Home » Posts tagged 'ANTIDEPRESSANT'
Tag Archives: ANTIDEPRESSANT
Maritupirdine
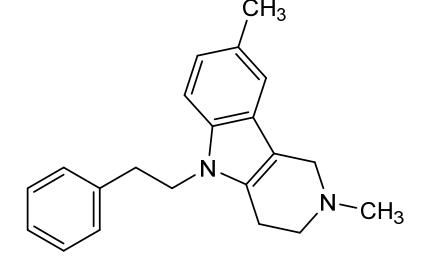
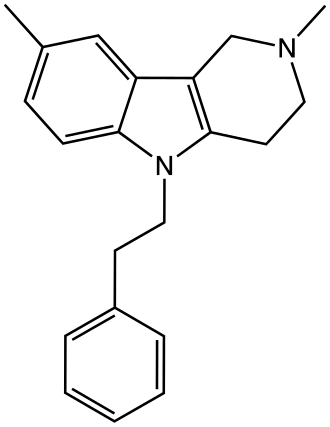
Maritupirdine
CAS 1025725-91-0
MF C21H24N2 MW304.4 g/mol
2,3,4,5-TETRAHYDRO-2,8-DIMETHYL-5-PHENETHYL-1H-PYRIDO(4,3-B)INDOLE
2,8-dimethyl-5-(2-phenylethyl)-2,3,4,5-tetrahydro-1H-pyrido[4,3-b]indole
serotonin receptor antagonist, antidepressant, AVN-101, AVN 101, CD 008 0045, 6MHR5IV28S
- OriginatorChemDiv
- DeveloperAllaChem; Avineuro Pharmaceuticals; ChemDiv
- ClassAntidementias; Indoles; Neuroprotectants; Pyridines; Small molecules
- Mechanism of ActionAdrenergic receptor antagonists; Dopamine receptor antagonists; Histamine receptor antagonists; Serotonin 6 receptor antagonists
- Phase IIAlzheimer’s disease; Anxiety disorders
- 27 Sep 2022No development reported – Phase-II for Anxiety disorders in Russia (PO)
- 22 Dec 2020Chemical structure information added
- 22 Oct 2020Avineuro Pharmaceuticals and Avineuro Pharmaceuticals plans a phase III trial in Anxiety disorders in December 2020 (PO, Capsules) (NCT04598867)
MARITUPIRDINE is a small molecule drug with a maximum clinical trial phase of II and has 1 investigational indication.
Maritupirdine (developmental code name AVN-101), a close structural analogue of latrepirdine, is a selective 5-HT6 receptor antagonist which is under development by Avineuro Pharmaceuticals for the treatment of Alzheimer’s disease and anxiety disorders.[1][2][3] As of November 2013, it was in phase II clinical trials for these indications.[2][3][4][needs update]
It was approved in Russia on May 31, 2023 under the brand name Aviandr for the treatment of generalized anxiety disorder, mild-to-moderate anxiety conditions (stress reactions and adjustment disorders) and anxiety after COVID-19.[5]
SYN
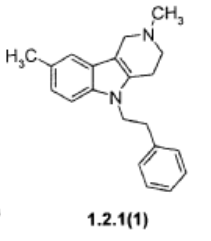
PAT
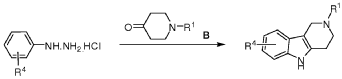
REF
- Alzheimer’s disease: Updated multi-targets therapeutics are in clinical and in progressPublication Name: European Journal of Medicinal ChemistryPublication Date: 2022-08-05PMID: 35635955DOI: 10.1016/j.ejmech.2022.114464
- Structure–Activity Relationships and Therapeutic Potentials of 5-HT7Receptor Ligands: An UpdatePublication Name: Journal of Medicinal ChemistryPublication Date: 2018-05-16PMID: 29767995DOI: 10.1021/acs.jmedchem.7b01898
- Serotonin 5-HT6Receptor Antagonists for the Treatment of Cognitive Deficiency in Alzheimer’s DiseasePublication Name: Journal of Medicinal ChemistryPublication Date: 2014-06-03PMID: 24850589DOI: 10.1021/jm5003952
- Synthesis and biological activity of 5-styryl and 5-phenethyl-substituted 2,3,4,5-tetrahydro-1H-pyrido[4,3-b]indolesPublication Name: Bioorganic & Medicinal Chemistry LettersPublication Date: 2010-01-01PMID: 19945877DOI: 10.1016/j.bmcl.2009.11.037
PAT
- Ligands of 5-ht6 receptors, a pharmaceutical composition, method for the production and use thereofPublication Number: EP-2184064-A2Priority Date: 2006-11-16
- Ligands of 5-ht6 receptors, a pharmaceutical composition, method for the production and use thereofPublication Number: US-2011046368-A1Priority Date: 2006-11-16
- Substituted 2,3,4,5-tetrahydro-1h-pyrido[4,3-b]indoles, methods for the production and use thereofPublication Number: AU-2008236872-A1Priority Date: 2007-04-05
- Substituted 2,3,4,5-tetrahydro-1h-pyrido[4,3-b]indoles, methods for the production and use thereofPublication Number: WO-2008123796-A2Priority Date: 2007-04-05
- Substituted 2,3,4,5-Tetrahyrdo-1H-Pyrido[4,3-B]Indoles, Methods for the Production and Use ThereofPublication Number: US-2010120792-A1Priority Date: 2007-04-05
- Sustituted 2,3,4,5-tetrahydro-1h-pyrido[4,3-b]indoles, methods for use thereofPublication Number: US-2013217703-A1Priority Date: 2007-04-05
- Ligands of 5-ht6 receptors, a pharmaceutical composition, method for the production and use thereofPublication Number: WO-2008060190-A2Priority Date: 2006-11-16



AS ON OCT2025 4.511 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Ivachtchenko V, Ivanenkov Y (2013). “Small Molecule 5-HT6R Ligands: A Comprehensive Insight into their Selectivity and Activity”. Current Bioactive Compounds. 9 (1): 64–100. doi:10.2174/1573407211309010007. ISSN 1573-4072.
