Home » Uncategorized (Page 204)
Category Archives: Uncategorized
Adamas claims success with new and improved Parkinson’s drug
See on Scoop.it – New drug approvals
amantadine Adamas claims success with new and improved Parkinson’s drug Adamas Pharmaceuticals is claiming success with a Phase II/III study of its extended-release version of amantadine for Parkin…
Medicinal Plants Used To Treat Sexual Dysfunction- A review
See on Scoop.it – New drug approvals
Medicinal Plants Used To Treat Sexual Dysfunction: A Review by Ramandeep et al. Int J Recent Adv Pharm Res, 2013;3(1):29-35. ISSN: 2230-9306; www.ijrapronline.com. 29. Medicinal Plants Used To Tr…
Medicinal Plants Used To Treat Sexual Dysfunction- A review
See on Scoop.it – New drug approvals
Medicinal Plants Used To Treat Sexual Dysfunction: A Review by Ramandeep et al. Int J Recent Adv Pharm Res, 2013;3(1):29-35. ISSN: 2230-9306; www.ijrapronline.com. 29. Medicinal Plants Used To Tr…
Adamas claims success with new and improved Parkinson’s drug
See on Scoop.it – New drug approvals
amantadine Adamas claims success with new and improved Parkinson’s drug Adamas Pharmaceuticals is claiming success with a Phase II/III study of its extended-release version of amantadine for Parkin…
Adamas claims success with new and improved Parkinson’s drug
amantadine
Adamas claims success with new and improved Parkinson’s drug
Amantadine (trade name Symmetrel, by Endo Pharmaceuticals) is a drug that has US Food and Drug Administration approval for use both as an antiviral and an antiparkinsonian drug. It is the organic compound 1-adamantylamine or 1-aminoadamantane, meaning it consists of an adamantane backbone that has an amino group substituted at one of the four methyne positions. Rimantadine is a closely related derivative of adamantane with similar biological properties.
Apart from medical uses, this compound is useful as a building block, allowing the insertion of an adamantyl group.
According to the US Centers for Disease Control and Prevention, 100% of seasonal H3N2 and 2009 pandemic flu samples tested have shown resistance to adamantanes, and amantadine is no longer recommended for treatment of influenza in the United States. Additionally, its effectiveness as an antiparkinsonian drug is undetermined, with a 2003 Cochrane Review concluding that there was insufficient evidence in support or against its efficacy and safety.
Synthesis
Amantadine may be prepared by reacting adamantane with bromine or nitric acid to give the bromide or nitroester at position one. Reaction of either compound with acetonitrile affords the acetamide, which is hydrolyzed to give 1-adamantylamine:
New Drug Approval Process: Regulatory View
DRUG REGULATORY AFFAIRS INTERNATIONAL
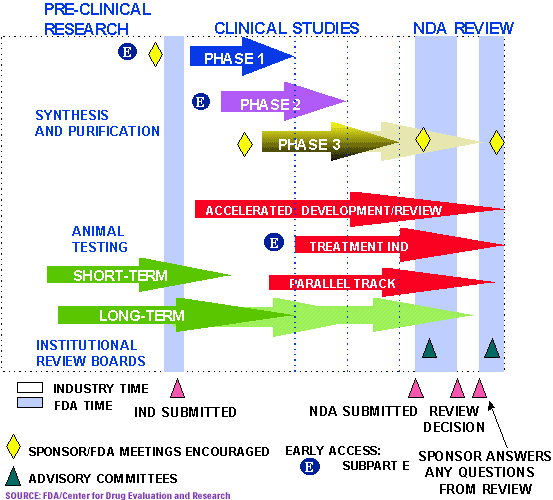
read all on
New Drug Approval Process: Regulatory View
http://www.pharmainfo.net/reviews/new-drug-approval-process-regulatory-view
Medicinal Plants Used To Treat Sexual Dysfunction- A review
Medicinal Plants Used To Treat Sexual Dysfunction: A Review
Ramandeep et al.

BMS and Simcere will co-develop and co-commercialize the subcutaneous formulation of Orencia® for the treatment of rheumatoid arthritis in China.

AsianScientist (Jun. 17, 2013) – Bristol-Myers Squibb Company and Simcere Pharmaceutical Group announced this week a new collaboration to co-develop and commercialize the subcutaneous (SC) formulation of BMS’s Orencia® (abatacept) for the treatment of rheumatoid arthritis in China.
Orencia SC is already on the market for the treatment of rheumatoid arthritis in the U.S., Europe, and Japan.
http://www.asianscientist.com/tech-pharma/bms-simcere-pharma-co-develop-orencia-sc-china-2013/
British Alliance Pharma plc has bought the complete rights for Novartis’ obstetric drug Syntometrine

oxytocin

ergometrine
Oxytocin/ergometrine (trade name Syntometrine) is an obstetric combination drug. The components are synthetically produced oxytocin, a human hormone produced in thehypothalamus, and ergometrine, an alpha-adrenergic, dopaminergic and serotonin (5-HT2) receptor agonist.
Both substances cause the uterus to contract. An injection of syntometrine is given in thethird stage of labor, just after the birth of the child, to facilitate delivery of the placenta and to prevent postpartum hemorrhage by causing smooth muscle tissue in the blood vessel walls to narrow, thereby reducing blood flow.
British Alliance Pharma plc has bought the complete rights for Novartis’ obstetric drug Syntometrine.
The Chippenham-based specialty company already owns the UK rights to Syntometrine, an obstetric drug used in the final stage of labour. With the US$11.5m deal, Alliance Pharma adds Novartis’ assets in other countries to its portfolio. read all at
http://www.eurobiotechnews.eu/news/news/2013-02/alliance-pharma-bags-novartis-drug.html
Constituents And Factors Of Technology Transfer In Pharmaceutical Industry

Technology transfer is helpful to develop dosage forms in various ways as it provides efficiency in process, maintains quality of product, helps to achieve standardized process which facilitates cost effective production. It is the process by which an original innovator of technology makes its technology available to commercial partner that will exploit the technology.
Technology transfer is both integral and critical to drug discovery and development for new medicinal products. The cost of product development raises during pilot scale-up and initial production batch i.e. the critical path for success is dependent on completion of technology transfer to the production site at an affordable cost.
Technology transfer is defined as “The processes that are needed for successful progress from drug discovery to product development to clinical trials to full-scale commercialization.”

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....














