| Country | Patent Number | Approved | Expires (estimated) |
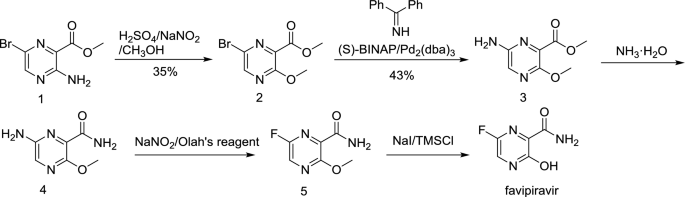
|---|---|---|---|
| United States | 5387603 | 1993-12-01 | 2013-12-01 |
| United States | 5403847 | 1992-11-13 | 2012-11-13 |
Home » Uncategorized (Page 94)
Category Archives: Uncategorized
FDA publishes List of Guidances planned for 2015
DRUG REGULATORY AFFAIRS INTERNATIONAL

At the beginning of each year the FDA always publishes a list of the guidances it plans to publish during that year. It has done so again in 2015. The document is relatively comprehensive, containing five pages. Find out more about the Guidances the FDA plans on publishing in 2015.
At the beginning of each year the FDA always publishes a list of the guidances it plans to publish during that year. It has done so again in 2015. The document is relatively comprehensive, containing five pages. The list is subdivided into different categories. It contains for example also guidances planned in connection with the topics Clinical Pharmacology or Clinical/Statistical.
CGMP is a category of its own for which “only” three new guidances are planned for 2015:
- A questions & answers (Q&A) paper on the topic data integrity
- CGMP rules for outsourced facilities (pharmacy compounding)
- Rules for the…
View original post 190 more words
GIVINOSTAT


Givinostat (INN[1]) or gavinostat (originally ITF2357) is a histone deacetylase inhibitor with potential anti-inflammatory, anti-angiogenic, and antineoplastic activities.[2] It is a hydroxamate used in the form of its hydrochloride.
Givinostat is in numerous phase II clinical trials (including for relapsed leukemias and myelomas),[3] and has been granted orphan drug designation in the European Union for the treatment of systemic juvenile idiopathic arthritis[4] and polycythaemia vera.[5]
In 2010, orphan drug designation was assigned in the E.U. for the treatment of systemic-onset juvenile idiopathic arthritis and for the treatment of polycythemia vera. In 2013, this designation was assigned by the FDA for the treatment of Duchenne’s muscular dystrophy and for the treatment of Becker’s muscular dystrophy.
ITF2357 was discovered at Italfarmaco of Milan, Italy. It was patented in 1997 and first described in the scientific literature in 2005.[6][7]
Givinostat hydrochloride, an orally active, synthetic inhibitor of histone deacetylase, is being evaluated in several early clinical studies at Italfarmaco, including studies for the treatment of myeloproliferative diseases, polycythemia vera, Duchenne’s muscular dystrophy and periodic fever syndrome. The company was also conducting clinical trials for the treatment of Crohn’s disease and chronic lymphocytic leukemia; however, the trials were terminated.
No recent development has been reported for research into the treatment of juvenile rheumatoid arthritis, for the treatment of multiple myeloma and for the treatment of Hodgkin’s lymphoma.
Muscular dystrophies (MDs) include a heterogeneous group of genetic diseases invariably leading to muscle degeneration and impaired function. Mutation of nearly 30 genes gives rise to various forms of muscular dystrophy, which differ in age of onset, severity, and muscle groups affected (Dalkilic I, Kunkel LM. (2003) Muscular dystrophies: genes to pathogenesis. Curr. Opin. Genet. Dev. 13:231-238). The most common MD is the Duchenne muscular dystrophy (DMD), a severe recessive X-linked disease which affects one in 3,500 males, characterized by rapid progression of muscle degeneration, eventually leading to loss of ambulation and death within the second decade of life.
Attempts to replace or correct the mutated gene, by means of gene or cell therapy, might result in a definitive solution for muscular dystrophy, but this is not easy to achieve. Alternative strategies that prevent or delay muscle degeneration, reduce inflammation or promote muscle metabolism or regeneration might all benefit patients and, in the. future, synergize with gene or cell therapy. Steroids that reduce inflammation are currently the only therapeutic tool used in the majority of DMD patients (Cossu G, Sampaolesi M . (2007) New therapies for Duchenne muscular dystrophy: challenges, prospects and clinical trials. TRENDS Mol . Med. 13:520-526).
Diethyl- [ 6- ( 4-hydroxycarbamoyl-phenyl-carbamoyloxy- methyl ) -naphthalen-2-yl-methyl ] -ammonium chloride , which is described in WO 97/43251 (anhydrous form) and in WO 2004/065355 (monohydrate crystal form), herein both incorporated by reference, is an anti-inflammatory agent which is able to inhibit the synthesis of the majority of pro-inflammatory cytokines whilst sparing anti-inflammatory ones. Diethyl- [ 6- ( 4-hydroxycarbamoyl-phenyl-carbamoyloxy- methyl ) -naphthalen-2-yl-methyl ] -ammonium chloride is also known as ITF2357.
The monohydrate crystal form of diethyl- [ 6- ( 4- hydroxycarbamoyl-phenyl-carbamoyloxy-methy1 ) – naphthalen-2-yl-methyl ] -ammonium chloride is known as Givinostat .
Givinostat is being evaluated in several clinical studies, including studies for the treatment of myeloproliferative diseases, polycythemia vera, periodic fever syndrome, Crohn’s disease and systemic- onset juvenile idiopathic arthritis. Orphan drug designation was assigned in the E.U. for the treatment of systemic-onset juvenile idiopathic arthritis and for the treatment of polycythemia vera.
Givinostat has been recently found to act also as a Histone Deacetylase inhibitor (WO 2011/048514).
Histone deacetylases ( HDAC ) are a family of enzymes capable of removing the acetyl group bound to the lysine residues in the N-terminal portion of histones or in other proteins.
HDACs can be subdivided into four classes, on the basis of structural homologies. Class I HDACs (HDAC 1, 2, 3 and 8) are similar to the RPD3 yeast protein and are located in the cell nucleus. Class II HDACs (HDAC 4, 5, 6, 7, 9 and 10) are similar to the HDA1 yeast protein and are located both in the nucleus and in the cytoplasm. Class III HDACs are a structurally distinct form of NAD-dependent enzymes correlated with the SIR2 yeast protein. Class IV (HDAC 11) consists at the moment of a single enzyme having particular structural characteristics. The HDACs of classes I, II and IV are zinc enzymes and can be inhibited by various classes of molecule: hydroxamic acid derivatives, cyclic tetrapeptides , short-chain fatty acids, aminobenzamides , derivatives of electrophilic ketones, and the like. Class III HDACs are not inhibited by hydroxamic acids, and their inhibitors have structural characteristics different from those of the other classes .
The expression “histone deacetylase inhibitor” in relation to the present invention is to be understood as meaning any molecule of natural, recombinant or synthetic origin capable of inhibiting the activity of at least one of the enzymes classified as histone deacetylases of class I, class II or class IV.
Although HDAC inhibitors, as a class, are considered to be potentially useful as anti-tumor agents, it is worth to note that, till now, only two of them (Vorinostat and Romidepsin) have been approved as drugs for the cure of a single tumor form (Cutaneous T-cell lymphoma ) .
It is evident that the pharmaceutical properties of each HDAC inhibitor may be different and depend on the specific profile of inhibitory potency, relative to the diverse iso-enzymes as well as on the particular pharmacokinetic behaviour and tissue distribution.
Some HDAC inhibitors have been claimed to be potentially useful, in combination with other agents, for the treatment of DMD (WO 2003/033678, WO 2004/050076, Consalvi S. et al. Histone Deacetylase Inhibitors in the Treatment of Muscular Dystrophies: Epigenetic Drugs for Genetic Diseases. (2011) Mol. Med. 17 : 457-465 ) .
The potential therapeutic use of HDAC inhibitors in DMD may however be hampered by the possible harmful effects of these relatively toxic agents, especially when used for long-term therapies in paediatric patients .
Givinostat, as anti-inflammatory agent, has been already used in a phase II study in children with Systemic Onset Juvenile Idiopathic Arthritis; Givinostat administered at 1.5 mg/kg/day for twelve weeks achieved ACR Pedi 30, 50 and 70 improvement of approximately 70% (Vojinovic J, Nemanja D. (2011) HDAC Inhibition in Rheumatoid Arthritis and Juvenile Idiopathic Arthritis. Mol. Med 17:397-403) showing only a limited number of mild or moderate but short lasting, adverse effects.
To date more than 500 patients (including 29 children) have been treated with Givinostat. Repeated dose toxicity studies were carried out in dogs, rats and monkeys. Oral daily doses of the drug were administered up to nine consecutive months. The drug was well tolerated with no overt toxicity at high doses. The “no adverse effect levels” (NOAEL) ranged from 10 to 25 mg/kg/day depending on the animal species and the duration of treatment.
In juvenile animals Givinostat at 60 mg/kg/day did not affect the behavioural and physical development and reproductive performance of pups.
No genotoxic effect was detected for Givinostat in the mouse lymphoma assay and the chromosomal aberration assay in vitro and in the micronucleus test and UDS test in vivo.
| Patent | Submitted | Granted |
|---|---|---|
| Monohydrate hydrochloride of the 4-hydroxycarbamoyl-phenyl)-carbamic acid (6-diethylaminomethyl-naphtalen-2-yl) ester [US7329689] | 2005-11-03 | 2008-02-12 |
Adverse effects
In clinical trials of givinostat as a salvage therapy for advanced Hodgkin’s lymphoma, the most common adverse reactions were fatigue (seen in 50% of participants), mild diarrhea or abdominal pain (40% of participants), moderate thrombocytopenia (decreased platelet counts, seen in one third of patients), and mild leukopenia (a decrease in white blood cell levels, seen in 30% of patients). One-fifth of patients experienced prolongation of the QT interval, a measure of electrical conduction in the heart, severe enough to warrant temporary suspension of treatment.[8]
Mechanism of action
Givinostat inhibits class I and class II histone deacetylases (HDACs) and several pro-inflammatory cytokines. This reduces expression of tumour necrosis factor (TNF), interleukin 1α and β, and interleukin 6.[7]
It also has activity against cells expressing JAK2(V617F), a mutated form of the janus kinase 2 (JAK2) enzyme that is implicated in the pathophysiology of many myeloproliferative diseases, including polycythaemia vera.[9][10] In patients with polycythaemia, the reduction of mutant JAK2 concentrations by givinostat is believed to slow down the abnormal growth of erythrocytes and ameliorate the symptoms of the disease.[5]
………………….

PATENT
https://www.google.com/patents/WO2004065355A1?cl=en
Hydrochloride of (6-diethylaminomethyl-naphthalen-2-yl)- methyl ester of (4-hydroxycarbamoylphenyl)-carbamic acid (II)
has been described in US patent 6,034,096 as a derivative of hydroxamic acid having anti-inflammatory and immunosuppressive activity, probably owing to the ability thereof to inhibit the production of pro-inflammatory cyto ines. This compound is obtained according to
Example 12 of the above-mentioned patent as an anhydrous, amorphous, hygroscopic, deliquescent solid which is difficult to handle.
crystalline form of monohydrous hydrochloride of
(6-diethylaminomethyl-naphthalen-2-yl)-methyl ester of
(4~hydroxycarbamoylphenyl)-carbamic acid (I).
This form is particularly advantageous from the industrial perspective because it is stable and simpler to handle than the anhydrous and amorphous form described above.
………………

PATENT
http://www.google.co.in/patents/US7329689
Hydrochloride of (6-diethylaminomethyl-naphthalen-2-yl)-methyl ester of (4-hydroxycarbamoylphenyl)-carbamic acid (II)
has been described in U.S. Pat. No. 6,034,096 as a derivative of hydroxamic acid having anti-inflammatory and immunosuppressive activity, probably owing to the ability thereof to inhibit the production of pro-inflammatory cytokines. This compound is obtained according to Example 12 of the above-mentioned patent as an anhydrous, amorphous, hygroscopic, deliquescent solid which is difficult to handle.
The 4-(6-diethylaminomethyl-naphthalen-2-ylmethoxycarbonylamino)-benzoic acid can be prepared as described in Example 12, point C, of U.S. Pat. No. 6,034,096.
The acid (1.22 kg, 3 moles) was suspended in THF (19 l) and the mixture was agitated under nitrogen over night at ambient temperature. The mixture was then cooled to 0° C. and thionyl chloride (0.657 l, 9 moles) was added slowly, still under nitrogen, with the temperature being maintained below 10° C. The reaction mixture was heated under reflux for 60 minutes, DMF (26 ml) was added and the mixture was further heated under reflux for 60 minutes.
The solvent was evaporated under vacuum, toluene was added to the residue and was then evaporated. This operation was repeated twice, then the residue was suspended in THF (11.5 l) and the mixture was cooled to 0° C.
The mixture was then poured into a cold solution of hydroxylamine (50% aq., 1.6 l, 264 moles) in 5.7 l of water. The mixture was then cooled to ambient temperature and agitated for 30 minutes. 6M HCl was added until pH 2 was reached and the mixture was partially evaporated under vacuum in order to eliminate most of the THF. The solid was filtered, washed repeatedly with water and dissolved in a solution of sodium bicarbonate (2.5%, 12.2 l). The solution was extracted with 18.6 l of a mixture of THF and ethyl acetate (2:1 v/v). 37% HCl (130 ml) were added to the organic layer in order to precipitate the monohydrate of the (6-diethylaminomethyl-naphthalen-2-yl)-methyl ester hydrochloride of the (4-hydroxycarbamoyl-phenyl)-carbamic acid. If necessary, this operation can be repeated several times to remove any residues of the original acid.
Finally, the solid was dried under vacuum (approximately 30 mbar, 50° C.), producing 0.85 kg (60%) of compound (I).
HPLC purity: 99.5%; water content (Karl Fischer method): 3.8%; (argentometric) assay: 99.8%.
| Elemental analysis | |||||
| C % | H % | Cl % | N % | ||
| Calculated for | 60.56 | 6.35 | 7.45 | 8.83 | |
| C24H30ClN3O5 | |||||
| Found | 61.06 | 6.48 | 7.48 | 8.90 | |
…..
PATENT
http://www.google.co.in/patents/US20120302633
The hydrochloride of the (4-hydroxycarbamoyl-phenyl)-carbamic acid (6-dimethylamino methyl-2-naphtalenyl) ester, also known as ITF 2357 and having the International Non Proprietary Name (INN) of Givinostat® is an organic compound with immunosuppressive and anti-inflammatory activity,
…………………..
http://www.google.com/patents/US6034096
EXAMPLE 12
4-[6-(Diethylaminomethyl)naphth-2-ylmethyloxycarbamoyl]-benzohydroxamic acid hydrochloride
A. 1-(3-Dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDCI) (22.2 g, 115 mmol) was added to a solution of 2,6-naphthalenedicarboxylic acid (25 g, 115 mmol) and hydroxybenzotriazole (15.6 g, 115 mmol) in dimethylformamide (1800 ml) and the mixture was stirred at room temperature for 2 hours. Diethyl amine (34.3 ml, 345 mmol) was added and the solution was stirred overnight at room temperature. The solvent was then evaporated under reduced pressure and the crude was treated with 1N HCl (500 ml) and ethyl acetate (500 ml), insoluble compounds were filtered off and the phases were separated. The organic phase was extracted with 5% sodium carbonate (3×200 ml) and the combined aqueous solutions were acidified with concentrated HCl and extracted with ethyl acetate (3×200 ml). The organic solution was then washed with 1N HCl (6×100 ml), dried over anhydrous sodium sulphate and the solvent was removed under reduced pressure yielding 18.5 g (Yield 60%) of pure 6-(diethylaminocarbonyl)-2-naphthalenecarboxylic acid; m.p.=122-124° C.
1 H-NMR d 8.67 (s, 1H), 8.25-8.00 (m, 4H), 7.56 (d, 1H), 3.60-3.20 (m, 4H), 1.30-1.00 (m, 6H).
B. A solution of 6-(diethylaminocarbonyl)-2- naphthalenecarboxylic acid (18 g, 66 mmol) in THF (200 ml) was slowly added to a refluxing suspension of lithium aluminium hydride (7.5 g, 199 mmol) in THF (500 ml). The mixture was refluxed for an hour, then cooled at room temperature and treated with a mixture of THF (25 ml) and water (3.5 ml), with 20% sodium hydroxide (8.5 ml) and finally with water (33 ml). The white solid was filtered off and the solvent was removed under reduced pressure. Crude was dissolved in diethyl ether (200 ml) and extracted with 1N HCl (3×100 ml). The aqueous solution was treated with 32% sodium hydroxide and extracted with diethyl ether (3×100 ml). The organic solution was dried over anhydrous sodium sulphate and the solvent was removed under reduced pressure yielding 12.7 g (79% yield) of pure 6-(diethylaminomethyl)-2-naphthalenemethanol as thick oil.
1 H-NMR d 7.90-7.74 (m, 4H), 7.49 (m, 2H), 5.32 (t, 1H, exchange with D2 O), 4.68 (d, 2H), 3.69 (s, 2H), 2.52 (q, 4H), 1.01 (t, 6H).
C. A solution of 6-(diethylaminomethyl)-2-naphthalene-methanol (12.5 g, 51 mmol) and N,N’-disuccinimidyl carbonate (13.2 g, 51 mmol) in acetonitrile (250 ml) was stirred at room temperature for 3 hours, then the solvent was removed and the crude was dissolved in THF (110 ml). This solution was added to a solution of 4-amino benzoic acid (7.1 g, 51 mmol) and sodium carbonate (5.5 g, 51 mmol) in water (200 ml) and THF (100 ml). The mixture was stirred overnight at room temperature, then THF was removed under reduced pressure and the solution was treated with 1N HCl (102 ml, 102 mmol). The precipitate was filtered, dried under reduced pressure, tritured in diethyl ether and filtered yielding 13.2 g (yield 64%) of pure 4-[6-(diethylaminomethyl)naphth-2-ylmethyloxycarbamoyl]-benzoic acid; m.p.=201-205° C. (dec.)
1 H-NMR d 10.26 (s, 1H), 8.13 (s, 1H), 8.05-7.75 (m, 6H), 7.63 (m, 3H), 5.40 (s, 2H), 4.32 (s, 2H), 2.98 (q, 4H), 1.24 (t, 6H).
D. A solution of 4-[6-(diethylaminomethyl)naphth-2-ylmethyloxycarbamoyl]benzoic acid (13.1 g, 32 mmol) and thionyl chloride (7 ml, 96 mmol) in chloroform (300 ml) was refluxed for 4 hours, then the solvent and thionyl chloride were evaporated. Crude was dissolved in chloroform (100 ml) and evaporated to dryness three times. Crude was added as solid to a solution of hydroxylamine hydrochloride (2.7 g, 39 mmol) and sodium bicarbonate (5.4 g, 64 mmol) and 1N sodium hydroxide (39 ml, 39 mmol) in water (150 ml) and THF (50 ml). The mixture was stirred overnight at room temperature, then THF was removed under reduced pressure and the aqueous phase was extracted with ethyl acetate (3×100 ml). The combined organic phases were dried over anhydrous sodium sulphate and the solvent was removed under reduced pressure. Crude was dissolved in THF and treated with a 1.5 N etheric solution of HCl. The solid product was filtered and dried yielding 6 g (yield 41%) of pure 4-[6-(diethylaminomethyl)naphth-2-ylmethyloxycarbamoyl]benzohydroxamic acid hydrochloride as white solid; m.p.=162-165° C., (dec.)
1 H-NMR d 11.24 (s, 1H, exchange with D2 O), 10.88 (s, 1H, exchange with D2 O), 10.16 (s, 1H), 8.98 (bs, 1H, exchange with D2 O), 8.21 (s, 1H), 8.10-7.97 (m, 3H), 7.89 (d, 1H), 7.80-7.55 (m, 5H), 5.39 (s, 2H), 4.48 (d, 2H), 3.09 (m, 4H), 1.30 (t, 6H).

