Home » Posts tagged 'Enclomiphene citrate'
Tag Archives: Enclomiphene citrate
Enclomiphene citrate, New patent, WO 2017182097, F.I.S. – FABBRICA ITALIANA SINTETICI S.P.A

Enclomiphene citrate, New patent, WO 2017182097, F.I.S. – FABBRICA ITALIANA SINTETICI S.P.A
WO-2017182097
F.I.S. – FABBRICA ITALIANA SINTETICI S.P.A
CARUANA, Lorenzo; (IT).
PADOVAN, Pierluigi; (IT).
DAL SANTO, Claudio; (IT)

Enclomiphene citrate is an active pharmaceutical ingredient currently under evaluation in clinical phase III for the treatment of secondary hypergonadism. Moreover, it also could be potentially used for an adjuvant therapy in hypogonadal men with Type 2 diabetes.
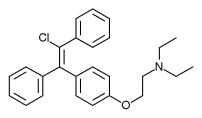
Enclomiphene citrate of formula (I):

has chemical name of Ethanamine, 2-[4-[(1 )-2-chloro-1 ,2-diphenyl ethenyl]phenoxy]-/V,/V-diethyl-, 2-hydroxy-1 ,2,3-propanetricarboxylate (1 : 1 ); has CAS RN. 7599-79-3, and it is also named trans-Clomiphene monocitrate, E-Clomiphene citrate or Enclomiphene monocitrate.
Enclomiphene is component of Clomiphene, an active pharmaceutical ingredient, having chemical name Ethanamine, 2-[4-(2-chloro-1 ,2- diphenylethenyl)phenoxy]-N,N-diethyl, since Clomiphene is a mixture of the geometric isomers trans-Clomiphene (i.e. Enclomiphene) and cis- Clomiphene.
The US patent 3,848,030, in examples 31 and 32, discloses a process for the resolution of the geometric isomers of Clomiphene through the preparation of salts with racemic binaphthyl-phosphoric acid.
In the later publication Acta Cryst. (1976), B32, pag. 291 -293, the actual geometric isomery has been definitely established by single crystal X-Ray diffraction.
Finally, in the publication “Analytical profiles of drug substances and excipients”, vol. 25, (1998), pag. 85-121 , in particular at pag. 99, it is stated that prior to 1976 the cis stereochemistry was wrongly assigned to the trans-isomer of Clomiphene (E-Chlomiphene or Enclomiphene), and only after the above publication on Acta Cryst. the correct geometric isomery has been definitively assigned.
These observations in the prior art have been confirmed by our experimentation. In particular, repeating the experiment 31 of US patent 3,848,030, the trans-Clomiphene salt with racemic binaphthyl-phosphoric acid was isolated and not the salt with cis-Clomiphene as stated in said patent, as confirmed by 2D H-NMR analysis (NOESY experiment). Thus, Example 31 of US3,848,030, provides, at the end, Enclomiphene citrate, crystallized from a mixture of ethyl ether and ethanol, having a m.p. of 133-135°C. Example 32, instead provided Cis-Clomiphene citrate, crystallized from a mixture of ethyl ether and ethanol, having a m.p. of 120-126°C.
Thus, with the aim of preparing Enclomiphene citrate, whole experiment 31 of US3,848,030 has been reworked also carrying out the crystallization of the product form a mixture of ethyl ether and ethanol, hence providing a not crystalline solid with two DSC peaks respectively at 1 14°C and 188°C, although the starting material used for the reworking example was quite a pure substance (HPLC Analysis (A A%) is 98.95% of Enclomiphene), and having a substantially the same chemical purity of that used in the prior art experiment (m.p. of our Enclomiphene BPA salt was 218°C versus 220- 222°C of the prior art Enclomiphene BPA salt of Example 31 ).
The patent US2,914,563, in example 3, discloses a process for the preparation of trans-Clomiphene citrate, containing from 30% to 50% of cis-Clomiphene, as citrate, by reaction of 1 -ρ-(β- diethylaminoethoxy)phenyl]-1 ,2-diphenylethylene hydrochloride with N- chlorosuccinimmide in dry chloroform under reflux.
