Home » Posts tagged 'firazorexton'
Tag Archives: firazorexton
Alixorexton
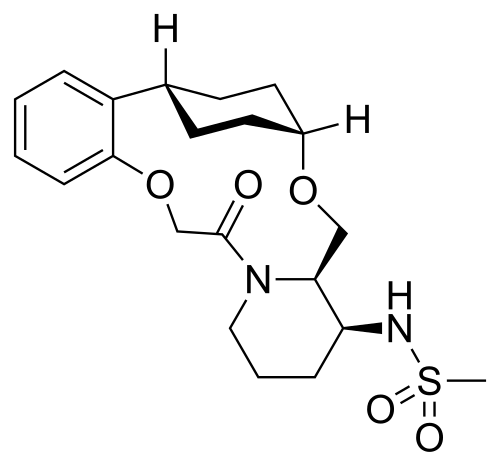
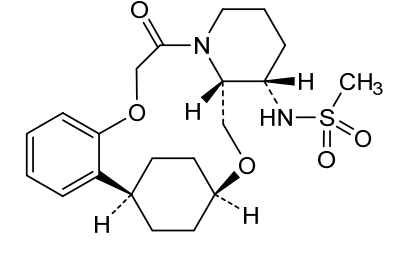
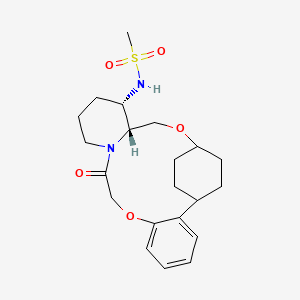
Alixorexton
CAS 2648347-56-0
MF C21H30N2O5S MW 422.5 g/mol
N-((21S,24S,52R,53S)-6-oxo-3,8-dioxa-5(2,1)-piperidina-1(1,2)-benzena-2(1,4)-cyclohexanacyclooctaphane-53-yl)methanesulfonamide
N-[(15S,16R)-10-oxo-8,18-dioxa-11-azatetracyclo[17.2.2.02,7.011,16]tricosa-2,4,6-trien-15-yl]methanesulfonamide
- Methanesulfonamide, N-[(4S,4aR,7alpha,10alpha)-1,2,3,4,4a,5,7,8,9,10,16,17-dodecahydro-17-oxo-7,10-ethanopyrido[1,2-d][1,7,4]benzodioxaazacyclotridecin-4-yl]-
- N-[(4S,4aR,7alpha,10alpha)-1,2,3,4,4a,5,7,8,9,10,16,17-Dodecahydro-17-oxo-7,10-ethanopyrido[1,2-d][1,7,4]benzodioxaazacyclotridecin-4-yl]methanesulfonamide
- N-[7,10-cis-(4S,4aR)-17-oxo-1,2,3,4,4a,5,7,8,9,10,16,17-dodecahydro-7,10-ethanopyrido[1,2-d][1,7,4]benzodioxaazacyclotridecin-4-yl]methanesulfonamide
N-[7,10-cis-(4S,4aR)-17-oxo-1,2,3,4,4a,5,7,8,9,10,16,17-dodecahydro-7,10-ethanopyrido[1,2-
d][1,7,4]benzodioxaazacyclotridecin-4-yl]methanesulfonamide
orexin-2 receptor agonist, ALKS 2680, Breakthrough Therapy designation, 3BRW4ARU66, TAK 994, firazorexton
Alixorexton (formerly ALKS 2680) is an investigational, once-daily, oral, selective orexin 2 receptor (OX2R) agonist being developed by Alkermes for treating narcolepsy and idiopathic hypersomnia. It has shown significant efficacy in Phase 2 trials (Vibrance-1, -2, -3) for improving wakefulness and received FDA Breakthrough Therapy designation in January 2026 for narcolepsy type 1.
- OriginatorAlkermes
- ClassSleep disorder therapies
- Mechanism of ActionOrexin receptor type 2 agonists
- Phase IIIdiopathic hypersomnia; Narcolepsy
- 09 Mar 2026Alkermes plans the phase III Brilliance NT1 trial for Narcolepsy in an undisclosed location (PO), in March 2026 (NCT07455383)
- 06 Jan 2026Alixorexton receives Breakthrough Therapy status for Narcolepsy in USA
- 12 Nov 2025Efficacy and adverse event data from the phase II VIBRANCE-2 trial in Narcolepsy released by Alkermes
Key Details About Alixorexton (ALKS 2680):
- Mechanism of Action: As an OX2R agonist, it targets the orexin system to treat the underlying cause of excessive daytime sleepiness by activating wake-promoting neurons.