Some nmr predictions
CAS NO. 497833-27-9, [6-(diethylaminomethyl)naphthalen-2-yl]methyl N-[4-(hydroxycarbamoyl)phenyl]carbamate H-NMR spectral analysis
![[6-(diethylaminomethyl)naphthalen-2-yl]methyl N-[4-(hydroxycarbamoyl)phenyl]carbamate NMR spectra analysis, Chemical CAS NO. 497833-27-9 NMR spectral analysis, [6-(diethylaminomethyl)naphthalen-2-yl]methyl N-[4-(hydroxycarbamoyl)phenyl]carbamate H-NMR spectrum](https://i0.wp.com/pic11.molbase.net/nmr/nmr_image/2015-01-20/001/566/1566912_1h.png)

13 C NMR PREDICTIONS
![[6-(diethylaminomethyl)naphthalen-2-yl]methyl N-[4-(hydroxycarbamoyl)phenyl]carbamate NMR spectra analysis, Chemical CAS NO. 497833-27-9 NMR spectral analysis, [6-(diethylaminomethyl)naphthalen-2-yl]methyl N-[4-(hydroxycarbamoyl)phenyl]carbamate C-NMR spectrum](https://i0.wp.com/pic11.molbase.net/nmr/nmr_image/2015-01-20/001/566/1566912_13c.png)

COSY NMR…..http://www.nmrdb.org/

HMBC /HSQC

References
- World Health Organization (2010). “International Nonproprietary Names for Pharmaceutical Substances (INN). Recommended INN: List 63”. WHO Drug Information 24 (1): 58–9.
- 2
- National Cancer Institute (2010). “Gavinostat”. NCI Cancer Dictionary. U.S. National Institutes of Health. Retrieved 2010-09-15.
- 3
- “Search results for ITF2357”. ClinicalTrials.gov.
- 4
- Committee for Orphan Medicinal Products (23 February 2010). “Public summary of opinion on orphan designation: Givinostat for the treatment of systemic-onset juvenile idiopathic arthritis”. European Medicines Agency. Retrieved 2010-09-15.
- 5
- Committee for Orphan Medicinal Products (3 March 2010). “Public summary of opinion on orphan designation: Givinostat for the treatment of polycythaemia vera”. European Medicines Agency.
- 6
- WO patent application 1997/043251, “Compounds with anti-inflammatory and immunosuppressive activities”, published 1997-11-20, assigned to Italfarmaco S.p.A.
- 7
- Leoni F, Fossati G. (2005). “The histone deacetylase inhibitor ITF2357 reduces production of pro-inflammatory cytokines in vitro and systemic inflammation in vivo”. Molecular Medicine 11: 1. doi:10.2119/2006-00005.Dinarello. PMID 16557334.
- 8
- Tan J, Cang S, Ma Y, Petrillo RL, Liu D (2010). “Novel histone deacetylase inhibitors in clinical trials as anti-cancer agents”. Journal of Hematology & Oncology 3: 5. doi:10.1186/1756-8722-3-5. PMID 20132536. Review.
- 9
- Vannucchi AM, Guglielmelli P, Pieri L, Antonioli E, Bosi A (2009). “Treatment options for essential thrombocythemia and polycythemia vera.” Expert Review of Hematology 2 (1): 41–55. doi:10.1586/17474086.2.1.41. Review.
- Guerini V, Barbui V, Spinelli O, et al. (April 2008). “The histone deacetylase inhibitor ITF2357 selectively targets cells bearing mutated JAK2(V617F)”. Leukemia 22 (4): 740–7. doi:10.1038/sj.leu.2405049. PMID 18079739.
Further reading
- Job-Deslandre, C (January 2007). “Idiopathic juvenile-onset systemic arthritis”. Orphanet. Orphan number: ORPHA85414.
| US6034096 | 12 May 1997 | 7 Mar 2000 | Italfarmaco S.P.A. | Compounds with anti-inflammatory and immunosuppressive activities |
| WO1997043251A1 | May 12, 1997 | Nov 20, 1997 | Italfarmaco Spa | Compounds with anti-inflammatory and immunosuppressive activities |
| WO2004063146A1 | Jan 7, 2004 | Jul 29, 2004 | Italfarmaco Spa | Hydroxamic acid derivatives having anti-inflammatory action |
| WO2004065355A1 | Jan 8, 2004 | Aug 5, 2004 | Italfarmaco Spa | Monohydrate hydrochloride of the 4-hydroxycarbamoyl-phenyl)-carbamic acid (6-diethylaminomethyl-naphtalen-2-yl) ester |
| WO2006003068A2 | Jun 7, 2005 | Jan 12, 2006 | Italfarmaco Spa | Alpha-amino acid derivatives with antiinflammatory activity |
| WO2008097654A1 | Feb 8, 2008 | Aug 14, 2008 | Nancie M Archin | Methods of using saha for treating hiv infection |
| Citing Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| US8518988 * | 3 Dec 2010 | 27 Aug 2013 | Chemi Spa | Polymorph of the hydrochloride of the (4-hydroxycarbamoyl-phenyl)-carbamic acid (6-dimethylamino methyl-2-naphthalenyl) ester |
| US20120302633 * | 3 Dec 2010 | 29 Nov 2012 | Chemi Spa | Novel polymorph of the hydrochloride of the (4-hydroxycarbamoyl-phenyl)-carbamic acid (6-dimethylamino methyl-2-naphthalenyl) ester |
| WO2011092556A1 | 3 Dec 2010 | 4 Aug 2011 | Chemi Spa | Novel polymorph of the hydrochloride of the (4-hydroxycarbamoyl-phenyl)-carbamic acid (6-dimethylamino methyl-2-naphtalenyl) ester |
 |
|
| Systematic (IUPAC) name | |
|---|---|
| {6-[(diethylamino)methyl]naphthalen-2-yl}methyl [4-(hydroxycarbamoyl)phenyl]carbamate | |
| Clinical data | |
|
|
| Legal status |
|
| Routes | Oral |
| Identifiers | |
| CAS number | 497833-27-9 |
| ATC code | None |
| PubChem | CID 9804992 |
| ChemSpider | 7980752 |
| UNII | 5P60F84FBH |
| Chemical data | |
| Formula | C24H27N3O4 |
| Molecular mass | 421.489 g/mol |

| Italfarmaco S.p.A. | |
|---|---|
| Stato | |
| Tipo | Società per azioni |
| Fondazione | 1938 a Milano |
| Fondata da | Gastone De Santis |
| Sede principale | Milano |
| Filiali | |
| Persone chiave | Francesco De Santis, [Presidente Holding] |
| Settore | sanità |
| Prodotti | Farmaci |
| Fatturato | >500 milioni di Euro (gruppo) (2011) |
| Dipendenti | >1900 (gruppo) (2011) |
| Sito web | www.italfarmaco.com |
MILAN ITALY


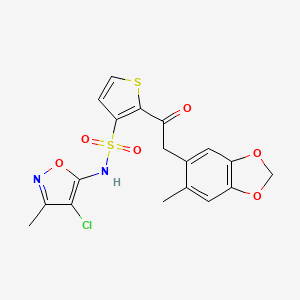
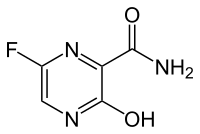
FAVIPIRAVIR, ファビピラビル

| Chemical Formula: | C5H4FN3O2 | |
| CAS #: | 259793-96-9 | |
| Molecular Weight: | 157.1 | |
ANTI-INFLUENZA COMPOUND |
||
| clinical trials | http://clinicaltrials.gov/search/intervention=Favipiravir | |
| Chemical Name: | 6-fluoro-3-hydroxy-2-pyrazinecarboxamide | |
| Synonyms: | T-705, T705, Favipiravir |
- Molecular FormulaC5H4FN3O2
- Average mass157.103 Da
6-Fluoro-3-hydroxypyrazine-2-carboxamide
C5H4FN3O2 : 157.1
[259793-96-9]
The drug substance is a white to light yellow powder. It is sparingly soluble in acetonitrile and in methanol, and slightly soluble in water and in ethanol (99.5). It is slightly soluble at pH 2.0 to 5.5 and sparingly soluble at pH 5.5 to 6.1. The drug substance is not hygroscopic at 25°C/51% to 93%RH. The melting point is 187°C to 193°C, and the dissociation constant (pKa) is 5.1 due to the hydroxyl group of favipiravir. Measurement results on the partition ratio of favipiravir in water/octanol at 25°C indicate that favipiravir tends to be distributed in the 1-octanol phase at pH 2 to 4 and in the water phase at pH 5 to 13.
Any batch manufactured by the current manufacturing process is in Form A. The stability study does not show any change in crystal form over time; and a change from Form A to Form B is unlikely.
Experimental Properties
| PROPERTY | VALUE | SOURCE |
|---|---|---|
| melting point (°C) | 187℃ to 193℃ | https://www.pmda.go.jp/files/000210319.pdf |
| water solubility | slightly soluble in water | https://www.pmda.go.jp/files/000210319.pdf |
| pKa | 5.1 | https://www.pmda.go.jp/files/000210319.pdf |
Favipiravir, also known as T-705, Avigan, or favilavir is an antiviral drug being developed by Toyama Chemical (Fujifilm group) of Japan with activity against many RNA viruses. Like certain other experimental antiviral drugs (T-1105 and T-1106), it is a pyrazinecarboxamide derivative. In experiments conducted in animals Favipiravir has shown activity against influenza viruses, West Nile virus, yellow fever virus, foot-and-mouth disease virus as well as other flaviviruses, arenaviruses, bunyaviruses and alphaviruses.[1]Activity against enteroviruses[2] and Rift Valley fever virus has also been demonstrated.[3] Favipiravir has showed limited efficacy against Zika virus in animal studies, but was less effective than other antivirals such as MK-608.[4] The agent has also shown some efficacy against rabies,[5] and has been used experimentally in some humans infected with the virus.[6]
In February 2020 Favipiravir was being studied in China for experimental treatment of the emergent COVID-19 (novel coronavirus)disease.[7][8] On March 17 Chinese officials suggested the drug had been effective in treating COVID in Wuhan and Shenzhen.[9][10]
Discovered by Toyama Chemical Co., Ltd. in Japan, favipiravir is a modified pyrazine analog that was initially approved for therapeutic use in resistant cases of influenza.7,9 The antiviral targets RNA-dependent RNA polymerase (RdRp) enzymes, which are necessary for the transcription and replication of viral genomes.7,12,13
Not only does favipiravir inhibit replication of influenza A and B, but the drug shows promise in the treatment of influenza strains that are resistant to neuramidase inhibitors, as well as avian influenza.9,19 Favipiravir has been investigated for the treatment of life-threatening pathogens such as Ebola virus, Lassa virus, and now COVID-19.10,14,15
Mechanism of action
The mechanism of its actions is thought to be related to the selective inhibition of viral RNA-dependent RNA polymerase.[11] Other research suggests that favipiravir induces lethal RNA transversion mutations, producing a nonviable viral phenotype.[12] Favipiravir is a prodrug that is metabolized to its active form, favipiravir-ribofuranosyl-5′-triphosphate (favipiravir-RTP), available in both oral and intravenous formulations.[13][14] Human hypoxanthine guanine phosphoribosyltransferase (HGPRT) is believed to play a key role in this activation process.[15] Favipiravir does not inhibit RNA or DNA synthesis in mammalian cells and is not toxic to them.[1] In 2014, favipiravir was approved in Japan for stockpiling against influenza pandemics.[16] However, favipiravir has not been shown to be effective in primary human airway cells, casting doubt on its efficacy in influenza treatment.[17]
Approval status
In 2014, Japan approved Favipiravir for treating viral strains unresponsive to current antivirals.[18]
In March 2015, the US Food and Drug Administration completed a Phase III clinical trial studying the safety and efficacy of Favipiravir in the treatment of influenza.[19]
Ebola virus trials
Some research has been done suggesting that in mouse models Favipiravir may have efficacy against Ebola. Its efficacy against Ebola in humans is unproven.[20][21][22] During the 2014 West Africa Ebola virus outbreak, it was reported that a French nurse who contracted Ebola while volunteering for MSF in Liberia recovered after receiving a course of favipiravir.[23] A clinical trial investigating the use of favipiravir against Ebola virus disease was started in Guéckédou, Guinea, during December 2014.[24] Preliminary results showed a decrease in mortality rate in patients with low-to-moderate levels of Ebola virus in the blood, but no effect on patients with high levels of the virus, a group at a higher risk of death.[25] The trial design has been criticised by Scott Hammer and others for using only historical controls.[26] The results of this clinical trial were presented in February 2016 at the annual Conference on Retroviruses and Opportunistic Infections (CROI) by Daouda Sissoko[27] and published on March 1, 2016 in PLOS Medicine.[28]
SARS-CoV-2 virus disease
In March 2020, Chinese officials suggested Favipiravir may be effective in treating COVID-19.[29]
SYN
https://link.springer.com/article/10.1007/s11696-018-0654-9

Electronic supplementary material
Ref
https://pdfs.semanticscholar.org/be8e/cb882b99204983d2f60077c7ab8b53f4d62c.pdf
Drug Discoveries & Therapeutics. 2014; 8(3):117-120.
As a RNA polymerase inhibitor, 6-fluoro-3-hydroxypyrazine-2-carboxamide commercially named favipiravir has been proved to have potent inhibitory activity against RNA viruses in vitro and in vivo. A four-step synthesis of the compound is described in this article, amidation, nitrification, reduction and fluorination with an overall yield of about 8%. In addition, we reported the crystal structure of the title compound. The molecule is almost planar and the intramolecular O−H•••O hydrogen bond makes a 6-member ring. In the crystal, molecules are packing governed by both hydrogen bonds and stacking interactions.


2.2.1. Preparation of 3-hydroxypyrazine-2-carboxamide To a suspension of 3-hydroxypyrazine-2-carboxylic acid (1.4 g, 10 mmol) in 150 mL MeOH, SOCl2 was added dropwise at 40°C with magnetic stirring for 6 h resulting in a bright yellow solution. The reaction was then concentrated to dryness. The residue was dissolved in 50 mL 25% aqueous ammonia and stirred overnight to get a suspension. The precipitate was collected and dried. The solid yellow-brown crude product was recrystallization with 50 mL water to get the product as pale yellow crystals (1.1 g, 78%). mp = 263-265°C. 1 H-NMR (300 MHz, DMSO): δ 13.34 (brs, 1H, OH), 8.69 (s, 1H, pyrazine H), 7.93-8.11 (m, 3H, pyrazine H, CONH2). HRMS (ESI): m/z [M + H]+ calcd for C5H6N3O2 + : 140.0460; found: 140.0457.
2.2.2. Preparation of 3-hydroxy-6-nitropyrazine-2- carboxamide In the solution of 3-hydroxypyrazine-2-carboxamide (1.0 g, 7 mmol) in 6 mL concentrate sulfuric acid under ice-cooling, potassium nitrate (1.4 g, 14 mmol) was added. After stirring at 40°C for 4 h, the reaction mixture was poured into 60 mL water. The product was collected by fi ltration as yellow solid (0.62 g, 48%). mp = 250-252°C. 1 H-NMR (600 MHz, DMSO): δ 12.00- 15.00 (br, 1H, OH), 8.97 (s, 1H, pyrazine H), 8.32 (s, 1H, CONH2), 8.06 (s, 1H, CONH2). 13C-NMR (75 MHz, DMSO): δ 163.12, 156.49, 142.47, 138.20, 133.81. HRMS (ESI): m/z [M + H]+ calcd for C5H5N4O4 + : 185.0311; found: 185.0304.
2.2.3. Preparation of 6-amino-3-hydroxypyrazine-2- carboxamide 3-Hydroxy-6-nitropyrazine-2-carboxamide (0.6 g, 3.3 mmol) and a catalytic amount of raney nickel were suspended in MeOH, then hydrazine hydrate was added dropwise. The resulting solution was refl uxed 2 h, cooled, filtered with diatomite, and then MeOH is evaporated in vacuo to get the crude product as dark brown solid without further purification (0.4 g, 77%). HRMS (ESI): m/z [M + H]+ calcd for C5H7N4O2 + : 155.0569; found:155.0509.
2.2.4. Preparation of 6-fluoro-3-hydroxypyrazine-2- carboxamide To a solution of 6-amino-3-hydroxypyrazine-2- carboxamide (0.4 g, 2.6 mmol) in 3 mL 70% HFpyridine aqueous at -20°C under nitrogen atmosphere, sodium nitrate (0.35 g, 5.2 mmol) was added. After stirring 20 min, the solution was warmed to room temperature for another one hour. Then 20 mL ethyl acetate/water (1:1) were added, after separation of the upper layer, the aqueous phase is extracted with four 20 mL portions of ethyl acetate. The combined extracts are dried with anhydrous magnesium sulfate and concentrated to dryness to get crude product as oil. The crude product was purified by chromatography column as white solid (0.12 g, 30%). mp = 178-180°C. 1 H-NMR (600 MHz, DMSO): δ 12.34 (brs, 1H, OH), 8.31 (d, 1H, pyrazine H, J = 8.0 Hz), 7.44 (s, 1H, CONH2), 5.92 (s, 1H, CONH2). 13C-NMR (75 MHz, DMSO): δ 168.66, 159.69, 153.98, 150.76, 135.68. HRMS (ESI): m/z [M + H]+ calcd for C5H5FN3O2 + : 158.0366; found: 158.0360.
SEE
Chemical Papers (2019), 73(5), 1043-1051.
PAPER
Medicinal chemistry (Shariqah (United Arab Emirates)) (2018), 14(6), 595-603
http://www.eurekaselect.com/158990/article
PATENT
CN 107641106
PAPER
Chemical Papers (2017), 71(11), 2153-2158.
https://link.springer.com/article/10.1007%2Fs11696-017-0208-6

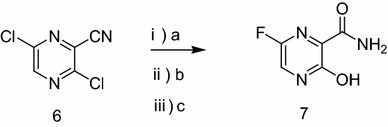
Below is the link to the electronic supplementary material.
References
- Furuta, Y.; Takahashi, K.; Shiraki, K.; Sakamoto, K.; Smee, D. F.; Barnard, D. L.; Gowen, B. B.; Julander, J. G.; Morrey, J. D. (2009). “T-705 (favipiravir) and related compounds: Novel broad-spectrum inhibitors of RNA viral infections”. Antiviral Research 82 (3): 95–102. doi:10.1016/j.antiviral.2009.02.198. PMID 19428599. edit

