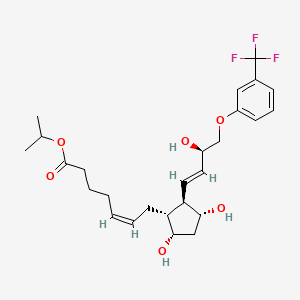
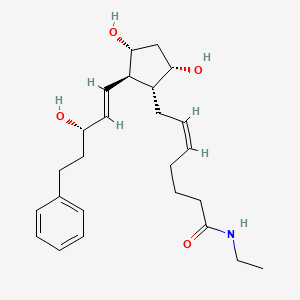
The drug maker is seeing great signs in the development of treatment for multiple myeloma, a bone marrow cancer. The results from its Phase 3 of Kyprolis’ clinical trial shows that patients can live almost nine months longer without worsening symptoms. According to Amgen, about 70,000 people in the U.S. are living with the disease and 24,000 new cases are diagnosed every year. With the good clinical trial result, Amgen plans to begin regulatory submissions around the world next year. Dr. Pablo Cagnoni, president of Amgen’s subsidiary Onyx Pharmaceuticals said, “The results demonstrate that Kyprolis can significantly extend the time patients live without their disease progressing. The ability of novel therapies to produce deep and durable responses may, one day, transform this uniformly fatal disease to one that is chronic and manageable.” Male patients over the age of 65 have the highest risk of developing it.
Home » Uncategorized (Page 111)
Category Archives: Uncategorized
Amgen’s Multiple Myeloma Drug Shows Promise in Phase 3 Trial
 Carfilzomib
Carfilzomib
Amgen’s Multiple Myeloma Drug Shows Promise in Phase 3 Trial
https://finance.yahoo.com/video/amgens-multiple-myeloma-drug-shows-195603222.html
Carfilzomib (marketed under the trade name Kyprolis, Onyx Pharmaceuticals, Inc.) is an anti-cancer drug acting as a selectiveproteasome inhibitor. Chemically, it is a tetrapeptide epoxyketone and an analog of epoxomicin.[1]
The U.S. Food and Drug Administration (FDA) approved it on 20 July 2012 for use in patients with multiple myeloma who have received at least two prior therapies, including treatment with bortezomib and an immunomodulatory therapy and have demonstrated disease progression on or within 60 days of completion of the last therapy. Approval is based on response rate. Clinical benefit, such as improvement in survival or symptoms, has not been verified.[2]
The abbreviation CFZ is common for referring to carfilzomib, but abbreviating drug names is not best practice in medicine.
Discovery, early development and regulatory approval
Carfilzomib is derived from epoxomicin, a natural product that was shown by the laboratory of Craig Crews at Yale University to inhibit the proteasome.[3] The Crews laboratory subsequently invented a more specific derivative of epoxomicin named YU101,[4] which was licensed to Proteolix, Inc. Craig Crews, Raymond Deshaies from Caltech, Phil Whitcome, the former CEO of Neurogen and Larry Lasky, a venture capitalist, founded Proteolix, and along with other researchers and scientists, advanced YU101. The scientists at Proteolix invented a new, distinct compound that had potential use as a drug in humans, known as carfilzomib. Proteolix advanced carfilzomib to multiple Phase 1 and 2 clinical trials, including a pivotal Phase 2 clinical trial designed to seek accelerated approval.[5]Clinical trials for carfilzomib continue under Onyx Pharmaceuticals, which acquired Proteolix in 2009.[5]
In January 2011, the FDA granted carfilzomib fast-track status, allowing Onyx to initiate a rolling submission of its new drug application for carfilzomib.[6] In December 2011, the FDA granted Onyx standard review designation,[7][8] for its new drug application submission based on the 003-A1 study, an open-label, single-arm Phase 2b trial. The trial evaluated 266 heavily-pretreated patients with relapsed and refractory multiple myeloma who had received at least two prior therapies, including bortezomib and either thalidomide or lenalidomide.[9] It costs approximately $10,000 per 28-day cycle, making it the most expensive FDA-approved drug for multiple myeloma.[10]
Mechanism
Carfilzomib irreversibly binds to and inhibits the chymotrypsin-like activity of the 20S proteasome, an enzyme that degrades unwanted cellular proteins. Inhibition of proteasome-mediated proteolysis results in a build-up of polyubiquinated proteins, which may cause cell cycle arrest, apoptosis, and inhibition of tumor growth.[1]
Clinical trials
Completed
A single-arm, Phase II trial (003-A1) of carfilzomib in patients with relapsed and refractory multiple myeloma showed that single-agent carfilzomib demonstrated a clinical benefit rate of 36 percent in the 266 patients evaluated and had an overall response rate of 22.9 percent and median duration of response of 7.8 months. The FDA approval of carfilzomib was based on results of the 003-A1 trial.[11]
In a Phase II trial (004), carfilzomib had a 53 percent overall response rate among patients with relapsed and/or refractory multiple myeloma who had not previously received bortezomib. This study also included a bortezomib-treated cohort. Results were reported separately.[12] This study also found prolonged carfilzomib treatment was tolerable, with approximately 22 percent of patients continuing treatment beyond one year. The 004 trial was a smaller study originally designed to investigate the impact of carfilzomib treatment in relationship to bortezomib treatment in less heavily pretreated (1-3 prior regimens) patients.[13]
A Phase II trial (005), which assessed the safety, pharmacokinetics, pharmacodynamics and efficacy of carfilzomib, in patients with multiple myeloma and varyi ng degrees of renal impairment, where nearly 50 percent of patients were refractory to both bortezomib and lenalidomide, demonstrated that pharmacokinetics and safety were not influenced by the degree of baseline renal impairment. Carfilzomib was tolerable and demonstrated efficacy.[14]
In another Phase II trial (006) of patients with relapsed and/or refractory multiple myeloma, carfilzomib in combination with lenalidomide and dexamethasone demonstrated an overall response rate of 69 percent.[15]
A Phase II trial (007) for multiple myeloma and solid tumors showed promising results.[16][17]
In Phase II trials of carfilzomib, the most common grade 3 or higher treatment-emergent adverse events were thrombocytopenia, anemia, lymphoenia, neutropenia, pneumonia, fatigue and hyponatremia.[18]
In a frontline Phase I/II study, the combination of carfilzomib, lenalidomide, and low-dose dexamethasone was highly active and well tolerated, permitting the use of full doses for an extended time in newly-diagnosed multiple myeloma patients, with limited need for dose modification. Responses were rapid and improved over time, reaching 100 percent very good partial response.[19]
Ongoing
A phase III confirmatory clinical trial, known as the ASPIRE trial, comparing carfilzomib, lenalidomide and dexamethasone versus lenalidomide and dexamethasone in patients with relapsed multiple myeloma is ongoing.[20] It is no longer recruiting and should report in 2014.

| Systematic (IUPAC) name | |
|---|---|
| (S)-4-Methyl-N-((S)-1-(((S)-4-methyl-1-((R)-2-methyloxiran-2-yl)-1-oxopentan-2-yl)amino)-1-oxo-3-phenylpropan-2-yl)-2-((S)-2-(2-morpholinoacetamido)-4-phenylbutanamido)pentanamide | |
| Clinical data | |
| Trade names | Kyprolis |
| Licence data | US FDA:link |
| Pregnancy cat. | D (US) |
| Legal status | ℞-only (US) |
| Routes | Intravenous |
| Identifiers | |
| CAS number | 868540-17-4 |
| ATC code | L01XX45 |
| PubChem | CID 11556711 |
| ChemSpider | 9731489 |
| KEGG | D08880 |
| ChEMBL | CHEMBL451887 |
| Synonyms | PX-171-007 |
| Chemical data | |
| Formula | C40H57N5O7 |
| Mol. mass | 719.91 g mol |
http://pubs.rsc.org/en/content/articlelanding/2013/np/c3np20126k/unauth#!divAbstract
The initial enthusiasm following the discovery of a pharmacologically active natural product is often fleeting due to the poor prospects for its ultimate clinical application. Despite this, the ever-changing landscape of modern biology has a constant need for molecular probes that can aid in our understanding of biological processes. After its initial discovery by Bristol-Myers Squibb as a microbial anti-tumor natural product, epoxomicin was deemed unfit for development due to its peptide structure and potentially labile epoxyketone pharmacophore. Despite its drawbacks, epoxomicin’s pharmacophore was found to provide unprecedented selectivity for the proteasome. Epoxomicin also served as a scaffold for the generation of a synthetic tetrapeptide epoxyketone with improved activity, YU-101, which became the parent lead compound of carfilzomib (Kyprolis™), the recently approved therapeutic agent for multiple myeloma. In this era of rational drug design and high-throughput screening, the prospects for turning an active natural product into an approved therapy are often slim. However, by understanding the journey that began with the discovery of epoxomicin and ended with the successful use of carfilzomib in the clinic, we may find new insights into the keys for success in natural product-based drug discovery.
References
- Carfilzomib, NCI Drug Dictionary
- “FDA Approves Kyprolis for Some Patients with Multiple Myeloma”. FDA. 2012-07-20. Retrieved 2013-07-23.
- Meng, L; Mohan, R.; Kwok, B.H.; Elofsson, M.; Sin, N.; Crews, C.M. (1999).“Epoxomicin, a potent and selective proteasome inhibitor, exhibits in vivo antiinflammatory activity”. Proc Natl Acad Sci USA 96 (18): 10403–8.doi:10.1073/pnas.96.18.10403. PMC 17900. PMID 10468620.
- Myung, J; Kim, K.B.; Lindsten, K.; Dantuma, N.P.; Crews, C.M. (2001). “Lack of proteasome active site allostery as revealed by subunit-specific inhibitors”. Mol Cell 7 (2): 411–20. doi:10.1016/S1097-2765(01)00188-5. PMID 11239469.
- ^ Jump up to:a b “Carfilzomib: From Discovery To Drug”. Chemical & Engineering News. 2012-08-27. Retrieved 2013-07-30.
- “Onyx multiple myeloma drug wins FDA fast-track status”. San Francisco Business Times. 2011-01-31. Retrieved 2011-09-01.
- “Beacon Breaking News – Carfilzomib to Get Standard, Not Priority, FDA Review”. The Myeloma Beacon. Retrieved 2012-02-27.
- “Fast Track, Accelerated Approval and Priority Review; Accelerating Availability of New Drugs for Patients with Serious Diseases”. FDA. Retrieved 2012-02-27.
- “PX-171-003-A1, an open-label, single-arm, phase (Ph) II study of carfilzomib (CFZ) in patients (pts) with relapsed and refractory multiple myeloma (R/R MM): Long-term follow-up and subgroup analysis”. ASCO 2011; Abstract 8027. 2011. Retrieved 2011-09-01.
- “FDA Approves Kyprolis (Carfilzomib) For Relapsed And Refractory Multiple Myeloma”. The Myeloma Beacon. Retrieved 2012-07-20.
- “Carfilzomib Prescribing Information”. NCI Drug Dictionary. Retrieved 2013-07-23.
- Vij, R (2012). “An open-label, single-arm, phase 2 study of single-agent carfilzomib in patients with relapsed and/or refractory multiple myeloma who have been previously treated with bortezomib”. Br J Haematol 158 (6): 739–748. doi:10.1111/j.1365-2141.2012.09232.x. PMID 22845873.
- Vij, R (2012). “An open-label, single-arm, phase ii (PX-171-004) study of single-agent carfilzomib in bortezomib-naive patients with relapsed and/or refractory multiple myeloma.”. Blood 119 (24): 5661–70. doi:10.1182/blood-2012-03-414359.PMID 22555973.
- Badros, AZ (2013). “Carfilzomib in multiple myeloma patients with renal impairment: pharmacokinetics and safety.”. Leukemia 27 (8): 1707–14. doi:10.1038/leu.2013.29.PMID 23364621.
- “European Hematology Association (EHA) 18th Congress. June 13-16, 2013.”. The Myeloma Beacon. 2013. Retrieved 2013-07-13.
- “Nikoletta Lendval, MD PhD et al. Phase II Study of Infusional Carfilzomib in Patients with Relapsed or Refractory Multiple Myeloma.”. Presented at: 54th ASH Annual Meeting and Exposition: December 2012. Retrieved 2013-07-23.
- “Phase II results of Study PX-171-007: A phase Ib/II study of carfilzomib (CFZ), a selective proteasome inhibitor, in patients with selected advanced metastatic solid tumors” – ASCO 2009; Abstract 3515.
- “Siegel DS, Martin T, Wang, M, et al. Results of PX-171- 003-A1, an open-label, single-arm, phase 2 study of carfilzomib in patients with relapsed and refractory multiple myeloma. Presented at: 52nd ASH Annual Meeting and Exposition; December 4-7, 2010; Orlando, Florida.”. OncLive.com. 2011-03-09. Retrieved 2011-09-01.
- “Final Results of a Frontline Phase 1/2 Study of Carfilzomib Lenalidomide, and Low-Dose Dexamethasone (CRd) in Multiple Myeloma (MM)”. ASH 20111; Abstract 631. Retrieved 2012-02-27.
- “Phase 3 Study Comparing Carfilzomib, Lenalidomide, and Dexamethasone (CRd) Versus Lenalidomide and Dexamethasone (Rd) in Subjects With Relapsed Multiple Myeloma”. ClinicalTrials.gov. 2011-08-04. Retrieved 2011-09-01.
External links
- “Carfilzomib Prescribing Information”. NCI Drug Dictionary.
Mangafodipir