- Target Indications: The drug is developed for Narcolepsy Type 1 (NT1), Narcolepsy Type 2 (NT2), and Idiopathic Hypersomnia (IH).
- Clinical Trial Results:
- Vibrance-1 (NT1): Demonstrated significant improvements in wakefulness, reduced cataplexy rates, and improved alertness in phase 2 trials.
- Vibrance-2 (NT2): Showed positive results, meeting primary endpoints in improving sleep latency and reducing daytime sleepiness.
- Safety: Generally well-tolerated, with most adverse events reported as mild to moderate.
- Development Status: Following positive Phase 2 data, Alkermes is preparing for phase 3 development, with plans to advance the program in early 2026.
Alixorexton is distinct as a potential once-daily treatment in a novel, rapidly advancing therapeutic category for sleep disorders
Alixorexton (INNTooltip International Nonproprietary Name; developmental code name ALKS-2680) is an orexin receptor agonist which is under development for the treatment of sleep disorders, including narcolepsy and idiopathic hypersomnia.[1][2][3] Alixorexton is being developed by Alkermes[4] As of July 2025, it is in phase 2 clinical trials with planned advancement to a phase 3 study for the treatment of narcolepsy type 1.[4]
Alixorexton, also known as TAK-994 or firazorexton, is a potent, orally bioavailable, brain-penetrant agonist that selectively targets the orexin 2 receptor (OX2R), with additional activity at the orexin 1 receptor (OX1R). Its mechanism of action involves mimicking the endogenous orexin neuropeptides, thereby promoting wakefulness. Preclinical studies have demonstrated that Alixorexton effectively suppresses fragmentation of wakefulness and reduces cataplexy-like episodes in mouse models of narcolepsy, indicating its potential as a therapeutic agent for sleep disorders.
- A Long-Term Study of ALKS 2680 in Subjects With Narcolepsy and Idiopathic HypersomniaCTID: NCT06767683Phase: Phase 2/Phase 3Status: RecruitingDate: 2026-03-04
- A Study to Evaluate the Safety and Effectiveness of ALKS 2680 in Subjects With Narcolepsy Type 2CTID: NCT06555783Phase: Phase 2Status: CompletedDate: 2026-02-27
- A Study to Evaluate the Safety and Effectiveness of ALKS 2680 in Subjects With Idiopathic HypersomniaCTID: NCT06843590Phase: Phase 2Status: RecruitingDate: 2026-02-12
- A Study to Evaluate the Safety and Effectiveness of ALKS 2680 in Subjects With Narcolepsy Type 1 (ALKS 2680-201)CTID: NCT06358950Phase: Phase 2Status: CompletedDate: 2025-10-14
SYN
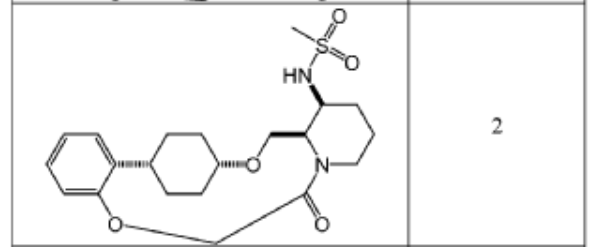
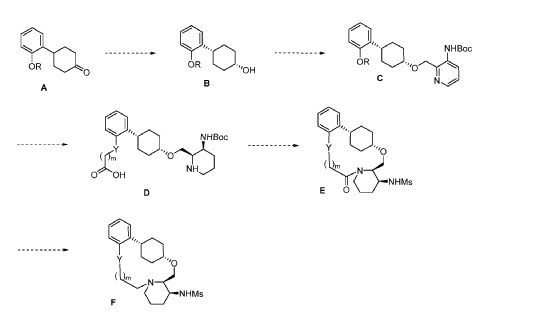
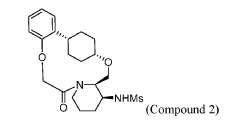
