Home » Posts tagged 'DO94E28WYW'
Tag Archives: DO94E28WYW
Amsulostat
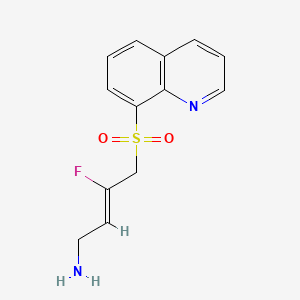
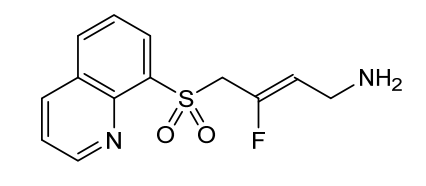
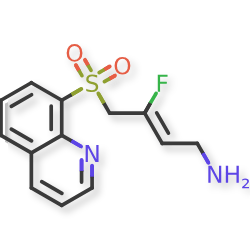
Amsulostat
CAS 2409963-83-1
MF C13H13FN2O2S MW280.32
2-Buten-1-amine, 3-fluoro-4-(8-quinolinylsulfonyl)-, (2Z)-
(2Z)-3-fluoro-4-(quinolin-8-ylsulfonyl)but-2-en-1-amine
(Z)-3-fluoro-4-quinolin-8-ylsulfonylbut-2-en-1-amine
(2Z)-3-fluoro-4-(quinolin-8-ylsulfonyl)but-2-en-1-amine
protein-lysine-oxidase (LOX) inhibitor, antifibrotic, PXS 5505, LOX inhibitor PXS-5505, LOX-IN-3, pan-LOX inhibitor PXS-5505, DO94E28WYW, SNT 5505
Amsulostat is an orally available, small-molecule, irreversible inhibitor of all lysyl oxidases (LOX) family members, with potential antifibrotic activity. Upon oral administration, amsulostat targets, binds to and inhibits the activity of all enzymes in the LOX family. This prevents the post-translational oxidative deamination of lysine residues on target proteins, including collagen and elastin, and reduces the formation of deaminated lysine (allysine), the formation of inter- and intramolecular cross-linkages and may prevent remodeling of the extracellular matrix (ECM), thereby reducing fibrotic tissue formation in certain chronic fibrotic diseases. LOX is often upregulated in fibrotic tissue and plays a key role in fibrosis.
Amsulostat (formerly PXS-5505) is an orally available, investigational, pan-lysyl oxidase (pan-LOX) inhibitor designed by Syntara to treat fibrotic diseases and solid tumors. It works by preventing collagen cross-linking and remodeling of the extracellular matrix, effectively reducing fibrosis. The drug is currently in Phase 2 clinical trials for myelofibrosis, showing promise in reducing symptom burden and spleen volume, and is also being studied for myelodysplastic syndrome (MDS) and pancreatic cancer.
Key Aspects of Amsulostat:
- Mechanism of Action: Irreversibly inhibits all LOX family members (LOX, LOXL1-4), reducing fibrotic tissue.
- Clinical Status (Myelofibrosis): Phase 2a data showed 73% of patients (who were suboptimal responders to ruxolitinib) achieved
reduction in total symptom score, with significant spleen volume reduction.
- Clinical Status (Other Cancers): Phase 2 trials (AZALOX) are evaluating its use in myelodysplastic syndrome (MDS) and chronic myelomonocytic leukemia (CMML). It is also being tested in combination with chemotherapy for pancreatic cancer to improve drug delivery to tumors.
- Regulatory Status: Has received Orphan Drug Designation for primary myelofibrosis from the FDA (USA) and EMA (Europe).
- Safety Profile: Clinical trials have reported it is well-tolerated with no treatment-related serious adverse events in early findings.
Amsulostat’s ability to target the stiff, fibrotic environment surrounding tumors makes it a promising “add-on” therapy to increase the effectiveness of existing cancer treatments, including chemotherapy and immunotherapy.