- Griebel G, Holmes A (September 2013). “50 years of hurdles and hope in anxiolytic drug discovery”. Nature Reviews. Drug Discovery. 12 (9): 667–87. doi:10.1038/nrd4075. PMC 4176700. PMID 23989795.
- Benhamú B, Martín-Fontecha M, Vázquez-Villa H, Pardo L, López-Rodríguez ML (September 2014). “Serotonin 5-HT6 receptor antagonists for the treatment of cognitive deficiency in Alzheimer’s disease”. Journal of Medicinal Chemistry. 57 (17): 7160–81. doi:10.1021/jm5003952. PMID 24850589.
- “AVN 101”. AdisInsight. Retrieved 2015-06-10.
- “Aviandr (2,8-dimethyl-5-(2-phenylethyl)-2,3,4,5-tetrahydro-1H-pyrido[4,3-b]indole hydrochloride) film-coated tablets. Full prescribing information”. Russian State Register of Medicines (in Russian). Avineuro Pharmaceuticals, Inc.
External links
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 1025725-91-0 1061354-48-0 (hydrochloride) |
| ChemSpider | 24643855 |
| UNII | 6MHR5IV28S |
| ChEMBL | ChEMBL592752 |
| CompTox Dashboard (EPA) | DTXSID901032403 |
| Chemical and physical data | |
| Formula | C21H24N2 |
| Molar mass | 304.437 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
////////////maritupirdine, serotonin receptor antagonist, antidepressant, AVN-101, AVN 101, CD 008 0045, 6MHR5IV28S
Melitracen

Melitracen
- Molecular FormulaC21H25N
- Average mass291.430 Da
10563-70-9[RN]
1568
1-Propanamine, 3-(10,10-dimethyl-9(10H)-anthracenylidene)-N,N-dimethyl-
225-858-5[EINECS], 234-150-5[EINECS]
3-(10,10-Dimethyl-9(10H)-anthracenyliden)-N,N-dimethyl-1-propanamine
Q7T0Y1109Z
Thymeol
мелитрацен[Russian][INN]
ميليتراسان[Arabic][INN]
美利曲辛[Chinese][INN]
Melitracen
CAS Registry Number: 5118-29-6
CAS Name: 3-(10,10-Dimethyl-9(10H)-anthracenylidene)-N,N-dimethyl-1-propanamine
Additional Names:N,N,10,10-tetramethyl-D9(10H),g-anthracenepropylamine; 9,10-dihydro-10,10-dimethyl-9-(3-dimethylaminopropylidene)anthracene; 9-[3-(dimethylamino)propylidene]-10,10-dimethyl-9,10-dihydroanthracene; N,N-dimethyl-3-(10,10-dimethyl-9(10H)-anthrylidene)propylamine
Molecular Formula: C21H25N, Molecular Weight: 291.43
Percent Composition: C 86.55%, H 8.65%, N 4.81%
Literature References: Prepn of the hydrochloride: Holm, Acta Chem. Scand.17, 2437 (1963); idem,GB939856 corresp to US3177209 (1963, 1965, both to Kefalas A/S). Crystal structure: J. Lopez de Lerma et al.,Acta Crystallogr.B35, 1739 (1979). Toxicity data: P. V. Petersen et al.,Acta Pharmacol. Toxicol.24, 121 (1966).
