Home » Posts tagged 'cardiac myosin inhibitor'
Tag Archives: cardiac myosin inhibitor
Ulacamten
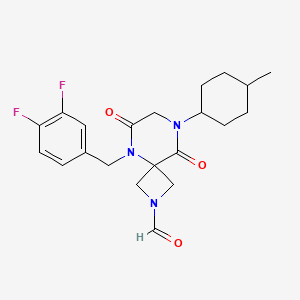
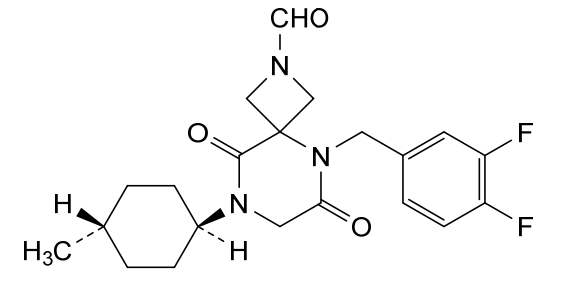
Ulacamten
CAS 2830607-59-3
MF C21H25F2N3O3 MW405.4 g/mol
5-[(3,4-difluorophenyl)methyl]-8-(4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde
5-[(3,4-difluorophenyl)methyl]-8-[(1r,4r)-4-methylcyclohexyl]-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde
cardiac myosin inhibitor, CK-586, CK-4021586, CK 586, CK 4021586, X325G97HZJ
Ulacamten (also known as CK-586 or CK-4021586) is an investigational drug developed by Cytokinetics that acts as a cardiac myosin inhibitor (CMI). It is currently being studied for the treatment of heart failure with preserved ejection fraction (HFpEF), a condition where the heart muscle is too stiff to fill properly
Key Characteristics and Development
- Mechanism of Action: Unlike earlier CMIs like mavacamten or aficamten, ulacamten is highly selective. It binds to the regulatory light chain (RLC) of myosin and only inhibits the “two-headed” form of cardiac myosin, potentially allowing for more precise control over heart muscle contraction.
- Clinical Status: As of March 2026, it is in Phase 2 clinical trials (specifically the AMBER-HFpEF study) to evaluate its safety, tolerability, and optimal dosage in patients with symptomatic HFpEF.
- Administration: It is designed as an orally active small molecule intended for once-daily dosing.