| Clinical data | |
|---|---|
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Pregnancy cat. | Not to be used |
| Routes | Intravenous infusion |
| Pharmacokinetic data | |
| Bioavailability | NA |
| Protein binding | 27% (manganese) Negligible (DPDP) |
| Half-life | 20 minutes (manganese) 50 minutes (DPDP) |
| Excretion | Renal and fecal (manganese) Renal (DPDP) |
| Identifiers | |
| ATC code | V08CA05 |
| PubChem | CID 3086672 |
| ChemSpider | 2343239 |
| UNII | N02W67RKJS |
| Chemical data | |
| Formula | C22H28MnN4O14P2 |
| Mol. mass | 689.362 g/mol |
Mangafodipir (sold under the brand name Teslascan as mangafodipir trisodium) is a contrast agent delivered intravenously to enhance contrast in magnetic resonance imaging (MRI) of the liver. It has two parts, paramagnetic manganese (II) ions and thechelating agent fodipir (dipyridoxyl diphosphate, DPDP). Normal liver tissue absorbs the manganese more than abnormal or cancerous tissue. The manganese shortens the longitudinal relaxation time (T1), making the normal tissue appear brighter in MRIs. This enhanced contrast allows lesions to be more easily identified.

The condensation of pyridoxal 5-phosphate (I) with ethylenediamine (II) in methanol by means of NaOH gives the corresponding diimine (III), which is reduced with hydrogen over Pt/C in methanol/water yielding the expected diamine (IV). The reaction of (IV) with bromoacetic acid (V) by means of NaOH in methanol/water affords the N,N’-diacetic acid derivative (VI), which is finally treated with MnCl2 in water containing NaOH.
References
C22H27MnN4Na3O14P2 ![]()
![]()
![]()
![]()
![]() 757.33
757.33
Trisodium trihydrogen (OC-6-13)-[[N,N¢-1,2-ethanediylbis[N-[[3-hydroxy-2-methyl-5-[(phosphonooxy)methyl]-4-pyridinyl]methyl]glycinato]](8-)] manganate(6-).
Trisodium trihydrogen (OC-6-13)-[[N,N¢-ethylenebis[N-[[3-hydroxy-5-(hydroxymethyl)-2-methyl-4-pyridyl]methyl]glycine] 5,5¢-bis(phosphato)](8-)]manganate(6-) ![]()
![]()
![]() [140678-14-4].
[140678-14-4].
NS 398 is a COX-2 inhibitor used in the study of the function of cyclooxygenases.

NS 398
N-[2-(Cyclohexyloxy)-4-nitrophenyl]methanesulfonamide
N-(2-cyclohexyloxy-4-nitrophenyl)methanesulfonamide.
123653-11-2, 123653-43-0 (Ca salt), 123653-44-1 (Na salt)
Cerebrovascular Diseases, Treatment of, NEUROLOGIC DRUGS, Stroke, Treatment of, Cyclooxygenase-2 Inhibitors
NS-398 is a COX-2 inhibitor used in the study of the function of cyclooxygenases.[2]
Selective cyclooxygenase-2 inhibitor (IC50 values are 3.8 and > 100 μM for COX-2 and COX-1 respectively). Orally active. Anti-inflammatory, anti-pyretic, analgesic and non-ulcerogenic in vivo. Induces apoptosis and cell cycle arrest
Cyclooxygenase (COX-2) has been recently suggested to play a role in hepatocarcinogenesis. However, the exact pathway by which COX-2 affects the growth of hepatocellular carcinoma (HCC) is not clear. This study investigated the effects of a specific COX-2 inhibitor, NS-398, on the cell proliferation and apoptosis of COX-2-expressing and non-expressing HCC cell lines.
In addition, the modulatory effect of NS-398 on apoptosis-regulating gene expression was examined. Semi-quantitative/quantitative reverse transcription-polymerase chain reaction and Western blot showed that Hep3B and HKCI-4 cells expressed COX-2 mRNA and protein, but HepG2 cells did not. NS-398 suppressed cell proliferation and induced apoptosis in the two COX-2-expressing cell lines in a dose-dependent manner, but not in HepG2 cells.
Fas ligand mRNA and protein expression were increased by the treatment with NS-398 (10 micro M) in COX-2-expressing cell lines. The expressions of Fas and Bcl-2 family genes (Bax, Bcl-2, Bcl-xL, Bcl-xS) were not affected by NS-398 treatment in all three cell lines. In conclusion, specific COX-2 inhibitor suppresses cell proliferation and induces apoptosis in HCC cell lines that express COX-2. Our finding suggests that COX-2 inhibition may offer a new approach for HCC chemoprevention.
| Identifiers | |
|---|---|
| CAS number | 123653-11-2 |
| PubChem | 4553 |
| Jmol-3D images | Image 1 |
| Properties | |
| Molecular formula | C13H18N2O5S |
| Molar mass | 314.36 g mol−1 |
| Appearance | Off-white solid |
| Solubility in water | Insoluble |
| Solubility in DMSO | 5 mg/mL |
| Hazards | |
| S-phrases | S22 S24/25 |

