Home » Posts tagged 'FLAX LAB'
Tag Archives: FLAX LAB
Atebimetinib
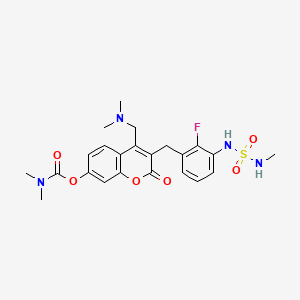
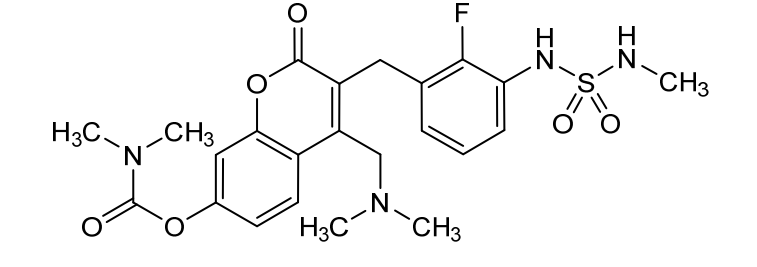
Atebimetinib
CAS 2669009-92-9
MF C23H27FN4O6S MW506.5 g/mol
[4-[(dimethylamino)methyl]-3-[[2-fluoro-3-(methylsulfamoylamino)phenyl]methyl]-2-oxochromen-7-yl] N,N-dimethylcarbamate
4-[(dimethylamino)methyl]-3-({2-fluoro-3-[(methylsulfamoyl)amino]phenyl}methyl)-2-oxo-2H-1-benzopyran-7-yl
dimethylcarbamate
MEK tyrosine kinase inhibitor, antineoplastic, IMM-104, IMM 104, Fast Track designation, TEL9243A3N
Atebimetinib (IMM-104) is an investigational oral, deep cyclic inhibitor (DCI) that targets the MAP kinase (MAPK) pathway in solid tumors, particularly RAS-mutant pancreatic cancer. Designed for rapid, pulsatile inhibition to minimize resistance and side effects, it is currently in Phase 2a trials, having shown promising, durable tumor shrinkage and high 1-year survival rates.
Key Aspects of Atebimetinib:
- Mechanism of Action: As a DCI, it works differently from standard inhibitors by targeting MAPK with a short half-life, allowing for rapid “pulsing” that suppresses tumor growth while permitting healthy cells to recover, thus improving tolerability.
- Targeted Cancers: Primarily aimed at RAS-mutant advanced or metastatic solid tumors, including pancreatic ductal adenocarcinoma (PDAC).
- Clinical Trial Results: In a Phase 2a study (NCT05585320), the combination of atebimetinib with modified chemotherapy showed a 64% overall survival (OS) rate at 12 months for first-line pancreatic cancer patients.
- Fast Track Designation: In 2024, the FDA granted fast track designation for atebimetinib to treat patients with pancreatic adenocarcinoma who have progressed after one line of therapy.
- Advantage over Traditional Inhibitors: It is designed to avoid typical MAP kinase inhibitor adverse events like pyrexia (fever) while overcoming the rapid resistance often seen in other therapies.
Atebimetinib is being developed by Immuneering Corporation.
Development Status
- FDA Designations: In 2024, the FDA granted atebimetinib Fast Track designation for patients with pancreatic adenocarcinoma (PDAC) who have progressed after one line of treatment.
- Future Plans: A global registrational Phase 3 trial, named MAPKeeper 301, is planned to begin dosing patients in mid-2026.
Clinical Trial Results (Phase 2a)
Recent data from the Phase 2a trial (as of early 2026) showed significant survival benefits when combined with modified chemotherapy (gemcitabine and nab-paclitaxel) for first-line pancreatic cancer:
- Overall Survival (OS): Reported at 94% at 6 months, 86% at 9 months, and 64% at 12 months. This is roughly double the 1-year survival rate typically seen with standard chemotherapy alone (~35%).