Khimiko-Farmatsevticheskii Zhurnal (1984), 18(1 1 ), 1318-24 English translation in the review Pharmaceutical Chemistry Journal November 1984, Volume 18, Issue 1 1 , pag. 758-764 (Title: Synthesis and biological study of the cis- and trans-isomers of Clomiphene citrate and some intermediates of its synthesis) discloses the trans-isomer of Clomiphene citrate, i.e. Enclomiphene citrate, characterized by:
1 H-NMR (MeOD) d 7.4-6.7 (m, 14H); 4.27 (t, 2H, -OCH2); 3.51 (t, 2H, CH2- N); 3.28 (q, 4H, 2xN-CH2)); 2.73 (2H); 2.78 (2H); 1.31 (t, 6H, 2xN-C-CHs)) Melting point: 138-139°C (98% purity by GLC);
IR spectrum, v cm-1 (suspension in mineral oil): 3640, 3430, 1720, 1710
(citrate), 1600-1555 (broad band, stilbene system); 750.
UV spectrum: λ max = 243 nm, ε 21 ,800 and λ max 300 nm, ε 1 1 ,400.
These prior art methods for the preparation of Enclomiphene citrate do not allow the preparation of Enclomiphene citrate having needle shaped crystal habit, indeed the crystallization by means of a mixture of ethyl ether and ethanol does not provide a crystalline solid having needle crystals.
Moreover, Enclomiphene citrate was described in literature with different melting points, in particular, 133-135°C and 138-139°C. Said solid forms of Enclomiphene citrate fail to comply with stabilities studies and furthermore show relatively poor solubility in water either in neutral or acid pH.
Furthermore, the prior art methods have the drawbacks related to the poor reproducibility of the process and of the solid form thus obtained.


EXPERIMENTAL SECTION
The starting material Clomiphene citrate can be prepared according to well-known prior art methods, or for example, as described in the example 1 of PCT/EP2015/074746 or can be purchased on the market.
[00190] Example 1 : Preparation of salt of Enclomiphene with racemic binaphthyl- phosphoric acid, starting from Clomiphene citrate.

Clomiphene citrate
[00191] A round bottom flask was charged 100 gr of Clomiphene Citrate (HPLC analysis (A/A%): 65.21 % Enclomiphene, 34.06% Z-Clomiphene) and 1000 mL of methanol. The suspension was stirred at 30°C up the complete
dissolution. Then a solution of racemic binaphthyl-phosphoric acid (abbreviated BPA) 30 gr (0.515 eq) in 30 ml_ of DMF was added. At the end of addition the mixture was stirred for 1 h at 30°C. The obtained suspension was filtered and the solid was washed with 100 ml_ of methanol.
[00192] 50.4 gr of Enclomiphene BPA salt (III) were obtained.
[00193] HPLC Analysis (A/A%): 97.04% Enchlomiphene, 2.5% Z-Clomiphene.
[00194] Example 1 b: Preparation of salt of Enclomiphene with racemic binaphthyl- phosphoric acid, starting from Clomiphene citrate.
[00195] A round bottom flask was charged 50 gr of Clomiphene Citrate and 500 ml_ of methanol. The suspension was heated at 40-45°C and stirred up to the complete dissolution. Then a solution of BPA 15 gr (0.515 eq) in 300 ml_ of methanol was added. At the end of addition the mixture was stirred for 1 h at 20°C. The obtained suspension was filtered and the solid was washed with 100 ml_ of methanol.
24.1 gr of Enclomiphene BPA salt were obtained.
HPLC Analysis (A/A%): 98.96% Enchlomiphene, 0.69% Z-Clomiphene.
[00196] Example 1 c: Preparation of salt of Enclomiphene with racemic binaphthyl- phosphoric acid, starting from Clomiphene citrate.
[00197] In a round bottom flask was charged 100 gr of Clomiphene Citrate and 1000 ml_ of methanol. The suspension was heated at 40-45°C and stirred up the complete dissolution. Then a solution of BPA 30 gr (0.515 eq) in 1000 ml_ of methanol was added. At the end of addition the mixture was stirred for 1 h at 20°C. the obtained suspension was filtered and the solid was wash with 100 ml_ of methanol.
47.9 gr of Enclomiphene BPA salt were obtained.
HPLC Analysis (A/A%): 98.81 % Enclomiphene, 0.79% Z-Clomiphene.
[00198] Example 1d: Preparation of salt of Enclomiphene with racemic binaphthyl- phosphoric, starting from Clomiphene citrate.
[00199] In a round bottom flask was charged 150 gr of Clomiphene citrate and 1500 mL of methanol. The suspension was heater at 40-45°C and stirred up the complete dissolution. Then a solution of BPA 45 gr (0.515 eq) in 900 mL of methanol was added. At the end of addition the mixture was
stirred for 1 h at 20°C. the obtained suspension was filtered and the solid was wash with 100 ml_ of methanol.