The condensation of 2-fluoronitrobenzene (I) with cyclohexanol (II) by means of NaH gives 2-(cyclohexyloxy)nitrobenzene (III), which is reduced with H2 over Pd/C in methanol yielding 2-(cyclohexyloxy)aniline (IV). The acylation of (IV) with methanesulfonyl chloride (V) in pyridine affords N-(2-cyclohexyloxy phenyl)methanesulfonamide (VI), which is finally nitrated with concentrated HNO3 in hot acetic acid.
EP 0317332
http://www.google.com/patents/EP0317332A2?cl=en
- Example 1
-
[0045](1) To 40 ml of a dioxane suspension containing 0.92 g of 60% sodium hydride was added 2.5 ml of cyclohexanol at room temperature over a 15-minute period, and the mixture was stirred at the same temperature for 1 hour and then at 50°C for 3.5 hours. The temperature of the reaction solution was returned to room temperature, 10 ml of a dioxane containing 3.2 g of 2-fluoronitrobenzene was added dropwise, and the mixture was stirred at room temperature overnight. The dioxane was evaporated, the residue was extracted with chloroform, and the chloroform layer was washed, in turn, with water and a saturated aqueous sodium chloride solution and then dried over anhydrous sodium sulfate. The solvent was evaporated to give an oil, which was then distilled under reduced pressure to give 3.8 g of 2-cyclohexyloxynitrobenzene.
b.p. 130 – 134°C/0.5 – 0.7 mmHg -
[0046](2) Fifty ml of a methanol solution containing 3.7 g of 2-cyclohexyloxynitrobenzene and 0.2 g of 5% palladium on carbon was stirred at room temperature under a hydrogen atmosphere for catalytic reduction. The catalyst was removed by filtration, and the filtrate was evaporated off to give 2.9 g of 2-cyclohexyloxyaniline as pale brown crystals.
m.p. 55 – 56°C -
[0047](3) To 20 ml of a pyridine solution containing 2.7 g of 2-cyclohexyloxyaniline was added dropwise 1.8 g of methanesulfonyl chloride under ice cooling with stirring. After completion of the addition, the mixture was stirred at room temperature for 2 hours. The reaction solution was poured into ice water and made acidic with dilute hydrochloric acid. The crystals which formed were collected by filtration, washed with water and dried to give 3.8 g of the crude crystals, which were then recrystallized from ethanol-n hexane to give 3.4 g of N-(2-cyclohexyloxyphenyl)methanesulfonamide.
m.p. 113 – 115°C -
[0048](4) To 20 ml of an acetic acid solution containing 3.4 g of N-(2-cyclohexyloxyphenyl)methanesulfonamide was added dropwise 1.5 g of 61% nitric acid on heating at 110°C over a 30-minute period, and then the mixture was stirred for 1 hour. The reaction solution was poured into ice water and neutralized with a dilute aqueous sodium hydroxide solution. The crystals which formed were collected by filtration, washed with water and dried to give 4.5 g of the crude crystals, which were then recrystallized from ethanol-n-hexane to give 3.3 g of N-(2-cyclohexyloxy-4-nitrophenyl)methanesulfonamide.
m.p. 136 – 137°C
| EP0093591A1 * | Apr 29, 1983 | Nov 9, 1983 | Eli Lilly And Company | Selective sulfonation process |
| FR2244473A1 * | Title not available | |||
| US3725451 * | Apr 13, 1970 | Apr 3, 1973 | Riker Laboratories Inc | Substituted benzoylhaloalkanesulfonanilides |
| US3840597 * | Jul 3, 1972 | Oct 8, 1974 | Riker Laboratories Inc | Substituted 2-phenoxy alkane-sulfonanilides |
| US3856859 * | Jun 8, 1973 | Dec 24, 1974 | Riker Laboratories Inc | Selective nitration process |
| Citing Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| EP1535614A2 * | Aug 22, 1997 | Jun 1, 2005 | University OofFlorida | Materials and methods for detection and treatment of immune system dysfunctions |
……………………………………………………..
The cortical collecting duct (CCD) is a major site of intrarenal prostaglandin E2 (PGE2) synthesis. This study examines the expression and regulation of the prostaglandin synthesizing enzymes cyclooxygenase-1 (COX-1) and -2 in the CCD. By indirect immunofluorescence using isoform-specific antibodies, COX-1 and -2 immunoreactivity was localized to all cell types of the murine M-1 CCD cell line. By immunohistochemistry, both COX-1 and COX-2 were localized to intercalated cells of the CCD on paraffin-embedded mouse kidney sections. When COX enzyme activity was measured in the M-1 cells, both indomethacin (COX-1 and -2 inhibitor) and the specific COX-2 inhibitor NS-398 effectively blocked PGE2 synthesis. These results demonstrate that COX-2 is the major contributor to the pool of PGE2synthesized by the CCD. By Western blot analysis, COX-2 expression was significantly upregulated by incubation with either indomethacin or NS-398. These drugs did not affect COX-1 protein expression. Evaluation of COX-2 mRNA expression by Northern blot analysis after NS-398 treatment demonstrated that the COX-2 protein upregulation occurred independently of any change in COX-2 mRNA expression. These studies have for the first time localized COX-2 to the CCD and provided evidence that the intercalated cells of the CCD express both COX-1 and COX-2. The results also demonstrate that constitutively expressed COX-2 is the major COX isoform contributing to PGE2synthesis by the M-1 CCD cell line. Inhibition of COX-2 activity in the M-1 cell line results in an upregulation of COX-2 protein expression.
http://jasn.asnjournals.org/content/10/11/2261.abstract
…………………………………………….
NS398 inhibits the growth of OSCC cells by mechanisms that are dependent and independent of suppression of PGE2 synthesis. Molecular targeting of COX-2, PGE2 synthase, or PGE2 receptors may be useful as a chemopreventive or therapeutic strategy for oral cancer.
http://clincancerres.aacrjournals.org/content/9/5/1885.full
…………………………………
References
- NS-398 at Sigma-Aldrich
- Wei Shen, Yong Li, Ying Tang, James Cummins and Johnny Huard (2005). “NS-398, a Cyclooxygenase-2-Specific Inhibitor, Delays Skeletal Muscle Healing by Decreasing Regeneration and Promoting Fibrosis”. American Journal of Pathology 167 (4): 1105–1117.doi:10.1016/S0002-9440(10)61199-6. PMC 1603662. PMID 16192645.
-
MORE References
Futaki et al (1993) NS-398, a novel non-steroidal anti-inflammatory drug with potent analgesic and antipyretic effects, which causes minimal stomach lesions. Gen.Pharmacol. 24 105. PMID: 8482483.
Futaki et al (1994) NS-398, a new anti-inflammatory agent, selectively inhibits prostaglandin G/H synthase/cyclooxygenase (COX-2) activity in vitro. Prostaglandins 47 55. PMID: 8140262.
Elder et al (2002) The MEK/ERK pathway mediates COX-2-selective NSAID-induced apoptosis and induced COX-2 protein expression in colorectal carcinoma cells. Int.J.Cancer 99 323. PMID: 11992399.
Gemoprost

Gemeprost, SC-37681, Ono-802, Cergem, Preglandin, Cervagem,
(E) -7 – [(1R, 2R, 3R-3-Hydroxy-2 – [(E) – (3R) -3-hydroxy-4,4-dimethyl-1-octenyl] -5-oxocyclopentyl] -2 -heptenoic acid methyl ester;
16,16-Dimethyl-DELTA2-trans-PGE1 methyl ester;
9-Oxo-11alpha, 15alpha-dihydroxy-16,16-dimethyl-2-trans, 13-trans-prostadiene-1-oic acid
Gemeprost (16, 16-dimethyl-trans-delta2 PGE1 methyl ester) is an analogue of prostaglandin E1.
Clinical use
It is used as a treatment for obstetric bleeding.
It is used with mifepristone to terminate pregnancy up to 24 weeks gestation. [1]
Side effects
Vaginal bleeding, cramps, nausea, vomiting, loose stools or diarrhea, headache, muscle weakness; dizziness; flushing; chills; backache; dyspnoea; chest pain; palpitations and mild pyrexia. Rare: Uterine rupture, severe hypotension, coronary spasms with subsequent myocardial infarctions
 |
|
| Systematic (IUPAC) name | |
|---|---|
| methyl (2E,11α,13E,15R)-11,15-dihydroxy-16,16-dimethyl-9-oxoprosta-2,13-dien-1-oate | |
| Clinical data | |
| AHFS/Drugs.com | International Drug Names |
| Legal status | ? |
| Routes | Pessary |
| Identifiers | |
| CAS number | 64318-79-2 |
| ATC code | G02AD03 |
| PubChem | CID 5282237 |
| ChemSpider | 4445416 |
| UNII | 45KZB1FOLS |
| KEGG | D02073 |
| Synonyms | methyl (E)-7-[(1R,2S,3R)-3-hydroxy-2-[(E,3R)-3-hydroxy-4,4-dimethyl-oct-1-enyl]-5-oxo-cyclopentyl]hept-2-enoate |
| Chemical data | |
| Formula | C23H38O5 |
| Mol. mass | 394.545 g/mol |
………………………………
http://www.chemdrug.com/databases/8_0_oqxuqtwlqgeukaaa.html

The reaction of 3-bromopropionic acid (I) with triphenylphosphine (II) in refluxing acetonitrile gives (2-carboxyethyl) -triphenylphosphonium bromide (III), which by a Wittig reaction with 2-oxa-3-hydroxy-6-syn- ( 3alpha-tetrahydropyranyloxy-4,4-dimethyl-1-trans-octen-1-yl) -7-anti-tetrahydropyranyloxybicyclo- [3.3.0] cis-octane (IV) (prepared according to reference 2) by means of sodium dimethylsulfinate in DMSO yields 9alpha-hydroxy-11alpha, 15alpha-bis (tetrahydropyranyloxy) -16,16-dimethyl-alpha-dinorprosta-5-cis-13-trans-dienoic acid (V). The reduction of (V) with H2 over Pd / C in methanol affords the 13-trans-prostenoic acid (VI), which is methylated with CH2N2 in ether yielding the methyl ester (VII). The reduction of (VII) with diisobutyl aluminum hydride in toluene affords the corresponding aldehyde (VIII) , which by a Wittig reaction with triethyl phosphonoacetate (IX) by means of NaH in THF is converted into 9alpha-hydroxy-11alpha, 15alpha-bis (tetrahydropyranyloxy) -16,16-dimethylprosta-2-trans-dienoic acid ethyl ester (X .) The hydrolysis of the ester (X) with KOH in ethanol-water gives the corresponding acid (XI), which is oxidized with CrO3, MnSO4 and H2SO4 in ether – water yielding the protected ketoacid (XII) The hydrolysis of (XII. ) with acetic acid-water at 80 C gives 9-oxo-11alpha, 15alpha-dihydroxy-16,16-dimethyl-prosta-2-trans-13-trans-dienoic acid (16,16-dimethyl-DELTA2-trans-PGE1 ) (XIII), which is finally methylated with CH2N2 in ether
References
- Bartley J, Brown A, Elton R, Baird DT (October 2001). “Double-blind randomized trial of mifepristone in combination with vaginal gemeprost or misoprostol for induction of abortion up to 63 days gestation”. Human reproduction (Oxford, England) 16 (10): 2098–102.doi:10.1093/humrep/16.10.2098. PMID 11574498. Retrieved 2008-10-29.
 |
|
: Gemeprost
CAS 64318-79-2
CAS Name: (2E,11a,13E,15R)-11,15-Dihydroxy-16,16-dimethyl-9-oxoprosta-2,13-dien-1-oic acid methyl ester
Additional Names: 16,16-dimethyl-trans-D2-PGE1 methyl ester
Manufacturers’ Codes: ONO-802
Trademarks: Cergem (Searle); Cervagem(e) (M & B); Preglandin (Ono)
Molecular Formula: C23H38O5
Molecular Weight: 394.54
Percent Composition: C 70.02%, H 9.71%, O 20.28%
Literature References:
Analog of prostaglandin E1, q.v. Prepn: M. Hayashi et al., DE 2700021; eidem, US 4052512 (both 1977 to Ono);
H. Suga et al., Prostaglandins 15, 907 (1978).
Effects on uterine contractility and steroid hormone plasma levels: K. Oshimaet al., J. Reprod. Fertil. 55, 353 (1979).
Effects on reproductive function: K. Matsumoto et al., Nippon Yakurigaku Zasshi 79, 15 (1982), C.A. 96, 98392 (1982).
Use in termination of first trimester pregnancy: O. Reiertsen et al., Prostaglandins Leukotrienes Med. 8, 31 (1982).
Therap-Cat: Abortifacient; oxytocic.
Keywords: Abortifacient/Interceptive; Oxytocic; Prostaglandin/Prostaglandin Analog
|
Latanoprost