To (21S,24S,52R, 535)-53-amino-3,8-dioxa-5(2,1)-piperidina-1(1,2)-benzena-2(1,4)-cyclohexanacyclooctaphan-6-one 32.1 g (900 mg, 2.613 mmol, 1 equiv.) and DIPEA (1.69 g, 13.064 mmol, 5 equiv.) in DCM (148 mL) was added MsCl (900 mg, 7.858 mmol, 3 equiv.) dropwise at room temperature under nitrogen atmosphere. The resulting solution was stirred for 2 hr at room temperature. The reaction was then quenched by the addition of 50 mL of water. The resulting solution was extracted with 3 x 200 mL of dichloromethane, dried over anhydrous sodium sulfate and concentrated under vacuum. The crude product was purified by Prep-HPLC to afford racemic crude product (800 mg, 72.46%) as a solid. The crude product was purified by prep-SFC to afford N-((21S,24S,52R,53S)-6-oxo-3,8-dioxa-5(2, 1)-piperidina-1(1, 2)-benzena-2(1, 4)-cyclohexanacyclooctaphane-53-yl)methanesulfonamide (270.6 mg, 27.1%) as a solid and its enantiomer, N-((21R,24R,52,53R)-6-oxo-3,8-dioxa-5(2, 1)-piperidina-1(1, 2)-benzena-2(1, 4)-cyclohexanacyclooctaphane-53-yl)methanesulfonamide (361.4mg, 36.1%) as a solid. LCMS (ESI): m/z calculated for C21H30N2O5S [M+H]+ = 423.19, found [M+H]+ = 423.15; 1H NMR (400 MHz, CDCl3): δ 7.19 (td, J = 7.7, 1.8 Hz,
1H), 7.11 (dd, J = 7.5, 1.7 Hz, 1H), 6.96 – 6.86 (m, 1H), 6.77 (dd, J = 8.0, 1.1 Hz, 1H), 5.24 (dd, J = 9.8, 4.9 Hz, 1H), 5.14 (d, J = 10.5 Hz, 1H), 4.34 (dd, J = 14.1, 9.4 Hz, 2H), 3.84 (t, J = 9.2 Hz, 1H), 3.73 (d, J = 14.9 Hz, 3H), 3.60 – 3.46 (m, 2H), 3.09 (s, 3H), 2.77 – 2.49 (m, 2H), 2.33 – 2.16 (m, 1H), 2.06 (d, J = 12.8 Hz, 2H), 2.00 – 1.82 (m, 2H), 1.68 (d, J = 11.5 Hz, 2H), 1.10 – 1.45 (m, 4H).
SYN
CREDIT MEDKOO
Synthetic Reference
Preparation of substituted macrocyclic derivatives for use in the treatment of narcolepsy or cataplexy
Assignee: Alkermes, Inc.
Inventors: Pennington, Lewis D.; et al
United States
Patent#US20210155636 A1
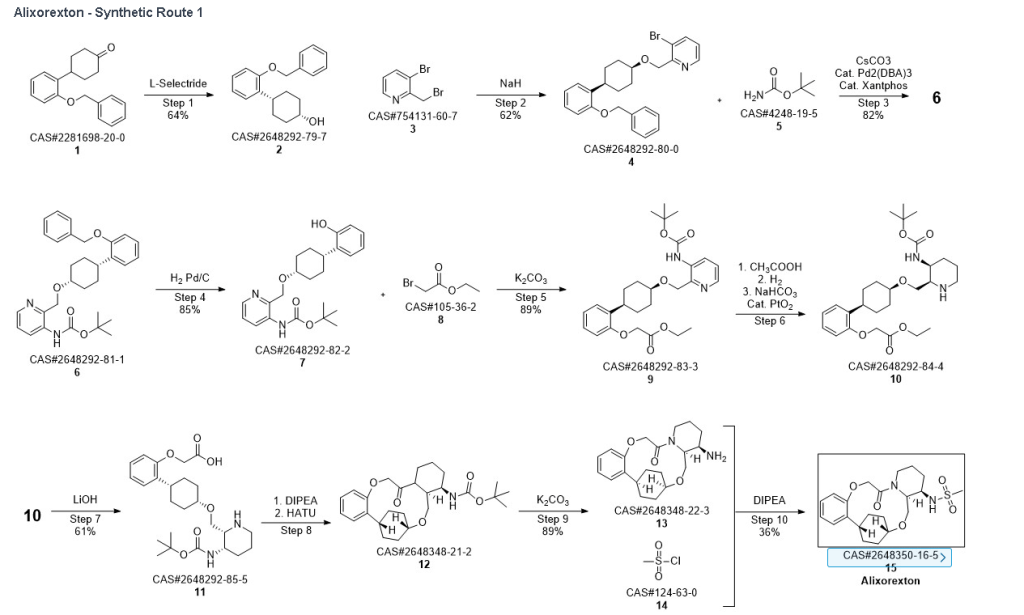
Example 1.1
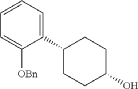
Into a 5-L 4-necked round-bottom flask purged and maintained with an inert atmosphere of nitrogen, was placed 4-[2-(benzyloxy)phenyl]cyclohexan-1-one (210 g, 749 mmol, 1.00 equiv.) in tetrahydrofuran (2.1 L). This was followed by the addition of L-selectride (1 mol/L in THF) (1123 mL, 5257 mmol, 1.50 equiv.) dropwise with stirring at 0 degrees C. The resulting solution was stirred for 4 hr at room temperature. The reaction was then quenched by the addition of water/ice. The resulting solution was extracted with ethyl acetate, and the organic phase was washed with brine. The mixture was dried over anhydrous sodium sulfate, filtered, concentrated under vacuum. The residue was purified by silica gel column chromatography with ethyl acetate/petroleum ether (1:100-1:5) to give 137 g (64%) of (1s,4s)-4-[2-(benzyloxy)phenyl]cyclohexan-1-ol as a solid. 1H NMR (400 MHz, CDCl 3): δ 7.45-7.26 (6H, m), 7.16 (1H, dd), 6.98-6.90 (2H, m), 5.09 (2H, s), 4.13 (1H, s), 3.12-3.02 (1H, m), 1.93-1.82 (4H, m), 1.73-1.41 (4H, m).
