Home » Posts tagged 'CU06-EYE'
Tag Archives: CU06-EYE
Rivasterat
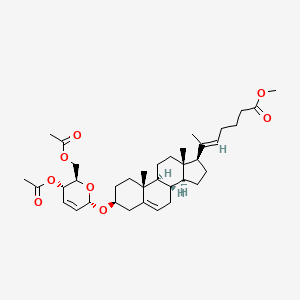
Rivasterat
CAS 2446590-96-9
MF C37H54O8 MW626.8 g/mol
methyl (E)-6-[(3S,8S,9S,10R,13S,14S,17R)-3-[[(2R,3S,6S)-3-acetyloxy-2-(acetyloxymethyl)-3,6-dihydro-2H-pyran-6-yl]oxy]-10,13-dimethyl-2,3,4,7,8,9,11,12,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-17-yl]hept-5-enoate
methyl (20E)-3β-[(4,6-di-O-acetyl-2,3-dideoxy-α-Derythro-hex-2-enopyranosyl)oxy]-27-norcholesta5,20(22)-dien-26-oate
cholesterol-derived steroid, anti-inflammatory, CURACLE, CU-06, CU-06-RE, CU06-1004, CU06-CERE/CV, CU06-EYE, CU06-HAE, CU06-IBD, CU06-ONCO, Sac-1004, 2X23JA5AKW
- OriginatorCURACLE
- ClassAnti-inflammatories; Anti-ischaemics; Antineoplastics; Eye disorder therapies; Ischaemic heart disorder therapies; Small molecules; Vascular disorder therapies
- Mechanism of ActionActin modulators; Chemokine CCL2 inhibitors; Histamine release inhibitors; Interleukin 1 beta inhibitors; Thrombin inhibitors; Vascular endothelial growth factors inhibitors
- Phase IIDiabetic macular oedema
- No development reportedAge-related macular degeneration; Cancer; Crohn’s disease; Diabetic retinopathy; Hereditary angioedema; Lung disorders; Macular degeneration; Myocardial infarction; Retinal oedema; Stroke; Ulcerative colitis; Unstable angina pectoris; Wet macular degeneration
- 28 Aug 2025No recent reports of development identified for research development in Unstable-angina-pectoris in South Korea (PO)
- 28 Jul 2025No recent reports of development identified for phase-I development in Wet macular degeneration in USA (PO)
- 28 May 2025No recent reports of development identified for phase-I development in Age-related-macular-degeneration in South Korea (PO)
Developer and Code Name
- Original code name: CU06-1004
- Developer: Curacle Co., Ltd. (South Korea)
- Drug class: Endothelial dysfunction blocker (EDB)
This class of drugs aims to restore endothelial barrier integrity rather than directly blocking VEGF like most retinal drugs.
The molecule contains:
- Steroid (cyclopenta[a]phenanthrene) core
- Unsaturated heptenoate side chain
- Acetylated sugar moiety (pyranose)
This glycosylated steroid structure is unusual for vascular-protective drugs.
Clinical Development
Phase I (Healthy Volunteers)
Key findings:
- Dose tested: 100–1200 mg
- Exposure increased more than dose proportional
- Food greatly increased absorption
- No significant drug accumulation after repeated dosing
- Minimal renal excretion detected
Phase II
Early clinical trials investigated oral therapy for diabetic macular edema with improvements in:
- Best-corrected visual acuity
- Inflammatory biomarkers
Summary
| Item | Details |
|---|---|
| Drug | Rivasterat (CU06-1004) |
| Originator | Curacle |
| Core patent | WO2013011939 |
| Chemistry | steroid glycoside |
| Key step | steroid glycosylation |
| Priority | ~2011 |
| Expiry | ~2031–2033 |
SYNTHESIS
WO2013011939
US20140148474
EP2741074
KR20130007373
SYN
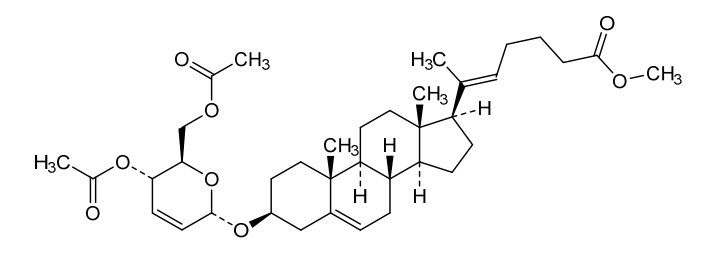
Manufacturing Example 1> Manufacturing of compound 1
[170]Compound 1 represented by the following chemical formula 1 can be manufactured using the manufacturing method described in Korean unpublished patent application number 10-2019-0166864. Specifically, it can be manufactured using the method according to the following reaction scheme 1 or reaction scheme 2.