- Progression-Free Survival (PFS): Median PFS reached 8.5 months.
- Disease Control Rate: Approximately 81% of patients achieved disease control.
SYN
WO2023076991 COMBINATION THERAPY FOR TREATING ABNORMAL CELL GROWTH
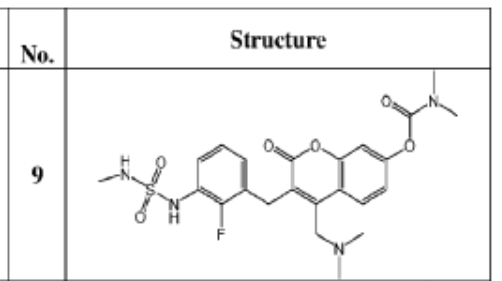
SYN
WO2025010293 MEK IMMUNE ONCOLOGY INHIBITOR PHARMACEUTICAL COMPOSITIONS
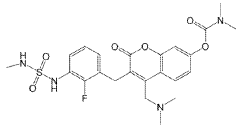
EXAMPLE 1A
Synthesis of Compound A
[0198] Compound A was prepared in 1 step:
[0199] 4-(bromomethyl)-3-(2-fluoro-3-((N-methylsulfamoyl)amino)benzyl)-2-oxo-2H-chromen-7-yl dimethylcarbamate (22.22 g, 34.79 mmol) was suspended in methanol. Dimethylamine 2M was added and the formed reaction mixture was stirred until full conversion was observed. After full conversion the reaction was concentrated under reduced pressure. IM HC1 was added to the residue and the water layer was extracted with CH2CI2. The water layer was made basic with solid Na CCE. The basic water layer was extracted with CH2CI2. The organic layer from the basic extraction was washed with brine, dried over Na2SO4, filtered and concentrated under reduced pressure to obtain the title compound (13.23 g, 25.7 mmol, yield: 74%) as a light yellow amorphous solid.
[0200] Yield: Compound A was isolated as a light yellow solid (74% over 1 step). Analysis: LCMS (Method T): tR = 1.53 min; m/z calculated for [M+H]+ = 507.2, found = 507.2; 1H NMR (400 MHz, DMSO) d 9.38 (s, 1H), 8.08 (d, J = 8.8 Hz, 1H), 7.28 (td, J = 8.0, 1.6 Hz, 1H), 7.25 – 7.18 (m, 2H), 7.15 (dd, J = 8.8, 2.4 Hz, 1H), 7.00 (t, J = 7.9 Hz, 1H), 6.90 – 6.77 (m, 1H), 4.04 (s, 2H), 3.64 (s, 2H), 3.06 (s, 3H), 2.93 (s, 3H), 2.52 (d, J = 4.9 Hz, 3H), 2.19 (s, 6H).
ADVT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Mek immune oncology inhibitor pharmaceutical compositionsPublication Number: WO-2025010293-A2Priority Date: 2023-07-03
- Inhibiting mitogen-activated protein (map)/erk kinase (mek)1 and mek2 and related methods of treatmentPublication Number: WO-2024220440-A1Priority Date: 2023-04-17
- Methods of treating cancer with a ras mutationPublication Number: WO-2024186693-A1Priority Date: 2023-03-03
- Combination therapy for treating abnormal cell growthPublication Number: WO-2023235356-A1Priority Date: 2022-06-03
- Combination therapy for treating abnormal cell growthPublication Number: WO-2023147297-A2Priority Date: 2022-01-25
- Methods of treating abnormal cell growthPublication Number: WO-2023081676-A1Priority Date: 2021-11-02
- Combination therapy for treating abnormal cell growthPublication Number: WO-2023076991-A1Priority Date: 2021-10-28
- Mek inhibitors and therapeutic uses thereofPublication Number: US-2023119327-A1Priority Date: 2020-01-10
//////atebimetinib, FLAX LAB, antineoplastic, IMM-104, IMM 104, Fast Track designation, TEL9243A3N

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