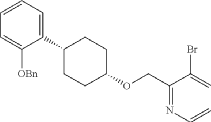
Into a 2-L 4-necked round-bottom flask purged and maintained with an inert atmosphere of nitrogen, was placed NaH (60% wt, 26.9 g, 2.00 equiv.) in tetrahydrofuran (200 mL). This was followed by the addition of a solution of (1s, 4s)-4-[2-(benzyloxy)phenyl]cyclohexan-1-ol (95 g, 336 mmol, 1.00 equiv.) in THF (200 mL) dropwise with stirring at 50-55 degrees C. After stirring for 2 hr, to this was added a solution of 3-bromo-2-(bromomethyl)pyridine (143.5 g, 571 mmol, 1.70 equiv.) in THF (550 mL) dropwise with stirring at 50-55 degrees C. The resulting solution was stirred for 14 hr at 50-55 degrees C. The reaction mixture was cooled. The reaction was then quenched by the addition of water. The resulting solution was extracted with ethyl acetate and the organic layers combined and dried over anhydrous sodium sulfate. The solids were filtered out. The resulting mixture was concentrated under vacuum. The residue was purified by silica gel column chromatography with ethyl acetate/petroleum ether (1:100-1:2) to give 94 g (62%) of 3-bromo-2-([[(1s,4s)-4-[2-(benzyloxy) phenyl]cyclohexyl]oxy]methyl)pyridine as a solid. 1H NMR (400 MHz, CDCl 3): δ 8.57 (1H, d), 7.90 (1H, dd), 7.48-7.26 (6H, m), 7.18-7.14 (2H, m), 6.98-6.91 (2H, m), 5.12 (2H, s), 4.77 (2H, s), 3.86 (1H, s), 3.17-3.10 (1H, m), 2.20-2.15 (2H, m), 1.98-1.88 (2H, m), 1.69-1.57 (4H, m).
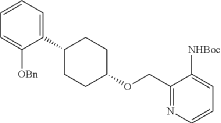
Into a 2-L 4-necked round-bottom flask purged and maintained with an inert atmosphere of nitrogen, was placed Xantphos (10.7 g, 18 mmol, 0.10 equiv.), Cs 2CO 3 (84 g, 258 mmol, 1.39 equiv.), 3-bromo-2-([[(1s,4s)-4-[2-(benzyloxy) phenyl]cyclohexyl]oxy]methyl)pyridine (84 g, 185 mmol, 1.00 equiv.), Pd 2(dba) 3 (8.5 g, 9 mmol, 0.05 equiv.) and tert-butyl carbamate (26 g, 222 mmol, 1.20 equiv.) in dioxane (840 mL). The resulting solution was stirred for 5 hr at 100 degrees C. The solids were filtered out. The filtrate was concentrated under vacuum. The residue was purified by silica gel column chromatography with ethyl acetate/petroleum ether (1:100-1:4) to provide 74 g (82%) of tert-butyl N-[2-([[(1s,4s)-4-[2-(benzyloxy)phenyl]cyclohexyl]oxy]methyl)pyridin-3-yl]carbamate as a solid.