Latanoprost
isopropyl-(Z)7[(1R,2R,3R,5S)3,5-dihydroxy-2-[(3R)-3-hydroxy-5-phenylpentyl]cyclopentyl]-5-heptenoate.
130209-82-4
XA41, PhXA34 [as 15 (R, S) -isomer], PhXA41, Xalatan
(Zanoni, G. et al., Tetrahedron 2010, 66, 7472)
Latanoprost (pronounced la-TA-noe-prost) ophthalmic solution is a medication administered into the eyes to control the progression of glaucoma or ocular hypertension by reducing intraocular pressure. It is a prostaglandin analogue (more specifically an analogue ofprostaglandin F2α[1]) that lowers the pressure by increasing the outflow of aqueous fluid from the eyes through the uvealsclearal tract.[2] Latanoprost is an isopropyl ester prodrug, meaning it is inactive until it is hydrolyzed by esterases in the cornea to the biologically active acid.[3]
It is also known by the brand name of Xalatan manufactured by Pfizer. Annual sales are approximately $1.6 billion. The patent for latanoprost expired in March 2011, and at least one generic version (manufactured by Mylan Inc.) is now widely available in the U.S. The Veterans Health Administration, part of the U.S. Department of Veterans Affairs, uses generic Latanoprost manufactured by Alcon Laboratories of Fort Worth, Texas distributed by Novartis generic brand Sandoz Pharmaceuticals.
Latanoprost was invented by Johan W. Stjernschantz and Bahram Resul, employees of the Pharmacia Corporation of Upsalla, Sweden.[4]
It is on the World Health Organization’s List of Essential Medicines, a list of the most important medication needed in a basic health system.[5]
 |
|
 |
|
| Systematic (IUPAC) name | |
|---|---|
| isopropyl (Z)-7-[(1R,2R,3R,5S)-3,5-dihydroxy-2- [(3R)3-hydroxy-5-phenylpentyl]-cyclopentyl] hept-5-enoate | |
| Clinical data | |
| Trade names | Xalatan |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a697003 |
| Pregnancy cat. | C (US) |
| Legal status | ℞-only (US) |
| Routes | Topical (eye drops) |
| Pharmacokinetic data | |
| Half-life | 17 minutes |
| Identifiers | |
| CAS number | 130209-82-4 |
| ATC code | S01EE01 |
| PubChem | CID 5311221 |
| IUPHAR ligand | 1961 |
| DrugBank | DB00654 |
| ChemSpider | 4470740 |
| UNII | 6Z5B6HVF6O |
| KEGG | D00356 |
| ChEBI | CHEBI:6384 |
| ChEMBL | CHEMBL1051 |
| Chemical data | |
| Formula | C26H40O5 |
| Mol. mass | 432.593 g/mol |
Medical uses
Ocular hypertension
- In well-controlled clinical trials including patients with open-angle glaucoma or ocular hypertension (IOP ≥21 mm Hg), monotherapy with latanoprost reduced IOP levels by 22 to 39% over 1 to 12 months’ treatment. Latanoprost was significantly more effective than timolol 0.5% twice daily in 3 of 4 large (n = 163 to 267) randomised, double-blind trials. Latanoprost demonstrated a stable long-term IOP-lowering effect in 1- or 2-year continuations of these trials, with no sign of diminishing effect during prolonged treatment.[6]
- Meta analysis suggests that latanoprost is more effective than timolol in lowering IOP. However, it often causes iris pigmentation. While current evidence suggests that this pigmentation is benign, careful lifetime evaluation of patients is still justified.[7]
Closed-angle glaucoma
- Patients who had elevated IOP despite iridotomy and/or iridectomy (including patients of Asian descent), latanoprost was significantly more effective than timolol in two double-blind, monotherapy trials (8.2 and 8.8 mm Hg vs 5.2 and 5.7 mm Hg for latanoprost vs timolol at 12 and 2 weeks, respectively).[8]
Method of administration
One drop in the affected eye(s) once daily in the evening; do not exceed the once daily dosage because it has been shown that more frequent administration may decrease the intraocular-pressure (IOP) lowering effect[2]
Adverse effects[
Listed from most to least common:
- >5% to 15%: Blurred vision, burning and stinging, conjunctival hyperemia, foreign body sensation, itching, increased pigmentation of the iris causing (heterochromia), punctate epithelial keratopathy
- 4%: Cold or upper respiratory tract infections, flu-like syndrome
- 1-4%: Dry eyes, excessive tearing, eye pain, lid crusting, lid edema, lid erythema (hyperemia), lid pain, photophobia (light intolerance)
- 1 % – 2%: Chest pain, allergic skin reactions, arthralgia, back pain, myalgia, thickening of the eyelashes.(used,also bimatoprost,in cosmetic industry as eyelash growth enhancers)
- <1% (Limited to important or life-threatening): Asthma, herpes keratitis, iritis, keratitis, retinal artery embolus, retinal detachment, toxic epidermal necrolysis, uveitis, vitreous hemorrhage from diabetic retinopathy
- A single case report links latanoprost use to the progression of keratoconus.[9]
Concerns related to adverse effects:
- Bacterial keratitis: Inadvertent contamination of multiple-dose ophthalmic solutions, has caused bacterial keratitis.
- Ocular effects: May permanently change/increase brown pigmentation of the iris, the eyelid skin, and eyelashes. In addition, may increase the length and/or number of eyelashes (may vary between eyes); changes occur slowly and may not be noticeable for months or years. Long-term consequences and potential injury to eye are not known.
- Ocular disease: Use with caution in patients with intraocular inflammation, aphakic patients, pseudophakic patients with a torn posterior lens capsule, or patients with risk factors for macular edema. Safety and efficacy have not been determined for use in patients with angle-closure-, inflammatory-, or neovascular glaucoma.
Special populations
Contact lens wearers: Contains benzalkonium chloride which may be absorbed by contact lenses; remove contacts prior to administration and wait 15 minutes before reinserting
Contraindications
Hypersensitivity to latanoprost, benzalkonium chloride, or any component of the formulation
Drug Interactions
Bimatoprost: The concomitant use of Latanoprost and Bimatoprost may result in increased intraocular pressure. Risk D: Consider therapy modification
Nonsteroidal Anti-Inflammatory Agents: May diminish the therapeutic effect of Prostaglandins (Ophthalmic). Nonsteroidal Anti-Inflammatory Agents may also enhance the therapeutic effects of Prostaglandins (Ophthalmic). Risk C: Monitor therapy
Pregnancy
Prescription of Latanoprost is limited in human studies due to high incidence of abortion shown in animal experiments. Because of this, Latanoprost is classified as Risk factor C (Adverse events were observed in animal reproduction studies at maternally toxic doses)according to United States Food and Drug Administration’s use-in-pregnancy ratings.[10]Lactation Excretion in breast milk unknown/use caution. Breast-Feeding Considerations It is not known if latanoprost is excreted in breast milk. The manufacturer recommends that caution be exercised when administering latanoprost to nursing women.[2]
Storage
Latanoprost is a substance exhibiting thermal and solar instability. Concentration of latanoprost will decrease by 10% when stored at 50 and 70 degrees Celsius every 8.25 and 1.32 days respectively. Reaction with ultraviolet radiation will cause rapid degradation of Latanoprost. It is therefor important to store Latanoprost ideally in temperature below room temperature and free from sunlight in order to attain acceptable drug quality. [11]
Latanoprost is a prostaglandin F2α analogue. Its chemical name is isopropyl-(Z)7[(1R,2R,3R,5S)3,5-dihydroxy-2-[(3R)-3-hydroxy-5-phenylpentyl]cyclopentyl]-5-heptenoate. Its molecular formula is C26H40O5and its chemical structure is:

XALATAN Sterile Ophthalmic Solution (latanoprost ophthalmic solution) is supplied as a sterile, isotonic, buffered aqueous solution of latanoprost with a pH of approximately 6.7 and an osmolality of approximately 267 mOsmol/kg. Each mL of XALATAN contains 50 micrograms of latanoprost. Benzalkonium chloride, 0.02% is added as a preservative. The inactive ingredients are: sodium chloride, sodium dihydrogen phosphate monohydrate, disodium hydrogen phosphate anhydrous, and water for injection. One drop contains approximately 1.5 μg of latanoprostLatanoprost is a colorless to slightly yellow oil that is very soluble in acetonitrile and freely soluble in acetone, ethanol, ethyl acetate, isopropanol, methanol, and octanol. It is practically insoluble in water.
………………………….
http://www.google.com/patents/EP2495235A1?cl=en
When Latanoprost is the desired product the double bond on the side chain of compound 9a is hydrogenated to form compound 11, then by Wittig reaction with 4-carboxybutyltriphenylphosphonium bromide compound 11 is converted into Latanoprost acid 12. By conversion of the carboxylic acid into isopropyl ester, the final product Latanoprost is obtained:
-
4-Carboxybutyltriphenylphosphonium bromide 15 (32.7 g, 0.074 mol) was suspended in tetrahydrofuran (75.0 mL) at 0°C under nitrogen atmosphere. A 1M solution of potassium tert-butoxide in tetrahydrofuran (296.0 mL, 0.296 mol) was added dropwise and the mixture turned into orange. After stirring for 45 minutes at 0°C the system was cooled to ―15°C. A solution of (3aR,4R,5R,6aS)-4-((R)-3-hydroxy-5-phenylpentyl)hexahydro-2H-cyclopenta[b]furan-2,5-diol (5.0 g, 0.016 mol) in tetrahydrofuran (23.0 mL) was added dropwise at a temperature lower than -10°C. After stirring overnight at -15°C no more starting was visible on TLC and water (100 mL) was added. The mixture was extracted with diisopropyl ether (70 mL) and after separation the aqueous phase was treated with 0.6 N HCl to pH 6.0. Three extractions with ethyl acetate (3x 125 mL) were then performed, each time adjusting the pH of the aqueous phase to 6.0. The combined organic layers were concentrated under vacuum at 35°C. An oil (13.87 g) was obtained which was used in the subsequent step without further purification.
-
1H-NMR {400 MHz, CDCl3, δ (ppm)}: 7.71 (m, 1H, Ph), 7.49 (m, 1H, Ph), 7.30-7.17 (m, 3H, Ph), 5.52-5.35 (m, 2H, -CH=CH-), 4.34 (bs, 4H, OH), 4.17 (m, 1H, -CH-OH(C-9)), 3.96 (m, 1H, -CH-OH (C-11)), 2.78 (m, 1H, -CH-OH (C-15)), 2.78 (m, 1H, -CH2Ph), 2.66 (m, 1H, -CH2Ph), 2.36-1.27 (m, 18H).
-
13C-NMR {400 MHz, CDCl3, δ (ppm)}: 176.5 (C), 142.2 (C), 130.8 (CH), 130.7 (CH), 129.4 (CH), 128.8 (CH), 128.7 (CH), 128.4 (CH), 125.7 (CH), 78.3 (CH), 74.2 (CH), 71.4 (CH), 52.2 (CH), 51.6 (CH), 42.4 (CH2), 38.8 (CH2), 35.2 (CH2), 33.4 (CH2), 32.0 (CH2), 29.1 (CH2), 26.6 (CH2), 26.4 (CH2), 24.7 (CH2).
-
HPLC-MS (ESI): [M+Na]+ = 413, [M+H]+ = 391.
EXAMPLE 17(Z)-isopropyl 7-((1R,2R,3R,5S)-3,5-dihydroxy-2-((R)-3-hydroxy-5-phenylpentyl)cyclopentyl)hept-5-enoate (Latanoprost)
-
Latanoprost acid (6.78 g, corresponding to 0.008 mol) was dissolved in N,N-dimethylformamide (108 mL) and cesium carbonate (8.48 g, 0.026 mol) was added at room temperature. 2-Iodopropane (3.46 mL, 0.035 mol) was added and the suspension was stirred at 40°C for 3 hours, checking the conversion on TLC. The mixture was then allowed to reach 25°C and a mixture of ice (184 g), water (40 mL), sodium thiosulfate (1M, 18 mL), was added stirring at -5/0°C for 15 minutes. The mixture was extracted with tert-butylmethylether (285 mL) and the phases were separated. The aqueous phase was extracted twice with tert-butylmethyl ether (2x 200 mL) and the combined organic layers were washed with brine (176 mL, 130 mL). The organic phase was concentrated under reduced pressure at 25°C and the crude product was obtained as a yellow oil (6.60 g). Purification by column chromatography on silica gel was performed eluting with dichloromethane:methanol increasing the percentage of methanol from 0 to 5%. A second purification on silica gel afforded Latanoprost (2.36 g, 0.005 mol, 68% over two steps).
-
1H-NMR {400 MHz, CDCl3, δ (ppm)}: 7.32-7.19 (m, 5H, Ph), 5.45-5.51 (m, 2H, H-5 e H-6 vinyl), 5.0 (hept, J=6.3 Hz, 1H, CH3CHCH3), 4,18 (bs, 1H, CHOH), 3.95 (bs, 1H, CHOH), 3.67 (bs, 1H, CHOH), 2.76 (m, 2H, CH 2Ph), 1.23 (d, J=6.3Hz, 6H, C(CH3)2), 2.55-1.3, 21H).
-
HPLC-MS (ESI): [M+Na]+ = 455, [M+H]+ = 432.
……………………………….
http://www.google.com/patents/EP2454227A1?cl=en
Example 3
Synthesis of Latanoprost
ether MTBE
8c-iso
Latanoprost
Scheme 5. Synthesis of Latanoprost
Synthesis of Latanoprost from 12c:
As shown in Scheme 5 in Example 3, a 250 ml.3-necked round-bottom flask equipped with a magnetic bar, a temperature probe, rubber septa, and a nitrogen gas inlet was charged at room temperature with 7.3 g (19.6 mmol) of deprotected lactone 12c in 70 mL of 2-propanol and 1.6 g (39.2 mmol) of sodium hydride, 60%, in mineral oil. The reaction mixture was heated at 35 0C for 18 h and TLC analysis indicated complete reaction. The mixture was diluted with 60 mL of water and the pH was adjusted to 6 with 1 N HCI. The layers were separated and the aqueous layer was back extracted with 40 mL of 2-propanol four times. The combined organic layers were washed with 50 mL of brine, dried over sodium sulfate, filtered, and concentrated.
The material was dissolved in 60 mL of THF, 5.0 mL (33.3 mmol) of DBU and 3.3 mL (33.3 mmol) of iodopropane. The reaction mixture was stirred at room temperature for 18 h and TLC analysis indicated complete reaction. The mixture was diluted with 60 mL of ethyl acetate and 60 mL of water. The layers were separated and the aqueous layer was back extracted with 40 mL of ethyl acetate for two times. The combined organic layers were washed with 50 mL of brine, dried over sodium sulfate, filtered, and concentrated.
The material was purified by using reverse phase biotage, 70 : 30 ACN :
H2O to obtain 4.1 g (49% yield) of Latanoprost, confirmed by 1H NMR.
………………….