Derivative Type: Hydrochloride
CAS Registry Number: 10563-70-9
Manufacturers’ Codes: U-24973A
Trademarks: Melixeran (Lusofarmaco); Trausabun (Promonta); Dixeran (Lundbeck)
Molecular Formula: C21H25N.HCl, Molecular Weight: 327.89
Percent Composition: C 76.92%, H 7.99%, N 4.27%, Cl 10.81%
Properties: Crystals from acetone, mp 245-248°. LD50 i.v. in mice: 52 mg/kg (Petersen).
Melting point: mp 245-248°
Toxicity data: LD50 i.v. in mice: 52 mg/kg (Petersen)
Therap-Cat: Antidepressant.
Keywords: Antidepressant; Tricyclics.
Melitracen (brand names Melixeran) is a tricyclic antidepressant (TCA), for the treatment of depression and anxiety.[1][2][3][4] In addition to single drug preparations, it is also available as Deanxit, marketed by Lundbeck, a combination product containing both melitracen and flupentixol.[5][6][7][8]
The pharmacology of melitracen has not been properly investigated and is largely unknown, but it is likely to act in a similar manner to other TCAs. Indeed, melitracen is reported to have imipramine and amitriptyline-like effects and efficacy against depression and anxiety, though with improved tolerability and a somewhat faster onset of action.[9][10]
- ATC:N06AA14
- MW:291.44 g/mol
- CAS-RN:5118-29-6
- InChI Key:GWWLWDURRGNSRS-UHFFFAOYSA-N
- InChI:InChI=1S/C21H25N/c1-21(2)19-13-7-5-10-17(19)16(12-9-15-22(3)4)18-11-6-8-14-20(18)21/h5-8,10-14H,9,15H2,1-4H3
- EINECS:225-858-5
- LD50:52 mg/kg (M, i.v.); 315 mg/kg (M, p.o.);
170 mg/kg (R, p.o.)
Derivatives
hydrochloride
- Formula:C21H25N • HCl
- MW:327.90 g/mol
- CAS-RN:10563-70-9
- EINECS:234-150-5
- LD50:52 mg/kg (M, i.v.); 315 mg/kg (M, p.o.);
170 mg/kg (R, p.o.)
| CAS-RN | Formula | Chemical Name | CAS Index Name |
|---|---|---|---|
| 90-44-8 | C14H10O | anthrone | 9(10H)-Anthracenone |
| 85118-29-2 | C21H27NO | 9-[3-(dimethylamino)propyl]-9,10-dihydro-10,10-dimethyl-9-anthracenol | 9-Anthracenol, 9-[3-(dimethylamino)propyl]-9,10-dihydro-10,10-dimethyl- |
| 19070-16-7 | C5H12ClMgN | 3-dimethylaminopropylmagnesium chloride | Magnesium, chloro[3-(dimethylamino)propyl]- |
| 5447-86-9 | C16H14O | 10,10-dimethylanthrone | 9(10H)-Anthracenone, 10,10-dimethyl- |
SYN

English: DOI number: 10.3891/acta.chem.scand.17-2437 GB 939856 corresp to US 3177209 (1963, 1965, both to Kefalas A/S).
SYN
https://pubs.rsc.org/en/content/articlehtml/2020/re/d0re00087f
| Fig. 10 Synthesis of melitracen HCl-(36) by Kiil and co-workers making use of a one-flow system. Adapted with permission from Org. Process Res. Dev., 2018, 22, 228–235. Copyright 2018 American Chemical Society.35 |
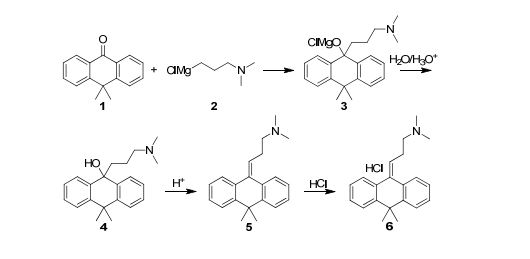
Grignard reactions are commonly used for the construction of carbon–carbon bonds and show exothermic behaviour which can be dangerous in large-scale batch processes. The use of Grignard reagents in flow can be beneficial because of the high control of reaction conditions, facile heat transport and small effective reaction volume.6,34 A recent example was published by Kiil and co-workers, who synthesised melitracen (36) in a one-flow system.35 Kiil hypothesised that the seven unit operations required in batch could be decreased by combining a hydrolysis and dehydration step, and removing a phase separation (Fig. 10).