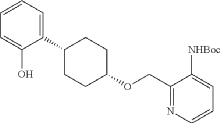
Into a 2-L 3-necked round-bottom flask, was placed tert-butyl N-[2-([[(1s,4s)-4-[2-(benzyloxy)phenyl]cyclohexyl]oxy]methyl)pyridin-3-yl]carbamate (74 g, 151 mmol, 1.00 equiv.) and Pd/C (7.4 g, 10% wt) in ethyl alcohol (740 mL), then, hydrogen gas was through in. The resulting solution was stirred for 14 hr at room temperature. The solids were filtered out. The filtrate was concentrated under vacuum. The residue was purified by silica gel column chromatography with ethyl acetate/petroleum ether (1:100-1:2) to provide 51.36 g (85%) of tert-butyl N-[2-([[(1s,4s)-4-(2-hydroxyphenyl)cyclohexyl]oxy]methyl)pyridin-3-yl]carbamate as a solid. LCMS (ESI): m/z [M+H] +=399.1; 1H NMR (300 MHz, CDCl 3): δ 8.65 (1H, s), 8.47 (1H, d), 8.19 (1H, q), 7.26-7.21 (1H, m), 7.09-7.03 (1H, m), 6.92-6.86 (1H, m), 6.75 (1H, q), 5.77 (1H, s), 4.84 (1H, s), 3.80 (1H, s), 2.94-2.93 (1H, m), 2.15-2.06 (2H, m), 1.88-1.47 (7H, m), 1.45 (9H, s), 1.26 (1H, d).
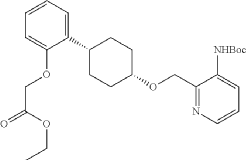
Into a 250-mL round-bottom flask purged and maintained with an inert atmosphere of nitrogen, was placed tert-butyl N-[2-([[(1s,4s)-4-(2-hydroxyphenyl)cyclohexyl]oxy]methyl)pyridin-3-yl]carbamate (8 g, 20.075 mmol, 1 equiv.), K 2CO 3 (13.97 g, 100.35 mmol, 5 equiv.), acetone (120 mL) and ethyl bromoacetate (5.03 g, 30.119 mmol, 1.5 equiv.). The resulting solution was stirred for 24 hr at 50 degrees C. The solids were filtered out. The filtrate was concentrated under vacuum. The residue was purified by silica gel column chromatography with ethyl acetate/petroleum ether (1:2) to provide ethyl 2-[2-[(1s,4s)-4-([3-[(tert-butoxycarbonyl)amino]pyridin-2-yl]methoxy)cyclohexyl]phenoxy]acetate (8.7 g, 89.43%) as a yellow oil. LCMS (ESI): m/z [M+H] +=485.
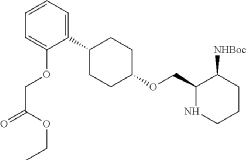
To a stirred mixture of ethyl 2-[2-[(1s,4s)-4-([3-[(tert-butoxycarbonyl)amino]pyridin-2-yl]methoxy)cyclohexyl]phenoxy]acetate (7.89 g, 16.268 mmol, 1 equiv.) in MeOH (142 mL) and AcOH (15.8 mL) were added PtO 2 (1.85 g, 8.142 mmol, 0.50 equiv.) at room temperature under hydrogen atmosphere. The resulting mixture was stirred for 2 hr at room temperature under hydrogen atmosphere. The resulting mixture was filtered, the filter cake was concentrated under reduced pressure. The reaction was quenched with sat. NaHCO 3 (aq.) at 0 degrees C. The resulting mixture was extracted with CH 2Cl 2 (3×500 mL). The combined organic layers were washed with brine (3×200 mL), dried over anhydrous Na 2SO 4. After filtration, the filtrate was concentrated under reduced pressure to afford diastereomeric cis and trans mixture (7 g, 88.7%) as a solid. The crude product was purified by Prep-TLC (DCM/MeOH=20:1) to afford cis-racemic mixture of ethyl 2-(2-((1S,4s)-4-((3-((tert-butoxycarbonyl)amino)piperidin-2-yl)methoxy)cyclohexyl)phenoxy)acetate (4.1 g) and trans-racemic mixture (1.7 g). LCMS (ESI): m/z [M+H] +=491.