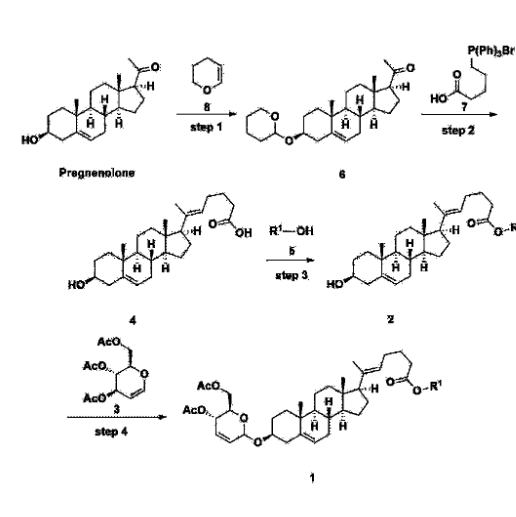
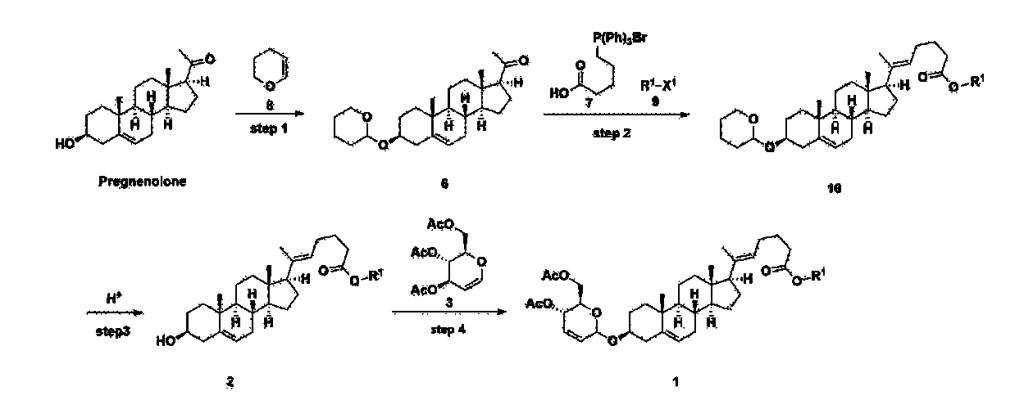
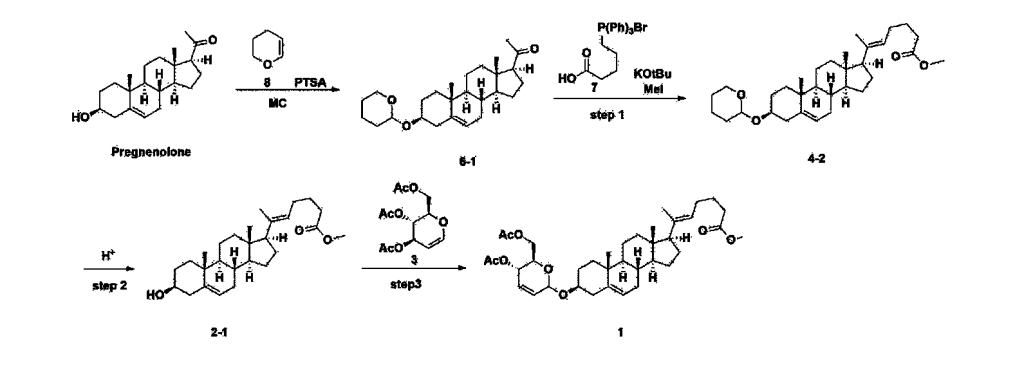
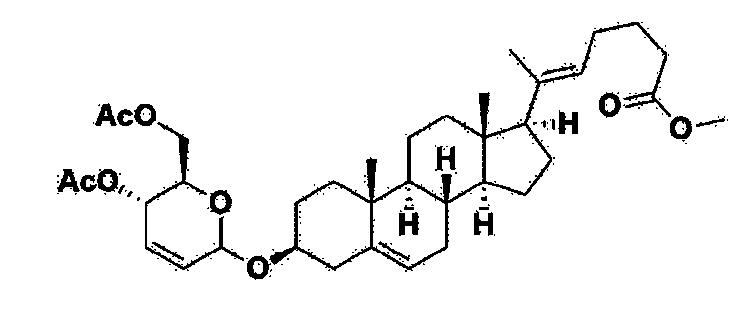
Step 1: Preparation of 6-1
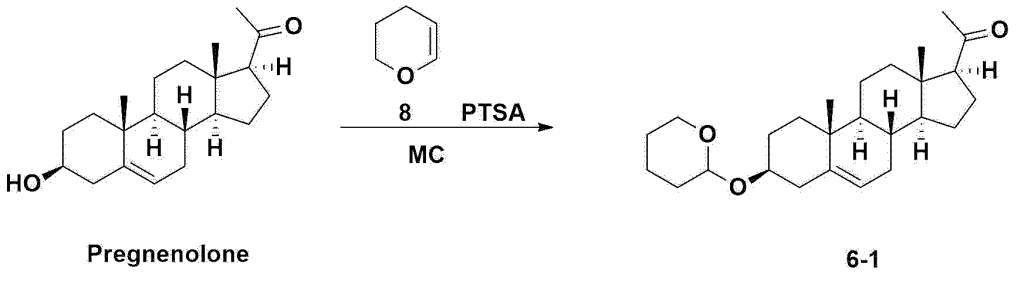
A thermometer was installed in a 5 L flask, and 200 g (0.632 mol) of pregnenolone was added to 2000 mL of dichloromethane, and 173 mL (1.896 mol) of 3,4-dihydro-2H-pyran was added. After lowering the temperature to 0-5 ℃, 3.0 g (15.8 mmol) of p-toluenesulfonic acid monohydrate dissolved in 50 mL of tetrahydrofuran (THF) was added dropwise and stirred at 0 ℃ for 1.5 hours. At 0 ℃, 800 mL of saturated sodium bicarbonate aqueous solution and 10 mL of triethylamine (TEA) were added to the reaction mixture and stirred. After separating the layers, the organic layer was washed with 800 mL of brine, and the aqueous layers were extracted again with 200 mL of dichloromethane, combined into the organic layers, dried over 200 g of anhydrous sodium sulfate, filtered, and distilled under reduced pressure. 1000 mL of MeOH and 5 mL of TEA were added to the obtained residue, heated to completely dissolve, and the temperature was lowered and stirred at -5 °C for 1 hour. The resulting solid was filtered and washed with 200 mL of MeOH to obtain 232.0 g (0.579 mol) of 6-1 (THP-Pregnenolone) as a pure white solid in a yield of 91.6%.
[204]1H-NMR (400 MHz, CDCl 3): δ 5.33-5.36 (m, 1H), 4.71-4.72 (m, 1H), 3.85-3.94 (m, 1H), 3.46-3.56 (m, 2H), 1.00-2.55 (m, 32H), 0.62 (s, 3H).