76.4 gr of E-Clomiphene BPA salt were obtained.
HPLC Analysis (A/A%): 98.82% Enchlomiphene, 0.80% Z-Clomiphene.
[00200] Example 2: Recrystallization of Enclomiphene BPA salt of formula (III) (the step A).

(Ill)
[00201] Into a proper 0.5 L reactor, equipped with propeller, temperature probes, condenser; Enclomiphene BPA salt (III) (50 g) and having Z-isomer of 1.64 % was suspended in DMF (2.1 L/Kg of Enclomiphene BPA (III)) and methanol (1.4 L/Kg of Enclomiphene BPA salt (III)). The suspension was heated to reflux (~ 76-79°C). Further DMF (0.1 L/Kg of Enclomiphene BPA (III)) might be required to improve the solubility of the starting material. Once the starting material was completely dissolved, methanol was added as anti-solvent (3.5 L/Kg of Enclomiphene BPA (III)). The temperature was decreased to 60°C and the mixture was stirred for 2 – 3 h. Then, the temperature was further decreased to 20 °C and filtered. The wet cake was washed twice with methanol (1.5 L/Kg of Enclomiphene BPA salt (III)). The product was dried under vacuum at 60 – 70 °C for 12 – 24 h. Time of drying could be prolonged until residual DMF is < 2500 ppm.
[00202] Analysis of quality of the final product of the above mentioned example and of the same product, obtained from repetition following the same process, it is shown in the following table:
Enclomiphene BPA (III) salt Enclomiphene BPA (III) salt rixx (Starting product) (finale product)
Z-isomer = 1.64 A/A% Z-isomer = 0.07 A/A%
Z-isomer = 0.79 A/A% Z- isomer = 0.03 A/A%
[00203] Example 3: Preparation of Enclomiphene citrate of formula (I), having needle shaped crystal habit, starting from Enclomiphene BPA salt formula (III).

(II)
[00204] Into a proper 4 L reactor, equipped with propeller, temperature probes, condenser; Enclomiphene BPA salt of formula (III) (400 g, assay 99.8 wt% 0.528 mol, 1 equiv.) was suspended in methyl-tert-butyl ether (MTBE, 2 L), isopropanol (IPA, 0.5 L) and water (2 L). The mixture was stirred for 15 minutes, then 0.48 L of ammonia solution 30 wt% was added and the mixture was further stirred for one hour. The aqueous phase was separated and the organic layer was washed with a solution of ammonia solution 30 wt% (0.12 L) and water (0.6 L). The aqueous phase was separated and the organic layer was finally washed with water (0.6 L). The organic solution was evaporated to residue under vacuum at 60-65°C. The residue was dissolved in 1.36 L of absolute ethanol. The assay of the solution was determined at this stage through a potentiometric titration and results in 15.125 wt% as Enclomiphene of formula (II) (0.466 mol). Then 0.24 L of water were added and the solution was heated to 65°C. Meanwhile, citric acid monohydrate (100.8 g, 0.475 mol, 1.02 equiv.) was dissolved in absolute ethanol (1.7 L) and water (0.3 L), the solution was heated to 65°C. The solution of citric acid was dropped into the solution of Enclomiphene (II), while maintaining 65°C. The dosage takes place in 30- 40 minutes. The inner temperature was decreased very slowly to 60°C over 80 minutes, then it was further decrease to 55°C over 40 minutes. When the inner temperature was in the range 60-55°C (typically at 58°C), the crystallization mixture was seeded with Enclomiphene citrate needle- shaped and a white product began to precipitate. Once reached 55°C the temperature was further decreased to 30°C over 30 minutes, then to 0°C over 30 minutes. The slurry was stirred at 0°C for at least two hours, then it was filtered and the wet cake was washed with 0.4 L of absolute ethanol. The product was dried under vacuum at 65°C. At the end of drying, 269 g of Enclomiphene citrate of formula (I) as needle crystal were isolated, corresponding to 91.8% molar yield.
[00205] HPLC Analysis (A/A%): 99.79% Enchlomiphene, 0.04% Z-Clomiphene (i.e. Z-isomer).
[00206] Example 4: Preparation of Enclomiphene citrate of formula (I), having a needle shaped crystal habit, with a mixture of ethanol and water, wherein the amount of water is 15%.