The investigation commenced with finding a suitable solvent for the Grignard reaction in which starting materials 34, 35 and intermediate products would dissolve. After having identified THF as the most suitable option, the next challenge was to find an acid that could induce both hydrolysis and dehydration in a single step. Hydrochloric acid was able to perform both transformations, however, precipitation was observed. Thus, hydrochloric acid molarities ranging from 1–12 M were tested. However, while even at the lowest molarity precipitation was observed, it also appeared that below 6 M the dehydration reaction did not proceed. Since the precipitation could not be prevented, a molarity of 12 M was eventually used. The individually optimised transformations were then combined in a one-flow continuous system. Most troublesome was that addition of HCl to the reaction mixture led to an exothermic reaction and boiling of the solvent. Therefore, a back-pressure regulator was employed so that melitracen (36) could be successfully synthesised as its HCl-salt in approximately 85% yield.
SYN
https://pubs.acs.org/doi/pdf/10.1021/acs.oprd.7b00368
A Grignard-based batch process, for the preparation of Melitracen HCl, has been redesigned to fit a continuous reactor system. The Grignard addition is carried out at room temperature, with subsequent hydrolysis of the magnesium alkoxide intermediate followed by dehydration of the resulting alcohol. The product undergoes further workup by simple gravimetric phase separation and then crystallization with 2 M HCl in diethyl ether to afford pure Melitracen HCl. All steps in the laboratory setup were concatenated, and the setup was proven capable of producing a significant portion of the commercial quantities of Melitracen HCl. The flow setup profits from a reduced footprint, lower energy consumption, fewer synthetic steps, and reduced raw material usage compared to the batch process.

As illustrated in Scheme 1, four synthetic steps are involved in the manufacturing of Melitracen HCl (6). The four steps are a classic Grignard addition to a ketone, a hydrolysis of a magnesium alkoxide, a dehydration of an alcohol and a salt precipitation to isolate the API. The Grignard addition is between 10,10-dimethylanthrone (10,10-DMA (1)) and 3-(N,N-dimethylamino)propylmagnesium chloride (DMPC-MgCl (2)), resulting in formation of the magnesium alkoxide 3. The magnesium alkoxide 3 is then hydrolyzed to the alcohol 4 and dehydrated to form product 5. The last step is a crystallization of the API as a salt, where HCl is added to obtain the Melitracen HCl (6)
Scheme 1: Syntheses of magnesium alkoxide 3, alcohol 4 and dehydrated product 5 in the manufacturing process of Melitracen HCl 6, from ketone 1 and Grignard reagent 2.
Current Batch Synthesis The current batch synthesis involves individual synthetic steps, as illustrated in Figure 1. DMPC-MgCl 2 is made in-house before it is used, due to its limited storage shelf life, in a toluene-THF solvent mixture. THF is present in trace amounts in order to stabilize the magnesium in the Grignard reagents.45 A solution of 10,10-DMA 1 is prepared in toluene and is slowly transferred to the DMPC-MgCl 2, maintaining a temperature of 50°C. DMPC-MgCl 2 is used in an equivalence of 1.6 compared to 10,10-DMA 1. The formed magnesium alkoxide 3 is hydrolyzed with water and acetic acid (80%). The aqueous phase is discarded and concentrated hydrochloric acid (37%) is used to dehydrate alcohol 4 to form dehydrated product 5. Toluene is replaced with ethanol by a solvent swap. Crystallization of the dehydrated product 5 from the ethanol phase is done with HCl gas to obtain the final Melitracen HCl (6), which is subsequently isolated by filtration.
Precipitation of Melitracen HCl from THF The dehydrated product 5 was crystallized as the final HCl salt in the THF in a batch experiment, in order to remove a solvent swap to ethanol. The crystallization was carried out with 2 M HCl in Et2O, as this was considered more suited for a later flow process and more easily implemented in the laboratory setup. An equivalence of 1.1 HCl was used and the requirement was an achievement of pH<2. The mixture was kept stirred during the crystallization and carried out at ambient temperature. After 10 minutes, fine white solids started to form, followed by a massive precipitation of Melitracen HCl 6. The Melitracen HCl 6 was filtered with a Büchner funnel and washed with THF. The isolated yield was 80% and within the specifications for the in-house analysis methods used in the routine production (CHN, TGA, UV-vis, HPLC, melting point). Figure 3 is a microscope picture of the isolated Melitracen HCl 6. For full-scale production, the HCl gas would still be more desirable for the crystallization and the 2 M HCl in Et2O merely serves as a proof of concept for the laboratory flow setup.
CLIP
PATENT
https://patents.google.com/patent/CN105418436B/en
Melitracen (Melitracen), is a kind of tricyclics, entitled 10, the 10- dimethyl -9- γ-two of chemistry Methylamino acrylic -9,10- dihydro-anthraquinone, Clinical practice is its hydrochloride.Melitracen can suppress in presynaptic membrane To the effect of the reuptake of norepinephrine and serotonin, and therefore improve containing for monoamine transmitterses in synaptic cleft Amount.