- Potential Benefits: Preclinical studies suggest it can reduce excessive myocardial contractility and improve left ventricular relaxation (diastolic function) without significantly compromising the heart’s overall pumping ability.
- AMBER-HFpEF: Assessment of CK-4021586 in a Multi-Center, Blinded Evaluation of Safety and Tolerability Results in HFpEFCTID: NCT06793371Phase: Phase 2Status: RecruitingDate: 2026-01-12
- A Single and Multiple Ascending Dose Study of CK-4021586 in Healthy Adult ParticipantsCTID: NCT05877053Phase: Phase 1Status: CompletedDate: 2025-05-01
SYN
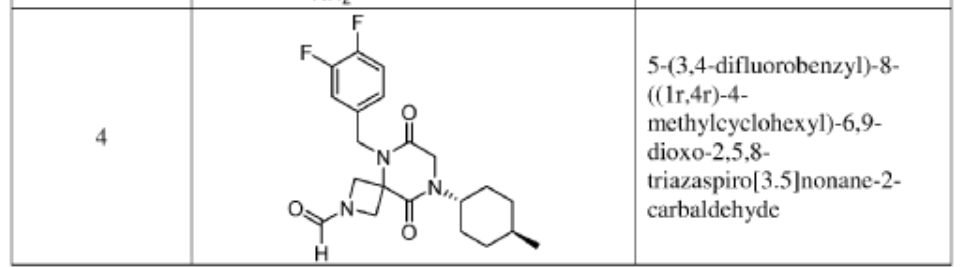
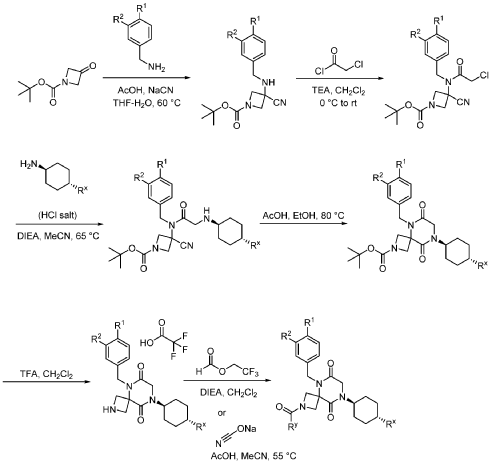
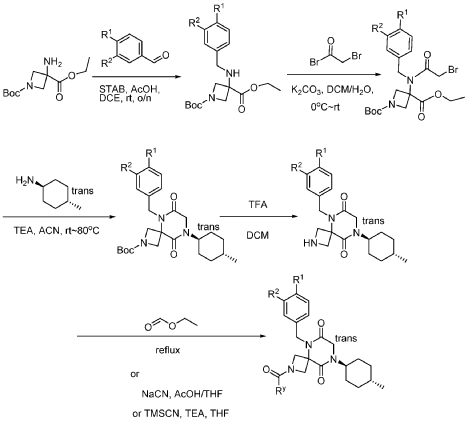
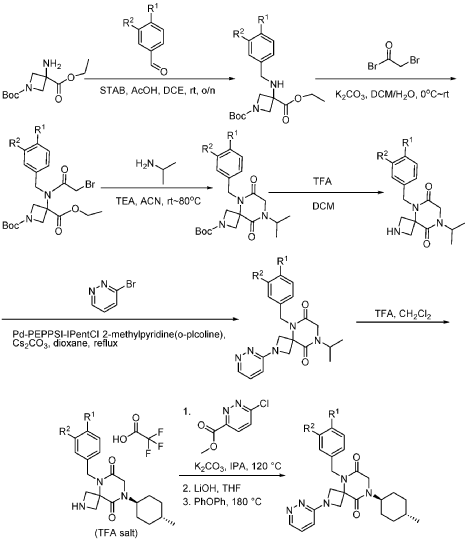
Example 1
Synthesis of 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde
(Compound 4)
Step 1: Synthesis of 1-(tert-butyl) 3-ethyl 3-((3,4-difluorobenzyl)amino)azetidine-1,3-dicarboxylate:
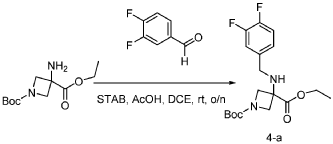
[0147] To a solution of 1-tert-butyl 3-ethyl 3-aminoazetidine-1,3-dicarboxylate (4.0 g, 16.4 mmol, 1.0 equiv) and 3,4-difluorobenzaldehyde (2.4 g, 19.6 mmol, 1.2 equiv) in DCE (40.0 mL) at 0 ˚C were added STAB (7.0 g, 32.8 mmol, 2.0 equiv) and AcOH (2.0 g, 32.8 mmol, 2.0 equiv). The resulting mixture was stirred at rt overnight, adjusted the pH to 8 with ammonium hydroxide, added water (50.0 mL) and extracted with DCM (50.0 mL) twice. The combined organic layers were washed with brine (50 mL) twice, dried over anhydrous Na 2 SO 4 , and concentrated under reduced pressure to afford 6.0 g of 1-tert-butyl 3-ethyl 3-(3,4-difluorobenzyl)amino)azetidine-1,3-dicarboxylate as a yellow oil. LRMS (ES) m/z 315 (M+H-56).