Step 2: Preparation of 4-2
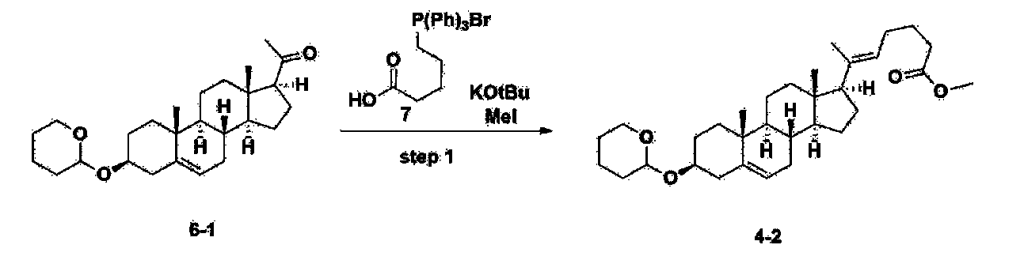
After installing a condenser, heating mantle, and mechanical stirrer in a 5L reactor, the reactor was heated to 119℃ (external temperature), cooled to room temperature while flowing nitrogen for 5 minutes, dried, and 332.5 g (0.75 mol) of 4-(carboxybutyl)triphenylphosphonium bromide and 168.1 g (1.50 mol) of potassium t-butoxide were added. Then, 2000 mL of anhydrous toluene and 750 mL of anhydrous tetrahydrofuran were added, and the reactor was heated to 119℃ (external temperature, internal mild reflux) and stirred for about 2 hours.
[209]6-1 100.0 g (0.250 mol) was dissolved in 500 mL of anhydrous toluene, added to the reaction solution, and reacted for about 20 hours. After the reaction was completed, the reaction mixture was cooled to room temperature, 320 mL (5.14 mol) of methyl iodide and 1000 mL of acetone were added, and stirred at room temperature for 15 hours. Most of the organic solvent was removed from the reaction mixture by distillation under reduced pressure, 1500 mL of ethyl acetate was added to dissolve, and the mixture was washed with 1000 mL of saturated ammonium chloride aqueous solution. The organic layer was washed twice with 1000 mL of water and 1000 mL of brine, dried with 100 g of sodium sulfate, filtered using 80 g of Celite, and concentrated.
[210]The obtained residue was dissolved in 2000 mL of methanol, stirred at 10°C for 13 hours and at 4-5°C for 1 hour, and the resulting solid was filtered, washed with 200 mL of methanol, and dried in vacuum to obtain 66.2 g of 4-2 as a white solid with a yield of 53.2%.
[211]
1H NMR(400MHz, CDCl 3): δ 5.36(t, J=5.80 Hz, 1H), 5.16(t, J=7.00 Hz, 1H), 4.71(m, 1H), 3.93(m, 1H), 3.66(s, 3H), 3.56(m, 2H), 2.37-0.88(m, 38H), 0.54(s, 3H).
Step 3: Preparation of 2-1
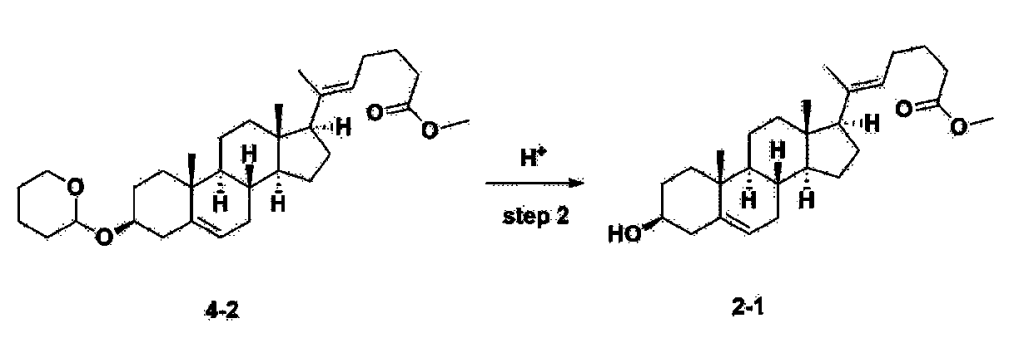
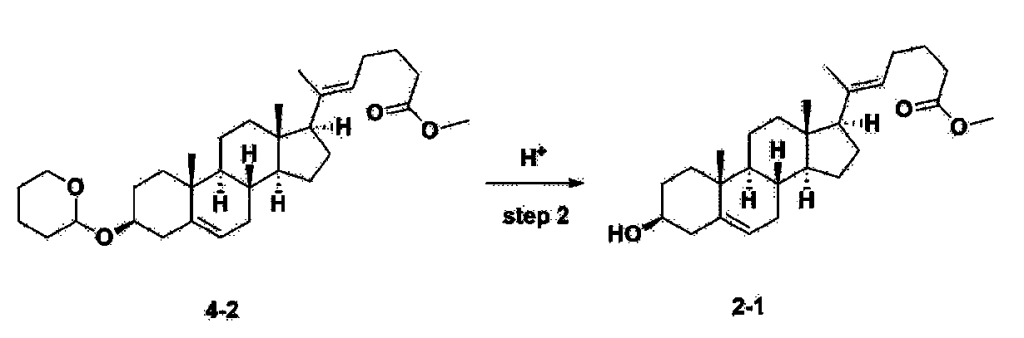
