Home » Posts tagged 'amezalpat'
Tag Archives: amezalpat
Amezalpat
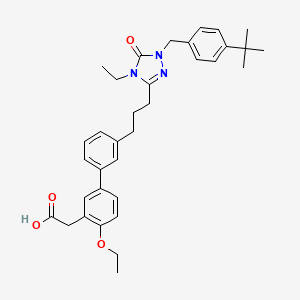
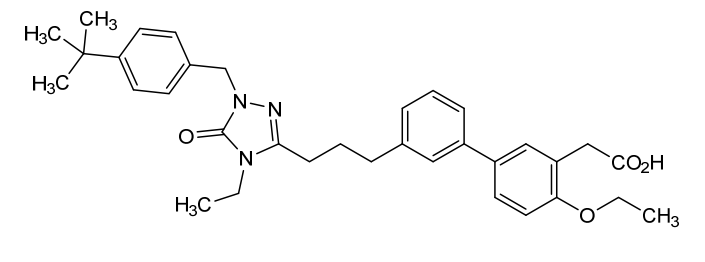
Amezalpat
CAS 1616372-41-8
MF C34H41N3O4 MW555.7 g/mol
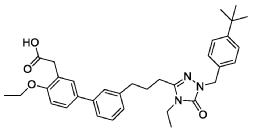
- [1,1′-Biphenyl]-3-acetic acid, 3′-[3-[1-[[4-(1,1-dimethylethyl)phenyl]methyl]-4-ethyl-4,5-dihydro-5-oxo-1H-1,2,4-triazol-3-yl]propyl]-4-ethoxy-
- 2-(3′-(3-(1-(4-(tert-Butyl)benzyl)-4-ethyl-5-oxo-4,5-dihydro-1H-1,2,4-triazol-3-yl)propyl)-4-ethoxy-[1,1′-biphenyl]-3-yl)acetic acid
- 3′-(3-(1-((4-(1,1-DIMETHYLETHYL)PHENYL)METHYL)-4-ETHYL-4,5-DIHYDRO-5-OXO-1H-1,2,4-TRIAZOL-3-YL)PROPYL)-4-ETHOXY(1,1′-BIPHENYL)-3-ACETIC ACID
2-[5-[3-[3-[1-[(4-tert-butylphenyl)methyl]-4-ethyl-5-oxo-1,2,4-triazol-3-yl]propyl]phenyl]-2-ethoxyphenyl]acetic acid

peroxisome proliferator-activated receptor alpha (PPARα) antagonist, antineoplastic, TPST 1120, FDA Fast Track, Orphan Drug, 1EQ4LQN9N3
Amezalpat (formerly TPST-1120) is an investigational, oral, small-molecule inhibitor targeting peroxisome proliferator-activated receptor alpha (PPAR being developed by Tempest Therapeutics. It works by directly targeting tumor cells and reducing immune suppression in the tumor microenvironment. In combination with atezolizumab and bevacizumab, it has shown improved survival in hepatocellular carcinoma (HCC) patients, receiving FDA Fast Track and Orphan Drug designations.
Key Details on Amezalpat
- Indication: Primarily being studied for unresectable or metastatic hepatocellular carcinoma (liver cancer).
- Mechanism: A selective, competitive antagonist of PPAR
, which plays a role in fatty acid metabolism in cancer cells.
- Clinical Efficacy: A phase 1b/2 study indicated that adding amezalpat to standard-of-care (atezolizumab + bevacizumab) improved median overall survival to 21 months compared to 15 months for the control, according to Tempest Therapeutics.
- Trial Status: A pivotal Phase 3 study (NCT06680258) to evaluate this combination as a first-line treatment is planned for 2025.
- Other Potential Uses: Preclinical data suggests potential activity in other advanced solid tumors, including renal cell carcinoma.
Disclaimer: Amezalpat is an investigational agent and is not yet approved by the FDA for widespread clinical use.
Amezalpat is an orally bioavailable, small molecule, selective and competitive antagonist of peroxisome proliferator activated receptor alpha (PPARa), with potential immunomodulating and antineoplastic activities. Upon oral administration, amezalpat targets, binds to and blocks the activity of PPARa, thereby blocking transcription of PPARa target genes leading to an intracellular metabolism shift from fatty acid oxidation (FAO) to glycolysis in FAO-dependent tumors and reducing the production of fatty acids in the tumor microenvironment (TME). As fatty acids are essential for tumor cell growth in FAO-dependent tumor cells and are needed for the metabolism of suppressive immune cells in the TME, including regulatory T-cells (Tregs), reducing the amount of fatty acids leads to a direct killing of FAO-dependent tumor cells. It also skews macrophages from the immune suppressive M2 phenotype to an effector M1 phenotype and facilitates the cytotoxicity of immune effector cells, thereby stimulating an anti-tumor immune response and further killing tumor cells. Amezalpat also restores the natural inhibitor of angiogenesis thrombospondin-1 (TSP-1) and stimulator of interferon genes (STING) in the TME. PPARa, a ligand-activated nuclear transcription factor and metabolic checkpoint, regulates the expression of FAO genes and lipid metabolism. It plays a key role in immunosuppression in the TME. FAO is a metabolic pathway essential to tumor growth, survival and immunosuppression.
