Home » Posts tagged '4RNU8C6XXW'
Tag Archives: 4RNU8C6XXW
Selonabant
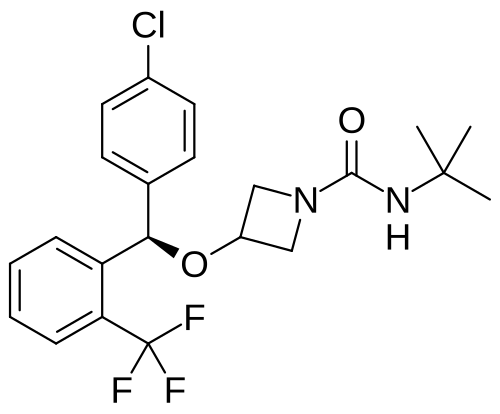
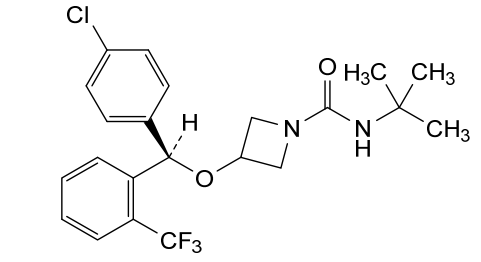
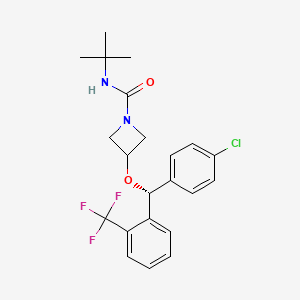
Selonabant
CAS 791848-71-0
MF C22H24ClF3N2O2 MW440.89 g/mol
N–tert-butyl-3-[(R)-(4-chlorophenyl)-[2-(trifluoromethyl)phenyl]methoxy]azetidine-1-carboxamide
N-tert-butyl-3-[(R)-(4-chlorophenyl)-[2-(trifluoromethyl)phenyl]methoxy]azetidine-1-carboxamide
(R)-N-(tert-butyl)-3-((4-chlorophenyl)(2-(trifluoromethyl)phenyl)methoxy)azetidine-1-carboxamide
N-tert-butyl-3-{(R)-(4-chlorophenyl)[2-(trifluoromethyl)phenyl]methoxy}azetidine-1-carboxamide
cannabinoid receptor 1 (CB1) antagonist, ANEB 001, V 24343, 4RNU8C6XXW, ANEB001
Drug class: Cannabinoid receptor antagonist (CB1)
The anti-obesity drug, V24343, acts by targeting the CB1 receptor in the brain and suppressing a person’s appetite.
Selonabant (INNTooltip International Nonproprietary Name, USANTooltip United States Adopted Name; developmental code names ANEB-001, V-24343) is a cannabinoid CB1 receptor antagonist which is under development for the treatment of acute cannabinoid intoxication.[1][2][3] It was also previously being developed to treat obesity, but development for this indication was discontinued.[1] The drug is administered by intravenous infusion.[1] It dramatically reduced the subjective effects of Δ9-tetrahydrocannabinol (THC) in a clinical trial.[3] Selonabant is being developed by Vernalis and Anebulo Pharmaceuticals.[1][2]
1 CLINICAL TRIALS
As of December 2024, it is in phase 2 trials.[1][2], I.V. Selonabant in Healthy Adult SubjectsCTID: NCT07211607Phase: Phase 1Status: RecruitingDate: 2026-02-12
Safety, Tolerability, Pharmacokinetics, and Pharmacodynamics of CB1 Antagonist ANEB-001 in a THC Challenge TestCTID: NCT05282797Phase: Phase 2Status: CompletedDate: 2023-08-29
Safety and Efficacy of Low Doses of V24343 in Obese SubjectsCTID: NCT00734201Phase: Phase 1Status: CompletedDate: 2011-07-25
A randomized, double-blind, placebo-controlled study to assess the safety, tolerability, pharmacokinetics and pharmacodynamics of single oral doses of CB1 antagonist ANEB-001 in healthy occasional cannabis users
EudraCT: 2021-000305-24
Phase: Phase 2, Status: Completed, Date: 2021-03-18
2. Chemical Structure Features
Key structural motifs:
• Azetidine ring (4-membered nitrogen heterocycle)
• Chiral benzhydryl ether center
• Trifluoromethyl phenyl group
• p-chlorophenyl group
• tert-butyl carboxamide
These motifs are typical of lipophilic GPCR ligands, enabling strong binding to the CB1 receptor.
Key descriptors:
| Property | Value |
|---|---|
| LogP | ~4.9 |
| H-bond donors | 1 |
| H-bond acceptors | 5 |
| TPSA | 41.6 Ų |
| Chiral centers | 1 |
3. Pharmacological Target
Primary target:
CB1 receptor (CNR1)
• GPCR located in the central nervous system
• Mediates effects of cannabinoids such as THC
Selonabant is a competitive CB1 antagonist.
