Home » Posts tagged 'becondogrel'
Tag Archives: becondogrel
Becondogrel
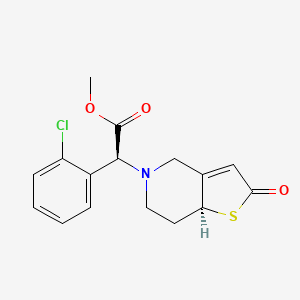
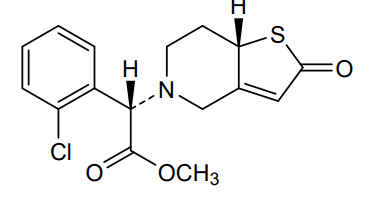
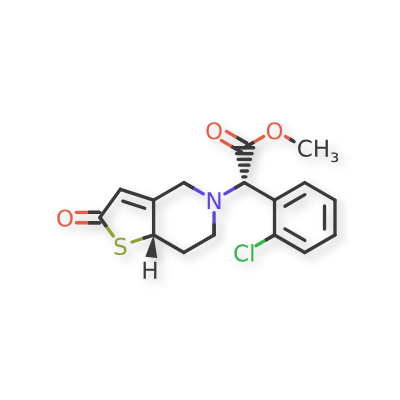
Becondogrel
CAS 1416696-44-0
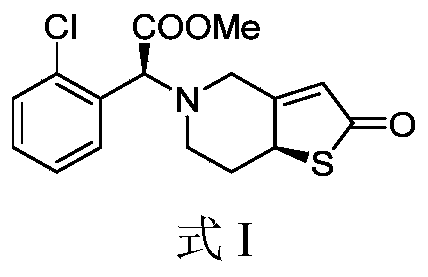
MF C16H16ClNO3S, MW 337.821
2-OXO-CLOPIDOGREL, (.ALPHA.S,7AS)-
(4S)-2-Oxoclopidogrel
Methyl (S)-(2-chlorophenyl)[(7aS)-2-oxo-2,6,7,7atetrahydrothieno[ 3,2-c]pyridin-5(4H)-yl]acetate
methyl (S)-(2-chlorophenyl)[(7aS)-2-oxo-2,6,7,7atetrahydrothieno[3,2-c]pyridin-5(4H)-yl]acetate
platelet aggregation inhibitor, D7X6820P1N, Copidogrel oxide, (4S)-2-Oxoclopidogrel
Becondogrel is an antiplatelet medication and an irreversible P2Y12 receptor antagonist. It is chemically known as 2-oxoclopidogrel, which is a direct metabolic intermediate of the widely used drug clopidogrel (Plavix).
Key Characteristics
- Mechanism of Action: It prevents blood cells (platelets) from sticking together, which helps inhibit the formation of blood clots (thrombosis).
- Relationship to Clopidogrel: Standard clopidogrel is a prodrug that requires two metabolic steps in the liver to become active. Becondogrel is designed to bypass the first of these steps, potentially reducing the individual variability in effectiveness seen with clopidogrel due to genetic differences in liver enzymes (CYP450).
- Clinical Status: As of early 2025, becondogrel was included in the World Health Organization’s (WHO) proposed International Nonproprietary Name (INN) list, indicating its development for medical use
SYN
- [EP3290423B1]
- https://patentscope.wipo.int/search/en/detail.jsf?docId=EP213389219&_cid=P22-MNPFAY-18756-1
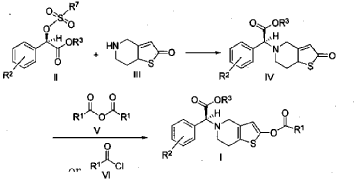
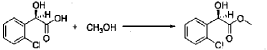
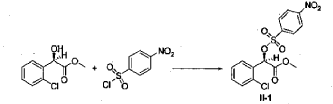
Example 3
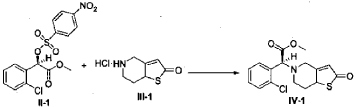
(2S)-Methyl 2-(2-oxo-7,7a-dihydrothieno[3,2-c]pyridin-5(2H,4H,6H)-yl)-2-(2-chlorophenyl)-acetate (IV-1)
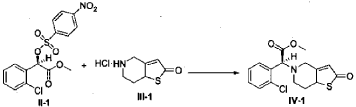
[0042] 58.1 g (0.15 mol) of (R)-methyl 2-(2-chlorophenyl)-2-(4-nitrophenylsulfonyloxy)-acetate (II-1), 32.3 g (0.17 mol) of 5,6,7,7a-tetrahydrothieno[3,2-c]pyridin-2(4H)-one hydrochloride (III-1), and 37.8g (0.38 mol) of potassium bicarbonate were added to 500 ml of acetonitrile. The reaction was stirred under a nitrogen atmosphere at room temperature for 26 hrs. The reaction solution was allowed to stand and the insoluble material was filtered off, to obtain a dark red mother liquor. The solvent was evaporated under reduced pressure, and 35.4 g of an oil product was obtained after purification by flash column chromatography (petroleum ether:ethyl acetate = 4:1). Yield 70%. Recrystalization from ethanol afforded 18.1 g of a pure product (IV-1) as a white solid. mp: 146-148°C, ee = 97.5%, [α] D 19 = +114.0° (c 0.5, MeOH); 1H-NMR (300 