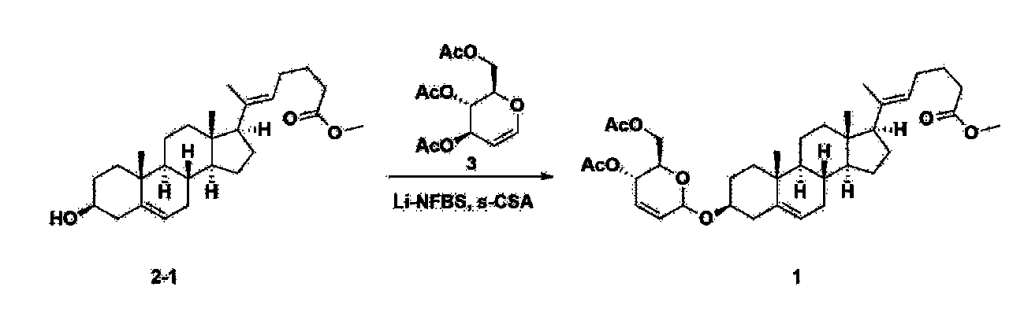
After installing a thermometer and a water bath in a 1 L flask, 42.0 g (0.101 mol) of compound 2-1 and 34.5 g (0.126 mol) of triiO-acetyl D-glucal were dissolved in 126 mL of anhydrous toluene and 252 mL of acetonitrile, and while maintaining the temperature at 30-35 ℃, 3.87 g (0.0130 mol) of lithium nonafluoro-1-butylsulfonate and 0.117 g (0.0005 mol) of (s)-camphor sulfonic acid were added and stirred for 2 hours. After completion of the reaction, the reaction was quenched with 504 mL of saturated sodium bicarbonate aqueous solution and extracted with 630 mL of heptane. The organic layer was washed twice with 504 mL of saturated sodium bicarbonate aqueous solution and then with 504 mL of brine. The organic layer was stirred with 42 g of anhydrous sodium sulfate and 34 g of charcoal, filtered with 34 g of celite, washed with 210 mL of methylene chloride, combined with the filtrate, concentrated, and dried under vacuum.
[223]
1H-NMR (400 MHz, CDCl3) : δ 5.79-5.88 (m, 2H), 5.35-5.36 (m, 1H), 5.27-5.29 (m, 1H), 5.12-5.16 (m, 2H), 4.15-4.24 (m, 3H), 3.66 (s, 3H), 3.54-3.57 (m, 1H), 0.91-2.32 (m, 38H), 0.54 (s, 3H).
PAT
- Crystalline form of vascular leakage blocker compoundPublication Number: US-12103945-B2Priority Date: 2020-05-04Grant Date: 2024-10-01
- New crystalline form of vascular leakage blocker compoundPublication Number: US-2022259256-A1Priority Date: 2020-05-04
- Preparation Method of Vascular Leakage Blockers With a High YieldPublication Number: US-2021395297-A1Priority Date: 2019-12-13
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

////////rivasterat, cholesterol-derived steroid, anti-inflammatory, CURACLE, CU-06, CU-06-RE, CU06-1004, CU06-CERE/CV, CU06-EYE, CU06-HAE, CU06-IBD, CU06-ONCO, Sac-1004, 2X23JA5AKW

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










