Home » Posts tagged 'ARRY 440'
Tag Archives: ARRY 440
Claturafenib
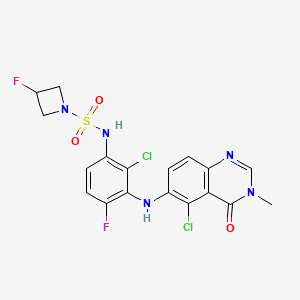
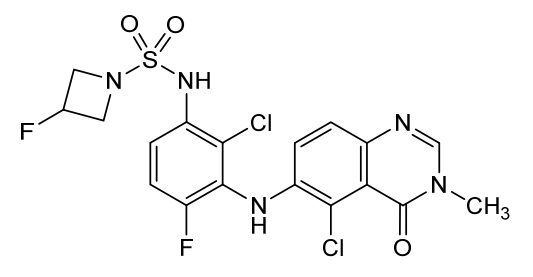
Claturafenib
CAS 2754408-94-9
MF C18H15Cl2F2N5O3S MW490.3 g/mol
N-[2-chloro-3-[(5-chloro-3-methyl-4-oxoquinazolin-6-yl)amino]-4-fluorophenyl]-3-fluoroazetidine-1-sulfonamide
N-{2-chloro-3-[(5-chloro-3-methyl-4-oxo-3,4-dihydroquinazolin-6-yl)amino]-4-fluorophenyl}-3-fluoroazetidine-1-sulfonamide
B-Raf (BRAF) inhibitor, antineoplastic, PF-07799933, PF 07799933, ARRY440, ARRY 440, PC35M52J8T
Claturafenib (development code PF-07799933) is an investigational cancer drug currently being developed by Pfizer. It is a selective, orally active pan-mutant BRAF inhibitor designed to treat advanced solid tumours with specific genetic alterations
Mechanism of Action
Claturafenib belongs to a class of drugs that target the MAPK/ERK signaling pathway, which is often hijacked by cancer cells to promote uncontrolled growth.
- Pan-Mutant Inhibition: Unlike first-generation BRAF inhibitors, claturafenib inhibits multiple classes of BRAF mutations, including Class 1 (V600), Class 2, and Class 3 alterations.
- Brain-Penetrant: It is designed to cross the blood-brain barrier, allowing it to potentially treat brain metastases or primary brain tumours.
- Dimer Disruption: It works by disrupting the formation of BRAF-containing dimers, which are responsible for signaling in many resistant or non-V600 mutant cancers.
- Selectivity: It is highly selective for mutant BRAF, significantly sparing normal (wild-type) cells to reduce off-target side effects.
🏥 Clinical Status
As of April 2026, claturafenib is in Phase 1 clinical trials.
- Target Indications: Advanced solid malignancies, including melanoma, colorectal cancer (CRC), and non-small cell lung cancer (NSCLC).
- Combination Therapy: It is being studied both as a single agent (monotherapy) and in combination with other drugs like binimetinib (a MEK inhibitor) or cetuximab (an EGFR inhibitor).
- Ongoing Study: Clinical trial NCT05355701 is currently evaluating its safety, dosage, and efficacy in patients whose disease has progressed on other treatments.
- A Study to Learn About the Study Medicine Called PF-07799933 in People With Advanced Solid Tumors With BRAF Alterations.CTID: NCT05355701Phase: Phase 1Status: RecruitingDate: 2026-03-27
- A Study to Learn About the Study Medicine Called PF-07799544 as Monotherapy or in Combination in People With Advanced Solid TumorsCTID: NCT05538130Phase: Phase 1Status: RecruitingDate: 2026-03-27
Claturafenib is an orally bioavailable class 1 and 2 inhibitor of the serine/threonine-protein kinase B-raf (BRAF) protein, with potential antineoplastic activity. Upon oral administration, claturafenib selectively binds to and inhibits the activity of class 1 and 2 BRAF alterations. This inhibits the proliferation of tumor cells which express these BRAF alterations. BRAF, a member of the raf family of serine/threonine protein kinases, plays a role in the regulation of mitogen-activated protein kinase (MAPK) and extracellular signal-regulated kinase (ERK) signaling pathways, which may be constitutively activated due to BRAF gene mutations. Mutated forms and fusions of BRAF are associated with a number of neoplastic diseases.
| Property | Value |
|---|---|
| Molecular Formula | |
| Molecular Weight | 490.31 g/mol |
| CAS Number | 2754408-94-9 |
| Other Names | ARRY-440, PF07799933 |
📍 Note: Claturafenib is an investigational compound and has not yet been approved by the FDA or other regulatory agencies for general use
SYN
SYN
Example 126
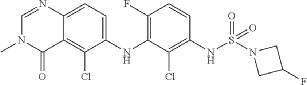
N-(2-chloro-3-((5-chloro-3-methyl-4-oxo-3,4-dihydroquinazolin-6-yl)amino)-4-fluorophenyl)-3-fluoroazetidine-1-sulfonamide
PAT
- 4-oxo-3,4-dihydroquinazolinon compounds for the treatment of braf-associated diseases and disordersPublication Number: CA-3186343-A1Priority Date: 2020-06-09
- Compounds for the treatment of BRAF-associated diseases and disordersPublication Number: US-12303509-B2Priority Date: 2020-06-09Grant Date: 2025-05-20
- 3,4-dihydro-2,7-naphthyridine-1,6(2H,7H)-dione compound as MEK inhibitorPublication Number: CN-117561255-APriority Date: 2021-03-31
- 4-oxo-3,4-dihydroquinazolinone compounds for the treatment of BRAF-related diseases and disordersPublication Number: KR-20230019944-APriority Date: 2020-06-09
- Compounds for the treatment of braf-associated diseases and disordersPublication Number: US-2022288074-A1Priority Date: 2020-06-09
- 4-oxo-3, 4-dihydroquinazolinone compounds for the treatment of BRAF related diseases and disordersPublication Number: CN-116096710-APriority Date: 2020-06-09
- 4-oxo-3,4-dihydroquinazolinon compounds for the treatment of braf-associated diseases and disordersPublication Number: EP-4161907-A1Priority Date: 2020-06-09
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
////////claturafenib, ANAX, B-Raf (BRAF) inhibitor, antineoplastic, PF-07799933, PF 07799933, ARRY440, ARRY 440, PC35M52J8T

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