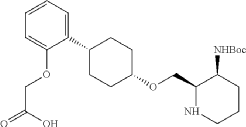
Into a 500 mL round-bottom flask purged and maintained with an atmosphere of nitrogen, was placed cis-racemic mixture of ethyl 2-(2-((1S,4s)-4-((3-((tert-butoxycarbonyl)amino)piperidin-2-yl)methoxy)cyclohexyl)phenoxy)acetate (4.1 g, 8.356 mmol, 1 equiv.), MeOH (30 mL), THF (60 mL), H 2O (30 mL) and lithium hydroxide (83 mg, 3.465 mmol, 5 equiv.). The reaction was stirred for 2 hr at room temperature. The reaction was concentrated and the residue was purified by reverse phase flash with the following conditions, then freezing-drying to afford 2-(2-((1s,4s)-4-((3-((tert-butoxycarbonyl)amino)piperidin-2-yl)methoxy)cyclohexyl)phenoxy)acetic acid (2.35 g, 60.8%) as a solid. LCMS (ESI): m/z [M+H] +=463
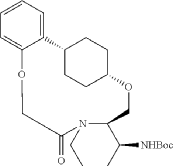
Into a 2000-mL round-bottom flask was added 2-(2-((1s,4s)-4-((3-((tert-butoxycarbonyl)amino)piperidin-2-yl)methoxy)cyclohexyl)phenoxy)acetic acid (100 mg, 0.216 mmol, 1 equiv.), MeCN (36 mL), DMF (9 mL), HATU (124 mg, 0.326 mmol, 1.51 equiv.) and DIPEA (56 mg, 0.436 mmol, 2.02 equiv.) under nitrogen atmosphere. The resulting solution was stirred for 3 hr at room temperature. LCMS showed full conversation. The resulting mixture was concentrated. The crude product tert-butyl ((2 1S,2 4S,5 2R,5 3S)-6-oxo-3,8-dioxa-5(2,1)-piperidina-1(1,2)-benzena-2(1,4)-cyclohexanacyclooctaphane-5 3-yl)carbamate was used directly for the next step without purification. LCMS (ESI): m/z [M+H] +=445.
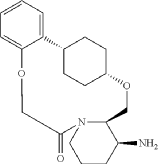
Into a 500-mL round-bottom flask purged and maintained with an inert atmosphere of nitrogen, was placed crude mixture tert-butyl ((2 1S,2 4S,5 2R,5 3S)-6-oxo-3,8-dioxa-5(2,1)-piperidina-1(1,2)-benzena-2(1,4)-cyclohexanacyclooctaphane-5 3-yl)carbamate (2 g, 4.499 mmol, 1 equiv.), DCM (120 mL), TFA (40 mL). The resulting solution was stirred for 1 hr at 25 degrees C. LCMS showed full conversation. The resulting mixture was concentrated under vacuum. The crude product was purified by Prep-HPLC to afford (2 1,S,2 4S,5 2R,5 3S)-5 3-amino-3,8-dioxa-5(2,1)-piperidina-1(1,2)-benzena-2(1,4)-cyclohexanacyclooctaphan-6-one 32.1 g (800 mg, 51.6%) as a solid. LCMS (ESI): m/z [M+H] +=345.
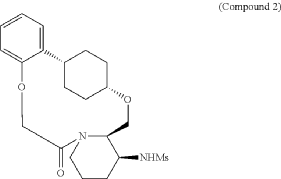
To (2 1S,2 4S,5 2R,5 3S)-5 3-amino-3,8-dioxa-5(2,1)-piperidina-1(1,2)-benzena-2(1,4)-cyclohexanacyclooctaphan-6-one 32.1 g (900 mg, 2.613 mmol, 1 equiv.) and DIPEA (1.69 g, 13.064 mmol, 5 equiv.) in DCM (148 mL) was added MSCl (900 mg, 7.858 mmol, 3 equiv.) dropwise at room temperature under nitrogen atmosphere. The resulting solution was stirred for 2 hr at room temperature. The reaction was then quenched by the addition of 50 mL of water. The resulting solution was extracted with 3×200 mL of dichloromethane, dried over anhydrous sodium sulfate and concentrated under vacuum. The crude product was purified by Prep-HPLC to afford racemic crude product (800 mg, 72.46%) as a solid. The crude product was purified by prep-SFC to afford N-((2 1S,2 4S,5 2R,5 3S)-6-oxo-3,8-dioxa-5(2,1)-piperidina-1(1,2)-benzena-2(1,4)-cyclohexanacyclooctaphane-5 3-yl)methanesulfonamide (270.6 mg, 27.1%) as a solid and its enantiomer, N-((2 1R,2 4R,5 2S,5 3R)-6-oxo-3,8-dioxa-5(2,1)-piperidina-1(1,2)-benzena-2(1,4)-cyclohexanacyclooctaphane-5 3-yl)methanesulfonamide (361.4 mg, 36.1%) as a solid. LCMS (ESI): m/z calculated for C 21H 30N 2O 5S [M+H] +=423.19, found [M+H] +=423.15; 1H NMR (400 MHz, CDCl 3): δ 7.19 (td, J=7.7, 1.8 Hz, 1H), 7.11 (dd, J=7.5, 1.7 Hz, 1H), 6.96-6.86 (m, 1H), 6.77 (dd, J=8.0, 1.1 Hz, 1H), 5.24 (dd, J=9.8, 4.9 Hz, 1H), 5.14 (d, J=10.5 Hz, 1H), 4.34 (dd, J=14.1, 9.4 Hz, 2H), 3.84 (t, J=9.2 Hz, 1H), 3.73 (d, J=14.9 Hz, 3H), 3.60-3.46 (m, 2H), 3.09 (s, 3H), 2.77-2.49 (m, 2H), 2.33-2.16 (m, 1H), 2.06 (d, J=12.8 Hz, 2H), 2.00-1.82 (m, 2H), 1.68 (d, J=11.5 Hz, 2H), 1.10-1.45 (m, 4H).