SYN
Example 6: 2-(3′-(3-(1-(4-(tert-Butyl)benzyl)-4-ethyl-5-oxo-4,5-dihydro-1H-1,2,4-triazol-3-yl)propyl)-4-ethoxy-[1,1′-biphenyl]-3-yl)acetic acid
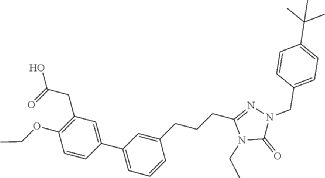
SYN
SYN
WO2014099503 TRIAZOLONE COMPOUNDS AND USES THEREOF
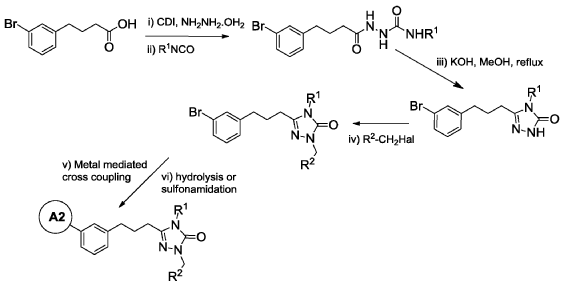
Example 6: 2-(3′-(3-(1-(4-(tert-Butyl)benzyl)-4-ethyl-5-oxo-4,5-dihydro-1H-1,2,4-triazol-3-yl)propyl)-4-ethoxy-[1, 1′-biphenyl]-3-yl)acetic acid
Pat
WO2025235527 CRYSTALLINE FORMS OF A PPAR ALPHA ANTAGONIST
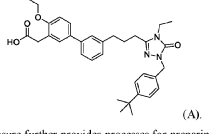
2-(3′-(3-(l-(4-(tertbutyl)benzyl)-4-ethyl-5-oxo-4,5-dihydro-lH-l,2,4-triazol-3-yl)propyl)-4-ethoxy-[1,T-biphenyl]-3-yl)acetic acid, depicted below as Compound A
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Triazolone compounds and uses thereofPublication Number: US-2017239223-A1Priority Date: 2012-12-20
- Triazolone compounds and uses thereofPublication Number: WO-2014099503-A1Priority Date: 2012-12-20
- Triazolone compounds and uses thereofPublication Number: US-10568871-B2Priority Date: 2012-12-20Grant Date: 2020-02-25
- Triazolone compounds and uses thereofPublication Number: US-2024041837-A1Priority Date: 2012-12-20
- Compound or pharmaceutically acceptable salt thereof, pharmaceutical composition and uses thereofPublication Number: BR-112015013350-B1Priority Date: 2012-12-20
- Triazolone compounds and uses thereofPublication Number: US-2015344446-A1Priority Date: 2012-12-20
- Triazolone compounds and uses thereofPublication Number: US-11666557-B2Priority Date: 2012-12-20Grant Date: 2023-06-06
- Triazolone compounds and uses thereofPublication Number: US-2020138790-A1Priority Date: 2012-12-20
- Triazolone compounds and uses thereofPublication Number: US-9676754-B2Priority Date: 2012-12-20Grant Date: 2017-06-13
- Triazolone compounds and uses thereofPublication Number: CA-2894281-CPriority Date: 2012-12-20Grant Date: 2021-04-20
- Triazolone compounds and uses thereofPublication Number: WO-2024102620-A2Priority Date: 2022-11-09
- Triazolone compounds and uses thereofPublication Number: AU-2013363398-B2Priority Date: 2012-12-20Grant Date: 2017-06-01
- Triazolone compounds and uses thereofPublication Number: EP-2935228-B9Priority Date: 2012-12-20Grant Date: 2017-12-06
- Triazolone compounds and uses thereofPublication Number: CA-2894281-A1Priority Date: 2012-12-20
- Triazolone compounds and uses thereofPublication Number: EP-2935228-B1Priority Date: 2012-12-20Grant Date: 2017-08-02
/////////////amezalpat, ANAX LAB, antineoplastic, TPST 1120, FDA Fast Track, Orphan Drug, 1EQ4LQN9N3

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