- WO 2000010569
- WO 2008099874
- WO 201009504
- WO 2010104170
- WO 2012063931





CLIP
Influenza virus is a central virus of the cold syndrome, which has attacked human being periodically to cause many deaths amounting to tens millions. Although the number of deaths shows a tendency of decrease in the recent years owing to the improvement in hygienic and nutritive conditions, the prevalence of influenza is repeated every year, and it is apprehended that a new virus may appear to cause a wider prevalence.
For prevention of influenza virus, vaccine is used widely, in addition to which low molecular weight substances such as Amantadine and Ribavirin are also used
CLIP
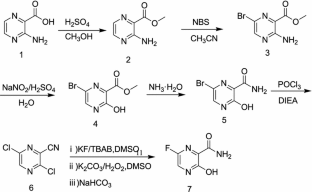
Synthesis of Favipiravir
ZHANG Tao1, KONG Lingjin1, LI Zongtao1,YUAN Hongyu1, XU Wenfang2*
(1. Shandong Qidu PharmaceuticalCo., Ltd., Linzi 255400; 2. School of Pharmacy, Shandong University, Jinan250012)
ABSTRACT: Favipiravir was synthesized from3-amino-2-pyrazinecarboxylic acid by esterification, bromination with NBS,diazotization and amination to give 6-bromo-3-hydroxypyrazine-2-carboxamide,which was subjected to chlorination with POCl3, fluorination with KF, andhydrolysis with an overall yield of about 22%.
PATENT
US6787544

| subs G1 | G2 | G3 | G4 | R2 |
| compd 32 N | CH | C—CF3 | N | H |
…………………
EP2192117
Example 1-1
To a 17.5 ml N,N-dimethylformamide solution of 5.0 g of 3,6-difluoro-2-pyrazinecarbonitrile, a 3.8 ml water solution of 7.83 g of potassium acetate was added dropwise at 25 to 35° C., and the solution was stirred at the same temperature for 2 hours. 0.38 ml of ammonia water was added to the reaction mixture, and then 15 ml of water and 0.38 g of active carbon were added. The insolubles were filtered off and the filter cake was washed with 11 ml of water. The filtrate and the washing were joined, the pH of this solution was adjusted to 9.4 with ammonia water, and 15 ml of acetone and 7.5 ml of toluene were added. Then 7.71 g of dicyclohexylamine was added dropwise and the solution was stirred at 20 to 30° C. for 45 minutes. Then 15 ml of water was added dropwise, the solution was cooled to 10° C., and the precipitate was filtered and collected to give 9.44 g of dicyclohexylamine salt of 6-fluoro-3-hydroxy-2-pyradinecarbonitrile as a lightly yellowish white solid product.
1H-NMR (DMSO-d6) δ values: 1.00-1.36 (10H, m), 1.56-1.67 (2H, m), 1.67-1.81 (4H, m), 1.91-2.07 (4H, m), 3.01-3.18 (2H, m), 8.03-8.06 (1H, m), 8.18-8.89 (1H, broad)
Example 1-2
4.11 ml of acetic acid was added at 5 to 15° C. to a 17.5 ml N,N-dimethylformamide solution of 5.0 g of 3,6-difluoro-2-pyrazinecarbonitrile. Then 7.27 g of triethylamine was added dropwise and the solution was stirred for 2 hours. 3.8 ml of water and 0.38 ml of ammonia water were added to the reaction mixture, and then 15 ml of water and 0.38 g of active carbon were added. The insolubles were filtered off and the filter cake was washed with 11 ml of water. The filtrate and the washing were joined, the pH of the joined solution was adjusted to 9.2 with ammonia water, and 15 ml of acetone and 7.5 ml of toluene were added to the solution, followed by dropwise addition of 7.71 g of dicyclohexylamine. Then 15 ml of water was added dropwise, the solution was cooled to 5° C., and the precipitate was filtered and collected to give 9.68 g of dicyclohexylamine salt of 6-fluoro-3-hydroxy-2-pyrazinecarbonitrile as a slightly yellowish white solid product.
Examples 2 to 5
The compounds shown in Table 1 were obtained in the same way as in Example 1-1.
| TABLE 1 | |||
 |
|||
| Example No. | Organic amine | Example No. | Organic amine |
| 2 | Dipropylamine | 4 | Dibenzylamine |
| 3 | Dibutylamine | 5 | N-benzylmethylamine |
Dipropylamine salt of 6-fluoro-3-hydroxy-2-pyrazinecarbonitrile
1H-NMR (DMSO-d6) 6 values: 0.39 (6H, t, J=7.5 Hz), 1.10 (4H, sex, J=7.5 Hz), 2.30-2.38 (4H, m), 7.54 (1H, d, J=8.3 Hz)
Dibutylamine salt of 6-fluoro-3-hydroxy-2-pyrazinecarbonitrile
1H-NMR (DMSO-d6) 6 values: 0.36 (6H, t, J=7.3 Hz), 0.81 (4H, sex, J=7.3 Hz), 0.99-1.10 (4H, m), 2.32-2.41 (4H, m), 7.53 (1H, d, J=8.3 Hz)
Dibenzylamine salt of 6-fluoro-3-hydroxy-2-pyrazinecarbonitrile
1H-NMR (DMSO-d6) δ values: 4.17 (4H, s), 7.34-7.56 (10H, m), 8.07 (1H, d, J=8.3 Hz)
N-benzylmethylamine salt of 6-fluoro-3-hydroxy-2-pyrazinecarbonitrile
1H-NMR (DMSO-d6) δ values: 2.57 (3H, s), 4.14 (2H, s), 7.37-7.53 (5H, m), 8.02-8.08 (1H, m)
Preparation Example 1
300 ml of toluene was added to a 600 ml water solution of 37.5 g of sodium hydroxide. Then 150 g of dicyclohexylamine salt of 6-fluoro-3-hydroxy-2-pyrazinecarbonitrile was added at 15 to 25° C. and the solution was stirred at the same temperature for 30 minutes. The water layer was separated and washed with toluene, and then 150 ml of water was added, followed by dropwise addition of 106 g of a 30% hydrogen peroxide solution at 15 to 30° C. and one-hour stirring at 20 to 30° C. Then 39 ml of hydrochloric acid was added, the seed crystals were added at 40 to 50° C., and 39 ml of hydrochloric acid was further added dropwise at the same temperature. The solution was cooled to 10° C. the precipitate was filtered and collected to give 65.6 g of 6-fluoro-3-hydroxy-2-pyrazinecarboxamide as a slightly yellowish white solid.
1H-NMR (DMSO-d6) δ values: 8.50 (1H, s), 8.51 (1H, d, J=7.8 Hz), 8.75 (1H, s), 13.41 (1H, s)
CLIP
jan 2014
First patient enrolled in the North American Phase 3 clinical trials for investigational flu treatment drug
Eur J Med Chem. 2013 Apr;62:534-44. doi: 10.1016/j.ejmech.2013.01.015. Epub 2013 Jan 29.
| US3631036 * | Nov 4, 1969 | Dec 28, 1971 | American Home Prod | 5-amino-2 6-substituted-7h-pyrrolo(2 3-d) pyrimidines and related compounds |
| US3745161 * | Apr 20, 1970 | Jul 10, 1973 | Merck & Co Inc | Phenyl-hydroxy-pyrazine carboxylic acids and derivatives |
| US4404203 * | May 14, 1981 | Sep 13, 1983 | Warner-Lambert Company | Substituted 6-phenyl-3(2H)-pyridazinones useful as cardiotonic agents |
| US4545810 * | Mar 25, 1983 | Oct 8, 1985 | Sds Biotech Corporation | Herbicidal and plant growth regulant diphenylpyridazinones |
| US4565814 * | Jan 18, 1984 | Jan 21, 1986 | Sanofi | Pyridazine derivatives having a psychotropic action and compositions |
| US4661145 * | Sep 20, 1984 | Apr 28, 1987 | Rohm And Haas Company | Plant growth regulating 1-aryl-1,4-dihydro-4-oxo(thio)-pyridazines |
| US5420130 | May 16, 1994 | May 30, 1995 | Synthelabo | 2-aminopyrazine-5-carboxamide derivatives, their preparation and their application in therapeutics |
| US5459142 * | Aug 23, 1993 | Oct 17, 1995 | Otsuka Pharmaceutical Co., Ltd. | Pyrazinyl and piperazinyl substituted pyrazine compounds |
| US5597823 | Jun 5, 1995 | Jan 28, 1997 | Abbott Laboratories | Tricyclic substituted hexahydrobenz [e]isoindole alpha-1 adrenergic antagonists |
| US6159980 * | Sep 15, 1997 | Dec 12, 2000 | Dupont Pharmaceuticals Company | Pyrazinones and triazinones and their derivatives thereof |
| EP0023358A1 * | Jul 28, 1980 | Feb 4, 1981 | Rohm And Haas Company | Process for the preparation of pyridazine derivatives |
| GB1198688A | Title not available | |||
| HU9401512A | Title not available | |||
| JPH09216883A * | Title not available | |||
| JPS5620576A | Title not available |
- ^ Jump up to:a b Furuta Y, Takahashi K, Shiraki K, Sakamoto K, Smee DF, Barnard DL, Gowen BB, Julander JG, Morrey JD (June 2009). “T-705 (favipiravir) and related compounds: Novel broad-spectrum inhibitors of RNA viral infections”. Antiviral Research. 82 (3): 95–102. doi:10.1016/j.antiviral.2009.02.198. PMID 19428599.
- ^ Furuta Y, Gowen BB, Takahashi K, Shiraki K, Smee DF, Barnard DL (November 2013). “Favipiravir (T-705), a novel viral RNA polymerase inhibitor”. Antiviral Research. 100 (2): 446–54. doi:10.1016/j.antiviral.2013.09.015. PMC 3880838. PMID 24084488.
- ^ Caroline AL, Powell DS, Bethel LM, Oury TD, Reed DS, Hartman AL (April 2014). “Broad spectrum antiviral activity of favipiravir (T-705): protection from highly lethal inhalational Rift Valley Fever”. PLoS Neglected Tropical Diseases. 8 (4): e2790. doi:10.1371/journal.pntd.0002790. PMC 3983105. PMID 24722586.
- ^ Mumtaz N, van Kampen JJ, Reusken CB, Boucher CA, Koopmans MP (2016). “Zika Virus: Where Is the Treatment?”. Current Treatment Options in Infectious Diseases. 8 (3): 208–211. doi:10.1007/s40506-016-0083-7. PMC 4969322. PMID 27547128.
- ^ Yamada K, Noguchi K, Komeno T, Furuta Y, Nishizono A (April 2016). “Efficacy of Favipiravir (T-705) in Rabies Postexposure Prophylaxis”. The Journal of Infectious Diseases. 213 (8): 1253–61. doi:10.1093/infdis/jiv586. PMC 4799667. PMID 26655300.
- ^ Murphy J, Sifri CD, Pruitt R, Hornberger M, Bonds D, Blanton J, Ellison J, Cagnina RE, Enfield KB, Shiferaw M, Gigante C, Condori E, Gruszynski K, Wallace RM (January 2019). “Human Rabies – Virginia, 2017”. MMWR. Morbidity and Mortality Weekly Report. 67(5152): 1410–1414. doi:10.15585/mmwr.mm675152a2. PMC 6334827. PMID 30605446.
- ^ Li G, De Clercq E. Therapeutic options for the 2019 novel coronavirus (2019-nCoV). Nature Reviews Drug Discovery 2020 Feb doi:10.1038/d41573-020-00016-0
- ^ BRIEF-Corrected-Zhejiang Hisun Pharma gets approval for clinical trial to test flu drug Favipiravir for pneumonia caused by new coronavirus. Reuters Healthcare, February 16, 2020.
- ^ [1]NHK World News ‘China: Avigan effective in tackling coronavirus’
- ^ Huaxia. Favipiravir shows good clinical efficacy in treating COVID-19: official. Xinhuanet.com, 17 March 2020
- ^ Jin Z, Smith LK, Rajwanshi VK, Kim B, Deval J (2013). “The ambiguous base-pairing and high substrate efficiency of T-705 (Favipiravir) Ribofuranosyl 5′-triphosphate towards influenza A virus polymerase”. PLOS ONE. 8 (7): e68347. Bibcode:2013PLoSO…868347J. doi:10.1371/journal.pone.0068347. PMC 3707847. PMID 23874596.
- ^ Baranovich T, Wong SS, Armstrong J, Marjuki H, Webby RJ, Webster RG, Govorkova EA (April 2013). “T-705 (favipiravir) induces lethal mutagenesis in influenza A H1N1 viruses in vitro”. Journal of Virology. 87 (7): 3741–51. doi:10.1128/JVI.02346-12. PMC 3624194. PMID 23325689.
- ^ Guedj J, Piorkowski G, Jacquot F, Madelain V, Nguyen TH, Rodallec A, et al. (March 2018). “Antiviral efficacy of favipiravir against Ebola virus: A translational study in cynomolgus macaques”. PLoS Medicine. 15 (3): e1002535. doi:10.1371/journal.pmed.1002535. PMC 5870946. PMID 29584730.
- ^ Smee DF, Hurst BL, Egawa H, Takahashi K, Kadota T, Furuta Y (October 2009). “Intracellular metabolism of favipiravir (T-705) in uninfected and influenza A (H5N1) virus-infected cells”. The Journal of Antimicrobial Chemotherapy. 64 (4): 741–6. doi:10.1093/jac/dkp274. PMC 2740635. PMID 19643775.
- ^ Naesens L, Guddat LW, Keough DT, van Kuilenburg AB, Meijer J, Vande Voorde J, Balzarini J (October 2013). “Role of human hypoxanthine guanine phosphoribosyltransferase in activation of the antiviral agent T-705 (favipiravir)”. Molecular Pharmacology. 84 (4): 615–29. doi:10.1124/mol.113.087247. PMID 23907213.
- ^ Koons C (7 August 2014). “Ebola Drug From Japan May Emerge Among Key Candidates”. Bloomberg.com.
- ^ Yoon JJ, Toots M, Lee S, Lee ME, Ludeke B, Luczo JM, et al. (August 2018). “Orally Efficacious Broad-Spectrum Ribonucleoside Analog Inhibitor of Influenza and Respiratory Syncytial Viruses”. Antimicrobial Agents and Chemotherapy. 62 (8): e00766–18. doi:10.1128/AAC.00766-18. PMC 6105843. PMID 29891600.
- ^ Hayden, Frederick. “Influenza virus polymerase inhibitors in clinical development”. Current Opinion in Infectious Diseases. doi:10.1097/QCO.0000000000000532.
- ^ “Phase 3 Efficacy and Safety Study of Favipiravir for Treatment of Uncomplicated Influenza in Adults – T705US316”. FDA. Retrieved 17 March 2020.
- ^ Gatherer D (August 2014). “The 2014 Ebola virus disease outbreak in West Africa”. The Journal of General Virology. 95 (Pt 8): 1619–24. doi:10.1099/vir.0.067199-0. PMID 24795448.
- ^ Oestereich L, Lüdtke A, Wurr S, Rieger T, Muñoz-Fontela C, Günther S (May 2014). “Successful treatment of advanced Ebola virus infection with T-705 (favipiravir) in a small animal model”. Antiviral Research. 105: 17–21. doi:10.1016/j.antiviral.2014.02.014. PMID 24583123.
- ^ Smither SJ, Eastaugh LS, Steward JA, Nelson M, Lenk RP, Lever MS (April 2014). “Post-exposure efficacy of oral T-705 (Favipiravir) against inhalational Ebola virus infection in a mouse model”. Antiviral Research. 104: 153–5. doi:10.1016/j.antiviral.2014.01.012. PMID 24462697.
- ^ “First French Ebola patient leaves hospital”. Reuters. 4 October 2016.
- ^ “Guinea: Clinical Trial for Potential Ebola Treatment Started in MSF Clinic in Guinea”. AllAfrica – All the Time. Retrieved 28 December 2014.
- ^ Fink S (4 February 2015). “Ebola Drug Aids Some in a Study in West Africa”. The New York Times.
- ^ Cohen J (26 February 2015). “Results from encouraging Ebola trial scrutinized”. Science. doi:10.1126/science.aaa7912. Retrieved 21 January 2016.
- ^ “Favipiravir in Patients with Ebola Virus Disease: Early Results of the JIKI trial in Guinea | CROI Conference”. croiconference.org. Retrieved 2016-03-17.
- ^ Sissoko D, Laouenan C, Folkesson E, M’Lebing AB, Beavogui AH, Baize S, et al. (March 2016). “Experimental Treatment with Favipiravir for Ebola Virus Disease (the JIKI Trial): A Historically Controlled, Single-Arm Proof-of-Concept Trial in Guinea”. PLoS Medicine. 13(3): e1001967. doi:10.1371/journal.pmed.1001967. PMC 4773183. PMID 26930627.
- ^ “Japanese flu drug ‘clearly effective’ in treating coronavirus, says China”. The Guardian. 2020-03-18. Retrieved 2020-03-18.\
- Beigel J, Bray M: Current and future antiviral therapy of severe seasonal and avian influenza. Antiviral Res. 2008 Apr;78(1):91-102. doi: 10.1016/j.antiviral.2008.01.003. Epub 2008 Feb 4. [PubMed:18328578]
- Hsieh HP, Hsu JT: Strategies of development of antiviral agents directed against influenza virus replication. Curr Pharm Des. 2007;13(34):3531-42. [PubMed:18220789]
- Gowen BB, Wong MH, Jung KH, Sanders AB, Mendenhall M, Bailey KW, Furuta Y, Sidwell RW: In vitro and in vivo activities of T-705 against arenavirus and bunyavirus infections. Antimicrob Agents Chemother. 2007 Sep;51(9):3168-76. Epub 2007 Jul 2. [PubMed:17606691]
- Sidwell RW, Barnard DL, Day CW, Smee DF, Bailey KW, Wong MH, Morrey JD, Furuta Y: Efficacy of orally administered T-705 on lethal avian influenza A (H5N1) virus infections in mice. Antimicrob Agents Chemother. 2007 Mar;51(3):845-51. Epub 2006 Dec 28. [PubMed:17194832]
- Furuta Y, Takahashi K, Kuno-Maekawa M, Sangawa H, Uehara S, Kozaki K, Nomura N, Egawa H, Shiraki K: Mechanism of action of T-705 against influenza virus. Antimicrob Agents Chemother. 2005 Mar;49(3):981-6. [PubMed:15728892]
- Furuta Y, Takahashi K, Fukuda Y, Kuno M, Kamiyama T, Kozaki K, Nomura N, Egawa H, Minami S, Watanabe Y, Narita H, Shiraki K: In vitro and in vivo activities of anti-influenza virus compound T-705. Antimicrob Agents Chemother. 2002 Apr;46(4):977-81. [PubMed:11897578]
- Furuta Y, Komeno T, Nakamura T: Favipiravir (T-705), a broad spectrum inhibitor of viral RNA polymerase. Proc Jpn Acad Ser B Phys Biol Sci. 2017;93(7):449-463. doi: 10.2183/pjab.93.027. [PubMed:28769016]
- Venkataraman S, Prasad BVLS, Selvarajan R: RNA Dependent RNA Polymerases: Insights from Structure, Function and Evolution. Viruses. 2018 Feb 10;10(2). pii: v10020076. doi: 10.3390/v10020076. [PubMed:29439438]
- Hayden FG, Shindo N: Influenza virus polymerase inhibitors in clinical development. Curr Opin Infect Dis. 2019 Apr;32(2):176-186. doi: 10.1097/QCO.0000000000000532. [PubMed:30724789]
- Madelain V, Nguyen TH, Olivo A, de Lamballerie X, Guedj J, Taburet AM, Mentre F: Ebola Virus Infection: Review of the Pharmacokinetic and Pharmacodynamic Properties of Drugs Considered for Testing in Human Efficacy Trials. Clin Pharmacokinet. 2016 Aug;55(8):907-23. doi: 10.1007/s40262-015-0364-1. [PubMed:26798032]
- Nguyen TH, Guedj J, Anglaret X, Laouenan C, Madelain V, Taburet AM, Baize S, Sissoko D, Pastorino B, Rodallec A, Piorkowski G, Carazo S, Conde MN, Gala JL, Bore JA, Carbonnelle C, Jacquot F, Raoul H, Malvy D, de Lamballerie X, Mentre F: Favipiravir pharmacokinetics in Ebola-Infected patients of the JIKI trial reveals concentrations lower than targeted. PLoS Negl Trop Dis. 2017 Feb 23;11(2):e0005389. doi: 10.1371/journal.pntd.0005389. eCollection 2017 Feb. [PubMed:28231247]
- de Farias ST, Dos Santos Junior AP, Rego TG, Jose MV: Origin and Evolution of RNA-Dependent RNA Polymerase. Front Genet. 2017 Sep 20;8:125. doi: 10.3389/fgene.2017.00125. eCollection 2017. [PubMed:28979293]
- Shu B, Gong P: Structural basis of viral RNA-dependent RNA polymerase catalysis and translocation. Proc Natl Acad Sci U S A. 2016 Jul 12;113(28):E4005-14. doi: 10.1073/pnas.1602591113. Epub 2016 Jun 23. [PubMed:27339134]
- Nagata T, Lefor AK, Hasegawa M, Ishii M: Favipiravir: a new medication for the Ebola virus disease pandemic. Disaster Med Public Health Prep. 2015 Feb;9(1):79-81. doi: 10.1017/dmp.2014.151. Epub 2014 Dec 29. [PubMed:25544306]
- Rosenke K, Feldmann H, Westover JB, Hanley PW, Martellaro C, Feldmann F, Saturday G, Lovaglio J, Scott DP, Furuta Y, Komeno T, Gowen BB, Safronetz D: Use of Favipiravir to Treat Lassa Virus Infection in Macaques. Emerg Infect Dis. 2018 Sep;24(9):1696-1699. doi: 10.3201/eid2409.180233. Epub 2018 Sep 17. [PubMed:29882740]
- Delang L, Abdelnabi R, Neyts J: Favipiravir as a potential countermeasure against neglected and emerging RNA viruses. Antiviral Res. 2018 May;153:85-94. doi: 10.1016/j.antiviral.2018.03.003. Epub 2018 Mar 7. [PubMed:29524445]
- Nature Biotechnology: Coronavirus puts drug repurposing on the fast track [Link]
- Pharmaceuticals and Medical Devices Agency: Avigan (favipiravir) Review Report [Link]
- World Health Organization: Influenza (Avian and other zoonotic) [Link]
 |
|
| Names | |
|---|---|
| IUPAC name
5-Fluoro-2-hydroxypyrazine-3-carboxamide
|
|
| Other names
T-705; Avigan; favilavir
|
|
| Identifiers | |
|
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
|
PubChem CID
|
|
| UNII | |
|
CompTox Dashboard (EPA)
|
|
| Properties | |
| C5H4FN3O2 | |
| Molar mass | 157.104 g·mol−1 |
| Pharmacology | |
| J05AX27 (WHO) | |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
////////////