An orally available, small-molecule, irreversible inhibitor of all lysyl oxidases (LOX) family members, with potential antifibrotic activity. Upon oral administration, amsulostat targets, binds to and inhibits the activity of all enzymes in the LOX family. This prevents the post-translational oxidative deamination of lysine residues on target proteins, including collagen and elastin, and reduces the formation of deaminated lysine (allysine), the formation of inter- and intramolecular cross-linkages and may prevent remodeling of the extracellular matrix (ECM), thereby reducing fibrotic tissue formation in certain chronic fibrotic diseases. LOX is often upregulated in fibrotic tissue and plays a key role in fibrosis.
SYN
syn

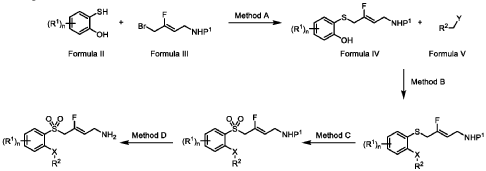
(Z)-3-fluoro-4-(quinolin-8-ylsulfonyl)but-2-en-1-amine dihydrochloride (Compound 33)
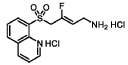
[0282] White solid; m.p.150-153 °C; 1H NMR (300 MHz, CD3OD) d ppm: 9.18 (d, J = 4.7 Hz, 1H), 8.70 (dd, J = 8.4, 2.6 Hz, 1H), 8.57 (d, J = 7.4 Hz, 1H), 8.45 (d, J = 8.5 Hz, 1H), 7.99– 7.68 (m, 2H), 5.22 (dt, J = 32.9, 7.4 Hz, 1H), 5.00 (d, J = 19.4 Hz, 2H), 3.60 (d, J = 7.7 Hz, 2H); LCMS: for C13H13FN2O2S calculated 280.1, found 281.1 [M+1]+.
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Haloallylamine sulfone derivative inhibitors of lysyl oxidases and uses thereofPublication Number: EP-3829520-B1Priority Date: 2018-08-03Grant Date: 2024-11-27
- Haloallylamine sulfone derivative inhibitors of lysyl oxidases and uses thereofPublication Number: US-2025082593-A1Priority Date: 2018-08-03
- Haloallylamine sulfone derivative inhibitors of lysyl oxidases and uses thereofPublication Number: US-2021353571-A1Priority Date: 2018-08-03
- Lox enzyme inhibiting methods and compositionsPublication Number: EP-4138824-A1Priority Date: 2020-04-21
- Haloallylamine sulfone derivative inhibitors of lysyl oxidases and uses thereofPublication Number: WO-2020024017-A1Priority Date: 2018-08-03
- Inhibitors of haloallylamine sulfone derivatives of lysyl oxidase and uses thereofPublication Number: KR-20210045984-APriority Date: 2018-08-03
- Halide allylamine derivate inhibitor for amine oxidase and its usePublication Number: TW-202019877-APriority Date: 2018-08-03
- Haloallylamine sulfone derivative inhibitors of lysyl oxidases and uses thereofPublication Number: US-12178791-B2Priority Date: 2018-08-03Grant Date: 2024-12-31
- Bithiazol deratives as inhibitors of lysyl oxidasesPublication Number: EP-4333843-A1Priority Date: 2021-05-13
- Inhibitors of lysyl oxidasesPublication Number: US-11712437-B2Priority Date: 2021-05-13Grant Date: 2023-08-01
- Method for selecting cancer patients for whom combination therapy of retinoid with cancer therapeutic agent is effective, and combination drug of retinoid with cancer therapeutic agentPublication Number: CN-115997122-APriority Date: 2020-06-26
- Method for selecting cancer patient for which combination therapy of retinoid and cancer treatment agent will be effective, and combined drug of retinoid and cancer treatment agentPublication Number: EP-4173639-A1Priority Date: 2020-06-26
- Method for selecting cancer patients for whom combination therapy with retinoid and cancer therapeutic agent is effective, and combination medicament with retinoid and cancer therapeutic agentPublication Number: US-2023301950-A1Priority Date: 2020-06-26
////////////amsulostat, ANAX LAB, protein-lysine-oxidase (LOX) inhibitor, antifibrotic, PXS 5505, LOX inhibitor PXS-5505, LOX-IN-3, pan-LOX inhibitor PXS-5505, DO94E28WYW, SNT 5505

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