Step 2: Synthesis of 1-(tert-butyl) 3-ethyl 3-(2-bromo-N-(3,4-difluorobenzyl)acetamido)azetidine-1,3-dicarboxylate:
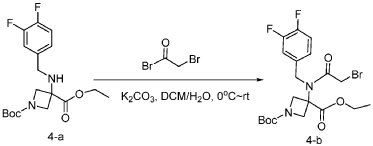
[0148] To a solution of 1-tert-butyl 3-ethyl 3-[[(3,4-difluorophenyl)methyl]amino]azetidine-1,3-dicarboxylate (6.0 g, 16.2 mmol, 1.0 equiv) in DCM (60.0 mL) at 0 ˚C were added a solution of K 2 CO 3 (3.4 g, 24.3 mmol, 1.50 equiv) in water (30 mL), and then bromoacetyl bromide (3.9 g, 19.4 mmol, 1.2 equiv) dropwise over a period of 10 min. The resulting mixture was stirred at rt overnight and extracted with DCM (50.0 mL) twice. The combined organic layers were washed with brine (100 mL) twice, dried over anhydrous Na 2 SO 4 , and concentrated under reduced pressure to afford 8.0 g of 1-(tert-butyl) 3-ethyl 3-(2-bromo-N-(3,4-difluorobenzyl)acetamido)azetidine-1,3-dicarboxylate as a yellow oil. LRMS (ES) m/z 435 (M+H-56).
Step 3: Synthesis of tert-butyl 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carboxylate:
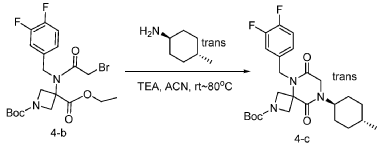
[0149] To a solution of 1-(tert-butyl) 3-ethyl 3-(2-bromo-N-(3,4-difluorobenzyl)acetamido)azetidine-1,3-dicarboxylate (8.0 g, 16.3 mmol, 1.0 equiv) in ACN (80 mL) were added TEA (4.9 g, 48.4 mmol, 3.0 equiv) and trans-(1r,4r)-4-methylcyclohexan-1-amine (2.8 g, 24.7 mmol, 1.5 equiv). The resulting mixture was stirred at rt for 1 h, gradually warmed to 80 ˚C, and stirred at 80 ˚C overnight. The mixture was cooled to rt, concentrated under reduced pressure, and triturated with a mixture of PE and EA (7/1; 80 mL) to afford 7 g (~80% purity) of tert-butyl 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carboxylate as an off-white solid. LRMS (ES) m/z 422 (M+H-56).
Step 4: Synthesis of 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-2,5,8-triazaspiro[3.5]nonane-6,9-dione:
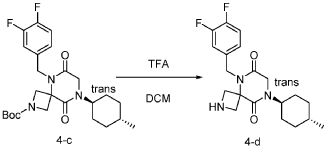
[0150] To a stirred solution of tert-butyl 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carboxylate (7.0 g, 14.7 mmol, 1.0 equiv) in DCM (70.0 mL) was added TFA (18.0 mL). The resulting mixture was stirred at rt for 3h, diluted with water (100.0 mL), adjusted the pH to 13-14 with aqueous NaOH solution (2 N), and extracted with DCM (100 mL) twice. The combined organic layers were washed with brine (100.0 mL) twice, dried over anhydrous Na 2 SO 4 , and concentrated under reduced pressure to afford 4.5 g (~80% purity) of 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-2,5,8-triazaspiro[3.5]nonane-6,9-dione as a yellow semi-solid. LRMS (ES) m/z 378 (M+H).