Want to know everything on vir series
click
http://drugsynthesisint.blogspot.in/p/vir-series-hep-c-virus-22.html
AND
http://medcheminternational.blogspot.in/p/vir-series-hep-c-virus.html
WORLD DRUG TRACKER
one time
$10.00
PALINAVIR
PALINAVIR, BILA-2011-BS
| Patent | Submitted | Granted |
|---|---|---|
| Substituted pipecolinic acid derivatives as HIV protease inhibitors [US5614533] | 1997-03-25 | |
| Substituted pipecolinic acid derivatives as HIV protease inhibitors. [EP0560268] | 1993-09-15 | 1995-01-04 |

……………………….
PATENT
http://www.google.com/patents/WO2013105118A1?cl=en
Scheme 5: Synthesis of Palinavir (6):
The organic solvent mentioned according to the invention is selected from the group consisting of organic solvents, wherein the organic solvents are polar aprotic such as DCM, THF, Ethyl acetate, acetone, DMF, acetonitrile, DMSO ; polar protic solvents such as lower alcohol particularly (C1-C6) alkyl alcohol, water, acetic acid ; non-polar solvents such as hexane, benzene, toluene, chloroform, pet. ether, 1,4-dioxane, heptane either alone or mixtures thereof . Additionally the purification or separation of crude product can be accomplished by known techniques viz. extraction, column chromatography in a suitable organic solvent with the aid of instruments such as TLC, HPLC, GC, mass spectroscopy, or distillation, crystallization, derivatization.
Want to know everything on vir series
click
http://drugsynthesisint.blogspot.in/p/vir-series-hep-c-virus-22.html
AND
http://medcheminternational.blogspot.in/p/vir-series-hep-c-virus.html
………………………….
J Org Chem 1997,62(11),3440

The reaction of tert-butoxycarbonyl-L-phenylalanine (I) with isobutyl chloroformate in THF gives the expected mixed anhydride which is treated with diazomethane and HCl yielding the corresponding chloromethyl ketone (II). The reduction of (II) with NaBH4 in THF affords the (S)-chlorohydrin (IV), which is treated with KOH in ethanol to obtain the chiral epoxide (V)(1,2). Ring opening of (V) with (?(cis)-N-tert-butyl-4-(4-pyridylmethoxy)piperidine-2-carboxamide (VI) by a treatment with LiCl in refluxing ethanol gives a mixture of diastereomers that is separated by chromatography giving the pure isomer (VII). The reaction of (VII) with tert-butoxycarbonyl-L-valine (VIII) by treatment first with trifluoroacetic acid (TFA), and condesation by means of BOP ((benzotriazol-1-yloxy)tris(dimethylamino)phosphonium hexafluorophosphate) and NMM (N-methylmorpholine) affords the expected condensation product (IX). Finally, this compound is condensed with quinoline-2-carboxylic acid (X) by means of BOP and NMM as before. 2) The piperidine (VI) has been obtained by condensation of (?(cis)-N-(tert-butoxycarbonyl)-4-hydroxypiperidine-2-carboxamide (XI) with 4-(chloromethyl)pyridine (XII) by means of NaH in DMS, followed by hydrolysis with HCl.

Palinavir can also be obtained as follows: The controlled oxidation of 2(S)-(dibenzylamino)-3-phenyl-1-propanol (XIII) with pyridine-SO3 complex in DMSO gives the corresponding aldehyde (XIV), which is condensed with bromochloromethane (XV) by means of Li in THF followed by hydrolysis with HCl yielding regioselectively the 1-chloro-2-butanol (XVI). The debenzylation of (XVI) by hydrogenation over Pd/C affords the free amine (XVII), which is treated with tert-butoxycarbonyl anhydride/triethylamine and dehydrochlorinated with KOH in methanol to give the desired chiral epoxide (V).

The chiral piperidine (2S,4R)(VI) has been obtained as follows: The cyclization of 3-buten-1-ol (XXII) with (S)-1-phenylethylamine (XXIII) and glyoxylic acid (XXIV) by means of tosyl chloride in THF gives a mixture of the (2S,4R) and (2R,4S) lactones (XXV), which is resolved by fractional crystallyzation of their salts with the chiral camphorsulfonic acid (XXVI), followed by elimination of the acid with ammonia to afford (2S,4R)(XXVII). The reaction of lactone (XXVII) with isopropylmagnesium chloride and tert-butylamine in THF gives (2S,4R)-N-tert-butyl-4-hydroxy-1-(1(S)-phenylethyl)piperidine-2-carboxamide (XXVIII), which is debenzylated by hydrogenation and protected with tert-butoxycarbonyl anhydride yielding (2S,4R)-N-(tert-butoxycarbonyl)-4-hydroxypiperidine-2-carboxamide (2S,4R)(XI), which is finally condensed with 4-(chloromethyl)pyridine (XII) as before to obtain the chiral piperidine (2S,4R)(VI), already reported.

The condendsation of epoxide (V) with (2S,4R)(VI) by means of basic alumina in THF, followed by elimination of the protecting group with HCl and NaOH yields directly the condensation product (XVIII) as a pure diastereomer and with a free amino group. Finally, this compound is condensed with N-(2-quinolylcarbonyl)-L-valine (XIX) through its activation compound with isobutyl chloroformate (the 4(S)-isopropyl-2-(2-quinolyl)oxazol-5(4H)-one (XX)). The N-acyl-L-valine (XIX) has been obtained by acylation of L-valine (XXI) with quinoline-2-carboxylic acid (X) through its acyl chloride obtained with SOCl2.
………………………..

Palinavir is an inhibitor with five chiral centers. It contains the amino acid valine and pipecolinin acid. The previous way to create this drug faced three major obstacles. First, the reaction from 2 to 3 used diazomethane. Therefore, is is difficult, if not impossible, to produce large quantities. Secondly, the steps included in going from 4 to 5 gave way to racemers which is very inefficient. Finally, chromatography is needed at two separate times.

Four issues were addresses in route to product 1. First, because of the number of chiral centers, stereochemical control was a concern. high chemical yields were a second concern. Also, multi step procedures were advantageous to cut down on purification steps. Finally, the synthesis tried to restrict the use of hazardous reagents. The following retrosynthesis reaction was conceived and three target molecules were identified as seen in figure 1.

Molecule 3 uses a diaseteroselective addition of in situ (chloromethyl)lithium to N,N-dibenzylphenylalaninol and is derived from a four step process.

Recrystallization of 13 is required. Molecule 14 was not reached because it posed a problem later in the reaction. The N-benzyl protection group could not be removed to react with 9.

8 is a derivative of naturally occurring pipocolic acid, 16, named 3-buten-1-ol. Selective crystallization of diastereomeric salts can lead to 17a, but a more efficient way is by having a 60:40 mixture of lactones 17a,b. This leads to 18a,b using a Brodroux process. Crystallization of 18a,b lead to a poor overall yield. Instead, 18a,b undergoes salt crystallization with (-)-camphorsulfonic acid. Finally, 18a underwent hydrolysis and then addition of di-tert butyl dicarbonate leads to 8.
8 was then transformed to 5 in a three step process.
 8 was added to NaOH and alkylated with 4-picolyl chloride. The protecting group was lost with the addition of acid.
8 was added to NaOH and alkylated with 4-picolyl chloride. The protecting group was lost with the addition of acid.
 Derivation of 9 was started by a simple substitution of 19, quinoline-2-carboxylic acid, to 20, an acid chloride, with the help of thionyl chloride. Acylation of amino acid L-valine to 20 was accomplished by a biphasic system.
Derivation of 9 was started by a simple substitution of 19, quinoline-2-carboxylic acid, to 20, an acid chloride, with the help of thionyl chloride. Acylation of amino acid L-valine to 20 was accomplished by a biphasic system.
 In the original synthesis of palinavir, a 2:1 mixture of 3 to 5 was needed to produce only ~35% of 6 and flash chromatography was needed. On a large scale without chromatography, 6 was produced with a 85% yield, but 21 was also produced. To keep the production of 21 to a minimum, the reaction was performed in a solution that was degassed. This insured that the pyridine ring would not react in the presence of air. With this precaution, only 1-2% of the yield was 21. A washing of the solution with 1 M KH2PO4 removed and left over 5. Deprotection was achieved with the addition of concentrated HCl and followed by adding NaOH. The product of 10 was a “viscous syrup”. 22 was 1-1.5% of the product and was not removed before the addition of 9 to form 80-85% palinavir.
In the original synthesis of palinavir, a 2:1 mixture of 3 to 5 was needed to produce only ~35% of 6 and flash chromatography was needed. On a large scale without chromatography, 6 was produced with a 85% yield, but 21 was also produced. To keep the production of 21 to a minimum, the reaction was performed in a solution that was degassed. This insured that the pyridine ring would not react in the presence of air. With this precaution, only 1-2% of the yield was 21. A washing of the solution with 1 M KH2PO4 removed and left over 5. Deprotection was achieved with the addition of concentrated HCl and followed by adding NaOH. The product of 10 was a “viscous syrup”. 22 was 1-1.5% of the product and was not removed before the addition of 9 to form 80-85% palinavir.

Coupling of 10 and 9 is the final step in the synthesis , although there are still some purification steps left.

Two recrystallizations were required for the final 99.6% purity.

………………………..

Palinavir is a potent peptidomimetic-based HIV protease inhibitor. We have developed a highly convergent and stereoselective synthesis which is amenable to the preparation of multikilogram quantities of this compound. The synthetic sequence proceeds in 24 distinct chemical steps (with several integrated, multistep operations) from commercially available starting materials. No chromatographies are required throughout the process, and the final product is purified by crystallization of its dihydrochloride salt to >99% homogeneity.
crude palinavir (1) as a thick brown oil (yield not determined). HPLC analysis (Supelcosil LZ-ABZ, 10−50% 1% TFA in MeCN/1% TFA in 25 min, 1 mL/min flow rate): 1, tR 17.80 min (84.1%); 24, tR 18.47 min (2.0%); 25, tR 19.97 min (1.45%).
palinavir dihydrochloride (1750 g, 51% yield) containing 0.25% w/w isopropanol (by 1H NMR):
mp 175−185 °C.
[α]25D −13.0° (c 1, MeOH). [α]25Hg365 +44.9° (c 1, MeOH).
IR (KBr) ν 3700−2300, 1660, 1555, 1520 cm-1.
1H NMR (DMSO-d6) δ 10.00 (broad s, 1H), 8.88 (d, J = 6.3 Hz, 2H), 8.61 (d, J = 8.4 Hz, 1H), 8.60 (s, 1H), 8.51 (d, J = 9.6 Hz, 1H), 8.35 (d, J = 8.7 Hz, 1H), 8.20 (d, J = 8.4 Hz, 1H), 8.16 (d, J = 8.7 Hz, 1H), 8.11 (d, J = 8.1 Hz, 1H), 7.94 (d, J = 6.0 Hz, 2H), 7.89 (t, J = 7.6 Hz, 1H), 7.74 (t, J = 7.5 Hz, 1H), 7.19 (d, J = 7.2 Hz, 2H), 7.08 (t, J = 7.5 Hz, 2H), 6.91 (t, J = 7.3 Hz, 1H), 4.86 (AB quartet, 2H), 4.37 (broad t, J = 7.8 Hz, 1H), 4.21 (d, J = 11.4 Hz, 1H), 4.11 (broad m, 1H), 3.96 (broad m, 1H), 3.80−3.65 (m, 2H), 3.26 (t, J = 7.4 Hz, 1H), 3.15−3.01 (m, 2H), 2.94 (broad d, J = 12.0 Hz, 1H), 2.62 (dd, J = 13.6, 10.6 Hz, 1H), 2.56 ((broad d, J = 12.0 Hz, 1H), 2.20−2.05 (m, 2H), 1.86 (m, 1H), 1.69 (q, J = 11.7 Hz, 1H), 1.31 (s, 9H), 0.81 (d, J = 6.3 Hz, 3H), 0.80 (d, J = 6.6 Hz, 3H).
13C NMR (DMSO-d6) δ 170.4, 166.4, 163.3, 158.3, 149.5, 145.9, 141.9, 138.6, 138.2, 130.7, 129.3, 129.1, 129.0, 128.3, 128.2, 128.0, 125.9, 124.1, 118.6, 72.3, 68.8, 67.2, 64.8, 58.0, 57.8, 54.4, 51.3, 51.1, 35.4, 34.1, 31.1, 28.2, 19.5, 17.9.
FAB-MS m/z 709 (MH+ of free base). Anal. Calcd for C41H54Cl2N6O5 (corrected for 8% water content as determined by Karl Fisher analysis and 0.25% w/w isopropanol as determined by 1H NMR): C, 58.31; H, 7.29; N, 9.93. Found: C, 57.76; H, 7.25; N, 9.89. Titration of HCl content using NaOH: 2.09 ± 0.03 mol HCl. HPLC homogeneity (Supelcosil LC-ABZ, 10−50% 1% TFA in MeCN/1% TFA in 25 min, 1 mL/min flow rate): palinavir dihydrochloride, tR 18.24 min (99.51%); 25 tR 20.39 min (0.33%). HPLC homogeneity (Nova-Pak C8, 20−80% MeCN/50 mM NaH2PO4 in 25 min, 1 mL/min flow rate): palinavir dihydrochloride, tR 15.52 min (99.67%); 25 tR 13.52 min (0.33%).
PURE palinavir (1) as a white amorphous powder (1902 g, 84% yield):
mp 100−107 °C. [α]25D −11.5° (c 1, MeOH).
IR (KBr) ν 3700−3100, 1660, 1520, 1495 cm-1.
1H NMR (CDCl3) δ 8.54 (d, J = 5.7 Hz, 2H), 8.48 (d, J = 8.6 Hz, 1H), 8.31 (d, J = 8.6 Hz, 1H, part of AB), 8.22 (d, J = 8.3 Hz, 1H, part of AB), 8.13 (d, J = 8.3 Hz, 1H), 7.90 (d, J = 8.0 Hz, 1H), 7.80 (t, J = 7.6 Hz, 1H), 7.65 (t, J = 7.6 Hz, 1H), 7.25 (d, J = 5.4 Hz, 2H), 7.13 (d, J = 7.3 Hz, 2H), 7.07 (t, J = 7.5 Hz, 1H), 6.92 (t, J = 7.3 Hz, 1H), 6.59 (d, J = 8.3 Hz, 1H), 6.57 (s, 1H), 4.61 (d, J = 13.4 Hz, 1H, part of AB), 4.51 (d, J = 13.4 Hz, 1H, part of AB), 4.32 (dd, J = 8.6, 6.4 Hz, 1H), 4.22 (m, 1H), 3.97 (m, 1), 3.47−3.33 (m, 2H), 2.94 (dd, J = 14.3, 4.1 Hz, 1H), 2.89 (d, J= 8.6 Hz, 1H), 2.79−2.72 (m, 1H), 2.77 (dd, J = 14.3, 10.8 Hz, 1H), 2.43 (dd, J = 13.4, 8.3 Hz, 1H), 2.40−2.25 (m, 3H), 1.95 (broad d, J = 12.4 Hz, 1H), 1.65 (q J = 11.8 Hz, 2H), 1.32 (s, 9H), 0.95 (d, J = 7.0 Hz, 3H), 0.83 (d, J = 6.7 Hz, 3H).
13C NMR (CDCl3) δ 171.6, 171.2, 165.0, 149.8, 148.8, 147.9, 146.5, 137.6, 137.5, 130.3, 129.9, 129.5, 129.4, 129.0, 128.8, 128.5, 128.2, 127.7, 126.4, 121.7, 118.8, 75.0, 71.9, 68.1, 66.7, 59.4, 56.9, 54.6, 50.9, 50.2, 34.8, 33.3, 29.8, 29.7, 28.7, 19.6, 17.5.
FAB-MS m/z 709 (MH+). Anal. Calcd for C41H52N6O5(corrected for 0.7% water content as determined by Karl Fisher analysis): C, 68.98; H, 7.42; N, 11.77. Found: C, 68.71; H, 7.47; N, 11.71. HPLC homogeneity (Supelcosil LC-ABZ, 10−50% 1% TFA in MeCN/1% TFA in 25 min, 1 mL/min flow rate): palinavir (1), tR 17.83 min (99.59%); 25 tR20.00 min (0.41%). HPLC homogeneity (Nova-Pak C8, 10−80% MeCN/50 mM NaH2PO4 in 25 min, 1 mL/min flow rate): palinavir (1), tR 17.37 min (99.51%); 25 tR 15.87 min (0.49%).
| Reference | ||
|---|---|---|
| 1 | * | ARUN K. GHOSH ET AL: “The Development of Cyclic Sulfolanes as Novel and High-Affinity P2 Ligands for HIV-1 Protease Inhibitors“, JOURNAL OF MEDICINAL CHEMISTRY, vol. 37, no. 8, 1 April 1994 (1994-04-01), pages 1177-1188, XP055057710, ISSN: 0022-2623, DOI: 10.1021/jm00034a016 |
| 2 | * | KAY BRICKMANN ET AL: “Synthesis of Conformationally Restricted Mimetics of [gamma]-Turns and Incorporation into Desmopressin, an Analogue of the Peptide Hormone Vasopressin“, CHEMISTRY – A EUROPEAN JOURNAL, vol. 5, no. 8, 2 August 1999 (1999-08-02), pages 2241-2253, XP055057517, ISSN: 0947-6539, DOI: 10.1002/(SICI)1521-3765(19990802)5:8<2241: :AID-CHEM2241>3.0.CO;2-L |
| 3 | * | KIRAN I N C ET AL: “A concise enantioselective synthesis of (+)-goniodiol and (+)-8-methoxygoniodiol via Co-catalyzed HKR of anti-(2SR, 3RS)-3-methoxy-3-phenyl-1, 2-epoxypropane“, TETRAHEDRON LETTERS, ELSEVIER, AMSTERDAM, NL, vol. 52, no. 3, 19 January 2011 (2011-01-19), pages 438-440, XP027558447, ISSN: 0040-4039 [retrieved on 2010-12-14] |
| 4 | * | M. TOKUNAGA: “Asymmetric Catalysis with Water: Efficient Kinetic Resolution of Terminal Epoxides by Means of Catalytic Hydrolysis“, SCIENCE, vol. 277, no. 5328, 15 August 1997 (1997-08-15), pages 936-938, XP055057541, ISSN: 0036-8075, DOI: 10.1126/science.277.5328.936 |
| 5 | * | PARKES K E B ET AL: “STUDIES TOWARD THE LARGE-SCALE SYNTHESIS OF THE HIV PROTEINASE INHIBITOR RO 31-8959“, JOURNAL OF ORGANIC CHEMISTRY, ACS, US, vol. 59, no. 13/16, 1 January 1994 (1994-01-01), pages 3656-3664, XP002011975, ISSN: 0022-3263, DOI: 10.1021/JO00092A026 |
| 6 | * | R. SANTHOSH REDDY ET AL: “Co(iii)(salen)-catalyzed HKR of two stereocentered alkoxy- and azido epoxides: a concise enantioselective synthesis of (S,S)-reboxetine and (+)-epi-cytoxazone“, CHEMICAL COMMUNICATIONS, vol. 46, no. 27, 1 January 2010 (2010-01-01), page 5012, XP055057537, ISSN: 1359-7345, DOI: 10.1039/c0cc00650e |
| 7 | * | SHINJI NAGUMO ET AL: “Intramolecular Friedel-Crafts type reaction of vinyloxiranes linked to an ester group“, TETRAHEDRON, vol. 65, no. 47, 1 November 2009 (2009-11-01), pages 9884-9896, XP055057655, ISSN: 0040-4020, DOI: 10.1016/j.tet.2009.09.037 |
| 8 | * | SUNITA K. GADAKH ET AL: “Enantioselective synthesis of HIV protease inhibitor amprenavir via Co-catalyzed HKR of 2-(1-azido-2-phenylethyl)oxirane“, TETRAHEDRON: ASYMMETRY, vol. 23, no. 11-12, 1 June 2012 (2012-06-01), pages 898-903, XP055057475, ISSN: 0957-4166, DOI: 10.1016/j.tetasy.2012.06.003
Want to know everything on vir series click http://drugsynthesisint.blogspot.in/p/vir-series-hep-c-virus-22.html AND http://medcheminternational.blogspot.in/p/vir-series-hep-c-virus.html |
Sitasentan TBC 11251
Sitasentan,TBC 11251
210421-64-0
N-(4-chloro-3-methyl-1,2-oxazol-5-yl)-2-[2-(6-methyl-1,3-benzodioxol-5-yl)acetyl]thiophene-3-sulfonamide
Sitaxentan sodium (TBC-11251) is a medication for the treatment of pulmonary arterial hypertension (PAH).[1] It was marketed as Thelin by Encysive Pharmaceuticals until Pfizer purchased Encysive in February 2008. In 2010, Pfizer voluntarily removed sitaxentan from the market due to concerns about liver toxicity.[2]
Sitaxentan belongs to a class of drugs known as endothelin receptor antagonists (ERAs). Patients with PAH have elevated levels of endothelin, a potent blood vessel constrictor, in their plasma and lung tissue. Sitaxentan blocks the binding of endothelin to its receptors, thereby negating endothelin’s deleterious effects.
Mechanism of action
Sitaxentan is a small molecule that blocks the action of endothelin (ET) on the endothelin-A (ETA) receptor selectively (by a factor of 6000 compared to the ETB).[3] It is a sulfonamide class endothelin receptor antagonist (ERA) and is undergoing Food and Drug Administration (FDA) review for treating pulmonary hypertension. The rationale for benefit compared to bosentan, a nonselective ET blocker, is negligible inhibition of the beneficial effects of ETB stimulation, such as nitric oxide production and clearance of ET from circulation. In clinical trials, the efficacy of sitaxentan has been much the same as bosentan, but the hepatotoxicity of sitaxentan outweighs its benefits. Dosing is once daily, as opposed to twice daily for bosentan.
Regulatory status
On December 10, 2010 Pfizer announced it would be withdrawing sitaxentan worldwide (both from marketing and from all clinical study use), citing that it is a cause of fatal liver damage.[2]
Sitaxentan was approved for marketing in the European Union in 2006, in Canada in 2006[4] and in Australia in 2007. By February 2008 it had been launched commercially in Germany, Austria, The Netherlands, the United Kingdom, Ireland, France, Spain and Italy.
In March 2006, the FDA recommended an approvable status to sitaxentan but said it would not yet approve the product. In July 2006, sitaxentan received a second approvable letter stating that efficacy outcome issues raised in the context of the STRIDE-2 study were still unresolved. In July 2007, Encysive commenced a formal dispute resolution process in a preliminary meeting with the FDA. In September 2007 the company announced that it was making preparations for another phase III clinical trial (intended to be named STRIDE-5) to overcome the FDA’s concerns.[5] The takeover by Pfizer resulted in a reconfiguration and extension of these plans, to include combination therapy with sildenafil. The Sitaxentan Efficacy and Safety Trial With a Randomized Prospective Assessment of Adding Sildenafil (SR-PAAS) was an ongoing program of three clinical trials conducted in the United States (ClinicalTtrials.gov identifiers: NCT00795639, NCT00796666 and NCT00796510) with anticipated completion dates between June 2010 and January 2014.