Wittig condensation of lactol (XIII) with (carboxybutyl) triphenylphosphonium bromide (XV) in the presence of potassium tert-butoxide produced the Z-olefin (XVI). Conversion of carboxylic acid (XVI) to the title isopropyl ester was then accomplished by alkylation with 2-iodopropane in the presence of DBU.
http://www.chemdrug.com/databases/8_0_xmmatmlqjiethrwn.html
………………………………
http://www.nature.com/nature/journal/v489/n7415/full/nature11411.html?WT.ec_id=NATURE-20120913

………………………

……………………………….
http://www.google.com/patents/WO2013186550A1?cl=en
(la) (Ic)
Example 6 – Experimental procedures for the synthesis of latanoprost
A synthesis of latanoprost is shown and described below.
latanoprost (77)
2-Phenethyloxirane, 61
m-CPBA,
A modified procedure of Woodward was used (Bernier, D. et al., The Journal of Organic Chemistry 2008, 73, 4229). A stirred solution of 4-phenyl-l-butene 62 (500 mg, 568 μΙ, 3.78 mmol) in CH2CI2 (20 ml) was cooled to 0 °C. m-CPBA (816 mg, 4.73 mmol) was added as a solid and the reaction mixture was stirred at 0 °C for 1.5 h, then r.t. for 24 h. The reaction mixture was poured into saturated K2C03 solution (50 ml) and extracted with CH2CI2 (2 x 50 ml). The combined organic phases were washed with saturated K2C03 solution (50 ml) before being dried (MgS04), filtered and concentrated to give a clear colourless liquid. This material was purified by column chromatography, eluting with petrol/EtOAc (9:1), to give the epoxide 61 (10.2 g, 91%) as a clear colourless liquid. The 13C, and IR data were consistent with the literature (Mitchell, J. M. et al., Journal of the American Chemical Society 2001, 123, 862; Elings, J. A. et al., European Journal of Organic Chemistry 1999, 1999, 837).
Rf = 0.42 (petrol :EtOAc, 9:1) vmax (neatycnrr1 3027, 2989, 2922, 2859, 1602, 1495, 1454, 1410, 835, 750, 699
*H NMR (400 MHz; CDCI3) δΗ = 1.83-1.99 (2 H, m, CH2), 2.53 (1 H, dd, J = 5.0, 2.7 Hz, CHH), 2.75-2.93 (2 H, m, CH2), 2.80 (1 H, dd, J = 5.0, 4.0 Hz, CHH), 3.01 (1 H, dddd, J = 6.5, 5.0, 4.0, 2.7 Hz, CH), 7.22-7.38 (5 H, m, Ar H’s)
13C NMR (100 MHz; CDCI3) 5C = 32.2 (CH2), 34.2 (CH2), 47.2 (CH2), 51.7 (CH), 126.0 (2 x ArCH), 128.3 (2 x ArCH), 128.4 (ArCH), 141.2 (ArC)
m/z (EI) 148.1 (M+, 10%), 130.1 (23%), 129.0 (18%), 118.1 (29%), 117.1 (83%), 115.0 (28%), 105.0 (22%), 104.0 (61%), 92.0 (22%), 91.0 (100%), 83.9 (37%), 77.0 (17%), 65.0 (31%)
(2S)-2-Phenethyloxirane, 63
A modified procedure of Jacobsen was used (Schaus, S. E. et al., Journal of the American Chemical Society 2002, 224, 1307). Racemic epoxide 61 (10.0 g, 67.5 mmol) was dissolved in THF (10 ml) and stirred at r.t.. (S^-i+J-^A/’-BisiS^-di-tert-butylsalicylidene)-!^- cyclohexanediaminocobalt(II) (204 mg, 0.34 mmol) was added and the resultant dark brown solution cooled to 0 °C. Acetic acid (77 μΙ, 1.35 mmol) and water (669 μΙ, 37.1 mmol) were added. The reaction was stirred at 0 °C for 1 h and then at r.t. for 23 h. The reaction mixture was concentrated under reduced pressure and purified by column chromatography (~200 g silica), eluting with petrol/EtOAc (9:1), to give the epoxide 3 as a dark red liquid. This was re- purified by column chromatography eluting with petrol/EtOAc (9.5:0.5 to 9:1), to give the epoxide 3 (4.62 g, 46%) as an orange liquid. The analytical data matched that of the racemic material described above. The enantioselectivity of the resolution was determined after subsequent conversion to the allylic alcohol 66. The optical rotation matched closely with that reported in the literature (Martynow, J. G. et al., European Journal of Organic Chemistry 2007, 2007, 689).
[a]D 21 -21.0 (c. 1.0, CHC ) (lit., [a]D 20 -22.5 (c. 1.0, CHCI3)) 5-Phenyl-l-penten-3-ol, 64
A modified procedure of Molander was used (Molander, G. A. et al., The Journal of Organic Chemistry 2009, 74, 1297). A stirred solution of hydrocinnamaldehyde 65 (2.50 g, 2.45 ml, 18.6 mmol) in THF (25 ml) was cooled to -78 °C. Vinyl magnesium bromide solution (1 M in THF) (22.4 ml, 22.4 mmol) was added dropwise over ~ 5 min. The reaction mixture was stirred at -78 °C for 1.5 h, then 0 °C for 3 h. The reaction mixture was poured into saturated NH4CI solution (50 ml) and extracted with Et20 (3 x 50 ml). The combined organic phases were washed with saturated NaCI solution (50 ml) before being dried (MgS04), filtered and concentrated to give a pale yellow liquid. This material was purified by column
chromatography, eluting with petrol/EtOAc (9: 1), to give the vinyl alcohol 64 (1.89 g, 63%) as a clear colourless liquid. The *H, 13C, and IR data were consistent with the literature (Molander, G. A. et al., The Journal of Organic Chemistry 2009, 74, 1297; Kim, J. W. et al., Chemistry – A European Journal 2008, 24, 4104). Rf = 0.40 (petrol :EtOAc, 4: 1)
max (CHC Vcnrr1 3335, 3026, 2923, 2859, 1496, 1454, 990, 922, 747, 698
*H NMR (400 MHz; CDCI3) δΗ = 1.55 (1 H, br.s, OH), 1.84-1.99 (2 H, m, CH2), 2.70-2.89 (2 H, m, CH2), 4.19 (1 H, app q, J = 6.0 Hz, CHO ), 5.20 (1 H, app dt, J = 10.5, 1.4 Hz, HC=C), 5.30 (1 H, app dt, J = 17.1, 1.4 Hz, HHC=C), 5.96 (1 H, ddd, J = 17.1, 10.5, 6.0 Hz, H2C=CH), 7.20-7.40 (5 H, m, ArCH’s)
13C NMR (100 MHz; CDCI3) 5C = 31.6 (CH2), 38.5 (CH2), 72.4 (HCOH), 114.9 (H2C=C), 125.8 (ArCH), 128.4 (2 x ArCH), 128.4 (2 x ArCH), 141.0 (H2C=Q, 141.8 (ArC)
m/z (EI) 162.1 (M+, 30%), 144.1 (52%), 129.1 (72%), 105.1 (68%), 92.1 (71%), 91.0 (100%), 57.0 (61%) 6D. (3S)-5-Phenyl-l-penten-3-ol, 66
A modified procedure of Falck was used (Alcaraz, L. et al., Tetrahedron Letters 1994, 35, 5449). A suspension of trimethylsulfonium iodide (18.2 g, 89.1 mmol) in anhydrous THF (220 ml) was stirred and cooled to -20 °C. 1.6 M n-BuLi (55.7 ml, 89.1 mmol) was added slowly and the reaction stirred at -20 °C for 1 h. A solution of epoxide 63 (4.40 g, 29.7 mmol) in anhydrous THF (50.0 ml) was added slowly. The reaction was stirred at -20 °C for 1 h and then allowed to warm to r.t. slowly. The reaction mixture was poured into water (200 ml) and extracted with Et20 (1 x 200 ml, 1 x 100 ml). The combined organic phases were washed with saturated NaCI solution (100 ml) before being dried (MgS04), filtered, and concentrated to give the crude material. This was purified by column chromatography (130 g silica), eluting with petrol/EtOAc (9:1), to give partially purified material. This was re-purified by column chromatography (50 g silica), eluting with petrol/EtOAc (9:1), to give allylic alcohol 66 (3.19 g, 66%) as a pale yellow liquid. The analytical data matched that described for the racemic material above.
[α]ο21 -11.0 (c. 1.0, CHCI3) (lit – Kanbayashi, N. et al., Angewandte Chemie International Edition 2011, 50, 5197, [a]D 25 -3.6 (for 85% ee (c. 0.4, CHCI3)))
Chiral-HPLC data: er = >99:1 (Chiralcel AD-H column, 210 nm, hexane/2-propanol: 98/2, flow rate: 0.5 mlVmin, room temperature; ¾: minor 41.0 min, major 43.7 min) 6E. tert-Butyl(dimethyl)[(lS)-l-phenethyl-2-propenyl]oxysilane, 67
67 A stirred solution of allylic alcohol 66 (3.00 g, 18.5 mmol) in CH2CI2 (53 ml) was cooled to 0 °C. Imidazole (2.27 g, 33.3 mmol) was added in one portion followed by t- butylchlorodimethylsilane (3.34 g, 22.2 mmol). The cooling bath was removed and the reaction mixture stirred at r.t. for 16 h before being poured into 10% aq. HCI (100 ml). The mixture was extracted with 40/60 petroleum ether (2 x 100 ml). The combined organics were washed with saturated NaCI solution (100 ml), dried (MgS04), filtered, and concentrated to give the crude material. This was purified by column chromatography, eluting with 40/60 petroleum ether, to give the protected alcohol 67 (4.68 g, 92%) as a colourless liquid. The *H NMR data and optical rotation matched that reported in the literature (Uenishi, J. i. et al., Organic Letters 2011, 13, 2350).
Rf = 0.25 (40/60 petroleum ether)
vmax (film)/cm-13064, 3027, 2952, 2929, 2886, 2856, 1497, 1472, 1462, 1455, 1361, 1251, 1122, 1083, 1030, 990, 921, 834, 774, 697