(I)
[00207] Into a proper 1 L reactor, equipped with propeller, temperature probes, condenser; Enclomiphene of fomula (II) (15,0 g, assay 99.9 wt% 0.0369 mol, 1 equiv.) was dissolved in absolute ethanol (102 ml_, 6.8 mL/g of free base), then 18 ml_ (1.2 mL/g of free base) of water were added and the solution was heated to 65°C. Meanwhile, citric acid monohydrate (7.92 g, 0.0377 mol, 1.02 equiv.) was dissolved in absolute ethanol (127 ml_) and water (23 ml_), the solution was heated to 65°C. The solution of citric acid was dropped into the solution of Enclomiphene (II), while maintaining 65°C. The dosage takes place in 30-40 minutes. The inner temperature was decreased very slowly to 60°C over 80 minutes, then it was further decrease to 55°C over 40 minutes. When the inner temperature was in the range 60-55°C (typically at 58°C), the crystallization mixture was seeded with Enclomiphene citrate needle-shaped and a white product began to precipitate. Once reached 55°C the temperature was further decreased to 30°C over 30 minutes, then to 0°C over 30 minutes. The slurry was stirred at 0°C for at least two hours, then it was filtered and the wet cake was washed with 30 ml_ of absolute ethanol. The product was dried under
vacuum at 65°C. At the end of drying, 20.2 g of Enclomiphene citrate of formula (I) as needle crystal were isolated, corresponding to 91.4% molar yield.
[00208] HPLC Analysis (A/A%): 99.86% Enchlomiphene, 0.03% Z-Clomiphene.
[00209] Example 4a: Preparation of Enclomiphene citrate of formula (I), having a needle shaped crystal habit, with a mixture of isopropanol and water, wherein the amount of water is 15%.
[00210] Into a proper 1 L reactor, equipped with propeller, temperature probes, condenser; Enclomiphene of fomula (II) (40,0 g, assay 99.9 wt% 0.0985 mol, 1 equiv.) was dissolved in isopropanol (272 ml_, 6.8 mL/g of free base), then 48 ml_ (1.2 mL/g of free base) of water were added and the solution was heated to 65°C. Meanwhile, citric acid monohydrate (21.10 g, 0.100 mol, 1.02 equiv.) was dissolved in isopropanol (340 ml_, 8.5 mL/g of free base) and water (60 mL, 1.5 mL/g of free base), the solution was heated to 65°C. The solution of citric acid was dropped into the solution of Enclomiphene (II), while maintaining 65°C. The dosage takes place in 30- 40 minutes. The inner temperature was decreased very slowly to 60°C over 80 minutes, then it was further decrease to 55°C over 40 minutes. When the inner temperature was in the range 60-55°C (typically at 58°C), the crystallization mixture was seeded with Enclomiphene citrate needle- shaped and a white product began to precipitate. Once reached 55°C the temperature was further decreased to 30°C over 30 minutes, then to 0°C over 30 minutes. The slurry was stirred at 0°C for at least two hours, then it was filtered and the wet cake was washed with 30 mL of isopropanol. The product was dried under vacuum at 65°C. At the end of drying, 56.5 g of Enclomiphene citrate of formula (I) as needle crystal were isolated, corresponding to 95.9% molar yield.
[0021 1] Example 4b: Preparation of Enclomiphene citrate of formula (I), having a needle shaped crystal habit, with a mixture of n-propanol and water, wherein the amount of water is 15%.
[00212] Into a proper 0.5 L reactor, equipped with propeller, temperature probes, condenser; Enclomiphene of fomula (II) (9,0 g, assay 99.9 wt% 0.0985 mol, 1 equiv.) was dissolved in 7-propanol (61 mL, 6.8 mL/g of free base), then 1 1 ml_ (1.2 mL/g of free base) of water were added and the solution was heated to 65°C. Meanwhile, citric acid monohydrate (4.70 g, 0.0224 mol, 1.02 equiv.) was dissolved in 7-propanol (77 ml_, 8.5 mL/g of free base) and water (14 ml_, 1.5 mL/g of free base), the solution was heated to 65°C. The solution of citric acid was dropped into the solution of Enclomiphene (II), while maintaining 65°C. The dosage takes place in 30- 40 minutes. The inner temperature was decreased very slowly to 60°C over 80 minutes, then it was further decrease to 55°C over 40 minutes. When the inner temperature was in the range 60-55°C (typically at 58°C), the crystallization mixture was seeded with Enclomiphene citrate needle- shaped and a white product began to precipitate. Once reached 55°C the temperature was further decreased to 30°C over 30 minutes, then to 0°C over 30 minutes. The slurry was stirred at 0°C for at least two hours, then it was filtered and the wet cake was washed with 30 mL of 7-propanol I. The product was dried under vacuum at 65°C. At the end of drying, 1 1.7 g of Enclomiphene citrate of formula (I) as needle crystal were isolated, corresponding to 88.1 % molar yield
[00213] Example 4c: Preparation of Enclomiphene citrate of formula (I), having a needle shaped crystal habit, with a mixture of n-butanol and water, wherein the amount of water is 15%.