N-(4-Chloro-3-methyl-5-isoxazolyl)-2-[2-(6-methyl-1,3-benzodioxol-5-yl)acetyl]-3-thiophenesulfonamide sodium salt, Sitaxsentan sodium salt, TBC-11251 sodium salt, Thelin
- CAS Number 210421-74-2
- Empirical Formula C18H14ClN2NaO6S2
- Molecular Weight 476.89
Adverse effects
Adverse effects observed with sitaxentan are class effects of endothelin receptor antagonists, and include :
- liver enzyme abnormalities (increased ALT and AST)
- headache
- edema
- constipation
- nasal congestion
- upper respiratory tract infection
- dizziness
- insomnia
- flushing
Because sitaxentan inhibits metabolism of warfarin, a decreased dose of warfarin is needed when co-administered with sitaxentan. This is because warfarin acts to prevent blood from clotting, and if it remains unmetabolized, it can continue to thin the blood.
http://www.google.com/patents/WO2007149568A2?cl=en
As used herein “sitaxsentan” refers to N-(4-chloro-3-methyl-5-isoxazolyl)-2-[2- methyl-4,5-(methylenedioxy)phenylacetyl]-thiophene-3-sulfonamide. Sitaxsentan is also known as TBCl 1251. Other chemical names for sitaxsentan include 4-chloro-3-methyl-5-(2- (2-(6-methylbenzo[d][l ,3]dioxol-5-yl)acetyl)-3-thienylsulfonamido)isoxazole and N-(4- chloro-3-methyl-5-isoxazolyl)-2-[3,4-(methylenedioxy)-6-methylphenylacetyl]-thiophene-3- sulfonamide.
The chemical name for sitaxsentan is N-(4-chloro-3-methyl-5-isoxazolyl)-2-[2- methyl-4,5-(methylenedioxy)phenylacetyl]-thiophene-3-sulfonamide, and its structural formula is as follows:
Sitaxsentan
Sitaxsentan is a potent endothelin receptor antagonist that has oral bioavailability in several species, a long duration of action, and high specificity for ETA receptors.
EXAMPLE 1
Preparation of 4-chloro-3-methyl-5-(2-(2-(6-methylbenzo[d] [l,3|dioxol-5-yl)aeetyl)-3- thienylsulfonamido)isoxazole, or N-(4-chloro-3-methyl-5-isoxazolyl)-2-[2-methy 1-4,5- (methylenedioxy)phenylacetyl]-thiophene-3-sulfonamide, or N-(4-chIoro-3-methyl-5- isoxazolyl)-2-[3,4-(methylenedioxy)-6-methylphenylacetyl]-thiophene-3-sulfonamide.
A. Preparation of (4-chIoro-3-methyl-5-(2-(2-(6-methylbenzo[d] [l,3]dioxol-5-yl)acetyl)- 3-thienylsuIfonamido)isoxazole 1. Preparation of 5-chloromethyI-6-methylbenzo[d][l,3]dioxole
To a mixture of methylene chloride (130 L), concentrated HCl (130 L), and tetrabuylammonium bromide (1.61 Kg) was added 5-methylbenzo[d][l,3]dioxole (10 Kg) followed by the slow addition of formaldehyde (14 L, 37 wt% in water). The mixture was stirred overnight. The organic layer was separated, dried with magnesium sulfate and concentrated to an oil. Hexane (180 L) was added and the mixture heated to boiling. The hot hexane solution was decanted from a heavy oily residue and evaporated to give almost pure 5-chloromethyl-6-methylbenzo[d][l,3]dioxole as a white solid. Recrystallization from hexane (50 L) gave 5-chloromethyl-6-methylbenzo[d][l,3]dioxole (80% recovery after recrystallization). 2. Formation of (4-chloro-3-methyl-5-(2-(2-(2-methyIbenzo[d][l,3]dioxol-5-yl) acetyl)-3-thienylsulfonamido)isoxazole
A portion of a solution of 5-chloromemyl-6-methylbenzo[d][l,3]di-oxole (16.8 g, 0.09 mol) in tetrahydrofuran (THF)(120 mL) was added to a well stirred slurry of magnesium powder, (3.3 g, 0.136 g-atom, Alfa, or Johnson-Mathey, -20 +100 mesh) in THF (120 mL) at room temperature. The resulting reaction admixture was warmed up to about 40-450C for about 2-3 min, causing the reaction to start. Once the heating activated the magnesium, and the reaction began, the mixture was cooled and maintained at a temperature below about 8 0C. The magnesium can be activated with dibromoethane in place of heat.
A flask containing the reaction mixture was cooled and the remaining solution of 5- chloromethlybenzo[d][l,3]dioxole added dropwise during 1.5 hours while maintaining an internal temperature below 8 0C. Temperature control is important: if the Grignard is generated and kept below 8 0C5 Wurtz coupling is suppressed. Longer times at higher temperatures promote the Wurtz coupling pathway. Wurtz coupling can be avoided by using high quality Mg and by keeping the temperature of the Grignard below about 8 0C and stirring vigorously. The reaction works fine at -20 0C, so any temperature below 8 0C is acceptable at which the Grignard will form. The color of the reaction mixture turns greenish.
The reaction mixture was stirred for an additional 5 min at 0 0C, while N2-methoxy- N2-methyl-3-(4-chloro-3-methyl-5-isoazolylsulfamoyl)-2-thiophenecarboxamide (6.6 g, 0.018 mol) in anhydrous THF (90 mL) was charged into the addition funnel. The reaction mixture was degassed two times then the solution of N2-methoxy-N2-methyl-3-(4-chloro-3- methyl-5-isoxazolylsulfamoyl)-2-thiophenecarboxamide was added at 0 0C over 5 min. TLC of the reaction mixture (Silica, 12% MeOHZCH2Cl2) taken immediately after the addition shows no N2-methoxy-N2-methyl-3-(4-chloro-3-methyl-5-isoxazolysulfamoyl)-2-thio- phenecarboxamide. The reaction mixture was transferred into a flask containing IN HCl (400 mL, 0.4 mol
HCl, ice-bath stirred), and the mixture stirred for 2 to 4 min, transferred into a separatory funnel and diluted with ethyl acetate (300 mL). The layers were separated after shaking. The water layer was extracted with additional ethyl acetate (150 mL) and the combined organics washed with half-brine. Following separation, THF was removed by drying the organic layer over sodium sulfate and concentrating under reduced pressure at about 39 0C to obtain the title compound. EXAMPLE 2
1.0 g Sitaxentan was dissolved in 10 ml ethyl acetate and 5 ml hexanes were added. The formed suspension was heated until a clear solution was obtained. Upon cooling light yellow plates were formed. After filtration and drying under vacuum 515 mg of sitaxentan polymorph A was obtained as light yellow plates in very high purity.
EXAMPLE 3
Preparation of 4-chloro-3-methyl-5-(2-(2-(6-methyIbenzo[dJ [l,3]dioxol-5-yl)acetyl)-3- thienylsulfonamido)isoxazole, Sodium Salt
The crystalline sitaxsentan from Example 2 is dissolved in ethyl acetate and washed with saturated NaHCO3 (5 x 10 mL). The solution is washed with brine, dried over Na2SO4 and concentrated in vacuo to obtain a solid residue. 10 mL OfCH2Cl2 is added and the mixture is stirred under nitrogen for 5 to 10 minutes. Ether (15 mL) is added and the mixture stirred for about 10 min. The product is isolated by filtration, washed with a mixture of CH2Cl2 /ether (1 :2) (10 mL) then with ether (10 mL) and dried under reduced pressure to obtain 4-Chloro-3-methyl-5-(2-(2-(6-methyIbenzo[d][l ,3]dioxol-5-yl)acetyl)-3- thienylsulfonamido)isoxazole, sodium salt.
………………………..
Yingyong Huaxue (2007), 24, (11), 1310-1313. Publisher: (Kexue Chubanshe, ) CODEN:YIHUED ISSN:1000-0518.
References
- 2
- Citing liver damage, Pfizer withdraws Thelin, Associated Press, December 12, 2010
- 3
- Girgis, RE; Frost, AE; Hill, NS; Horn, EM; Langleben, D; McLaughlin, VV; Oudiz, RJ; Robbins, IM et al. (2007). “Selective endothelinA receptor antagonism with sitaxsentan for pulmonary arterial hypertension associated with connective tissue disease”. Annals of the rheumatic diseases 66 (11): 1467–72. doi:10.1136/ard.2007.069609. PMC 2111639. PMID 17472992.
- 4
- “UPDATE 1-Encysive gets Canadian approval for hypertension drug”. Reuters. 30 May 2007. Retrieved 2007-07-08.
- 5
- “Encysive Pharmaceuticals to Conduct Phase III Study With Thelin (Sitaxsentan Sodium) in Pulmonary Arterial Hypertension”. PrimeNewswire via COMTEX News Network. 29 September 2007. Retrieved 2007-12-12.
![]()
External links
- http://www.drugs.com/nda/thelin_050714.html
- Mucke HAM: Sitaxsentan for the Oral Treatment of Pulmonary Arterial Hypertension: Benefits from Endothelin Receptor Subtype Selectivity? Clinical Medicine: Therapeutics 2009:1 111–121.
- ATS 2005. The International Conference of the American Thoracic Society. 20–25 May 2005. San Diego, CA.
- Robyn J. Barst, MD; Stuart Rich, MD, FCCP; Allison Widlitz, MS, PA; Evelyn M. Horn, MD; Vallerie McLaughlin, MD and Joyce McFarlin, RN : Clinical Efficacy of Sitaxsentan, an Endothelin-A Receptor Antagonist, in Patients With Pulmonary Arterial Hypertension Chest. 2002;121:1860-1868.
- American Heart Association. Primary or Unexplained Pulmonary Hypertension
| US20010021714 * | Apr 4, 1996 | Sep 13, 2001 | Ming Fai Chan | Compounds such as n-(4-bromo-3-methyl-5-isoxazolyl)-2-n-benzylbenzo(b)thiophene-3-sufonamide administered as endothelin peptide receptor antagonists |
| Reference | ||
|---|---|---|
| 1 | * | WU C ET AL: “Discovery of TBC11251, a Potent, Long Acting, Orally Active Endothelin Receptor-A Selective Antagonist” JOURNAL OF MEDICINAL CHEMISTRY, AMERICAN CHEMICAL SOCIETY. WASHINGTON, US, vol. 40, no. 11, 23 May 1997 (1997-05-23), pages 1690-1697, XP002164198 ISSN: 0022-2623 |
| Patent | Submitted | Granted |
|---|---|---|
| ANTIHYPERTENSIVE THERAPY METHOD [US2007293552] | 2007-12-20 | |
| Crystalline N-(4-chloro-3-methyl-5-isoxazolyl)-2-[2-methyl-4.5-(methylenedioxy)phenylacetyl]-thiophene-3-sulfonamide [US2008026061] | 2008-01-31 | |
| Gnrh agonist combination drugs [US2005215528] | 2005-09-29 | |
| THIENYL-, FURYL-, PYRROLYL- AND BIPHENYLSULFONAMIDES AND DERIVATIVES THEREOF THAT MODULATE THE ACTIVITY OF ENDOTHELIN [WO9631492] | 1996-10-10 | |
| SULFONAMIDES FOR TREATMENT OF ENDOTHELIN-MEDIATED DISORDERS [WO9849162] | 1998-11-05 |
| Patent | Submitted | Granted |
|---|---|---|
| Respiratory Drug Condensation Aerosols and Methods of Making and Using Them [US2009258075] | 2009-10-15 | |
| Method and Composition for Treating Alzheimer’s Disease and Dementias of Vascular Origin [US2010173872] | 2010-07-08 | |
| Method and Composition for Treating Alzheimer’s Disease and Dementias of Vascular Origin [US2010184725] | 2010-07-22 | |
| Formulations of sitaxsentan sodium [US2008076812] | 2008-03-27 | |
| Methods and compositions for treatment of sleep apnea [US2008085313] | 2008-04-10 | |
| Processes for the preparation of 4-chloro-3-methyl-5-(2-(2-(6-methylbenzo[d][1,3]dioxol-5-yl)acetyl)-3-thienylsulfonamido)isoxazole [US2008086010] | 2008-04-10 | |
| Method and composition for treating alzheimer’s disease and dementias of vascular origin [US2004092427] | 2004-05-13 | |
| Method for preventing or treating pulmonary inflammation by administering an endothelin antagonist [US2003004199] | 2003-01-02 | |
| Methods and Compositions for Treatment of an Interstitial Lung Disease [US2009004268] | 2009-01-01 | |
| Methods and compositions for treatment of diastolic heart failure [US2007232671] | 2007-10-04 | |
| Patent | Submitted | Granted |
|---|---|---|
| Isoxazolyl endothelin antagonists [US6043265] | 2000-03-28 | |
| Aminoguanidine hydrazone derivatives, process for producing the same and drugs thereof [US6350749] | 2002-02-26 | |
| Method for preventing or treating pain by administering an endothelin antagonist [US6573285] | 2002-06-27 | 2003-06-03 |
| Method for preventing or treating erectile dysfunction by administering an endothelin antagonist [US6268388] | 2001-07-31 | |
| Method and composition for potentiating the antipyretic action of a nonopioid analgesic [US7351692] | 2003-12-25 | 2008-04-01 |
| Method and Composition for Potentiating an Opiate Analgesic [US8114896] | 2010-05-06 | 2012-02-14 |
| SUBSTITUTED THIOPHENES [US7863308] | 2008-10-16 | 2011-01-04 |
| Respiratory drug condensation aerosols and methods of making and using them [US7550133] | 2004-06-03 | 2009-06-23 |
| SUBSTITUTED THIOPHENES [US2010280086] | 2010-11-04 | |
| Method and Composition for Potentiating an Opiate Analgesic [US2010311665] | 2010-12-09 |
 |
|
| Systematic (IUPAC) name | |
|---|---|
| N-(4-chloro-3-methyl-1,2-oxazol-5-yl)-2-[2-(6-methyl-2H-1,3-benzodioxol-5-yl)acetyl]thiophene-3-sulfonamide | |
| Clinical data | |
| AHFS/Drugs.com | International Drug Names |
| Licence data | EMA:Link |
| Legal status | |
| Routes | Oral |
| Pharmacokinetic data | |
| Bioavailability | 70 to 100% |
| Protein binding | >99% |
| Metabolism | Hepatic (CYP2C9– and CYP3A4-mediated) |
| Half-life | 10 hours |
| Excretion | Renal (50 to 60%) Fecal (40 to 50%) |
| Identifiers | |
| CAS number | 184036-34-8 210421-64-0 (sodium salt) |
| ATC code | C02KX03 |
| PubChem | CID 216235 |
| IUPHAR ligand | 3950 |
| DrugBank | DB06268 |
| ChemSpider | 21106381 |
| UNII | J9QH779MEM |
| KEGG | D07171 |
| ChEMBL | CHEMBL282724 |
| Synonyms | Sitaxsentan; TBC-11251 |
| Chemical data | |
| Formula | C18H15ClN2O6S2 |
| Molecular mass | 454.906 g/mol |
![]()