On the preparation method of melitracen, document report both domestic and external is seldom, existing as described below:
US3177209, GB939856, DK97400, are the compound patents of Lundbeck drugmaker of Denmark, it is mentioned that Synthetic method is that, with 10,10- dimethylanthracene -9- ketone and N, TMSDMA N dimethylamine base propyl group magnesium chloride is generated in the middle of melitracen Body, then by intermediate be dissolved under chloroform, reflux state lead to hydrogen chloride prepare melitracen crude product, then crystallized again with acetone Melitracen is obtained, this method needs to be passed through hydrogen chloride at reflux, there is substantial amounts of smog to produce, and reaction condition is not yet It is easy to control, it there is larger safety factor.
CN103877088A is Lundbeck drugmaker of Denmark in a kind of safe melitracen group disclosed in 2014 Compound, wherein the purity to melitracen in drug regimen proposes more strict requirements, especially to that may make in clinic Cause the impurity (formula I, formula II) of the adverse reactions such as anxiety, irritated and excitement in, even more propose:Formula I<0.1%, formula II< 0.1, I+formula of formula II<0.1% rigors.The melitracen of patent US3177209, GB939856, DK97400 method synthesis Impurity is more, and primary purification can not obtain satisfactory active pharmaceutical ingredient (API).
It is also mentioned that the preparation method of melitracen hydrochloride, this method is with 10,10- diformazans in patent CN103877088A The γ of base-9-dimethylaminopropyl-9- anthrols are raw material, add dichloromethane and hydrochloric acid, are heated to reflux, reaction system alkaline hydrolysis from The free alkali obtained afterwards, is re-dissolved in acetone and leads to hydrogen chloride into salt, obtain melitracen crude product, then isolated and purified with column chromatography Obtain the melitracen of high-purity.The melitracen yield that it is prepared into is low, and purifies and separates process needs column chromatography, it is impossible to meet The need for large-scale production.
Embodiment 1
A kind of preparation method of melitracen hydrochloride, comprises the following steps:
(1) melitracen intermediate is prepared
10,10- dimethylanthracene -9- ketone carry out grignard reaction with 3- dimethylaminos-n-propyl chloride in the presence of initiator, obtain To melitracen intermediate, detailed process is as follows:
340g magnesium rods and 17.5L absolute ethers are added in 20L glass reaction kettles, stirring is warming up to 30~35 DEG C, addition 1.75kg 3- dimethylaminos-n-propyl chloride, finish insulated and stirred, add 1g iodine and 2mL 1,2- Bromofume as initiator, 9h is stirred at reflux, magnesium rod disappears completely, reaction system is cooled into 10~20 DEG C, 1.5kg 10,10- dimethyl is slowly added to Anthracene -9- ketone, then it is warming up to 30~35 DEG C, back flow reaction 1 hour;TLC monitoring reactions are complete, and reaction system is cooled into 10~20 DEG C, then add 5.5L water, ether layer is separated, anhydrous sodium sulfate is added and is concentrated under reduced pressure drying, obtain melitracen intermediate 2.03kg, receive The ﹪ of rate 97.2, purity 98.5%.
TLC monitoring methods:Add water and be quenched after sampling, take organic layer point plate;Solvent is petroleum ether:Ethyl acetate=2:1 (volume ratio);The Rf of 10,10- dimethylanthracene -9- ketone is 0.6, and the Rf of melitracen intermediate is 0.1.
(2) melitracen crude product is prepared
2kg melitracens intermediate, 10L chloroforms and 2.4L concentrated hydrochloric acids are put into 20L glass reaction kettles, stirred molten Solution, obtains pale yellow solution, and 60 DEG C of heating stirring reaction 2 hours, TLC monitoring reactions are complete, and separate aqueous layer, organic phase is concentrated under reduced pressure Dry, it is melitracen crude product 2.03kg, yield 95.7%, purity 99.41%, containing Formulas I to obtain white solid:0.20%, formula II:0.13%;Formulas I, II1HNMR spectrograms, melitracen crude product liquid phase spectrogram are shown in accompanying drawing 1,2,3 respectively;
TLC monitoring methods:Organic phase point plate is extracted reaction solution, solvent is dichloromethane:Methanol:Acetic acid=150:10:2 (volume ratio).