Mechanism:
- THC activates CB1 receptor
- Leads to euphoria, altered cognition, tachycardia
- Selonabant blocks CB1 binding
- Reverses or prevents THC effects
4. Therapeutic Indication
Main indication
Acute cannabinoid intoxication
Potential clinical uses:
• Cannabis overdose in emergency rooms
• Pediatric accidental cannabis ingestion
• Edible THC overdose
• Severe psychiatric reactions to cannabis
A CB1 antagonist could act as a “cannabis antidote.”
5. Clinical Development
Developer:
Anebulo Pharmaceuticals (with earlier work by Vernalis)
Development timeline
| Phase | Details |
|---|---|
| Preclinical | Demonstrated CB1 antagonism |
| Phase I | Safety and PK studies |
| Phase II | THC challenge studies |
In clinical trials, the drug significantly reduced subjective effects of THC in volunteers.
Status (2024–2025): Phase II development.
6. Administration
Originally studied as:
• Intravenous infusion for rapid reversal of THC effects
Some reports indicate oral activity in research settings.
The IV route is preferred in emergency settings.
7. Comparison with Earlier CB1 Antagonists
Selonabant belongs to the same pharmacological class as:
| Drug | Status |
|---|---|
| Rimonabant | Withdrawn (psychiatric side effects) |
| Taranabant | Development stopped |
| Selonabant | Designed for short-term antidote use |
Key difference:
Earlier CB1 antagonists were chronic obesity drugs, which caused depression and suicidality.
Selonabant is designed for acute use only, reducing psychiatric risk.
8. Pharmacological Profile
Approximate characteristics (reported in literature):
• High CB1 receptor affinity
• CNS-penetrant
• Rapid onset
• Short therapeutic exposure
Effects in THC challenge studies:
• Reduced intoxication score
• Reduced cognitive impairment
• Reduced subjective “high”
9. Chemical Class
Selonabant can be classified as:
Diarylmethoxy-azetidine carbamides
Key scaffold:
Diarylmethanol → ether → azetidine carbamate.
This scaffold appears in several CB1 inverse agonists.
PATENT
WO2005080345
Title: Cannabinoid CB1 receptor antagonists
Assignee: Vernalis (R&D) Ltd
Priority: ~2004
This is the primary patent family covering the scaffold used for Selonabant.
Related Continuation Patent
WO2005080328
Follow-up Patent on CB1 Antagonists
WO2005075450
Later Use Patent (Anebulo)
US11141404
Assignee: Anebulo Pharmaceuticals
Coverage:
• use of Selonabant (ANEB-001)
• treatment of acute cannabinoid overdose
• IV formulations
The patent protection extends to ~2040
Patent Family Map
Main Selonabant IP family:
| Patent | Type | Notes |
|---|---|---|
| WO2005080345 | Composition of matter | core scaffold |
| WO2005080328 | analog compounds | synthetic examples |
| WO2005075450 | optimized CB1 antagonists | SAR |
| US11141404 | method of use | overdose treatment |
| PCT follow-ups | formulation / delivery | IV use |
SYN
https://patents.google.com/patent/US11141404B1/en
SYN
US 7,504,522
Preparation of 2-(trifluoromethyl)-4-chlorobenzhydrol (96)
Preparation of 1-benzhydryl-3-[2-(trifluoromethyl)-4′-chlorobenzhydryloxy]azetidine (97)
| This material was prepared from 1-benzhydryl-3-azetidinol (1) (40.1 mmol) and 2-(trifluoromethyl)-4-chlorobenzhydrol (96) (80.2 mmol) using the procedure described for compound (3) (13.5 g, 66%). |
Preparation of 3-[2-(trifluoromethyl)-4′-chlorobenzhydryloxy]azetidine hydrochloride (98)
| This material was prepared from 1-benzhydryl-3-[2-(trifluoromethyl)-4′-chlorobenzhydryloxy]azetidine (97) (25.6 mmol) using the procedure described for compound (9) (8.2 g, 85%). |
| NMR (400 MHz, d 6-DMSO) δ H 3.78 (1H, m), 3.97 (3H, m), 4.89 (1H, q, J 6.0 Hz), 5.85 (1H, s), 7.33 (2H, m), 7.44 (2H, m), 7.59 (1H, m), 7.76 (3H, m), 8.97 (1H, bs) |
