Home » Posts tagged 'dabogratinib'
Tag Archives: dabogratinib
Dabogratinib
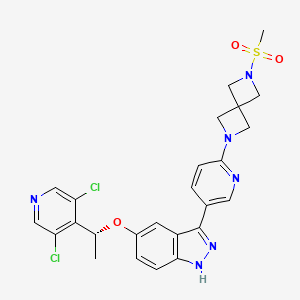
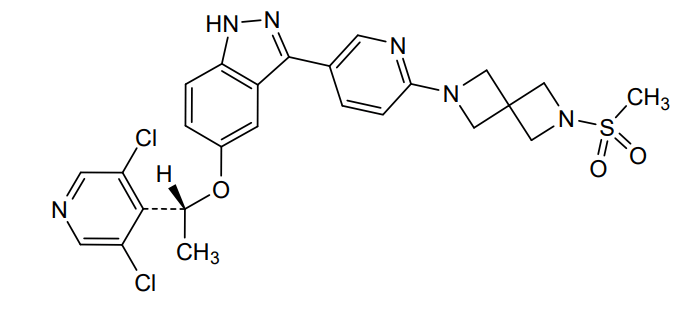
Dabogratinib
CAS 2800223-30-5
MF C25H24Cl2N6O3S, 559.5 g/mol
5-[(1R)-1-(3,5-dichloro-4-pyridinyl)ethoxy]-3-[6-(2-methylsulfonyl-2,6-diazaspiro[3.3]heptan-6-yl)-3-pyridinyl]-1H-indazole
(R)-5-(1-(3,5-Dichloropyridin-4-yl)ethoxy)-3-(6-(6-(methylsulfonyl)-2,6-diazaspiro[3.3]heptan-2-yl)pyridin-3-yl)-1H-indazole
[6-(5-{5-[(1R)-1-(3,5-dichloropyridin-4-yl)ethoxy]-1H-indazol-3-yl}pyridin-2-yl)-2,6-diazaspiro[3.3]heptan-2-yl](methyl)-λ6sulfanedioneTYRA-300
fibroblast growth factor receptor inhibitor, antineoplastic, TYRA-300, TYRA 300, A1AV2, FH245S2JZJ
Dabogratinib (TYRA-300) is an orally active, highly selective inhibitor of fibroblast growth factor receptor 3 (FGFR3), designed to treat cancers with FGFR3 alterations and genetic diseases like achondroplasia. It shows potent tumor growth inhibition in preclinical studies and early phase I/II (SURF301) clinical activity against advanced bladder cancer and metastatic urothelial carcinoma.
Key Aspects of Dabogratinib (TYRA-300)
- Mechanism: It acts as a selective inhibitor of FGFR3 with a high selectivity over other isoforms (FGFR1/2/4), which helps minimize toxicity.
- Target Indications: It is being developed for FGFR3-mutant cancers, including non-muscle invasive bladder cancer (NMIBC) and metastatic urothelial carcinoma, as well as pediatric achondroplasia.
- Preclinical Performance: Studies showed that it reduces tumor growth and drives tumor regression, especially in xenograft models with FGFR3-activating mutations (e.g., S249C).
- Clinical Trials:
- SURF301 (Phase I/II): Ongoing study, Tyra Biosciences reported early efficacy in patients with advanced metastatic urothelial carcinoma (mUC) harboring FGFR3 mutations/fusions.
- SURF302 (Phase II): Evaluating the drug in patients with FGFR3-altered, low-grade, intermediate-risk non–muscle invasive bladder cancer (NMIBC).
- BEACH301 (Phase II): Studying the drug in children with achondroplasia, as it is designed to increase long-bone growth.
- Properties: It is an orally bioavailable molecule with an IC50 of
for FGFR3.
Dabogratinib is an orally bioavailable, selective inhibitor of human fibroblast growth factor receptor 3 (FGFR3), with potential antineoplastic activity. Upon oral administration, dabogratinib specifically targets and binds to certain FGFR3 activating gene alterations, and specifically the gatekeeper mutants V555L/M. This blocks FGFR3-mediated signaling and leads to an inhibition of tumor cell proliferation in FGFR3-overexpressing cells. FGFR3, a receptor tyrosine kinase, is involved in angiogenesis and in the proliferation, differentiation, and survival of tumor cells. FGFR3 expression is associated with poor prognosis. It is overexpressed by certain tumor cell types.