Formulas I:1H NMR(400MHz,DMSO)δ7.78-7.82(m,2H),δ7.50-7.53(m,2H),δ7.28-7.35 (m, 4H), δ 2.11 (S, 6H), δ 2.08 (d, J=6.8Hz, 2H), δ 1.96 (t, J=6.4Hz, 2H), δ 1.72 (s, 3H), δ 1.61(s,3H),δ1.26(brs,1H),δ1.02-1.09(m,2H)
Formula II:1H NMR(400MHz,DMSO)δ8.95(s,2H),δ7.47-7.63(m,4H),δ7.27-7.37(m, 4H), δ 6.06 (t, J=7.2Hz, 1H), δ 3.09 (t, J=7.2Hz, 2H), δ 2.91 (m, 2H), δ 2.54 (s, 3H), δ 1.53 (s,6H)
(3) purifying of melitracen crude product
Take 2.03kg melitracens crude product (purity 99.41%, Formulas I:0.20%, Formula II:0.13%) 4 times of amount (W/, are added V isopropanol), 20~25 DEG C of stirring 4h (mashing), is filtered, and is dried, is obtained product 2.0kg, yield is 98.5%, and purity is 99.61%, containing Formulas I:0.054%, without Formula II;Melitracen crude product is shown in accompanying drawing 4 through isopropanol mashing sample liquid chromatography(LC figure;
Product after 2kg is beaten is added in 30L glass reaction kettle, adds 16kg isopropanols, and backflow is dissolved, then Cool to 10 DEG C and stir crystallization and stay overnight, suction filtration is dried under reduced pressure, and obtains melitracen 1.89kg, and yield 94.5%, purity 99.98% contains Formulas I:0.0026%, without Formula II;See accompanying drawing 5 through isopropanol recrystallization liquid phase spectrogram.
Embodiment 2
A kind of preparation method of melitracen hydrochloride, comprises the following steps:
(1) melitracen intermediate is prepared
This step is identical with the step (1) in embodiment 1;
(2) melitracen crude product is prepared
This step is identical with the step (2) in embodiment 1;
(3) purifying of melitracen crude product
Take 10g melitracens crude product (purity 99.41%, Formulas I:0.20%, Formula II:0.13%) 4 times of amounts (W/V), are added Ethanol, 20~25 DEG C stirring 4h (mashing), filtering, drying, obtain product 9.79g, yield is 97.9%, purity 99.69% contains Formula I 0.047%, containing formula II 0.005%;Melitracen crude product is shown in accompanying drawing 6 through ethanol mashing sample liquid chromatography(LC figure;
Product after 9.0g ethanol is beaten is added in 250mL round-bottomed flask, adds the dissolving of 230mL alcohol refluxs, Then 10 DEG C are cooled to stir crystallization and stay overnight, suction filtration is dried under reduced pressure, obtain melitracen 8.4g, yield 93.3%, purity 99.98%, Containing Formulas I:0.0041%, without Formula II;See accompanying drawing 7 through ethanol recrystallization liquid phase spectrogram.
Embodiment 3
A kind of preparation method of melitracen hydrochloride, comprises the following steps:
(1) melitracen intermediate is prepared
This step is identical with the step (1) in embodiment 1;
(2) melitracen crude product is prepared
100g melitracens intermediate, 500mL chloroforms and 120mL concentrated hydrochloric acids are put into 1L three-necked bottles, stirred molten Solution, obtains pale yellow solution, and 60 DEG C of heating stirring reaction 2 hours, TLC monitoring reactions are complete, and separate aqueous layer, organic phase is concentrated under reduced pressure Dry, it is melitracen crude product 104g, yield 98.3%, purity 99.38%, containing Formulas I to obtain white solid:0.22%, Formula II: 0.15%;Melitracen crude product liquid phase spectrogram is shown in accompanying drawing 8;
TLC monitoring methods:Organic phase point plate is extracted reaction solution, solvent is dichloromethane:Methanol:Acetic acid=150:10:2 (volume ratio).
(3) purifying of melitracen crude product
Above-mentioned melitracen crude product is taken, the methanol of 4 times of amounts (W/V) is added, 20~25 DEG C of stirring 4h (mashing) obtain product Weight is 18.48g, and yield is 92.4%, and purity is 99.66%, containing Formulas I:0.05%, Formula II:0.008%, see accompanying drawing 9.
Embodiment 4
A kind of preparation method of melitracen hydrochloride, comprises the following steps:
(1) melitracen intermediate is prepared
This step is identical with the step (1) in embodiment 3;
(2) melitracen crude product is prepared
This step is identical with the step (2) in embodiment 3;
(3) purifying of melitracen crude product
Above-mentioned melitracen crude product is taken, the n-butanol of 4 times of amounts (W/V) is added, 20~25 DEG C of stirring 5h (mashing) are produced Thing weight is 19.6g, and yield is 98%, and purity is 99.54%, containing Formulas I:0.05%, Formula II:0.009%, see accompanying drawing 10.
