mice”. Lethbridge News Now. 5 August 2020. Retrieved 19 March 2021.
- ^ Jump up to:a b c d e f g h Tasker, John Paul (30 April 2021). “COVID-19 vaccine maker Providence says it’s leaving Canada after calls for more federal support go unanswered”. CBC News. Retrieved 1 May 2021.
- ^ Jump up to:a b c Stephenson, Amanda (26 January 2021). “Made-in-Canada COVID vaccine to be manufactured in Calgary”. Calgary Herald. Retrieved 22 March 2021.
- ^ Clinical trial number NCT04765436 for “PTX-COVID19-B, an mRNA Humoral Vaccine, is Intended for Prevention of COVID-19 in a General Population. This Study is Designed to Evaluate Safety, Tolerability, and Immunogenicity of PTX-COVID19-B Vaccine in Healthy Seronegative Adults Aged 18-64” at ClinicalTrials.gov
- ^ “Providence Therapeutics Holdings Inc: PTX-COVID19-B”. Montreal: McGill University. Retrieved 19 March 2021.
- ^ “Made-in-Canada coronavirus vaccine starts human clinical trials”. Canadian Broadcasting Corporation. 26 January 2021.
- ^ Jump up to:a b “Company Profile”. PitchBook.
- ^ Jump up to:a b “Company Profile”. DNB.
- ^ Jump up to:a b Code, Jillian (5 February 2021). “‘Do something’ Made-In-Canada vaccine CEO pleads for federal government to respond”. CTV News. Calgary, Alberta. Retrieved 22 March 2021.
- ^ Jump up to:a b Fieldberg, Alesia (26 January 2021). “Providence Therapeutics begins first clinical trials of Canadian-made COVID-19 vaccine”. CTV. Retrieved 2 May 2021.
- ^ Jump up to:a b c d “Manitoba Supports Made-In-Canada COVID-19 Vaccine to Protect Manitobans” (Press release). 11 February 2021. Retrieved 3 May 2021.
- ^ Providence Therapeutics Holdings Inc.: a Phase I, First-in-Human, Observer-Blinded, Randomized, Placebo Controlled, Ascending Dose Study to Evaluate the Safety, Tolerability, and Immunogenicity of PTX-COVID19-B Vaccine in Healthy Seronegative Adults Aged 18-64 (Report). Clinical Trials via U.S. National Library of Medicine. 19 February 2021. Retrieved 1 May2021.
- ^ Jump up to:a b c Gibson, Shane (11 February 2021). “Manitoba agrees to purchase 2M doses of Providence Therapeutics coronavirus vaccine”. Global News. Retrieved 2 May 2021.
- ^ “Providence Therapeutics begins first clinical trials of Canadian-made COVID-19 vaccine”. CTV. Retrieved 2 May 2021.
- ^ Jump up to:a b c Evidence (PDF), 43rd Parliament, 2nd Session. Standing Committee on International Trade, 29 April 2021, retrieved 2 May2021
- ^ Sorenson, Brad (5 February 2021). “An Open Letter to the Government of Canada”. Retrieved 3 May 2021.
- ^ Dyer, Steven. “‘Canada had an opportunity’, Calgary company explores taking vaccine development out of Canada”. CTV. Retrieved 2 May 2021.
| Vaccine description | |
|---|---|
| Target | SARS-CoV-2 |
| Vaccine type | mRNA |
| Clinical data | |
| Routes of administration | Intramuscular |
| Part of a series on the |
| COVID-19 pandemic |
|---|
| COVID-19 (disease)SARS-CoV-2 (virus) |
| showTimeline |
| showLocations |
| showInternational response |
| showMedical response |
| showImpact |
| COVID-19 portal |
////////PTX-COVID19-B, canada, hyderabad, providence, Gennova Biopharmaceuticals, biological e, COVID-19, SARS-CoV-2 , corona virus, covid 19, phase 1
NEW DRUG APPROVALS
ONE TIME
$10.00

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