Structures and observed activities of the ETA receptor antagonists for the HipHop training set
 COCK WILL TEACH YOU NMR
COCK WILL TEACH YOU NMR COCK SAYS MOM CAN TEACH YOU NMR
COCK SAYS MOM CAN TEACH YOU NMR

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE amcrasto@gmail.com
amcrasto@gmail.com
TAKE A TOUR
KISUMU, KENYA

Kisumu – Wikipedia, the free encyclopedia
Kisumu is a port city in Kisumu County, Kenya 1,131 m (3,711 ft), with a population of 409,928 (2009 census). It is the third largest city in Kenya, the principal city …
Kisumu CountyKisumu County is one of the new devolved counties of Kenya. Its …
|
Kisumu International AirportKisumu International Airport is an airport in Kisumu, Kenya (IATA …
|
Boat riding in Kisumu
Local inhabitants near Kisumu, 1911


Clockwise: Lake Victoria Panorama, Kisumu Panorama, sunset at Oginga Odinga street, Downtown, Kiboko Point, Nighttime in Kisumu and Jomo Kenyatta Stadium.
|
|
Coordinates:  0°6′S 34°45′E 0°6′S 34°45′E |
|
| Country | |
|---|---|
| County | Kisumu County |
Kisumu panorama, viewed from Lake Victoria
WANT TO KNOW ON SENTAN SERIES
MEDICINAL CHEMISTRY AT ITS BEST, Tracks information on drugs on worldwide basis by Dr Anthony Melvin Crasto, worlddrugtracker, helping millions with websites, 6 million hits on google, one lakh connections worldwide, email amcrasto@gmail.com, call +91 9323115463 India
READ MORE ON SENTAN SERIES……http://medcheminternational.blogspot.in/p/sentan-series.html
| Antagonists of Endothelin type A receptor ETA | |
| Name | Structure |
| BQ-123 |  |
| Bosentan |  |
| Atrasentan |  |
| Tezosentan |  |
| Sitaxsentan |  |
| Darusentan |  |
| Clazosentan |  |
| ZD-4054 (Zibotentan) |  |
| Ambrisentan |  |
| Tak-044 |  |
| Avosentan |  |
MAHABALIPURAM, INDIA
Mahabalipuram – Wikipedia, the free encyclopedia
Mahabalipuram, also known as Mamallapuram is a town in Kancheepuram district in the Indian state of Tamil Nadu. It is around 60 km south from the city of …Shore Temple – Seven Pagodas – Pancha Rathas –
.
.



Krishna’s Butter Ball in Mahabalipuram, India. The surface below the rock is …




http://www.weather-forecast.com/locations/Mamallapuram
 Come to Mahabalipuram (also known as Mammallapuram), an enchanting beach that is located on the east coast of India.
Come to Mahabalipuram (also known as Mammallapuram), an enchanting beach that is located on the east coast of India.
 Moonraikers Restaurant, Mamallapuram
Moonraikers Restaurant, Mamallapuram


 Hotel Mamalla Bhavan – Mahabalipuram Chennai – Food, drink and entertainment
Hotel Mamalla Bhavan – Mahabalipuram Chennai – Food, drink and entertainment
 .
.
A carving at the Varaha Temple, Mahabalipuram

/////////////
SWINE FLU ; AYURVEDA SUCCESSFUL TREATMENT ; स्वाइन प्लू का सुरक्षित आयुर्वेदिक इलाज
स्वाइन प्लू के लक्षणो पर आधारित सभी रोगियो का आयुर्वेदिक इलाज करने के बाद यह अनुभव मे आया है कि महामारी की तरह फैल रही बीमारी का बहुत सटीक और अचूक इलाज आयुर्वेद मे है /
वायरल / अथवा स्वाइन फ्लू के रोगियो के इलाज मे मैने निम्न दवाये दी है उन्हे मै सार्वजनिक तौर पर देश के सभी नागरिको के लिये यहा बता रहा हू /
स्वाइन फ्लू या इस जैसी बीमारी के इलाज के लिये मेरा नुस्खा इस तरह है /
महामृत्युन्जय रस दो गोली
कफ कुठार रस चार गोली
सुदर्शन घन वटी दो गोली
सप्त पर्ण घन वटी दो गोली
वयस्क व्यक्ति के लिये यह एक खुराक है /
सभी ऊपर लिखी गयी दवओ की गोलियो को गुन्गुने पानी से तीन तीन घन्टे के अन्तर से खिलाना चाहिये three hourly a with lukwarm water
कम उम्र के किशोरो को ऊपर लिखी दवाओ की एक एक गोली…
View original post 646 more words
Detailed Requirements concerning the DOE in the Regulatory Submission Dossier: EMA’s and FDA’s Recommendations
DRUG REGULATORY AFFAIRS INTERNATIONAL

The EMA has published together with the FDA a new question & answer (Q&A) paper at the end of 2014. This document answers questions on detailed requirements in connection with the documents concerning regulatory submissions. Among others it contains the answer to the question “What level of detail should be considered for design of experiments (DOEs) in a regulatory submission?”
GMP News
25/02/2015
In our News dated 18 February we reported on a question & answer (Q&A) paper which was published by EMA and FDA together at the end of 2014. This document answers questions on detailed requirements in connection with the documents concerning regulatory submissions. It also answers a question on the topic design of experiments (DOE).

The document answers the question “What level of detail should be considered for design of experiments (DOEs) in a regulatory submission?” as follows:
The level of detail should be commensurate…
View original post 165 more words
SILODOSIN………For treatment of benign prostatic hypertophy

SILODOSIN
Alpha 1A adrenoceptor antagonist
Prostate hyperplasia
Kissei Pharmaceutical Co Ltd INOVATOR
CAS 160970-54-7
2,3-Dihydro-1-(3-hydroxypropyl)-5-[(2R)-2-[[2-[2-(2,2,2-trifluoroethoxy)phenoxy]ethyl]amino]propyl]-1H-indole-7-carboxamide
160970-64-9 (racemate)
169107-04-4 (diHBr)
In February 2008, the FDA accepted for review an NDA for silodosin for the treatment of dysuria associated with BPH . In October 2008, the FDA approved the drug . In April 2009, Actavis launched silodosin for the treatment of the signs and symptoms of BPH .
1-(3-hydroxypropyl)-5-[(2R)-2-[2-[2-(2,2,2-trifluoroethoxy)phenoxy]ethylamino]propyl]-2,3-dihydroindole-7-carboxamide
Kissei Pharmaceutical, Daiichi Sankyo (formerly Daiichi Seiyaku), Actavis (formerly Watson) and Recordati have developed and launched silodosin (Urief; Trupass; Rapaflo; Thrupas; Silodyx; Urorec; KMD-3213; Youlifu), an oral alpha 1A adrenoceptor antagonist selective for prostatic receptors . The product is comarketed in Europe by several licensees. The drug is indicated for the treatment of the signs and symptoms of benign prostatic hyperplasia (BPH).
Silodosin, a highly selective alpha1A-adrenoceptor antagonist, was launched in May 2006 in Japan for the oral treatment of urinary disturbance associated with benign prostatic hyperplasia (BPH). The product was launched in the U.S. for the treatment of signs and symptoms of benign prostatic hyperplasia in 2009. In 2009, a positive opinion was received in the E.U. for this indication and final approval was obtained in 2010. Launch in the E.U. took place the same year.
In May 2006, silodosin was launched as a capsule formulation in Japan. Actavis launched the drug in the US in April 2009. In June 2010, EU launched began, initially with Germany ; in November 2010, the drug was launched in France; by December 2010, the drug was launched in Spain.
In 2001, Kissei established an agreement with Daiichi Pharmaceutical to codevelop and comarket silodosin. An oral, once-daily formulation of silodosin filed in the U.S. by Watson (now Actavis) was approved in 2008. Watson (now Actavis) obtained exclusive rights in 2004 to develop and market the drug in the U.S.
PRODUCT Was developed and launched byKissei Pharmaceutical, Daiichi Sankyo, Actavis and Recordati. Family members of the product case EP0600675 have SPC protection in most EU states until 2018; while its Orange Book listed equivalent, US5387603, expire in the US in 2018 with US156 extension.
Silodosin (trade names Rapaflo (USA), Silodyx (Europe and South Africa), Rapilif (India), Silodal (India), Urief (Japan), Urorec (Russia)) is a medication for the symptomatic treatment of benign prostatic hyperplasia. It acts as an α1–adrenoceptor antagonist with high uroselectivity (selectivity for the prostate).
 |
|
| Systematic (IUPAC) name | |
|---|---|
| 1-(3-hydroxypropyl)-5-[(2R)-({2-[2-[2-(2,2,2-trifluoroethoxy)phenoxy]ethyl}amino)propyl]indoline-7-carboxamide | |
| Clinical data | |
|
|
| Legal status |
|
| Routes | Oral |
| Pharmacokinetic data | |
| Bioavailability | 32% |
| Protein binding | 97% |
| Metabolism | Hepatic glucuronidation (UGT2B7-mediated); also minor CYP3A4 involvement |
| Half-life | 13±8 hours |
| Excretion | Renal and fecal |
| Identifiers | |
| CAS number | 160970-54-7 |
| ATC code | G04CA04 |
| PubChem | CID 5312125 |
| IUPHAR ligand | 493 |
| ChemSpider | 4471557 |
| UNII | CUZ39LUY82 |
| ChEMBL | CHEMBL24778 |
| Synonyms | KAD-3213, KMD-3213 |
| Chemical data | |
| Formula | C25H32F3N3O4 |
| Molecular mass | 495.534 g/mol |
History
Silodosin received its first marketing approval in Japan in May 2006 under the tradename Urief, which is jointly marketed by Kissei Pharmaceutical Co., Ltd. and Daiichi Sankyo Pharmaceutical Co., Ltd.
Kissei licensed the US, Canadian, and Mexican rights for silodosin to Watson Pharmaceuticals, Inc. in 2004.
On February 12, 2008, Watson announced that the New Drug Application submitted to the United States Food and Drug Administration for silodosin has been accepted for filing. FDA approved this drug on October 9, 2008.[1] Silodosin is marketed under the trade names Rapaflo in the US and Silodyx in Europe.[2] and Rapilif in India (Ipca Urosciences)
Pharmacology
Since silodosin has high affinity for the α1A adrenergic receptor, it causes practically no orthostatic hypotension (in contrast to other α1 blockers). On the other side, the high selectivity seems to cause more problems with ejaculation.[3]
As α1A adrenoceptor antagonists are being investigated as a means to male birth control due to their ability to inhibit ejaculation but not orgasm, a trial with 15 male volunteers was conducted. While silodosin was completely efficacious in preventing the release of semen in all subjects, 12 out of the 15 patients reported mild discomfort upon orgasm. The men also reported the psychosexual side effect of being strongly dissatisfied by their lack of ejaculation.[4]