[00214] Into a proper 0.5 L reactor, equipped with propeller, temperature probes, condenser; Enclomiphene of fomula (II) (9,0 g, assay 99.9 wt% 0.0985 mol, 1 equiv.) was dissolved in 7-butanol (61 mL, 6.8 mL/g of free base), then 1 1 mL (1.2 mL/g of free base) of water were added and the solution was heated to 65°C. Meanwhile, citric acid monohydrate (4.70 g, 0.0224 mol, 1.02 equiv.) was dissolved in 7-butanol (77 mL, 8.5 mL/g of free base) and water (14 mL, 1.5 mL/g of free base), the solution was heated to 65°C. The solution of citric acid was dropped into the solution of Enclomiphene (II), while maintaining 65°C. The dosage takes place in 30- 40 minutes. The inner temperature was decreased very slowly to 60°C over 80 minutes, then it was further decrease to 55°C over 40 minutes. When the inner temperature was in the range 60-55°C (typically at 58°C), the crystallization mixture was seeded with Enclomiphene citrate needle- shaped and a white product began to precipitate. Once reached 55°C the temperature was further decreased to 30°C over 30 minutes, then to 0°C over 30 minutes. The slurry was stirred at 0°C for at least two hours, then it was filtered and the wet cake was washed with 30 ml_ of 7-butanol. The product was dried under vacuum at 65°C. At the end of drying, 1 1.6 g of Enclomiphene citrate of formula (I) as needle crystal were isolated, corresponding to 87.4% molar yield.
[00215] Example 4d: Preparation of Enclomiphene citrate of formula (I), having a needle shaped crystal habit, with a mixture of tert-butanol and water, wherein the amount of water is 15%.
[00216] Into a proper 0.5 L reactor, equipped with propeller, temperature probes, condenser; Enclomiphene of fomula (II) (9,0 g, assay 99.9 wt% 0.0985 mol, 1 equiv.) was dissolved in te T-butanol (61 ml_, 6.8 mL/g of free base), then 1 1 ml_ (1.2 mL/g of free base) of water were added and the solution was heated to 65°C. Meanwhile, citric acid monohydrate (4.70 g, 0.0224 mol, 1.02 equiv.) was dissolved in te T-butanol (77 ml_, 8.5 mL/g of free base) and water (14 mL, 1.5 mL/g of free base), the solution was heated to 65°C. The solution of citric acid was dropped into the solution of Enclomiphene (II), while maintaining 65°C. The dosage takes place in 30- 40 minutes. The inner temperature was decreased very slowly to 60°C over 80 minutes, then it was further decrease to 55°C over 40 minutes. When the inner temperature was in the range 60-55°C (typically at 58°C), the crystallization mixture was seeded with Enclomiphene citrate needle- shaped and a white product began to precipitate. Once reached 55°C the temperature was further decreased to 30°C over 30 minutes, then to 0°C over 30 minutes. The slurry was stirred at 0°C for at least two hours, then it was filtered and the wet cake was washed with 30 mL of te T-butanol. The product was dried under vacuum at 65°C. At the end of drying, 1 1.2 g of Enclomiphene citrate of formula (I) as needle crystal were isolated, corresponding to 84.4% molar yield.
[00217] Example 5: Preparation of Enclomiphene citrate of formula (I), having a needle shaped crystal habit. Preparation of the seed crystal.