Step 5: Synthesis of 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde (Compound 4):
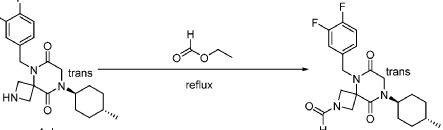
[0151] A solution of tert-butyl 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carboxylate (1.5 g, 4.0 mmol, 1.0 equiv) in ethyl formate (15.0 mL) was stirred at 80
o C overnight. The mixture was cooled to rt, concentrated under reduced pressure, and purified by C18 column chromatography, eluted with a mixture of water (0.05% NH
4 HCO
3 )/CH
3 CN (3:2) to afford 1.3 g (81%) of 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde as an amorphous white solid. An experimental X-ray powder diffraction (XRPD) pattern of this amorphous white solid is shown in FIG. 1 LRMS (ES) m/z 406 (M+H);
1 H NMR (300 MHz, DMSO-d6) δ 7.96 (s, 1H), 7.47 – 7.29 (m, 2H), 7.10 (ddd, J = 9.4, 4.4, 2.0 Hz, 1H), 4.82 (s, 2H), 4.50 (d, J = 9.6 Hz, 1H), 4.15-4.28 (m, J = 3H), 4.01 (s, 2H), 3.96 (d, J = 10.8 Hz, 1H), 1.80 – 1.69 (m, 2H), 1.65 – 1.48 (m, 4H), 1.35 (d, J = 10.9 Hz, 1H), 1.13 – 0.93 (m, 2H), 0.88 (d, J = 6.5 Hz, 3H).
PAT
5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde, also called compound 1, having the structure shown below,
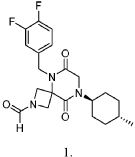
Example 1
Synthesis of 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde (Compound 1)
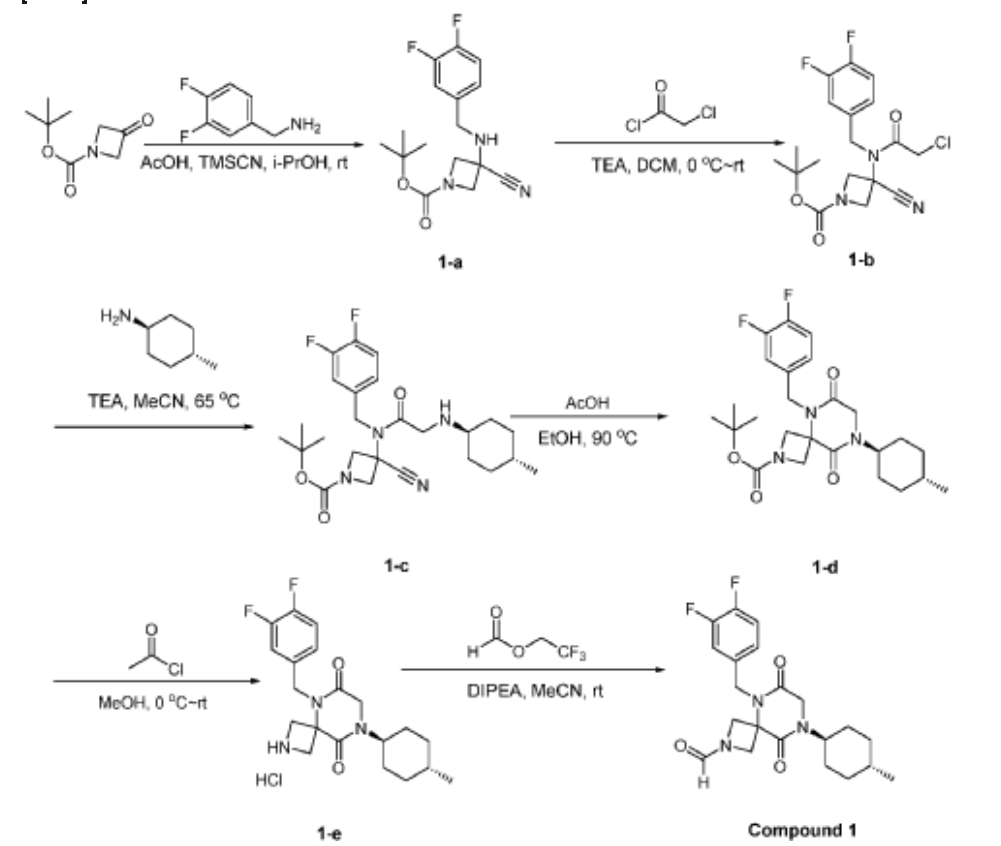
Step 6: Synthesis of 5-[(3,4-difluorophenyl)methyl]-6,9-dioxo-8-[(1r,4r)-4-methylcyclohexyl]-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde (Compound 1):
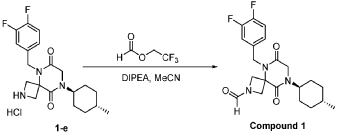
[0165] To a solution of 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde (4.0 kg, 10.60 mol, 1 equiv) in MeCN (20 L) at r.t. were added 2,2,2-trifluoroethyl formate (1.63 kg, 12.72 mol, 1.2 equiv) and DIPEA (3.42 kg, 26.50 mol, 2.5 equiv) . The resulting mixture was stirred overnight at rt. The reaction was monitored by LCMS. The resulting mixture was concentrated under reduced pressure. The resulting mixture was diluted with EtOAc (10 L). The resulting mixture was quenched with NH 4Cl (6 L, sat.) and water (6 L), extracted with EtOAc (3×15 L). The combined organic layers were washed with NH 4Cl (aq.) (10 L) and brine (10 L), dried over anhydrous Na 2SO 4, concentrated under reduced pressure to give a crude brown oil, the crude oil was re-crystallized from cyclohexane and