///////////////////////////////
CN 103848772
http://www.google.com/patents/CN103848772A?cl=en
silodosin (Silodosin) is 〃 2 Japanese orange Johnson invented – receptor antagonist, for the treatment of benign prostatic hyperplasia or hypertrophy, and other related symptoms. Clinical trials showed that 25% of patients with benign prostatic hyperplasia need for drugs or surgery. Although prostatectomy is better, the mortality rate is not high, but patients bring varying degrees of damage. So look for an effective and safe non-surgical treatment, not only can control the further development of the disease, while relieving the symptoms of the patient.
benign prostatic hyperplasia in older male patients have a higher prevalence, and clinical alternative drugs rarely, so the development of a benign prostatic hyperplasia drug treatment, not only has good social benefits, but also to bring good economic benefits. The study confirmed that silodosin is the treatment of benign prostatic hyperplasia in an important class of drugs.
Currently, the research reported in the published literature on the preparation of compounds of silodosin, are:
Early 1995, Kitazawa M et al patent US5387603, the reporter silodosin total synthesis method, but the method reaction step is long, the yield is not too high, not suitable for our industrial production.
In 2009, 翟富民 et al patent CN102115455A, which reported a method for preparing Sailuoduoxin key intermediates. The appropriate method for improving existing methods, although shorter than the previous method step in the step, but low synthesis yield of the process, we can not meet the needs of industrial production.
In summary, the compounds prepared silodosin more synthetic methods are constantly improved, but there are still a lot of flaws. Therefore, there is need for further research on the preparation of compounds of silodosin to get simple process, product yield, product easy separation of the new preparation. SUMMARY
The present invention is to overcome the above problems of the prior art, there is provided a method for preparing important intermediates silodosin, the present invention is simple process, high yield, easy separation of the product, the method suitable for industrial production .
To achieve the above technical object, to achieve the above technical result, the present invention is realized by the following technical scheme:
One kind of silodosin preparation of important intermediate, comprising the steps of:
Step I) in a flask, 282g of raw materials 1-acetyl-5- (2-bromo-propyl) indoline, 222g phthalimide potassium salt and 700mL DMF, was heated at 110 ° C for 2h; After completion of the reaction, to which was added the right amount of water to wash away the excess solvent DMF and salt extraction desolventizing after EA, was 296g crude;
Step 2) In a flask was added 296g crude product obtained in step I, dissolved 800mL ethanol, was added 165mL of hydrazine hydrate, 50 ° C is heated to precipitate a white solid; After completion of the reaction, cooling suction filtered, the filter cake washed with ethanol, and then the mother liquor removing solvent under reduced pressure; After dissolving EA, washed with water to wash away the excess hydrazine, and finally the organic phase the solvent was removed to give 165g intermediate, i.e. 1-acetyl-5- (2-aminopropyl) indoline;
Step 3) In the three-necked flask, 165g of Intermediate 1-acetyl-5- (2-aminopropyl) indoline, dissolved 600mL methanol, stirred at room temperature, and thereto was slowly added dropwise bromine; the addition was complete After stirring at room temperature 5-6h; After completion of the reaction, slowly poured into saturated NaHSO3, and wash away excess bromine; extracted with ethyl acetate, washed with water and saturated brine, dried over anhydrous sodium sulfate; After filtration, the solvent was removed in vacuo to give the crude product recrystallized from toluene to give 177g pure product;
Step 4) In a flask was added 177g of pure product obtained in Step 3 and 65g CuCN, after use 700mL DMF, was heated at 110 ° C for 3 to 5h; After completion of the reaction, the amount of water was added thereto, washing off excess solvent DMF and salt, EA desolventizing crude extract, after recrystallization 121g pure 1-acetyl-5- (2-bromo-propyl) -7-cyano-indoline that silodosin important intermediates;
Step (1), (2), (3), (4) synthesis reaction is:
Further, the step I) to step 4) by TLC plate tracking point detection reaction.
The beneficial effects of the present invention are:
Preparation silodosin important intermediate of the present invention, mention of the method is simple, high reaction yield, product easily separated, suitable for industrial production and so on.
Preparation Method A silodosin important intermediate, comprising the following steps: Step I) in a flask, 282g of raw materials 1-acetyl-5- (2-bromo-propyl) indoline, 222g o phthalimide potassium and 700mL DMF, heated at 110 ° C for 2h; After completion of the reaction, to which was added the right amount of water to wash away the excess solvent DMF and salt extraction desolventizing after EA, was 296g crude;
Step 2) In a flask was added 296g crude product obtained in step I, dissolved 800mL ethanol, was added 165mL of hydrazine hydrate, 50 ° C is heated to precipitate a white solid; After completion of the reaction, cooling suction filtered, the filter cake washed with ethanol, and then the mother liquor removing solvent under reduced pressure; After dissolving EA, washed with water to wash away the excess hydrazine, and finally the organic phase the solvent was removed to give 165g intermediate, i.e. 1-acetyl-5- (2-aminopropyl) indoline;
Step 3) In the three-necked flask, 165g of Intermediate 1-acetyl-5- (2-aminopropyl) indoline, dissolved 600mL methanol, stirred at room temperature, and thereto was slowly added dropwise bromine; the addition was complete After stirring at room temperature 5-6h; After completion of the reaction, slowly poured into saturated NaHSO3, and wash away excess bromine; extracted with ethyl acetate, washed with water and saturated brine, dried over anhydrous sodium sulfate; After filtration, the solvent was removed in vacuo to give the crude product recrystallized from toluene to give 177g pure product;
Step 4) In a flask was added 177g of pure product obtained in Step 3 and 65g CuCN, after use 700mL DMF, was heated at 110 ° C for 3 to 5h; After completion of the reaction, the amount of water was added thereto, washing off excess solvent DMF and salt, EA desolventizing crude extract, after recrystallization 121g pure 1-acetyl-5- (2-bromo-propyl) -7-cyano-indoline that silodosin important intermediates;
Step (1), (2), (3), (4) synthesis reaction is:
Further, the step I) to step 4) by TLC plate tracking point detection reaction.
…………………………………………………
WO2013056842
http://www.google.com/patents/WO2013056842A1?cl=en
Silodosin is commercially available under the tradenames RAPAFLO® or
UROPvEC as a capsule formulation for oral use containing 4 mg or 8 mg of the drug. The capsules are to be taken orally once daily for the treatment of the signs and symptoms of benign prostatic hyperplasia. US 5,387,603 and EP 0 600 675 disclose silodosin as a therapeutic agent for the treatment for dysurea associated with benign prostatic hyperplasia. The molecular structure of silodosin (XXV) is shown below.
(XXV)
The synthesis of silodosin is relatively complex and requires a sequence of multiple steps. A key intermediate compound in the synthesis of silodosin is the optically active amine compound represented by the general formula R-Y:
1
wherein, R represents a protecting group and R represents a cyano (CN) or carbamoyl (CONH2) group. The intermediate compound R-Y bears the asymmetric carbon atom that imparts the optical activity to silodosin. Therefore, it is important to obtain the compound R-Y with high optical purity, because according to the methods reported in the state of the art the optical purity of the compound R-Y determines the optical purity of the final product silodosin.
JP 2001-199956 discloses a process for the preparation of a compound of formula R-Y, wherein l-(3-benzoyloxypropyl)-7-cyano-5-(2-oxopropyl)-2,3- dihydroindole or the corresponding 7-carbamoyl derivative is reacted with an optically active amine, namely L-2-phenylglycinol or L-l-phenylethanamine, to afford an imine compound of formula III as depicted in the below scheme 1. Scheme l . JP 2001-199956
R1 = COPh; R2 = CN or CONH2; R3 = H or OH a = 1. cat. deprotection
2. frational crystallization with L-tartaric acid
b = 1. chromatographic separation
2. cat. deprotection
The optically active imine III is subjected to catalytic hydrogenation using platinum(IV) oxide as a catalyst affording the diastereomers IV in a ratio of 3.8:1. The chiral auxiliary II is subsequently removed by catalytic hydrogenation using 10% palladium on carbon, i. e. under the typical conditions which lead to the cleavage and removal of benzylic protecting groups from nitrogen or oxygen atoms. The catalytic deprotection reaction affords the desired intermediate compound R-Y with an optical purity corresponding to the ratio of the diasteromers obtained in the previous step, i. e. the ratio of compound R-Y to S-Y is approximately 3.8: 1, which corresponds to an optical purity of approximately 58.3% enantiomeric excess (e.e.).
In order to increase the optical purity of the intermediate R-Y JP 2001-199956 suggests to conduct a fractional crystallization of the desired enantiomer with L-tartaric acid. After a series of fractional crystallizations the compound R-Y is obtained with an optical purity of 97.6% enantiomeric excess. Alternatively, the diastereomers of the compound of formula IV are separated using chromatographic techniques as column chromatography on silicagel. The pure diastereomer R-TV affords the desired enantiomer R-Y with an optical purity of 100% e.e. after removal of the chiral auxiliary II with hydrogen using 10% palladium on carbon as catalyst.
Another approach for the synthesis of the key intermediate compound R-Y is reported in JP 2002-265444. The route of synthesis disclosed in said document is depicted in the below scheme 2.
Scheme 2. JP 2002-265444
R1 = CH2Ph (Bn); R2 = CN The process involves the reaction of an enantiomeric mixture of the compound of formula VI with (I S, 2R)-2-benzylaminocyclohexane methanol (VII) to obtain a diastereomeric mixture containing the salt VIII. After a series of crystallizations the diastereomer VIII was obtained with an optical purity of 92.8% diastereomeric excess (d.e.). Subsequently, the salt VIII was treated with an acidic aqueous solution to release the acid R-Vl from the salt. After extraction from the aqueous solution with ethyl acetate the acid R-Vl is converted into its amide IX. The compound IX is finally subjected to a Hofmann type rearrangement reaction to obtain the desired intermediate compound R-V.
WO 201 1/030356 discloses a process for the preparation of the intermediate compound R-V, which avoids the resolution of the enantiomers of specific intermediate compounds using chiral auxiliaries or optically active bases. The route of synthesis described in WO 201 1/030356 starts from L-alanine (X), which is a naturally occurring optically active amino acid. The process described in
WO 2011/030356 is depicted in the below scheme 3.
R1 = trimethylsilyl (TMS), tert-butyl dimethylsilyl (TBDMS), allyl, benzyl, propargyl R2 = CN or CONH2 The amino acid is protected by the addition of ethyl chloroformate and subsequently activated by the addition of oxalyl chloride to afford i?-(N-ethoxycarbonyl)alanine as an acyl chloride (XI). Said acyl chloride is reacted with hydroxy protected l-(3- hydroxypropyl)-7-cyano-2,3-dihydroindole of formula XII in a Friedel-Crafts acylation reaction, which gives a compound of formula XIII. The oxo group in compound XIII is reduced to afford a compound of formula XIV that is subsequently subjected to a hydrolysis reaction to yield the key intermediate compound R-Y. It is an object of the present invention to provide a process for preparing silodosin or a pharmaceutically acceptable salt thereof, which process affords the drug with high optical purity and with better yield compared to the prior art processes. This object is solved by the subject matter as defined in the claims.
Scheme 5. Conversion of com ound V to silodosin
R = protecting group
R2 = CN or CONH2
X = leaving group
Example 11. Silodosin (XXV)
A. The compound XXIV (18.0 g) was dissolved in methanol (150 ml) and 5% aqueous sodium hydroxide solution (50 ml). The reaction mixture was stirred at room temperature for 2 h. The deprotected compound XXIV, i. e. a compound of formula XXIV with R = hydrogen and R = cyano, was extracted with toluene. Subsequently, a 10% lactic acid solution (25 ml) was added to the toluene phase in order to extract the product in the aqueous phase. The aqueous solution was separated and then basified. The deprotected product was finally extracted with ethyl acetate. Removal of the solvent gives the deprotected compound to XXIV (R1 = H and R2 = CN; 1 1.0 g) as an oily mass.
B. A mixture of compound XXIV (R1 = H and R2 – CN; 10.0 g), DMSO (80 ml) and 5N NaOH solution (9.0 ml) was stirred for 15 min. at room temperature. An aqueous H202 (30%) solution (1 1.0 ml) was added to the reaction mixture, which was stirred at room temperature for additional 2 h after completion of the addition. Water was added to the reaction mixture, the product was extracted with ethyl acetate, and the solvent was subsequently evaporated to afford 9.0 g crude silodosin.
Example 12. Silodosin (XXV)
10.0 g of crude silodosin (optical purity = 85.0% e.e.) was dissolved in ethyl acetate (120 ml) at 55°C. The resulting clear solution was gradually cooled to 25°C under stirring. The suspension was further cooled to 15°C and stirred for 2 hours. The precipitated solid was filtered and dried at 50°C under vacuum to obtain 7.2 g of XXV with an optical purity of 97.5% e.e.
Example 13. Silodosin (XXV)
10.0 g of crude silodosin (optical purity = 98.5% e.e.) was dissolved in ethyl acetate (120 ml) at 55°C. The resulting clear solution was gradually cooled to 25 °C under stirring. The suspension was further cooled to 15°C and stirred for 2 hours. The precipitated solid was filtered and dried at 50°C under vacuum to obtain 7.2 g of XXV with an optical purity of 99.9% e.e.
Example 14. Silodosin (XXV)
10.0 g of crude silodosin (optical purity = 90.0 %e.e.) was dissolved in ethyl acetate (120 ml) at 55°C. The resulting clear solution was gradually cooled to 25°C under stirring. The suspension was further cooled to 15°C and stirred for 2 hours. The precipitated solid was filtered and dried at 50°C under vacuum to obtain 7.2 g of XXV with an optical purity of 97.0% e.e.
Example 15. Silodosin (XXV)
10.0 g of crude silodosin (optical purity = 92.0% e.e.) was dissolved in isopropyl acetate (160 ml) at 55°C. The resulting clear solution was gradually cooled to 25°C under stirring. The suspension was further cooled to 15°C and stirred for 2 hours. The precipitated solid was filtered and dried at 50°C under vacuum to obtain 8.2 g of XXV with an optical purity of 98.0% e.e. Example 16. Silodosin (XXV)
10.0 g of crude silodosin (optical purity = 98.0% e.e.) was dissolved in isopropyl acetate (160 ml) at 55°C. The resulting clear solution was gradually cooled to 25°C under stirring. The suspension was further cooled to 15°C and stirred for 2 hours. The precipitated solid was filtered and dried at 50°C under vacuum to obtain 8.0 g of XXV with an optical purity of 99.5% e.e.
………………………………
EP2475634
http://www.google.com/patents/EP2475634A2?cl=en
Scheme- 1.
Example-14
Preparation of Preparation of l-(3-Hydroxy-propyl)-5-(2(R)-{2-[2-(2, 2, 2-trifluoro- ethoxy)-phenoxy]-ethyIamino}-propyl)-2,3-dihydro-lH-indol-7-carboxylic acid amide (I)(Silodosin)
To a solution of Benzoic acid 3-[5(R)-(2-amino-propyl)-7-cyano-2, 3-dihydro-indol-l- yl]-propyl ester (XV) (3.5 g, 10 mmole) in Dimethyl sulphoxide (60 ml), charged Hydrogen peroxide (10% w/w) (11 ml). Then added 5 N sodium hydroxide solution (12.3 ml) and reaction mass was stirred for 2 hours. After completion of reaction water was added and extracted the product in ethyl acetate. Organic layer was washed with brine and dried over sodium sulphate. The solvent was evaporated below 40°C under reduced pressure and added methanol (25 ml). To this solution charged glacial acetic acid (0.25 g, 4mmole) and [2-(2, 2, 2-Trifluoro-ethoxy)-phenoxy]-acetaldehyde (VIII) (3 g, 0.0125 mole). Reaction mixture was stirred at 25-30°C for 1 hour. Then reacted with sodium cyanoborohydride (0.15 g, 2.8 mmoles) and heated at 40-45°C for 2 hours. After the completion of reaction solvent was distilled off below 40°C under reduced pressure and added water to the residue. Reaction mass was then acidified with aqueous mineral acid. The aqueous layer was then basified and product was extracted in ethyl acetate. Organic layer was washed with water and dried over sodium sulphate. The solvent was evaporated under reduced pressure and the residue was purified by column chromatography on silica gel using a mixture of ethyl acetate and hexane (5/95) as eluent to give 0.8g of (I) as yellow solid. Purity (by HPLC) = 98%
Example 15
Preparation of l-(3-hydroxypropyl)-5-[(2R)-({2-[2-(2, 2, 2-trifIuoroethoxy) phenoxy]-ethyl} amino) propyl]-2, 3-dihydro-lH-indole-7-carbonitriIe (XVII) A mixture of 3-[7-Cyano-5 (R)-[-2-{2-[2-(2,2,2-trifluoroethoxy)-phenoxy] ethyl} amino) propyl]-2,3-dihydro-lH-indol-l-yl}propyl benzoate (XVI) (6.0 g , 0.010 mole), methanol (30 ml) and aqueous solution of Sodium hydroxide ( 1.6 g in 8 ml of water) was stirred at ambient temperature for 6 hours. To the reaction mixture water (90ml) was added and product was extracted with ethyl acetate (90 ml). The organic layer was washed with saturated sodium bicarbonate solution followed by brine wash and dried over anhydrous sodium sulfate. The solvent was evaporated under reduced pressure to give 3.85 g of (XVII). Example 16
Preparation of l-(3-Hydroxy-propyl)-5(R)-(2-{2-[2-(2, 2, 2-trifluoro-ethoxy)- phenoxy]-ethylamino}-propyl)-2, 3-dihydro-lH-indol-7-carboxylic acid amide (I) (Silodosin)
To a solution of l-(3-hydroxypropyl)-5(R)-[2-({2-[2-(2,2,2-trifluoroethoxy)phenoxy]- ethyl}amino)propyl]-2,3-dihydro-lH-indole-7-carbonitrile (XVII) (6.0 g , 0.013 mole) in dimethylsulfoxide (75 ml) was added 5 N sodium hydroxide solution (4.5 ml). To this reaction mixture, 30 % hydrogen peroxide (2.63 ml) was added slowly below 25°C. Reaction mixture was stirred at ambient temperature for 6 hours. Aqueous solution of sodium sulfite (2.1 in 150 ml water) was added to the reaction mixture. The reaction mixture was extracted with ethyl acetate. The combined ethyl acetate layer was extracted 2N hydrochloric acid. The aqueous layer was neutralized with sodium bicarbonate and extracted the product in ethyl acetate. The organic layer was washed with saturated sodium bicarbonate solution followed by brine wash and dried over anhydrous sodium sulfate. The solvent was evaporated under reduced pressure, and the residue was dissolved in ethyl acetate. The resulting solution was cooled to 5°C and filtered to get 4.51 g of (I) as solid.
…………………………………………………
WO2012147019
http://www.google.com/patents/WO2012147019A1?cl=en
The present invention provides a process for the preparation of Silodosin of formula (I). More particularly, the present invention provides the process for preparation of tartrate salt of 3-[7-cyano-5[(2R)-2-({2-[2-(2,2,2- trifluoroethoxy)phenoxy ] ethyl } amino)propyl] -2, 3 -dihydro- 1 H-indol- 1 -y 1 } propyl benzoate of formula (IV), which is a precursor in the preparation of Silodosin.
Background of the Invention:
A compound of 3-[7-cyano-5[(2R)-2-({2-[2-(2,2,2-trifluoroethoxy) phenoxy] ethyl}amino)propyl]-2,3-dihydro-lH-indol-l-yl}propyl benzoate (IV) is a key intermediate for preparation of Silodosin. The chemical name of Silodosin is l-(3- hydroxypropyl)-5-[(2R)-2-({2-[2-(2,2,2-trifluoroethoxy)phenoxy]ethyl} amino) propyl]-2,3-dihydro-lH-indole-7-carboxamide and structurally represented as
(IV)
(I)
U.S.Pat. No. 5,387,603 discloses Silodosin as therapeutic agents for the treatment of dysuria, urinary disturbance associated with benign prostatic hyperplasia.
U.S.Pat. No. 6,310,086 discloses a process for preparing a Silodosin analogue compound from reaction of (R)-3-{5-(2-aminopropyl)-7-cyano-2,3- dihydro- 1 H-indol- 1 -yl jpropylbenzoate with 2-(2-Ethoxyphenoxy)ethyl methane sulfonate and finally isolated as residue and purified by column chromatography on silicagel. The said literature process has certain drawbacks like use of column chromatography.
U.S.Pat. No. 7,834,193 (IN 3178/DELNP/2007) discloses the process for preparation of monooxalate salt of 3-{7-cyano-5-[(2R)-2-({2-[2-(2,2,2- trifluoroethoxy)phenoxy ] ethyl } amino)propyl] -2, 3 -dihydro- 1 H-indol- 1 -y 1 } propyl benzoate (IV). This patent specifically discloses the preparation of monooxalate salt of formula (IV) helps to remove N,N-dialkyl impurity to certain extend. CN 101993405 A discloses the reaction of (R)-5-(2-aminopropyl)-l-(3-(4- fluorobenzoyloxy)propyl)-7-cyanoindoline with 2-(2-(2,2,2-trifluoroethoxy) phenoxy)ethyl methane sulfonate followed by oxalic acid salt preparation.
The main drawback in the prior art process, the formation of N,N-dialkyl impurity compound of formula (VI), as disclosed in detailed description, in the preparation of Silodosin, during condensation of compound of formula (II) with compound of formula (III), the impurity which is not removable by crystallization method or precipitation technique and column chromatography purification is not suitable for commercial purpose. So considering the commercial importance of Silodosin, the present invention focus on the preparation of pure Silodosin, and surprisingly found that the isolation of formula (IV) as tartrate salt helps to prepare Silodosin having less than 0.2 % of N,N dialkyl impurity and with good yield. None of the prior arts teaches or motivates isolation of tartaric acid addition salt of formula (IV). The preparation of Silodosin from tartrate salt of 3-{7-cyano-5-[(2R)- 2-({2-[2-(2,2,2-trifluoroethoxy)phenoxy] ethyl}amino)propyl]-2,3-dihydro-lH- indol-l-yl} propyl benzoate (IV) or its freebase of the present invention has purity of greater than 99.6 %.

Example 3
Preparation of l-(3-hydroxypropyl)-5-[(2R)-2-({2-[2-(2,2,2-trifluoroethoxy) phenoxy] ethyl-} amino) propyl]-2,3-dihydro-lH-indole-7-carboxamide (Silodosin)
Method A: The compound of l-(3-hydroxypropyl)-5-[(2R)-2-({2-[2-(2,2,2- trifluoroethoxy)phenoxy] ethyl- } amino)propyl] -2,3 -dihydro- 1 H-indole-7- carbonitrile of formula (V) in dimethylsulfoxide was treated with 48% hydrogen peroxide and 20% sodium hydroxide solution and stirred at room temperature till completion of reaction. After completion of reaction, reaction mass quenched with 5% sodium bisulphite solution and ethylacetate was added over it. The ethylacetate layer was separated and treated with 20 % aqueous hydrochloric acid. The aqueous layer separated, neutralized with sodium bicarbonate solution and extracted with ethylacetate. The separated organic layer was washed with 10% sodium bicarbonate solution, brine solution and dried under vacuum. The organic layer distilled upto residue under vacuum at 50-55°C. The obtained residue was crystallized in ethylacetate.
Method B: To the tartrate salt of 3-[7-cyano-5[(2R)-2-({2-[2-(2,2,2- trifluoroethoxy) phenoxy] ethyl}amino)propyl]-2,3-dihydro-lH-indol-l-yl}propyl benzoate (IV) (100 grams) in methanol, aqueous potassium hydroxide solution (38.38 grams) was added and stirred at room temperature till reaction completion. After completion of reaction, DM water and dichloromethane was added over it under stirring. Organic layer separated, washed with brine solution distilled under vacuum upto less than 1 volume. To the solution, dimethyl sulphoxide, 20% sodium hydroxide and hydrogen peroxide was added and stirred till completion of reaction. After completion of reaction, water containing sodium bisulfite was added to the reaction mass. The pH of the reaction mixture adjusted to about 8.5 using 10% sodium hydroxide and extracted in dichloromethane twice, washed with water, dried and concentrated upto 1-2 volume under vacuum. To the obtained solution, toluene was added over it at room temperature under stirring. The reaction mixture maintained for complete solid formation, filtered and dried under vacuum. Yield 58 grams. Example 4
Purification of Silodosin:
Method A: To the mixture of toluene and acetonitrile solvent, Silodosin was added over it and heated to 50° – 55 °C for complete dissolution. The reaction mass gradually cooled to room temperature and maintained for completion of solid formation. The obtained solid is filtered, washed with toluene and dried under vacuum. Method B: To the mixture of ethyl acetate and toluene solvent, Silodosin was added over it and heated to 60° – 65 °C for complete dissolution. The reaction mass gradually cooled to room temperature and maintained for completion of solid formation. The obtained solid is filtered, washed with toluene and dried under vacuum.
//////////////////////////////////////////
CN101993407
http://www.google.com/patents/CN101993407B?cl=en
silodosin for selective inhibition of urethral smooth muscle contraction and reduce the pressure within the urethra, but no significant impact on blood pressure, for the treatment of benign prostatic hyperplasia. At present, the method of synthesis Silodosin many reports, but the lack of high yield method for industrial production.
JP200199956 reported that benzoic acid as a starting material, 1_ (3_ benzoyloxy-propyl) indoline hydrochloride (structural formula (1), R is a hydrogen atom) in 60% yield, then through the multi-step reaction was further prepared silodosin intermediate 1- (3-benzoyloxy-propyl) -5- (2-nitro-propyl) -7-cyano-indoline (structural formula VIII ), the total yield is low, and only 20 percent. Compound (VIII) with potassium carbonate, the reaction of hydrogen peroxide to yield compound (IX), impurities, and purified by column chromatography to be not suitable for industrial production. Compound (IX) under catalysis of molybdenum oxide, and L- (S) – benzyl glycyl alcohol asymmetric reactions, protecting groups may be due to steric hindrance is small, low chiral induction, is 3.8: I.