Example 81 FINAL MOLECULE
3-[2-(trifluoromethyl)-4′-chlorobenzhydryloxy]-N-(tert-butyl)azetidine-1-carboxamide (99)
| NMR (400 MHz, d 6-DMSO) δ H 1.20 (9H, s), 3.51 (1H, m), 3.65 (1H, m), 3.84 (2H, m), 4.25 (1H, m), 5.62 (1H, s), 5.73 (1H, s), 7.31 (2H, m), 7.39 (2H, m), 7.57 (1H, m), 7.75 (3H, m) |
SYN
(R)-N-(tert-butyl)-3-((4-chlorophenyl)(2-(trifluoromethyl)phenyl)methoxy)azetidine-1-carboxamide (Compound 1):
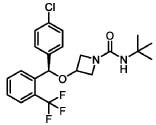
SYN
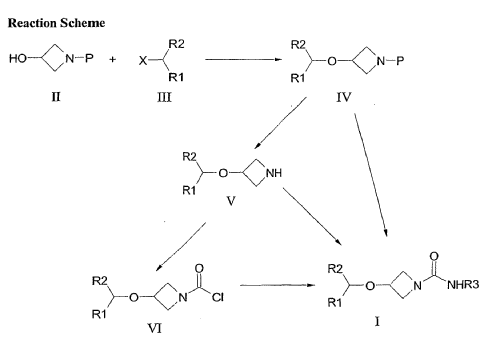
PAT
The core patent describing the chemistry for Selonabant-type molecules is:
WO2005080345
Title: Cannabinoid receptor antagonists
Assignee: Vernalis plc
Priority: ~2004
for SAR exploration
PATENTS
- Azetidinecarboxamide derivatives and their use in the treatment of cb1 receptor mediated disordersPublication Number: WO-2004096763-A1Priority Date: 2003-05-01
- Azetidinecarboxamide derivatives and their use in the treatment of CB1 receptor mediated disordersPublication Number: NZ-543016-APriority Date: 2003-05-01
- Botulinum neurotoxins for use in therapyPublication Number: US-11260114-B2Priority Date: 2017-03-22Grant Date: 2022-03-01
- Azetidinecarboxamide derivatives and their use in the treatment of cb1 receptor mediated disordersPublication Number: EP-1620395-A1Priority Date: 2003-05-01
- Azetidinecarboxamide derivatives and their use in the treatment of cb1 receptor mediated disordersPublication Number: EP-1620395-B1Priority Date: 2003-05-01Grant Date: 2009-12-30
- Azetidinecarboxamide derivatives and their use in the treatment of cb1 receptor mediated disordersPublication Number: US-7504522-B2Priority Date: 2003-05-01Grant Date: 2009-03-17
- Azetidinecarboxamide derivatives and their use in the treatment of cb1 receptor mediated disordersPublication Number: US-2007054891-A1Priority Date: 2003-05-01
- Botulinum neurotoxins production methodsPublication Number: US-2023016041-A1Priority Date: 2017-04-28
- Initiating neurotoxin treatmentsPublication Number: US-2023058666-A1Priority Date: 2017-04-21
- Initiating neurotoxin treatmentsPublication Number: EP-4137149-A1Priority Date: 2017-04-21
- Botulinum neurotoxins for treating hyperhidrosisPublication Number: US-2022370574-A1Priority Date: 2017-04-20
- Botulinum neurotoxins for use in therapyPublication Number: US-2023027850-A1Priority Date: 2017-03-22
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

| Clinical data | |
|---|---|
| Other names | ANEB-001; ANEB001; V-24343; V24343 |
| Drug class | Cannabinoid receptor antagonist; Cannabinoid CB1 receptor antagonist; Cannabinoid antidote |
| Identifiers | |
| IUPAC name | |
| CAS Number | 791848-71-0 |
| PubChem CID | 68902536 |
| DrugBank | DB18908 |
| ChemSpider | 128922145 |
| UNII | 4RNU8C6XXW |
| KEGG | D12875 |
| ChEMBL | ChEMBL5095165 |
| Chemical and physical data | |
| Formula | C22H24ClF3N2O2 |
| Molar mass | 440.89 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
References
- “Anebulo Pharmaceuticals”. AdisInsight. 26 December 2024. Retrieved 25 February 2025.
- “Delving into the Latest Updates on Selonabant with Synapse”. Synapse. 23 January 2025. Retrieved 25 February 2025.
- Gorbenko AA, Heuberger JA, Juachon M, Klaassen E, Tagen M, Lawler JF, et al. (February 2025). “CB1 Receptor Antagonist Selonabant (ANEB-001) Blocks Acute THC Effects in Healthy Volunteers: A Phase II Randomized Controlled Trial”. Clinical Pharmacology and Therapeutics. 117 (5): 1427–1436. doi:10.1002/cpt.3581. PMC 11993283. PMID 39898464.
//////////selonabant, cannabinoid receptor 1 (CB1) antagonist, ANEB 001, V 24343, 4RNU8C6XXW, ANEB001

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