[00218] Into a proper 1 L reactor, equipped with propeller, temperature probes, condenser; Enclomiphene of fomula (II) (15,0 g, assay 99.9 wt% 0.0369 mol, 1 equiv.) was dissolved in absolute ethanol (102 ml_, 6.8 mL/g of free base), then 18 ml_ (1.2 mL/g of free base) of water were added and the solution was heated to 65°C. Meanwhile, citric acid monohydrate (7.92 g,
0.0377 mol, 1.02 equiv.) was dissolved in absolute ethanol (127 ml_, 8.5 mL/g of free base) and water (23 mL 1.5 mL/g of free base), the solution was heated to 50°C. The solution of citric acid was dropped into the solution of Enclomiphene (II), while maintaining 50°C. The dosage takes place in 30-40 minutes. At the end of the dosage, the stirring was turned off and the mixture was allowed to cool down to room temperature without stirring. The product began to crystallize at 40-30°C. Once reached 20- 25°C the stirring was turned on and the temperature was further decreased to 0°C over 30 minutes. The slurry was stirred at 0°C for at least two hours, then it was filtered and the wet cake was washed with 30 mL of absolute ethanol. The product was dried under vacuum at 65°C. At the end of drying, 13.9 g of Enclomiphene citrate of formula (I) were isolated, corresponding to 62.3% molar yield
[00219] Example 6: Preparation of Enclomiphene citrate of formula (I), having a non-needle shaped crystals, with a mixture of acetone and water, wherein the amount of water is 15%.
Comparative example (see Fig. 8) and evidence example of the invention. Following the same process described in the example 4, substituting ethanol solvent with acetone solvent. Starting from 15,0 g of Enclomiphene of formula (II), following the above mentioned process, 22.3 g of Enclomiphene citrate of formula (I) were isolated, corresponding to 94.2% molar yield product. For the morphology of the crystal see fig. 8.
[00220] Indeed, the microscopy analysis provides a better further evidence of the crystal habit of Enclomiphene citrate (I) of the example 6 (see Fig.8) which has a form more different than/to Enclomiphene citrate (I) having a needle shaped crystal habit, obtained according to above described examples,
1. e. 4, 4a, 4b, 4c, 4d (see Fig. 5, 6 and 7).
[00221] HPLC Analysis (A/A%): 99.63% Enchlomiphene, 0.20% Z-Clomiphene.
[00222] Example 7: Analytical method to identify and quantify Z-Clomiphene of formula (IV) into Enclomiphene of formula (II) or Enclomiphene citrate of formula (I) or Enclomiphene BPA salt of formula (III) and for determining the chemical purity.
[00223] Chromatographic conditions:
Dim. Column: 250 mm x 4.6 mm , 5 pm
Stationaly phase: Butyl sylane (USP phase L26, Vydac 4C is suggested) Temp. Column: room temperature
Mobile Phase: Methanol / water / triethylamine 55 : 45 : 0.3 v/v
Adjust at pH 2.5 with phosphoric acid
Flow: 1.0 mL/min
Detector UV a 233 nm,
Injection Volume: 10 μΙ_
Sample diluent: mobile phase.
Applying the conditions described above the expected retention times are as indicated below:

/////////////////Enclomiphene citrate, New patent, WO 2017182097, F.I.S. – FABBRICA ITALIANA SINTETICI S.P.A

Enclomiphene
citrate); 7619-53-6 ((Z)-clomifene citrate)
 Chemical Structure of Clomifene
Chemical Structure of Clomifene
NOTE:
Clomifene may be separated into its Z-and E-isomers, zuclomifene and enclomifene
.
энкломифен Enclomiphene citrate إينكلوميفان

Enclomiphene citrate
NDA FILED Hypogonadism, Repros Therapeutics
An estrogen receptor (ER) antagonist potentially for treatment of hypogonadotropic hypogonadism.

ICI-46476; RMI-16289
CAS No.15690-57-0(free)
7599-79-3(Enclomiphene citrate)
| Molecular Weight | 598.08 |
| Formula | C26H28ClNO▪C6H8O7 |
Ethanamine, 2-[4-[(1E)-2-chloro-1,2-diphenylethenyl]phenoxy]-N,N-diethyl-, 2-hydroxy-1,2,3-propanetricarboxylate (1:1)
- Ethanamine, 2-[4-(2-chloro-1,2-diphenylethenyl)phenoxy]-N,N-diethyl-, (E)-, 2-hydroxy-1,2,3-propanetricarboxylate (1:1)
- Triethylamine, 2-[p-(2-chloro-1,2-diphenylvinyl)phenoxy]-, citrate (1:1), (E)-
- (E)-Clomiphene citrate
- Androxal
- Clomiphene B citrate
- Enclomid
- Enclomiphene citrate
- trans-Clomiphene citrate

Clomifene is a mixture of two geometric isomers, enclomifene (E-clomifene) and zuclomifene (Z-clomifene). These two isomers have been found to contribute to the mixed estrogenic and anti-estrogenic properties of clomifene.