Silodosin Preparation: 12 Example
Example 11 to give 8 g solid, dissolved in DMSO 100ml, was added 5mol / L NaOH 12ml, 18 ~ 20 ° C was added dropwise slowly with 30% H2027 grams, then 30 ° C, the reaction ended 4h. Extracted with ethyl acetate, the combined organic layer was washed 2N HCl and then the organic layer, the aqueous layer was neutralized with sodium hydroxide, and then extracted with ethyl acetate, washed with saturated sodium bicarbonate, dried over anhydrous sodium sulfate, and evaporated concentrated and then dissolved in ethyl acetate, natural cooling crystallization, filtration, drying 5 g (87%), purity> 99%.
Mp 105 ~ 108 ° C
[a] 20d = -16.2 C = I, MeOH
1NMR spectrum (DMS0-d6): δ ppm 0.9-1.0 (3H, d), 1.5-1.6 (1H, s), 1.6-1.7 (2H, m),
2.3-2.4 (1H, dd), 2.6-2.7 (1H, dd), 2.8-3.0 (5H, m), 3.1-3.2 (2H, m), 3.3-3.4 (2H, m),
3.4-3.5 (2H, t), 4.0-4.1 (2H, t), 4.2-4.3 (1H, s), 4.6-4.8 (2H, t), 6.9-7.15 (6H, m),
7.2-7.3 (1H, s), 7.5-7.6 (1H, s)
…………………………………………………..
see
Silodosin is (I) (formula 1, claim 1, page 31).
Process for the prepartion of pure polymorphic gamma form of silodosin – comprising dissolving any polymorphic form of silodosin in a solvent and seeding gamma form of silodosin.
Crude (I) (50 g) was dissolved in methanol, filtered and solvent was distilled under vacuum. The residue was dissolved in isopropanol at 50 degreeC, cooled and seed of (I) gamma form was added and further cooled and cyclohexane (500 mL) was added, solid was filtered, washed and dried to obtain pure polymorphic form gamma of (I) having a toluene content of 12 ppm (example 10, pages 29-30).
A process for the preparation of silodosin and/or its salt is claimed, comprising the reaction of 3-[5-((2R)-2-aminopropyl)-7-cyano-2,3-dihydro-1H-indol-1-yl]propyl benzoate(2R,3R)-monotartrate with 2-[2-(2,2,2-trifluoroethoxy)phenoxy]ethyl methanesulfonate to form a cyano benzyloxy intermediate, followed by hydrolysis to form a cyano hydroxy intermediate, which is then reacted with tartaric acid and hydrolyzed in the presence of an oxidizing agent to obtain the product. An alternate method of preparation of silodosin comprising the hydrolyses of tartrate salt of cyano hydroxy intermediate in the presence of an oxidizing agent, pure polymorphic form gamma of silodosin, and the cyano hydroxy intermediate are also claimed. Further processes for the prepartion of the pure polymorphic form gamma of silodosin are claimed, wherein the process involves the dissolution of of any polymorphic form of silodosin in a solvent by heating at 30-100 degree C, cooling before and after seeding with gamma form of silodosin, adding an antisolven, isolating the polymorph and optionally micronizing.
The present invention provides an improved and efficient process for the preparation of

It acts as an selective ai -adrenoceptor antagonist and is useful in the symptomatic treatment of benign prostatic hyperplasia (BPH). Chemically it is known as l-(3-hydroxypropyl)-5-[(2R)- ( { 2-[2-[2-(2,2,2-trifluoroethoxy)phenoxy]ethylamino) propyl] indoline-7-carboxamide.
Silodosin and its pharmaceutically acceptable salts are first disclosed in US patent 5,387,603. Synthetic approach for the production of silodosin, is described in patent ‘603 can be represented as shown below in scheme 1.
l

Scheme 1
As represented in scheme 1, silodosin is prepared by the reaction of l-acetyl-5-(2r aminopropyl)indoline-7-carbonitrile with 2-[2-(2,2,2-trifiuoroethoxy)phenoxy] ethyl methanesulfonate in the presence of sodium bicarbonate in ethanol to give l-acetyl-5-[2-[2-[2-(2,2,2-trifluoroethoxy)phenoxy]ethylamino]propyl]indoline-7-carbonitrile, which upon reaction with di-tert-butyldicarbonate in methylene chloride produces protected acetyl indoline carbonitrile compound. Further deacetylation with sodium hydroxide in ethanol followed by treatment with acetic acid provides protected indoline carbonitrile compound, which upon hydrolysis using dimethyl sulfoxide, 30% hydrogen peroxide, sodium hydroxide and acetic acid gives protected indoline carboxamide, which upon further reaction with 2-tert-butyldimethylsiloxy)ethyl-4-nitrobenzene sulfonate in the presence of cis-dicyclohexano-18 crown-6 and potassium carbonate in dioxane gives protected (tert-butyl-dimethylsiloxy) ethyl indoline carbonitrile. Further treatment with tetrabutylammonium fluoride in tetrahydrofuran produces N-boc protected hydroxy deprotected propyl indoline carbonitrile, which under goes facile deprotection of boc group upon treatment with trifluoroacetic acid, in methylene chloride to yield silodosin. The complete process is very complex, make use of pyrophoric reagents
which are very difficult to handle in large scale and have many extra steps involving protection and depfotection. Further in US patent ‘603, concrete detail of preparation and purification of silodosin have not been reported. Furthermore, isolated silodosin is characterized using IR, NMR and specific rotation but the patent is silent on product appearance and crystalline nature. There are several processes known for the preparation of silodosin and its intermediates viz; in JP 4634560; JP 4921646; JP-2006- 188470; WO2011/124704 and WO2011/101864. In most of the inventions, silodosin is prepared by following reaction as shown in scheme 2. Major disadvantages of these processes are the formation of N,N dialkyl impurity, and other impurities which forms during the condensation of 3-[5-((2/?)-2-aminopropyl)-7-cyano-2,3-dihydro-lH-indol-l-yl]propyl benzoate or its salts like monotartrate with 2-[2-(2,2,2-trifluoroethoxy)phenoxy] ethyl methanesulfonate. N,N dialkyl impurity forms in about 12-15% and may form due to reaction of one molecule of benzoate compound with two molecules of methanesulfonate compound. Removal of this impurity is not possible by simple purification

wherein R is benzoyl, benzyl, tetrahydropyranyl, 2-trimethylsilylethyl, dinitrophenyl, diphenyl methyl and the like
Scheme 2
US patent 7,834,193 discloses a process for preparation of silodosin with similar condensation of 3-[5-((2R)-2-arriinopropyl)-7-cyano-2,3-dihydro-lH-indol-l-yl]pfopyl benzoate or its salts like monotartrate with 2-[2-(2,2,2-trifluoroethoxy)phenoxy]ethyl methanesulfonate, but 3-{7-cyano-5-[(2R)-2-({2-(2,2,2-trifluoroethoxy)-phenoxy]ethyl}amino)propyl)-2,3-dihydro-lH-indol-l-yl)-propylbenzoate is purified by preparing monooxalate salt as shown below in
scheme 3. This patent specifically prepares monooxalate salt of 3- {7-cyano-5-[(2R)-2-({ 2- (2,2,2-trifluoroethoxy)-phenoxy]ethyl }amino)propyl)-2,3-dihydro-lH-indol-l-yl)-propyl benzoate to remove N,N÷dialkyl impurity, but impurity has not been removed completely, only a certain % of it, has been removed.

Scheme 3
In PCT publication WO2012/131710, preparation of silodosin is described wherein improved processes for preparation of 3-[5-((2R)-2-aminopropyl)-7-cyano-2,3-dihydro-lH-indol-l- yl]propyl benzoate have been disclosed which is then converted to silodosin by condensation with 2-[2-(2,2,2-trifluoroethoxy)phenoxy]ethyl methanesulfonate. In exemplified process, 3-[5- ((2R)-2-aminopropyl)-7-cyano-2,3-dihydro-lH-indol-l-yl]propyl benzoate is condensed with 2-[2-(2,2,2-trifluoroethoxy)phenoxy]ethyl methanesulfonate and the resulting benzoate compound is hydrolyzed to give l-(3-hydroxy propyl)-5-[(2R)-2-({ 2-[2,2,2-trifluoroethoxy) phenoxy] ethyl }amino)propyr]-2,3-dihydro-lH-indol-7-carbonitrile.The carbonitrile compound is treated with oxalic acid to prepare its oxalate salt having purity greater than 99%, which is then hydrolyzed using a base to prepare free carbonitrile compound having purity greater than 99%, but this patent is silent about N, N- dialkyl impurity or its removal.
In PCT publication WO2012/147019, preparation of silodosin using 3-{ 7-cyano-5-[(2R)-2-({2- (2,2,2-trifluoroethoxy)-phenoxy]ethyl}amino)propyl)-2,3-dihydro-lH-indol-l-yl)-propyl benzoate tartrate salt has been described as shown below in scheme 4.

Scheme 4
One other PCT publication WO2012/147107 describes preparation of silodosin by preparing hydrochloride and acetic acid salts of l-(3-hydroxypropyl)-5-[(2R)-2-({2-[2,2,2-trifluoroethoxy) phenoxy] ethyl }amino)propyl]-2,3-dihydro-lH-indol-7-carbonitrile to remove N,N dialkyl impurity. It has been observed that in exemplified process, wherein hydroxy compound namely l-(3-hydroxy propyl)-5-[(2R)-2-({2-[2,2,2-trifluoroethoxy) phenoxy] ethyl }amino)propyl]-2,3-dihydro-lH-indol-7-carbonitrile is purified by preparing its acetate salt to, remove the impurities but still N, N-dialkyl impurity remains in an amount of 0.6%, which is difficult to remove in next stage or require extra purifications.
Beside to use highly pure silodosin, use of a pure polymorphic form of API is an essential requirement of drug formulation, these both aspects when address jointly, and obtained silodosin can be converted to pure polymorph then only a complete solution of prior art problems can be achieved. Apart from above mentioned process patents/publications which aimed to prepare the pure silodosin, there are exist some polymorph patents/publications which also aims to prepare pure polymorphic form of silodosin.
Polymorphism is considered as one of the- most important solid-state property of drug substance, since different polymorph have different physiochemical and biological properties and in pharmaceutical chemistry it is often desired to obtain one particular form that is biologically active and also offers ease of handling during formulation. The available literature references related to polymorph of silodosin are incorporated herein.
Japanese patent 3331048 (publication No.H07-330726), discloses a process for purification of silodosin wherein silodosin is dissolved in ethyl acetate, dried over anhydrous magnesium sulfate, solvent is distilled off and again dissolved in ethyl acetate at 70°C and crystallizes below room temperature. The resulting product is characterized by melting point, IR, NMR and specific rotation. Here also disclosure is silent about polymorphic form of product.
US patent publication US2006/0142374A1 (equivalent European patent EP1541554B 1) discloses polymorphic forms of silodosin including three crystalline polymorphic form of silodosin which are named as alpha (a), beta (β) and gamma (γ) and one amorphous form. These polymorphic forms have been characterized by X-ray powder diffraction pattern. In the patent publication, processes for the preparation of all these three crystalline forms have been disclosed. In. a given process, form alpha is prepared by dissolving crude silodosin in appropriate amount of ethyl acetate, ethyl formate, acetone, methyl ethyl ketone, acetonitrile, tetrahydrofuran or mixture of acetone and acetonitrile (1: 1), preferably ethyl acetate under heating, allowing to stand at room temperature to precipitate the crystal gradually. Similarly, form beta is prepared by dissolving crude silodosin in appropriate amount of methanol under heating, adding petroleum ether as a anti-solvent, crystal precipitation is ensured using vigorous stirring.
In a second process, to prepare the form beta, crude silodosin is dissolved in ethanol or 1-propanol and the reaction mass is cooled quickly. The crystalline form gamma is prepared by dissolving crude silodosin in appropriate amount of toluene or a mixture of acetonitrile and toluene (1:4) or ethyl acetate and toluene (1: 19), preferably in toluene, under heating, cooling to room temperature and allowing to precipitate gradually upon standing. In a second process to prepare form gamma, crude silodosin is dissolved in 2-propanol and the crystals are precipitated by adding an appropriate amount of toluene. In spite of disclosing three crystalline polymorphic forms, the patent publication prefers preparation and use of form alpha by highlighting the problems faced for preparation and use of other forms. It is disclosed that crystal form beta has manufacturing difficulties at industrial scale since precipitation occurs only when the nonpolar antisolvent is added to warm solution which leads to inconsistency in quality of crystals.
With the second process for preparation of form beta, desired level of yield and purity has not been achieved. Further, according to this publication, preparation of gamma form involves use of toluene which can not be removed completely from final product, because of its high boiling point and raises the problem of residual solvent. In the case of toluene, a class 2 solvent, its limits should not be more than 890 ppm. In the exemplified process, toluene content has not been disclosed, which clearly reflects that product was not suitable for pharmaceutical composition having problem of high residual content of toluene. Furthermore patent publication also states that all the three crystal forms donot have any difference in hygroscopicity and stabilities.
Thereafter, several patents/publications disclose preparation of polymorphic forms alpha and beta. For example a PCT publication WO2012/147107 discloses a process for preparation of beta form using isopropyl acetate and methyl isobutylketone. In another PCT publication WO2012/077138, preparation of alpha and beta forms are disclosed using various solvent , system. Similarly, in a Chinese patent CN102010359, crystalline form beta is prepared by dissolving the crude silodosin in alcoholic solvent by heating and the product is crystallized by cooling or by adding an antisolvent such as ketone or ether.
European patent EP2474529 discloses new polymorphic forms delta (δ) and eta (ε) of silodosin by using a solvent (tetrahydrofuran) and antisolvent (n-heptane, n-hexane, cyclohexane, tert butylmethyl ether).Further it discloses conversion of delta form to beta form by just heating the delta form at a particular temperature. The form delta can also be transformed into form eta by. slurrying in aqueous methanol. One new crystalline form designated as delta has also been disclosed in a Chinese patent publication CN102229558. An Indian patent application 478/MUM/2010, also discloses a new polymorphic form Zy-S which is prepared by using solvent such as esters, aromatic hydrocarbons, ketones, and alcohols.
All the above disclosures are silent about the preparation of gamma form of silodosin and only available disclosure reports that gamma form have problem of residual solvent, as impurity and is not suitable for pharmaceutical compositions.
Method C: l-(3-HydroxypropyI)-5-[(2R)-2-({2-[2,2,2-trifIuoroethoxy)phenoxy] ethyl} amino) propyl]-2,3-dihydro-lH-indol-7-carbonitrile tartrate (lOg) dissolved in dimethylsulfoxide (120 ml) and to this solution, was added 5 mol/L aqueous sodium hydroxide solution (15ml). To the reaction mixture, 30% hydrogen peroxide (5ml) was added and keeping the temperature below 25°C. The reaction mixture was stirred at 20-25°C, for 5 hours. To the reaction mixture, sodium sulfite (5g) dissolved in water (100ml) was added slowly. The reaction mixture was extracted with ethyl acetate (1x200ml) and ethyl acetate layer was concentrated under reduced pressure. The resulting product was dissolved in methanol and clear solution was filtered through micron filter paper of size 0.22 micron two times and filtrate was concentrated.The resulting compound was dissolved in toliiene (70ml) and isopropyl alcohol (7ml) at 50-55°C and the solution was cooled to 20-25°C, cyclohexane was added and stirred for further 4 hours, filtered and dried to give title compound having purity 99.86% and N,N-dialkyl impurity not detected by HPLC. Example 5: Preparation of pure Polymorphic Form Gamma (γ) of Silodosin
Silodosin (15g) having toluene content 1872 ppm, was micronized under air pressure. The micronized product was dried under vacuum at 55°C-60°C for 23.0 hours to afford pure polymorphic form gamma of silodosin having toluene content 460 ppm.
Example 6: Preparation of pure Polymorphic Form Gamma (γ) of Silodosin
Silodosin [having toluene content 1327 ppm] was micronized under air pressure. The micronized product was dried under vacuum at 55°C-60°C for 16 hours to afford pure polymorphic form gamma of silodosin having toluene content 350 ppm.
Example 7: Preparation of pure Polymorphic Form Gamma (γ) of Silodosin
Silodosin crude (3.0g) was dissolved in isopropanol (12ml) at 50°C and reaction mass was cooled to 35°C and seed of silodosin gamma form (O.lg) was added. Thereafter reaction mass was again cooled to 15-20°C and cyclohexane (30ml) was added to the reaction mass and stirred for further 0.5 hour. The resulting solid, thus obtained, was filtered, washed with cyclohexane and dried to afford pure polymorphic form gamma of silodosin having toluene content 34 ppm.
References
- “Drugs.com, Watson Announces Silodosin NDA Accepted for Filing by FDA for the Treatment of Benign Prostatic Hyperplasia”. Retrieved 2008-02-13.
- European Medicines Agency: Assessment report for Silodyx
- Schubert-Zsilavecz, M, Wurglics, M, Neue Arzneimittel 2008/2009
- Kobayashi K, Masumori N, Kato R, Hisasue S, Furuya R, Tsukamoto T. (December 2009). “Orgasm is preserved regardless of ejaculatory dysfunction with selective alpha1A-blocker administration.”. Int J Impot Res. 21 (5): 306–10. doi:10.1038/ijir.2009.27. PMC 2834370. PMID 19536124.
External links
a1a-Adrenoceptor antagonist. Prepn: M. Kitazawa et al., EP 600675; eidem, US 5387603 (1994, 1995 both to Kissei).PRODUCT PATENT
Adrenoceptor binding study: K. Shibata et al., Mol. Pharmacol. 48, 250 (1995); and tissue selectivity: S. Murata et al., J. Urol. 164, 578 (2000).
Pharmacology: K. Akiyama et al., Pharmacology 64, 140 (2002).
Series of articles on pharmacology, pharmacokinetcs and toxicology: Yakugaku Zasshi 126, 187-263 (2006).
Review of development and therapeutic potential: F. Kamali, Curr. Opin. Cent. Peripher. Nerv. Syst. Invest. Drugs 1, 248-252 (1999)
| CN101993405A * | Aug 27, 2009 | Mar 30, 2011 | 浙江华海药业股份有限公司;上海医药工业研究院 | Indoline derivative as well as preparation method and application thereof |
| JP2006188470A * | Title not available | |||
| US7834193 * | Apr 16, 2007 | Nov 16, 2010 | Kissei Pharmaceutical Co., Ltd. | industrial production of silodosin (for treating dysuria associated with benign prostatic hyperplasia) via mixing 3-{7-cyano-5-[(2R)-2-({2-[2-(2,2,2-trifluoroethoxy)-phenoxy]ethyl}amino]propyl]-2,3-dihydro-1H-indol-1-yl}-propyl benzoate and oxalic acid, nitrilizing, hydrolyzing |
| WO2011030356A2 * | Sep 13, 2010 | Mar 17, 2011 | Sandoz Ag | Process for the preparation of indoline derivatives and their intermediates thereof |
| WO2011124704A1 * | Apr 8, 2011 | Oct 13, 2011 | Ratiopharm Gmbh | Process for preparing an intermediate for silodosin |
| WO2012131710A2 * | Mar 27, 2012 | Oct 4, 2012 | Panacea Biotec Ltd | Novel process for the synthesis of indoline derivatives |
| JP2006188470A * | Title not available |
| Patent | Submitted | Granted |
|---|---|---|
| Combination therapy for the treatment of benign prostatic hyperplasia [US6410554] | 2002-06-25 | |
| Indoline compound and process for producing the same [US7834193] | 2007-08-23 | 2010-11-16 |
| Agents and crystals for improving excretory potency of urinary bladder [US8252814] | 2009-10-22 | 2012-08-28 |
| METHODS FOR TREATING BENIGN PROSTATIC HYPERPLASIA [US2011319464] | 2011-12-29 | |
| PREVENTIVE AND/OR THERAPEUTIC AGENT FOR URINE COLLECTION DISORDER ACCOMPANYING LOWER URINARY TRACT OBSTRUCTION [US2009227651] | 2009-09-10 | |
| PREVENTIVE AND/OR THERAPEUTIC AGENT FOR URINE COLLECTION DISORDER ACCOMPANYING LOWER URINARY TRACT OBSTRUCTION [US2010137399] | 2010-06-03 | |
| Agents for improving excretory potency of urinary bladder [US2004116457] | 2004-06-17 | |
| Medicinal Composition for Prevention of Transition to Operative Treatment for Prostatic Hypertrophy [US2008090893] | 2008-04-17 | |
| METHODS FOR TREATING BENIGN PROSTATIC HYPERPLASIA [US2008242717] | 2008-10-02 | |
| Agents and crystals for improving excretory potency of urinary bladder [US2006281725] | 2006-12-14 |