- Efficacy and Safety of TYRA-300 in Participants With FGFR3 Altered Low Grade, Intermediate Risk Non-Muscle Invasive Bladder CancerCTID: NCT06995677Phase: Phase 2Status: RecruitingDate: 2026-04-09
- A Study of TYRA-300 in Children With Achondroplasia: BEACH301CTID: NCT06842355Phase: Phase 2Status: RecruitingDate: 2026-03-06
- Safety and Preliminary Anti-Tumor Activity of TYRA-300 in Advanced Urothelial Carcinoma and Other Solid Tumors With FGFR3 Gene AlterationsCTID: NCT05544552Phase: Phase 1/Phase 2Status: Active, not recruitingDate: 2026-01-12
PAT
Example 46. 5-[(1R)-1-(3,5-dichloro-4-pyridyl)ethoxy]-3-[6-(2-methylsulfonyl-2,6-diazaspiro[3.3]heptan-6-yl)-3-pyridyl]-1H-indazole
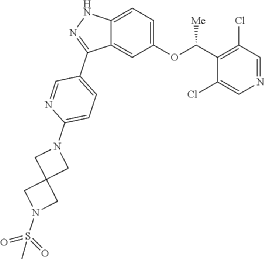
(5-[(1R)-1-(3,5-dichloro-4-pyridyl)ethoxy]-3-[6-(2-methylsulfonyl-2,6-diazaspiro[3.3]heptan-6-yl)-3-pyridyl]-1H-indazole. Triethylamine (20.5 uL, 0.148 mmol, 1.2 equiv) and methylsulfonyl chloride (9.5 uL, 0.123 mmol, 1.0 equiv) were sequentially added at room temperature to a solution of example 45 (59.0 mg, 0.123 mmol, 1 equiv) in anhydrous THE (3 mL). After stirring for 2 hours, the reaction mixture was concentrated under reduced pressure and diluted with saturated brine (30 mL) and dichloromethane (30 mL). The layers were separated. The organic layer was dried over sodium sulfate, filtered and concentrated under reduced pressure on to Celite (1 g). The product was purified on an Interchim automated chromatography system (RediSep Rf Gold HP C18, 15.5 g cartridge), eluting with a gradient of 0 to 100% acetonitrile in water. The fractions containing product were collected and lyophilized to give a white solid (45.0 mg, 65% yield). Analysis: LCMS: m/z=559.2 (M+H); 1H NMR (400 MHz, DMSO-d6) δ 13.02 (br s, 1H), 8.59 (s, 2H), 8.52 (dd, J=0.6, 2.2 Hz, 1H), 7.87 (dd, J=2.4, 8.6 Hz, 1H), 7.46 (d, J=8.9 Hz, 1H), 7.16 (d, J=2.1 Hz, 1H), 7.09 (dd, J=2.3, 9.0 Hz, 1H), 6.54 (dd, J=0.4, 8.6 Hz, 1H), 6.10 (q, J=6.6 Hz, 1H), 4.17 (s, 4H), 4.12 (s, 4H), 3.03 (s, 3H), 1.76 (d, J=6.6 Hz, 3H).
PAT
- Indazole compoundsPublication Number: TW-202241906-APriority Date: 2020-12-30
- Indazole compounds as kinase inhibitorsPublication Number: EP-4271673-A1Priority Date: 2020-12-30
- Indazole compounds as kinase inhibitorsPublication Number: WO-2022147246-A1Priority Date: 2020-12-30
- Indazole compounds as kinase inhibitorsPublication Number: US-12264149-B2Priority Date: 2020-12-30Grant Date: 2025-04-01
- Polymorphic compounds and uses thereofPublication Number: EP-4547670-A1Priority Date: 2022-06-29
- Indazole compounds as kinase inhibitorsPublication Number: US-12071428-B2Priority Date: 2020-12-30Grant Date: 2024-08-27
- Indazole Compounds as Kinase InhibitorsPublication Number: KR-20230152654-APriority Date: 2020-12-30
- Indazole compounds as kinase inhibitorsPublication Number: US-2024109865-A1Priority Date: 2020-12-30
- Indazole compounds as kinase inhibitorsPublication Number: US-2024208941-A1Priority Date: 2020-12-30
- Tyra-300 (5-[(1r)-1-(3,5-dichloro-4-pyridyl)ethoxy]-3-[6-(2-methylsulfonyl-2,6-diazaspiro[3.3]heptan-6-yl)-3-pyridyl]-1h-indazole ) in combination with a pd-1 or pd-l1 antagonist for use in the treatment of cancerPublication Number: WO-2025064744-A1Priority Date: 2023-09-22
- Fgfr inhibitors and methods of use thereofPublication Number: WO-2025061029-A1Priority Date: 2023-09-18
- Polymorphic compounds and uses thereofPublication Number: AU-2023300357-A1Priority Date: 2022-06-29
- Polymorphic compounds and uses thereofPublication Number: WO-2024006883-A1Priority Date: 2022-06-29
- Polymorphic compounds and uses thereofPublication Number: TW-202408493-APriority Date: 2022-06-29
ADVT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
Publication Name: Journal of Medicinal Chemistry
Publication Date: 2024-09-11
PMID: 39258897
DOI: 10.1021/acs.jmedchem.4c01531
////////dabogratinib, anax lab, fibroblast growth factor receptor inhibitor, antineoplastic, TYRA-300, TYRA 300, A1AV2, FH245S2JZJ

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