REF
CHEMICAL.AI
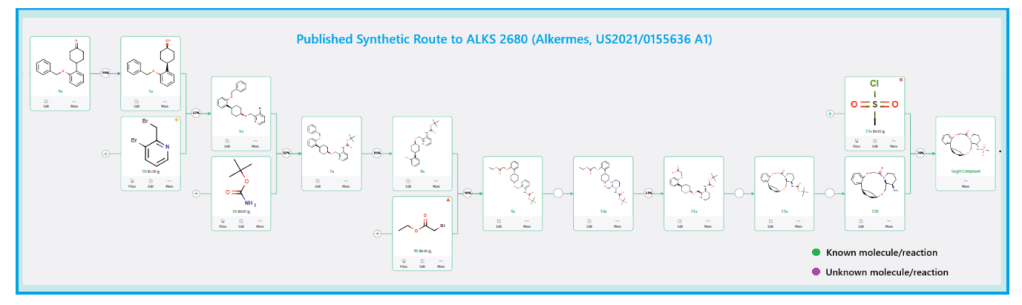
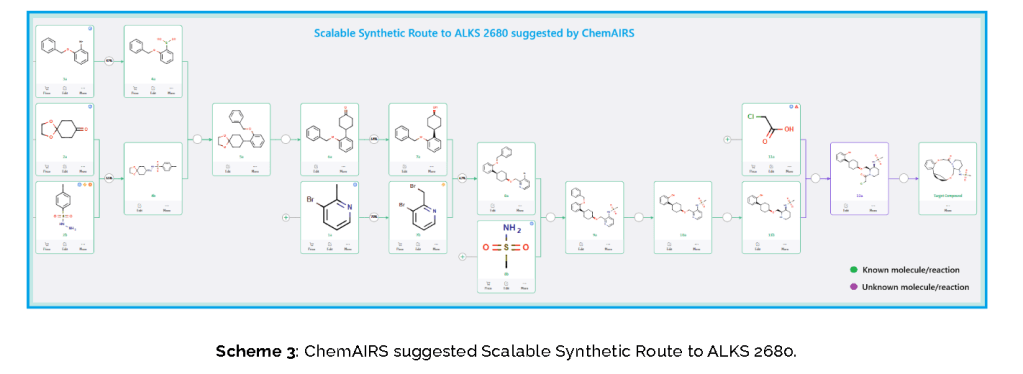
Published Synthetic Route to ALKS 2680 (Alkermes)
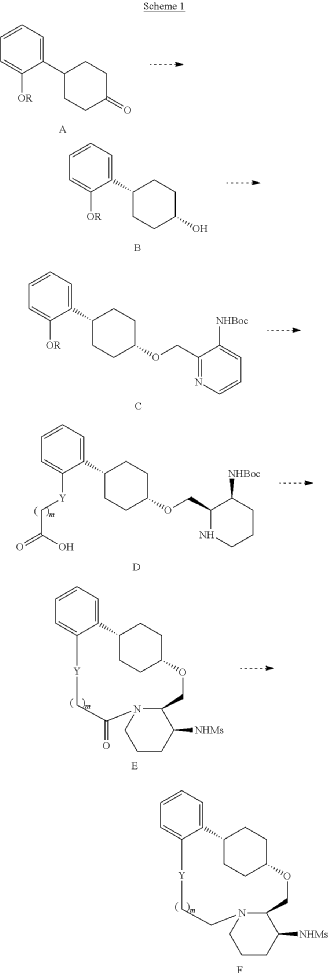
The total synthesis of ALKS 2680 was originally reported in US2021/0155636 A1, beginning with a stereoselective reduction of ketone 4a to afford alcohol 5a (Scheme 1). The patented route employed L-selectride for facial selectivity.
To optimize this step, ChemAIRS proposed an alternative asymmetric hydrogenation utilizing an Ir/f-AmphBINOL catalyst system. This enantioselective methodology has demonstrated excellent control over both enantio- and diastereoselectivity, delivering chiral alcohols in up to 99% ee and a cis/trans ratio of 99:1. [DOI: 10.1021/acs.orglett.3c03550]
Another key transformation in the Alkermes route is the macrocyclization of intermediate 11a using HATU/DIPEA in a mixed MeCN/DMF solvent system to deliver macrocycle 12a.