MHz, CDCl 3) δ 1.79-1.93 (m, 1 H), 2.30-2.40 (m, 1 H), 2.56-2.70 (m, 1 H), 3.00-3.27 (m, 2 H), 3.72 (s, 3 H), 3.79-3.93 (m, 1 H), 4.12-4.19 (m, 1 H), 4.89 (d, 1 H, J= 5.6 Hz), 6.00 (d, 1 H, J = 5.2 Hz), 7.26-7.50 (m, 4 H); 13C-NMR (75 MHz, CDCl 3) δ 33.9, 34.0, 49.0, 49.7, 51.1, 51.6, 52.2, 52.4, 67.3, 76.6, 77.0, 77.4, 126.6, 126.8, 127.2, 129.8, 130.1, 132.7, 134.8, 167.2, 167.4, 170.8, 198.6; ESI-MS m/ z 338.1 [M+H] +; HRMS Calcd for C 16H 17NO 3SCl [M+H] + m/ z 338.0618, found 338.0626.
SYN
- [US2022028976]
- https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2023204830&_cid=P22-MNPFG3-21704-1
PAT
. The chemical name of the compound with the Equation I structure is: (S)-2-(2-chlorophenyl)-2-((S)-2-oxo-2,6,7,7a-tetrahydrothiophene[3,2-c]and pyridine-5(4H))yl)methyl acetate.



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Synthesis of Biologically Active Piperidine Metabolites of Clopidogrel: Determination of Structure and Analyte Development.Publication Name: The Journal of organic chemistryPublication Date: 2015-07-07PMID: 26151079DOI: 10.1021/acs.joc.5b00632
- Antiplatelet Agents Aspirin and Clopidogrel Are Hydrolyzed by Distinct Carboxylesterases, and Clopidogrel Is Transesterificated in the Presence of Ethyl AlcoholPublication Name: The Journal of Pharmacology and Experimental TherapeuticsPublication Date: 2006-12PMID: 16943252DOI: 10.1124/jpet.106.110577
- Contribution of Hepatic Cytochrome P450 3A4 Metabolic Activity to the Phenomenon of Clopidogrel ResistancePublication Name: CirculationPublication Date: 2004-01-20PMID: 14707025DOI: 10.1161/01.cir.0000112378.09325.f9
PAT
- Solid preparation containing oxidized clopidogrel and preparation method thereforPublication Number: EP-4628079-A1Priority Date: 2022-12-02
- Method for treating blood plasma sample containing clopidogrel oxide, and measurement methodPublication Number: EP-4394372-A1Priority Date: 2021-08-23
- Crystalline form b of tetrahydrothienopyridine compound, preparation method, composition and application thereofPublication Number: US-2022289761-A1Priority Date: 2019-05-17
- B crystal form of tetrahydrothienopyridine compound, preparation method therefor, composition and applicationPublication Number: WO-2020233226-A1Priority Date: 2019-05-17
- B crystal form of tetrahydrothienopyridine compound, preparation method therefor, composition and applicationPublication Number: EP-3985009-A1Priority Date: 2019-05-17
- Optically active 2-hydroxy tetrahydrothienopyridine derivatives, preparation method and use in manufacture of medicament thereof
- Publication Number: EP-3290423-B1
- Priority Date: 2010-02-02
- Grant Date: 2021-07-21
//////////becondogrel, platelet aggregation inhibitor, D7X6820P1N, Copidogrel oxide, (4S)-2-Oxoclopidogrel

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