EXAMPLE 1
Preparation of trans-clomiphene citrate from
1- {4- [2-(Oiethylamino)ethoxy| phenylj-1 ,2-diphenylethanol
Dehydration
[0023] l-{4-[2-(Diethylamino)ethoxy]phenyl}-l,2-diphenylethanol (6) dissolved in ethanol containing an excess of hydrogen chloride was refluxed 3 hours at 50 °C. The solvent and excess hydrogen chloride were removed under vacuum and the residue was dissolved in dichloromethane. 2-{4-[(Z)-l,2-diphenylvinyl]phenoxy}-N,N- diethylethanaminium hydrogen chloride (7) was obtained.
Chlorination
The hydrochloride salt (7) solution obtained above was treated with 1.05 equivalents of N-chlorosuccinimide and stirred at room temperature for about 20 hours. Completion of the reaction was confirmed by HPLC. The hydrochloride salt was converted to the free base by addition of saturated aqueous bicarbonate solution. The mixture was stirred at room temperature for 30 minutes after which the phases were separated and the organic phase was evaporated in vacuo. 2-{4-[2-chloro-l,2- diphenylvinyl]phenoxy}-N,N-diethylethanamine (clomiphene -1.8:1 E:Z mixture) (8) was obtained.
Separation of clomiphene isomers
Clomiphene (8) obtained above is dissolved in methanol and racemic binaphthyl- phosphoric acid (BPA) is added under stirring. When the precipitate begins separating from the solution, stirring is stopped and the mixture is allowed to settle at room temperature for 2 hours. The precipitate is filtered, washed with methanol and ether and dried. Trans-clomiphene-BPA salt (3) is obtained.
The enclomiphene-BPA salt (3) obtained above is extracted with ethyl acetate and NH3 solution. To the organic solution washed with water and dried, citric acid dissolved in ethanol is added. The solution is allowed to settle for about one hour at room temperature; the precipitate is then filtered and dried under vacuum. The obtained precipitate, trans-clomiphene citrate (1) is dissolved in 2-butanone for storage.
EXAMPLE 2
Synthesis of Clomiphene Using a Single Solvent
Step 1 – Dehydration of l-i4-r2-(Diethylamino)ethoxy1phenyl|-l,2- diphenylefhanol to form 2-{4-[(Z)-l,2-diphenylvinyllphenoxy}-N,N-diethylethanaminium hydrogen sulfate (7) [0030] The synthesis route described in Example 1 utilized HC1 for the dehydration step and utilized ethanol at 50 °C as the solvent. Sulfuric acid was investigated as an alternative to HC1 for the dehydration step (as described in Example 1) in part due to the more favorable corrosion profile of sulfuric acid. Dichloromethane (methylene chloride) was investigated as an alternative solvent for the dehydration step as this would render removal of the ethanol solvent prior to the chlorination step unnecessary.
A 100 mL 3-neck round bottom flask, fitted with a temperature probe and a stir bar, was charged with l- {4-[2-(Diethylamino)ethoxy]phenyl}-l,2-diphenylethanol (6) (6.60 g, 16.9 mmol) and 66 mL (lxlO3 mmol) of methylene chloride to give a yellow solution which was cooled in an ice bath to 0 °C. Concentrated sulfuric acid (H2S04, 0.96 mL, 18.1 mmol) was added at a rate such that the internal temperature did not exceed 5 °C. Upon completion of the addition, the mixture was allowed to stir one hour at ambient temperature. Completion of the reaction was confirmed by high performance liquid chromatography (HPLC). The reaction resulted in 7.96 grams of 2- (4-[(Z)- 1 ,2- diphenylvinyl]phenoxy}-N,N-diethylethanaminium hydrogen sulfate (7), a yield of 100%. Thus, sulfuric acid was demonstrated to be a suitable acid for the dehydration step.
[0042] Using these HPLC conditions, starting material has a retention time of 3.30 min and product has a retention time of 4.05 min.