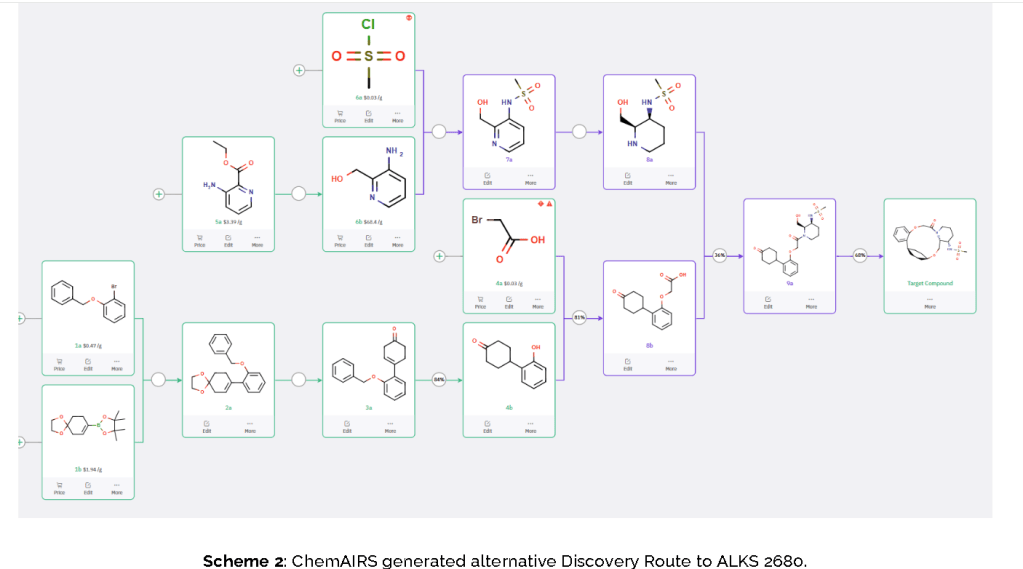
The conversion of (pyridin-3-yl)methanesulfonamide 7a to (piperidin-3-yl)methanesulfonamide 8a was facilitated by Rh/C-catalyzed hydrogenation in EtOH/AcOH, followed by kinetic resolution using (+)-mandelic acid for stereochemical enrichment (WO2017/135306).
The macrocycle was constructed via intramolecular reductive etherification using Et₃SiH/TMSOTf in DCM (doi.org/10.1002/ejoc.201300135), providing the final scaffold in a late-stage cyclization strategy conducive to structural diversification.
PAT
- Substituted macrocyclic compounds and related methods of treatmentPublication Number: US-11542276-B2Priority Date: 2019-11-25Grant Date: 2023-01-03
- Substituted Macrocyclic Compounds and Related Methods of TreatmentPublication Number: US-2021155636-A1Priority Date: 2019-11-25
- Substituted macrocyclic compounds and related methods of treatmentPublication Number: US-2023339969-A1Priority Date: 2019-11-25
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
References
- “Alixorexton – Alkermes”. AdisInsight. Springer Nature Switzerland AG. Retrieved 2025-07-30.
- Morse AM (May 2025). “Enhancing the Management of Hypersomnia: Examining the Role of the Orexin System”. Seminars in Neurology. 45 (3): 410–419. doi:10.1055/a-2589-3825. PMID 40239951.
- Prakash BA, Shah I, Ni G, Vasudevan S, Jagannath A, Foster RG (May 2025). “Dreaming of Better Treatments: Advances in Drug Development for Sleep Medicine and Chronotherapy”. Journal of Sleep Research. 34 (5) e70087. doi:10.1111/jsr.70087. PMID 40346938.
- Chen E (2025-07-21). “Alkermes drug helped narcolepsy patients stay awake in Phase 2 trial”. STAT. Retrieved 2025-07-30.
| Clinical data | |
|---|---|
| Other names | ALKS-2680; ALKS2680 |
| Routes of administration | Oral |
| Drug class | Orexin OX2 receptor agonist |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2648347-56-0 |
| PubChem CID | 156417714 |
| IUPHAR/BPS | 13879 |
| UNII | 3BRW4ARU66 |
| Chemical and physical data | |
| Formula | C21H30N2O5S |
| Molar mass | 422.54 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
/////////alixorexton, anax lab, orexin-2 receptor agonist, ALKS 2680, Breakthrough Therapy designation, 3BRW4ARU66, TAK 994, firazorexton, CHEMICAL AI

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