*H NMR (400 MHz; CDCI3) δΗ = 0.05 (3 H, s, SiCH3), 0.08 (3 H, s, SiCH3), 0.93 (9 H, s, C(CH3)3), 1.82 (2 H, m, CH2), 2.66 (2 H, m, CH2), 4.17 (1 H, m, OCH), 5.08 (1 H, ddd, J = 10.4, 1.5, 1.3 Hz, HA =CH), 5.19 (1 H, app dt, J = 17.2, 1.5 Hz, HHC=CH), 5.86 (1 H, ddd, J = 17.2, 10.4, 6.0 Hz, H2C=CH), 7.18 (3 H, m, ArH’s), 7.28 (2 H, m, ArH’s)
13C NMR (100 MHz; CDCI3) 5C = -4.8 (SiCH3), -4.3 (SiCH3), 18.3 (C(CH3)3), 25.9 (C(CH3)3), 31.5 (CH2), 39.8 (CH2), 73.3 (CHOSi), 114.0 (H2C=C), 125.7 (ArCH), 128.3 (2 x ArCH), 128.4 (2 x ArCH), 141.4 (H2C=Q, 142.5 (ArC).
[a]D” 12.0 (c. 1.0, CHCI3) (lit., [a]D 20 14.5 (c. 1.0, CHCI3)) 6F. (3S)-3-[l-(tert-Butyl)-l,l-dimethylsilyl]oxy-5-phenylpentan-l-ol, 68
67
A modified procedure of Denmark was used (Denmark, S. E. et al., Organic Letters 2005, 7, 5617). Compound 67 (2.00 g, 7.23 mmol) was added to a flame dried schlenk flask under N2. 9-BBN (0.5 M in THF) (15.9 ml, 7.96 mmol) was added via syringe and the resulting solution stirred at r.t. for 1 h. A further 1.1 eq. (15.9 ml, 7.96 mmol) of 9-BBN was added and the reaction stirred at r.t. for 2 h. Water (16.0 ml) and NaB03.4H20 (5.56 g, 36.2 mmol) were added and the reaction stirred at r.t. for 2 h. The reaction mixture was poured into saturated NH4CI solution (60 ml) and extracted with Et20 (3 x 100 ml). The combined organic phases were washed with sat. NaCI solution (100 ml), dried (MgS04), filtered, and concentrated to give the crude material. This was purified 3 times by column chromatography (twice eluting with petrol/EtOAc (6:1) and once with petrol/ EtOAc/Eti) (9:0.5:0.5)) to give the alcohol 68 (672 mg, 32%) as a clear colourless oil.
Rf = 0.18 (petrol :EtOAc, 9:1)
vmax (film)/cm-13351 (broad), 3063, 2950, 2928, 2885, 2856, 1496, 1471, 1462, 1454, 1360, 1253, 1092, 1057, 1028, 1005, 834, 773, 746, 698
*H NMR (400 MHz; CDCI3) δΗ = 0.09 (3 H, s, SiCH3), 0.10 (3 H, s, SiCH3), 0.92 (9 H, s, C(CH3)3), 1.75 (1 H, m, CHH), 1.83-1.94 (3 H, m, CH2, CHH), 2.32 (1 H, app t, J = 5.2 Hz, OH), 2.64 (2 H, m, CH2), 3.75 (1 H, app dq, J = 10.8, 5.5 Hz, OCHH), 3.87 (1 H, app ddt, J = 10.8, 8.1, 4.8 Hz, OCHtf), 3.99 (1 H, app qd, J = 6.1, 4.4 Hz, HCOTBDMS), 7.16-7.23 (3 H, m, ArCH’s), 7.27-7.33 (2 H, m, ArCH’s)
13C NMR (100 MHz; CDCI3) 5C = -4.7 (SiCH3), -4.4 (SiCH3), 18.0 (C(CH3)3), 25.8 (C(CH3)3), 31.7 (CH2), 37.8 (CH2), 38.7 (CH2), 60.1 (CH2), 71.2 (SiOCH), 125.8 (ArCH), 128.2 (2 x ArCH), 128.4 (2 x ArCH), 142.1 (ArC)
HRMS (ESI) calcd for Ci7H30O2SiNa [MNa+] 317.1907, found 317.1906
[a]D 23 23.0 (c. 1.0, CHCI3) 6G. tert-Butyl[(lS)-3-iodo-l-phenethylpropyl]oxydimethylsilane, 69
68 A modified procedure of Rychnovsky was used (Dalgard, J. E. et al., Organic Letters 2004, 6, 2713). Alcohol 68 (600 mg, 2.04 mmol) was added to a flame dried schlenk flask under N2. CH2CI2 (10 ml) was added via syringe and the resulting solution stirred at r.t..
Triphenylphosphine (695 mg, 2.65 mmol) and imidazole (222 mg, 3.26 mmol) were added as solids in one portion. Iodine (672 mg, 2.65 mmol) was added to the resulting solution. A slight exotherm was noted and the solution changed from a light yellow colour to a brown colour with the formation of a precipitate. The reaction was stirred at r.t. for 1 h. The reaction mixture was dry loaded onto silica (2 g) and purified by column chromatography (14 g silica), eluting with petrol to petrol/EtOAc (9: 1). This gave the iodide 69 (725 mg, 88%) as a clear, colourless oil.
Rf = 0.20 (40/60 petroleum ether)
ifiln /cnr^OeS, 3026, 2951, 2928, 2886, 2856, 1495, 1471, 1461, 1360, 1253, 1187, 1165, 1140, 1092, 1063, 1005, 975, 931, 833, 773, 697
*H NMR (400 MHz; CDCI3) δΗ = 0.09 (3 H, s, SiCH3), 0.10 (3 H, s, SiCH3), 0.92 (9 H, s, (C(CH3)3), 1.79 (2 H, m, CH2), 2.05 (2 H, m, CH2), 2.64 (2 H, m, CH2), 3.24 (2 H, m, CH2), 3.82 (1 H, quin., J = 5.7 Hz, OCH), 7.16-7.23 (3 H, m, ArCH’s), 7.27-7.33 (2 H, m, ArCH’s) 13C NMR (100 MHz; CDCI3) 5C = -4.3 (SiCH3), -4.3 (SiCH3), 3.0 (CH2), 18.1 (C(CH3)3), 25.9 (C(CH3)3), 31.3 (CH2), 38.7 (CH2), 40.8 (CH2), 71.7 (OCH), 125.8 (ArCH), 128.3 (2 x ArCH), 128.4 (2 x ArCH), 142.1 (ArC)
HRMS (ESI) calcd for Ci7H30OSiI [MH+] 405.1108, found 405.1105
[a]D 23 26.0 (c. 1.0, CHCI3)
6H. [(lR)-3-((3aR,4R,6aS)-2-Methoxy-5-(£)-l-[(l,l,l- trimethylsilyl)oxy]methylideneperhydrocyclopenta[d]furan-4-yl)-l- phenethylpropyl]oxy(tert-butyl)dimethylsilane, 70
70 Iodide 69 (1.32 g, 3.27 mmol, 1.1 eq.) was added via syringe to a flame dried schlenk flask (evacuated and purged with nitrogen several times and allowed to cool). Anhydrous Et20 (13.3 ml) was added via syringe and the resulting solution cooled to -78 °C. 1.63 M t-BuLi (4.01 ml, 6.54 mmol, 2.2 eq.) was added dropwise and the reaction mixture stirred at -78 °C for 2 h and -40 °C for 2 h before being cooled back to -78 °C. Meanwhile, thiophene (275 mg, 262 μΙ, 3.27 mmol, 1.1 eq.) was added via syringe to a flame dried schlenk flask (evacuated and purged with nitrogen several times and allowed to cool). Anhydrous THF (13.3 ml) was added via syringe and the resulting solution cooled to -30 °C. 1.63 M n-BuLi (2.01 ml, 3.27 mmol, 1.1 eq.) was added dropwise and the solution stirred at -30 °C for 30 min. CuCN (293 mg, 3.27 mmol, 1.1 eq.) was added as a solid, in one portion. The cooling bath was removed and the suspension allowed to warm to r.t. The resulting tan/brown solution of cuprate was added dropwise via syringe to the schlenk flask containing the alkyl lithium and anhydrous THF (13.3 ml) added. The mixture was stirred at -20 °C for 1 h to allow formation of mixed cuprate 71. This was cooled to -78 °C and a solution of enal 24 (500 mg, 2.97 mmol, 1.0 eq.) in anhydrous THF (13.3 ml) was added dropwise. The mixture was stirred at -78 °C for 1 h and then allowed to warm slowly to -20 °C. TMSCI (1.61 g, 1.89 ml, 14.9 mmol, 5.0 eq.) was added via syringe followed by NEt3 (1.80 g, 2.49 ml, 17.8 mmol, 6 eq.). The reaction was quenched by the addition of saturated NH4CI solution (50 ml) and extracted with Et20 (3 x 50 ml). The combined organic phases were washed with saturated NH4CI solution (50 ml) and saturated NaCI solution (50 ml) before being dried (MgS04), filtered, and concentrated to give the crude material as a yellow oil. This was used directly in the next step.
61. (3aR 4R,5R,6aS)-4-((3R)-3-[l-(tert-Butyl)-l,l-dimethylsilyl]oxy-5- phenylpentyl)-2-methoxyperhydrocyclopenta[d]furan-5-ol, 73
70 73 The crude material from the conjugate addition / trapping experiment, containing 70, was dissolved in CH2Cl2/MeOH (3: 1) (30 ml) and cooled to -78 °C. A stream of ozone was passed through the stirred solution. The reaction was monitored periodically by TLC in order to judge completion of the ozonolysis (judged by consumption of silyl enol ether). The reaction mixture was flushed with a stream of N2, for 15 min, to remove excess 03. NaBH4 (202 mg, 5.35 mmol) was added in one portion. The reaction mixture was stirred at -78 °C for 2 h before the cooling bath was removed and the reaction allowed to warm to r.t.. The reaction was stirred at r.t. for 1 h. NaBH4 (67.4 mg, 1.78 mmol) was added and the reaction stirred at r.t. for a further 15 min. The reaction mixture was poured into saturated NaCI solution (25 ml) and extracted with EtOAc (3 x 25 ml). The combined organic phases were dried (MgS04), filtered, and concentrated to give the crude product as a pale yellow oil. This was purified by column chromatography on silica, eluting with petrol/EtOAc (4: 1), giving the alcohol 73 (as an approximately 2:1 mixture of diastereoisomers) as a clear, colourless oil (800 mg, 62% (2 steps from enal 24)).
Rf = 0.23 (petrol :EtOAc, 4:1)
vmax (neatycnrr1 3434 (broad), 3026, 2928, 2856, 1496, 1471, 1454, 1360, 1343, 1254, 1098, 1053, 1004, 937, 833, 773, 698