Embodiment 5
A kind of preparation method of melitracen hydrochloride, comprises the following steps:
(1) melitracen intermediate is prepared
This step is identical with the step (1) in embodiment 3;
(2) melitracen crude product is prepared
This step is identical with the step (2) in embodiment 3;
(3) purifying of melitracen crude product
Above-mentioned melitracen crude product is taken, the isopropanol of 4 times of amounts (W/V) is added, 30~35 DEG C of stirring 5h (mashing) are produced Thing weight is 18.06g, and yield is 90.3%.
Embodiment 6
A kind of preparation method of melitracen hydrochloride, comprises the following steps:
(1) melitracen intermediate is prepared
This step is identical with the step (1) in embodiment 3;
(2) melitracen crude product is prepared
This step is identical with the step (2) in embodiment 3;
(3) purifying of melitracen crude product
Above-mentioned melitracen crude product is taken, the isopropanol of 4 times of amounts (W/V) is added, 50 DEG C of stirring 3h (mashing) obtain product weight Measure as 14.2g, yield is 71%.
Embodiment 7
A kind of preparation method of melitracen hydrochloride, comprises the following steps:
(1) melitracen intermediate is prepared
This step is identical with the step (1) in embodiment 3;
(2) melitracen crude product is prepared
This step is identical with the step (2) in embodiment 3;
(3) purifying of melitracen crude product
Above-mentioned melitracen crude product is taken, the isopropanol of 4 times of amounts (W/V) is added, 5-10 DEG C of stirring 5h (mashing) obtains product Weight is 19.7g, and yield is 98.5%, and purity is 99.53%, containing Formulas I:0.054%, Formula II:0.014%, see accompanying drawing 11.
Embodiment 8
With reference to CN103877088A, crystallized using acetone, that is, take 10g melitracens intermediate and 24mL dichloromethane, 6.7mL concentrated hydrochloric acids are heated to reflux 2h and are cooled to room temperature, and pH is to 8-9 for regulation, then are extracted with dichloromethane and product, are concentrated to give free Alkali cpd, acetone is dissolved in by the free alkali compound, concentrated hydrochloric acid is added dropwise to pH=0.1, stirring, cooling separate out solid 7.1g, This solid crystallizes to obtain sample 6.4g with acetone again, and total recovery is 60.9%, and purity is 99.64%, containing Formulas I:0.09%, Formula II: 0.04%.Melitracen is shown in accompanying drawing 12 only with acetone crystallization liquid chromatography(LC figure.
Repeat literature method crystallized only with acetone obtained by product in impurity Formulas I, Formula II impurity summation be 0.13%, The adverse reactions such as anxiety, irritated and excitement may be caused in Clinical practice.
In summary, the effect of mashing is to make melitracen crude product rapid dispersion, and the effect of methanol mashing is similar with ethanol, But it is good without isopropanol effect, but methanol mashing yield is decreased obviously trend;N-butanol mashing needs the extension time to reach To the effect same with ethanol, but be not as good as isopropanol effect, and because the viscosity of n-butanol is slightly larger, melitracen crude product is at it In disperse slightly worse, invention has the granular solids that not readily dissolve after filtering, and the removal effect to other impurities is also poor;Isopropanol Temperature is raised during mashing, yield is decreased obviously, and reduces temperature, yield has no raising, though to the removal effect of impurity Formula II It can so control in the range of conforming to quality requirements, but compared to being decreased obviously in embodiment 1.

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
References
- ^ Swiss Pharmaceutical Society (2000). Index Nominum 2000: International Drug Directory (Book with CD-ROM). Boca Raton: Medpharm Scientific Publishers. ISBN 3-88763-075-0.
- ^ Hall, Chapman and; Chemical Abstracts Service, American Chemical Society; Rhodes, P. H (1996). Dictionary of organic compounds. London: Chapman & Hall. ISBN 0-412-54090-8.
- ^ O’Neil, Maryadele J. (2001). The Merck index: an encyclopedia of chemicals, drugs, and biologicals. Rahway, NJ: Merck Research Laboratories. ISBN 0-911910-13-1.
- ^ José Miguel Vela; Helmut Buschmann; Jörg Holenz; Antonio Párraga; Antoni Torrens (2007). Antidepressants, Antipsychotics, Anxiolytics: From Chemistry and Pharmacology to Clinical Application. Weinheim: Wiley-VCH. ISBN 978-3-527-31058-6.
- ^ Muller, Niels F; Dessing, Rudolf P; Pharmacy, European Society of Clinical (1998). European Drug Index, 4th Edition. Boca Raton: CRC Press. ISBN 3-7692-2114-1.
- ^ Van Moffaert M, Dierick M, De Meulemeester F, Vereecken A (1983). “Treatment of depressive anxiety states associated with psychosomatic symptoms. A double-blind multicentre clinical study: mianserin versus melitracen-flupentixol”. Acta Psychiatrica Belgica. 83 (5): 525–39. PMID 6670581.