It was determined that removal of water produced by the dehydration reaction was important before performing the chlorination step. When ethanol is used as the solvent for this reaction, as in Example 1, the water is removed azeotropically upon removal of the ethanol. Several methods of drying the dichloromethane solution were attempted. Drying with MgS04 had a deleterious effect on the subsequent chlorination step, rendering the chlorination process very messy with a number of new impurities observed following HPLC analysis which were determined to be the corresponding chlorohydrins. On the other hand, a wash with brine was sufficient to remove enough water and had no deleterious effect on the chlorination step. Accordingly, the solution was stirred vigorously with brine (66 ml) for 30 minutes and then the phases were separated prior to chlorination step.
Step 2- Synthesis of 2-|4-r2-chloro-L2-diphenylvinyl1phenoxyl-N,N- diethylethanamine 8
The solution of 2-{4-[(Z)-l,2-diphenylvinyl]phenoxy}-N,N-diethylethanaminium hydrogen sulfate (7.94 grams) in methylene chloride obtained in step 1 is stirred at room temperature and treated with N-chlorosuccinimide (2.37 g, 17.7 mmol, 1.05 equivalents) in a single portion and left to stir at room temperature for 12 hours. The yellow solution became orange and then went back to yellow. After 12 hours, a sample was removed, concentrated and assayed by HPLC to confirm the extent of reaction. HPLC analysis revealed that the reaction had proceeded but not to completion. Accordingly, an additional 0.09 equivalents of N-chlorosuccinimide (203 mg, 1.52 mmol) was added and the solution stirred at room temperature for an additional 4 hours. The reaction was again assayed by HPLC which revealed that the reaction was near completion. Accordingly, an additional 0.09 equivalents of N-chlorosuccinimide (203 mg, 1.52 mmol) was added and the solution stirred for an additional 12 hours at room temperature. The reaction was again assayed by HPLC and an additional 0.058 equivalents of N-chlorosuccinimide (131 mg, 0.98 mmol) was added and the solution stirred for an additional 4 hours. HPLC indicated that the reaction was complete at that point. The reaction was carefully quenched by slow addition of 66 mL (600 mmol) of saturated aqueous sodium bicarbonate solution and the quenched mixture was stirred for 30 minutes at room temperature – the reaction mixture pH should be about 8-9 after addition of saturated aqueous sodium bicarbonate solution. The reaction yielded 6.86 grams of 2-{4-[2-chloro-l,2-diphenylvinyl]phenoxy}-N,N- diethylethanamine (8). The phases were separated and the organic phase was evaporated in vacuo. The resulting light brown oil was transferred to a tared amber bottle using a small volume of dichloromethane.
[0055] Using these HPLC conditions, the retention time of product is 15 minutes.
Chromatographic Separation of Clomiphene Isomers
Clomiphene (mixture of isomers) in free base form obtained by steps 1 and 2 is loaded onto a chromatographic column (e.g. batch high pressure chromatography or moving bed chromatography) using the same solvent as used in steps 1 and 2 (here DCM) in order to separate the cis- and trans-clomiphene isomers. Trans-clomiphene is preferably eluted using a solvent suitable for recrystallization.
Journal of Chromatography (1984), 298, (1), 172-4.
US2914562https://www.google.co.in/patents/US2914562PATENTUS2914529
http://www.google.co.in/patents/US2914529
PAPER
J. Med. Chem.1967, 10, 84–86.
DOI: 10.1039/C5CC01968K
| CN103351304A * | Jul 1, 2013 | Oct 16, 2013 | 暨明医药科技(苏州)有限公司 | Synthesis method of clomiphene |
| US2914563 * | Aug 6, 1957 | Nov 24, 1959 | Wm S Merrell Co | Therapeutic composition |
| US3848030 * | Mar 10, 1972 | Nov 12, 1974 | Richardson Merrell Spa | Optical isomers of binaphthyl-phosphoric acids |
| US5681863 * | Dec 5, 1994 | Oct 28, 1997 | Merrell Pharmaceuticals Inc. | Non-metabolizable clomiphene analogs for treatment of tamoxifen-resistant tumors |
| Reference | ||
|---|---|---|
| 1 | * | RAO ET AL.: “Synthesis of carbon-14 labeled clomiphene.“, JOUMAL OF LABELLED COMPOUNDS AND RADIOPHARMACEUTICALS, vol. 22, no. 3, 1985, pages 245 – 255, XP055180053, Retrieved from the Internet <URL:http://onlinelibrary. wiley .com/doi/10.1002/jlcr.2580220306/abstract> [retrieved on 20150504] |
//////////энкломифен, Enclomiphene citrate, إينكلوميفان , ICI-46476, RMI-16289, nda filed, Hypogonadism, Repros Therapeutics

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....