1H NMR (400 MHz; CDCI3) 5H = (mixture of 2 diastereoisomers, signals of minor diastereoisomer indicated by *) 0.05 (3 H, s, CH3), 0.06* (3 H, s, CH3), 0.07 (3 H, s, CH3), 0.07* (3 H, s, CH3), 0.91 (9 H, s, C(CH3)3), 0.92* (9 H, s, C(CH3)3), 1.12-1.80 (7 H, m), 1.12- 1.80* (7 H, m), 1.90-2.38 (5 H, m), 1.90-2.38* (5 H, m), 2.53-2.75 (2 H, m, CH2), 2.53-2.75* (2 H, m, CH2), 3.32 (3 H, s, OCH3), 3.39* (3 H, s, OCH3), 3.72 (1 H, m, CHOTBDMS), 3.72* (1 H, m, CHOTBDMS), 3.79* (1 H, m, CHOH), 3.89 (1 H, m, CHOH), 4.55 (1 H, app td, J = 6.3, 2.5 Hz, CH), 4.64* (1 H, app td, J = 6.8, 2.7 Hz, CH), 5.06* (1 H, d, J = 5.5 Hz, OCHO), 5.11 (1 H, d, J = 4.9 Hz, OCHO), 7.19 (3 H, m, ArCH’s), 7.19* (3 H, m, ArCH’s), 7.29 (2 H, m, ArCH’s), 7.29* (2 H, m, ArCH’s)
13C NMR (100 MHz; CDCI3) 5C = (observed signals, mixture of 2 diastereoisomers) -4.42 (SiCH3), -4.41 (SiCH3), -4.33 (SiCH3), 18.1 (2 x C(CH3)3), 25.9 (2 x C(CH3)3), 29.3 (CH2), 30.3 (CH2), 31.7 (CH2), 35.1 (CH2), 38.8 (CH2), 39.9 (CH2), 40.0 (CH2), 41.1 (CH2), 42.7 (CH2),
46.5, 47.2, 54.4, 55.1, 55.3, 55.7, 71.8, 71.8, 79.3, 79.7, 82.5, 85.8, 106.5 (OCHOCH3), 108.0 (OCHOCH3), 125.7 (ArCH), 125.7 (ArCH), 128.3 (4 x ArCH), 128.3 (4 x ArCH), 142.6 (ArC), 142.6 (ArC). One SiCH3 and three CH2‘s could not be assigned due to overlapping signals. 6J. (3aR,4R,5R,6aS)-4-[(3R)-3-Hydroxy-5- phenylpentyl]perhydrocyclopenta[b]furan-2,5-diol, 74
Alcohol 73 (400 mg, 0.920 mmol) was stirred with 1.5% aqueous HQ / THF (3:2) (18 ml) at r.t. for 16 h. The mixture was neutralised with 1 M NaOH and extracted with CH2CI2 (5 x 30 ml). The combined organic phases were dried (MgS04), filtered, and concentrated to give the triol 74 and silanol by-product as a clear, colourless oil (~400 mg). This material was taken forward for the subsequent transformation without purification.
(4-Carboxybutyl)(triphenyl)phosphonium bromide 29 (2.45 g, 5.52 mmol) was added to a flame dried schlenk flask, under N2, and anhydrous THF (20.0 ml) added. The resulting suspension was cooled to 0 °C. KOt-Bu (1.24 g, 11.0 mmol) was added in one portion and the resulting orange mixture stirred at 0 °C for 40 min. A solution of crude triol 74 (282 mg, 0.920 mmol) in anhydrous THF (5.0 ml) was added dropwise via syringe. After complete addition the cooling bath was removed and the mixture was stirred at r.t. for 1.5 h. The reaction was quenched with H20 (30 ml) and washed with Et20 (2 x 30 ml) to remove triphenylphosphine oxide. The aqueous phase was made acidic with 1 M HQ (~10 ml) and extracted with CH2CI2 (5 x 25 ml). The combined organic phases were dried (MgS04), filtered, and concentrated to give the crude material as solids. These were placed on a sinter funnel and washed with petrol/EtOAc (1: 1) (4 x 20 ml) and then EtOAc (2 x 40 ml). The filtrate was concentrated under vacuum and purified by column chromatography on silica, eluting with CH2Cl2/MeOH (9.5:0.5 to 9:1) to give acid 75 (163 mg, 45% over 2 steps from alcohol 73) as a clear, colourless oil. The *Η data and optical rotation were consistent with the literature (Martynow, J. G. et al., European Journal of Organic Chemistry 2007, 2007, 689).
Rf = 0.27 (CH2CI2:MeOH, 9:1)
vmax (neatycnrr1 3338 (broad), 2930, 2857, 1704, 1452, 1407, 1254, 1028, 747, 699, 636 *H NMR (400 MHz; CDCI3) δΗ = 1.39 (2 H, m, CH2), 1.47-1.97 (10 H, m, 4 x CH2, 2 x CH), 2.07-2.48 (6 H, m, 3 x CH2), 2.67 (1 H, m, CH ), 2.80 (1 H, m, CH/-/), 3.60-4.85 (6 H, broad signal, 2 x OCH, 3 x OH, COOH), 3.72 (1 H, m, OCH), 5.40 (1 H, m, =CH), 5.49 (1 H, m, =CH), 7.15-7.24 (3 H, m, ArCH’s), 7.25-7.32 (2 H, m, ArCH’s)
[a]D 24 29.0 (c. 1.0, MeOH) (lit, [a]D 20 29.7 (c. 1.0, MeOH)) 6L. Isopropyl (Z)-7-(lR,2R,3R,5S)-3,5-dihydroxy-2-[(3R)-3-hydroxy-5- phenylpentyl]cyclopentyl-5-heptenoate, latanoprost, 77
A modified procedure of Zanoni and Vidari was used (Zanoni, G. et al., Tetrahedron 2010, 66, 7472). Carboxylic acid 75 (100 mg, 0.256 mmol) was dissolved in DMF (2.0 ml) and stirred at r.t.. Cs2C03 (125 mg, 0.384 mmol) was added in one portion followed by 2- iodopropane (51 μΙ, 0.512 mmol). The reaction was stirred at r.t. for 18 h. The reaction mixture was poured into 3% citric acid solution (10 ml) and extracted with TBME (4 x 10 ml). The combined organic phases were washed with 10% NaHC03 solution (10 ml) and saturated NaCI (2 x 10 ml) before being dried (MgS04), filtered, and concentrated to give the crude product as a clear, colourless oil (95 mg). This was purified by column chromatography (3 g silica), eluting with petrol/EtOAc (2: 1 to 1:2), to give latanoprost 77 (71 mg, 64 %) as a clear colourless oil. The IR, 13C, and optical rotation data were consistent with the literature (Zanoni, G. et al., Tetrahedron 2010, 66, 7472). Rf = 0.44 (EtOAc)
vmax (neatVcm“1 3360 (broad), 2980, 2931, 2857, 1712, 1495, 1454, 1374, 1311, 1247, 1180, 1106, 1030, 966, 910, 820, 731, 699
*H NMR (400 MHz; CDCI3) δΗ = 1.23 (6 H, d, J = 6.4 Hz, 2 x CH3), 1.30-1.90, (14 H, m, 5 x CH2, 2 x CH, 2 x OH), 2.07-2.39 (6 H, m, 3 x CH2), 2.45 (1 H, d, J = 5.5 Hz, OH), 2.63- 2.86 (2 H, m, CH2), 3.68 (1 H, br.s, CHO ), 3.95 (1 H, br.s, CHOH), 4.18 (1 H, br.s, CHO ), 5.01 (1 H, sept., J = 6.4 Hz, OCH(CH3)2), 5.35-5.52 (2 H, m, 2 x =CH), 7.16-7.24 (3 H, m, ArH’s), 7.25-7.32 (2 H, m, ArH’s)
13C NMR (125 MHz; CDCI3) 5C = 21.9 (2 x CH3), 24.9 (CH2), 26.6 (CH2), 26.8 (CH2), 29.6 (CH2), 32.1 (CH2), 34.0 (CH2), 35.7 (CH2), 39.0 (CH2), 42.5 (CH2), 51.8 (CH), 52.7 (CH), 67.6 (OCH), 71.2 (OCH), 74.5 (OCH), 78.6 (OCH), 125.7 (CH), 128.3 (2 x ArCH), 128.3 (2 x ArCH), 129.3 (CH), 129.5 (CH), 141.1 (ArC), 173.5 (C=0)
[a]D 23 33.0 (c. 1.0, MeCN) (lit, [a]D 20 32.7 (c. 1.0, MeCN))
References
- Ishikawa H, Yoshitomi T, Mashimo K, Nakanishi M, Shimizu K (February 2002). “Pharmacological effects of latanoprost, prostaglandin E2, and F2alpha on isolated rabbit ciliary artery”. Graefes Arch. Clin. Exp. Ophthalmol. 240 (2): 120–5. doi:10.1007/s00417-001-0412-4. PMID 11931077.
- Patel SS, Spencer CM (1996). “Latanoprost. A review of its pharmacological properties, clinical efficacy and tolerability in the management of primary open-angle glaucoma and ocular hypertension”. Drugs Aging 9 (5): 363–378. doi:10.2165/00002512-199609050-00007. PMID 8922563.
- Huttunen et al. (2011) Prodrugs—from Serendipity to Rational Design. Pharmacol Rev 63:750–771
- “Patent US5296504 – Prostaglandin derivatives for the treatment of glaucoma or ocular hypertension – Google Patents”.
- “WHO Model List of EssentialMedicines”. World Health Organization. October 2013. Retrieved 22 April 2014.
- Perry CM, McGavin JK, Culy CR, Ibbotson T (2003). “Latanoprost. An Update of its Use in Glaucoma and Ocular Hypertension”. Drugs Aging 20 (8): 1170–2229.PMID 12795627.
- Zhang WY, Wan Po AL, Dua HS, Azuara-Blanco A (2001). “Meta-analysis of randomised controlled trials comparing latanoprost with timolol in the treatment of patients with open angle glaucoma or ocular hypertension”. British Journal of Ophthalmology 85: 983–990. doi:10.1136/bjo.85.8.983. PMID 11466259.
- Aung T; Wong HT; Yip CC; et al. (2000). “Comparison of the intraocular pressure-lowering effect of latanoprost and timolol in patients with chronic angle closure glaucoma: a preliminary study.”. Ophthalmology 107 (6): 1178–83. doi:10.1016/s0161-6420(00)00073-7. PMID 10857840.
- Amano S, Nakai Y, Ko A, Inoue K, Wakakura M (2008). “A case of keratoconus progression associated with the use of topical latanoprost”. Japanese Journal of Ophthalmology 52 (4): 334–6. doi:10.1007/s10384-008-0554-6. PMID 18773275.
- De Santis, M., Lucchese, A., Carducci, B., Cavaliere, A., De Santis, L., & Merola, A. et al. (2004). Latanoprost exposure in pregnancy. American Journal Of Ophthalmology, 138(2), 305.pmid=15289149.1
- Morgan, P., Proniuk, S., Blanchard, J., & Noecker, R. (2001). Effect of temperature and light on the stability of latanoprost and its clinical relevance. Journal Of Glaucoma, 10(5), 401–405.
External links
- LATANOPROST solution [Greenstone LLC] (Nov 2011), Daily Med, U.S. National Library of Medicine, National Institutes of Health
Travoprost