- ^ Bin Yaacob H (April 1985). “Flupenthixol and Melitracen in the management of trigeminal neuralgia”. Dental Journal of Malaysia. 8 (2): 37–8. PMID 3917005.
- ^ Hashash JG, Abdul-Baki H, Azar C, et al. (June 2008). “Clinical trial: a randomized controlled cross-over study of flupenthixol + melitracen in functional dyspepsia”. Alimentary Pharmacology & Therapeutics. 27 (11): 1148–55. doi:10.1111/j.1365-2036.2008.03677.x. PMID 18331614. S2CID 40714136.
- ^ Aronson, Jeffrey Kenneth (2008). Meyler’s Side Effects of Psychiatric Drugs (Meylers Side Effects). Amsterdam: Elsevier Science. ISBN 978-0-444-53266-4.
- ^ Author Unknown (1970). Ann Reports Medicinal Chem V5 (v. 5). Boston: Academic Press. ISBN 0-12-040505-9.
{{cite book}}:|author=has generic name (help)
| Clinical data | |
|---|---|
| Trade names | Adaptol, Dixeran, Melixeran, Thymeol, Trausabun |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral, intramuscular injection |
| ATC code | N06AA14 (WHO) |
| Legal status | |
| Legal status | In general: ℞ (Prescription only) |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 5118-29-6 |
| PubChem CID | 25382 |
| ChemSpider | 23697 |
| UNII | Q7T0Y1109Z |
| KEGG | D08171 |
| ChEMBL | ChEMBL110094 |
| CompTox Dashboard (EPA) | DTXSID4048274 |
| ECHA InfoCard | 100.023.507 |
| Chemical and physical data | |
| Formula | C21H25N |
| Molar mass | 291.438 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
| (what is this?) (verify) |
//////////Melitracen, Q7T0Y1109Z, Thymeol, мелитрацен , ميليتراسان , 美利曲辛 , U 24973A, Antidepressant, Tricyclics,

NEW DRUG APPROVALS
ONE TIME
$10.00
US OK for Forest/Pierre Fabre antidepressant fetzima, levomilnacipran

levomilnacipran
The US Food and Drug Administration has approved Forest Laboratories and Pierre Fabre’s Fetzima for major depressive disorder.
Fetzima (levomilnacipran extended-release), a once-daily serotonin and norepinephrine reuptake inhibitor, has been given the green light based on Phase III studies of adults with MDD and statistically significant and clinically meaningful improvement in depressive symptoms across three doses (40, 80, and 120 mg).
read all at
http://www.pharmatimes.com/Article/13-07-26/US_OK_for_Forest_Pierre_Fabre_antidepressant.aspx


Levomilnacipran (F2695) is an antidepressant currently under development by Forest Laboratories for the treatment of depression in the United States and Canada.[1][2][3] As of 2009 it is in phase III clinical trials.[4] Levomilnacipran is an active enantiomer of milnacipran and therefore has similar effects and pharmacology, acting as a serotonin-norepinephrine reuptake inhibitor.[2][5] On 20 January 2011, Forest and Pierre Fabre Medicament announced that levomilnacipran was no better than placebo in a late-stage clinical trial. Two other late-stage trials will be finished in mid-2011.
References
- “Future Treatments for Depression, Anxiety, Sleep Disorders, Psychosis, and ADHD — Neurotransmitter.net”.
- “Pierre Fabre Medicament and Forest Laboratories to Collaborate on Development and Commercialization of F2695 for Depression – FierceBiotech”.
- “News: Forest Buys CNS Disease-Related Drug for $75M Upfront.”.
- “Search of: F2695 – List Results – ClinicalTrials.gov”.
- Deprez D, Chassard D, Baille P, Mignot A, Ung HL, Puozzo C (1998). “Which bioequivalence study for a racemic drug? Application to milnacipran”. European Journal of Drug Metabolism and Pharmacokinetics 23 (2): 166–71. PMID 9725476.
NDA-US Marketing by Ranbaxy, Alembic has announced that it has received an NDA approval for extended release version of Pfizer’s anti depressant drug Pristiq, Desvenlafaxine Base

DESVENLAFAXINE
read at
5 march 2013
Alembic has announced that it has received an NDA approval for extended release version of Pfizer’s anti depressant drug Pristiq. Pristiq sell approximately $550m in the US. Alembic has outlicensed rights to Ranbaxy for marketing in the US. The company will start marketing the product immediately.
Alembic will manufacture and supply the drug to Ranbaxy for marketing in the US. Vadodara-based pharma player, Alembic Pharmaceuticals Limited has received the approval from the US Food and Drug Administration (USFDA) for a bioequivalent version of Pristiq by Pfizer.

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