EtOAc (5:1, 4L, 80 °C to r.t.), filtered to afford 3 kg (1 st batch) 5-[(3,4-difluorophenyl)methyl]-6,9-dioxo-8-[(1r,4r)-4-methylcyclohexyl]-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde light yellow solid. The filtrate was concentrated under reduced pressure, re-crystallized with petroleum ether and EtOAc (10:1, 3 L, rt) to afford 800 g (2 nd batch) of light yellow solid. Two batches were combined, dried to afford 3.8 kg of Form I (m.p. at 133 °C) 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde light yellow solid. The overall yield of this step is 97%.
PAT
- Cardiac sarcomere inhibitorsPublication Number: US-12286437-B2Priority Date: 2021-03-04Grant Date: 2025-04-29
- Cardiac sarcomere inhibitorsPublication Number: US-11919909-B2Priority Date: 2021-03-04Grant Date: 2024-03-05
- Cardiac sarcomere inhibitorsPublication Number: US-2024309011-A1Priority Date: 2021-03-04
- Cardiac sarcomere inhibitorsPublication Number: WO-2022187501-A1Priority Date: 2021-03-04
- Cardiac sarcomere inhibitorsPublication Number: US-2022306642-A1Priority Date: 2021-03-04
- cardiac sarcomere inhibitorPublication Number: CN-117083275-APriority Date: 2021-03-04
- Cardiac sarcomere inhibitorsPublication Number: EP-4301760-A1Priority Date: 2021-03-04
- Crystalline forms of 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehydePublication Number: US-2024132498-A1Priority Date: 2022-09-02
- Crystalline forms of 5- (3, 4-difluorobenzyl) -8- ((1 r,4 r) -4-methylcyclohexyl) -6, 9-dioxo-2, 5, 8-triazaspiro [3.5] nonane-2-carbaldehydePublication Number: CN-119998292-APriority Date: 2022-09-02
- Crystalline forms of 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehydePublication Number: WO-2024050539-A1Priority Date: 2022-09-02
- Crystalline forms of 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehydePublication Number: EP-4581034-A1Priority Date: 2022-09-02
- Cardiac sarcomere inhibitorsPublication Number: TW-202302594-APriority Date: 2021-03-04
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

REF
Publication Name: Scientific Reports
Publication Date: 2024-05-27
PMCID: PMC11130313
PMID: 38802475
DOI: 10.1038/s41598-024-62840-3
///////////////ulacamten, ANAX, cardiac myosin inhibitor, CK-586, CK-4021586, CK 586, CK 4021586, X325G97HZJ

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