Travoprost
cas 157283-68-6
[1R-[lα(Z),2β(lE,3R*),3α,5α]]-7-[3,5-Dihydroxy-2-[3-hydroxy-4-[3-(trifluoromethyl)phenoxy]-1 -butenyl]cyclopentyl]-5-heptenoic acid, 1 -methylethylester
(+)-16-m-trifluoromethylphenoxy tetranor Prostaglandin F2α isopropyl ester; (+)-Fluprostenol ispopropyl ester
(+)-(5Z,9α,1α,13E,15R)-trihydroxy-16-(3-(trifluoromethyl)phenoxy)-17,18,19,20-tetranor-prosta-5,13-dien-1-oic acid, isopropyl ester
(+) – Fluprostenol isopropyl ester,

Ophthalmic solution used for the reduction of elevated intraocular pressure in patients with open-angle glaucoma or ocular hypertension who are intolerant of other intraocular pressure lowering medications or insufficiently responsive (failed to achieve target IOP determined after multiple measurements over time) to another intraocular pressure lowering medication.
Travoprost free acid is a selective FP prostanoid receptor agonist and is believed to reduce intraocular pressure by increasing the drainage of aqueous humor, which is done primarily through increased uveoscleral outflow and to a lesser extent, trabecular outflow facility.
Travoprost, an isopropyl ester prodrug, is a synthetic prostaglandin F2 alpha analogue that is rapidly hydrolyzed by esterases in the cornea to its biologically active free acid. The travoporst free acid is potent and highly selective for the FP prostanoid receptor.
Travoprost ophthalmic solution is a topical medication used for controlling the progression of glaucoma or ocular hypertension, by reducing intraocular pressure. It is a synthetic prostaglandin analog (or more specifically, an analog of prostaglandin F2α)[1][2] that works by increasing the outflow of aqueous fluid from the eyes.[3] It is also known by the brand names of Travatan and Travatan Z, manufactured by Alcon, and Travo-Z, manufactured by Micro Labs.
Travoprost is a synthetic prostaglandin F analogue. Its chemical name is [1R-[lα(Z),2β(lE,3R*),3α,5α]]-7-[3,5-Dihydroxy-2-[3-hydroxy-4-[3-(trifluoromethyl)phenoxy]-1 -butenyl]cyclopentyl]-5-heptenoic acid, 1 -methylethylester. It has a molecular formula of C26H35F3O6 and a molecular weight of 500.55. The chemical structure of travoprost is:
 |
Travoprost is a clear, colorless to slightly yellow oil that is very soluble in acetonitrile, methanol, octanol, and chloroform. It is practically insoluble in water.
TRAVATAN® (travoprost ophthalmic solution) 0.004% is supplied as sterile, buffered aqueous solution of travoprost with a pH of approximately 6.0 and an osmolality of approximately 290 mOsmol/kg.
TRAVATAN® contains Active: travoprost 0.04 mg/mL; Preservative: benzalkonium chloride 0.15 mg/mL; Inactives: polyoxyl 40 hydrogenated castor oil, tromethamine, boric acid, mannitol, edetate disodium, sodium hydroxide and/or hydrochloric acid (to adjust pH) and purified water.
| Country | Patent Number | Approved | Expires (estimated) |
|---|---|---|---|
| Canada | 2181172 | 2003-04-29 | 2015-12-19 |
| Canada | 2129287 | 2002-05-14 | 2014-08-02 |
| United States | 5631287 | 1994-12-22 | 2014-12-22 |
| United States | 6503497 | 1995-05-06 | 2012-05-06 |
|
7-25-2012
|
TOPICAL APPLICATION OF TRAVOPROST FOR COMBATING HAIR LOSS
|
|
|
12-28-2011
|
Stable prostaglandin-containing compositions
|
|
|
7-22-2011
|
IMPROVED PROCESS FOR THE PRODUCTION OF BIMATOPROST
|
|
|
6-3-2011
|
Process for the Preparation of Prostaglandin Analogues and Intermediates Thereof
|
|
|
9-17-2010
|
Compositions and Methods for Reducing Body Fat
|
|
|
5-28-2010
|
COMPLEXES OF PROSTAGLANDIN DERIVATIVES AND MONOSUBSTITUTED, CHARGED BETA-CYCLODEXTRINS
|
|
|
4-30-2010
|
AMINO ACID SALTS OF PROSTAGLANDINS
|
|
|
4-30-2010
|
AMINO ACID SALTS OF PROSTAGLANDINS
|
|
|
2-24-2010
|
Compositions and methods for reducing body fat
|
|
|
1-15-2010
|
Process for the Production of Prostaglandins and Prostaglandin Analogs
|
|
4-3-2009
|
METHOD FOR SCREENING OF PROSTAGLANDIN COMPOUNDS COMPRISING AN OPTIMAL FORMULATION FOR THE ENHANCEMENT OF HAIR GROWTH AND THE STIMULATION OF FOLLICULAR ANAGEN AND FORMULATIONS RESULTING THEREFROM
|
|
|
3-9-2005
|
9,11-cycloendoperoxide pro-drugs of prostaglandin analogues for treatment of ocular hypertension and glaucoma
|
|
|
10-8-2004
|
Use of cloprostenol and fluprostenol analogues to treat glaucoma and ocular hypertension
|
|
|
4-21-2004
|
Use of cloprostenol and fluprostenol analogues to treat glaucoma and ocular hypertension
|
Side effects
Possible side effects of this medication are:
- May cause blurred vision
- May cause eyelid redness
- May permanently darken eyelashes
- May cause eye discomfort
- May eventually cause permanent darkening of the iris to brown (heterochromia)
- May cause a temporary burning sensation during use
- May cause thickening of the eyelashes
- May cause inflammation of the prostate gland, restricting urine flow (BPH)
 |
|
| Systematic (IUPAC) name | |
|---|---|
| propan-2-yl 7-[3,5-dihydroxy-2-[3-hydroxy-4-[3-(trifluoromethyl) phenoxy]-but-1-enyl]-cyclopentyl]hept-5-enoate |
|
| Clinical data | |
| Trade names | Travatan |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a602027 |
| Pregnancy cat. | C US |
| Legal status | Rx only (US) |
| Routes | Topical (eye drops) |
| Identifiers | |
| CAS number | 157283-68-6 |
| ATC code | S01EE04 |
| PubChem | CID 5282226 |
| DrugBank | DB00287 |
| ChemSpider | 4445407 |
| UNII | WJ68R08KX9 |
| Chemical data | |
| Formula | C26H35F3O6 |
| Mol. mass | 500.548 g/mol |


The condensation of 2- [3- (trifluoromethyl) phenoxy] acetyl chloride (I) with methylphosphonic acid dimethyl ester (II) by means of BuLi in THF gives 2-oxo-3- [3- (trifluoromethyl) phenoxy] propylphosphonic acid dimethyl ester (III), which is condensed with the known bicyclic aldehyde (IV) by means of BuLi in dimethoxyethane, yielding the unsaturated ketone (V). The reduction of (V) with zinc borohydride in dimethoxyethane affords the unsaturated alcohol (VI), which is treated with K2CO3 to give a diastereomeric mixture of unsaturated diols, resolved by chromatography to yield the chiral unsaturated diol (VII). The protection of (VII) with dihydropyran and TsOH in dichloromethane provides the bis (tetrahydropyranyl) ether (VIII), which by reduction of the lactone ring with diisobutylaluminum hydride in THF gives the lactol (IX). The condensation of (IX) with the phosphonium bromide (X) by means of NaH in DMSO yields the prostenoic acid (XI), which is esterified with isopropyl iodide and 1,8-diazabicyclo [5.4.0] undec-7-ene (DBU) in acetone to afford the corresponding isopropyl ester (XII). Finally, this compound is deprotected with acetic acid in hot THF / water.
http://www.chemdrug.com/databases/8_0_qkvreurfepijmjcf.html
…………………………………………..
Org Process Res Dev2002,6, (2): 138
http://pubs.acs.org/doi/abs/10.1021/op010097p

A commercial synthesis of the antiglaucoma agent, travoprost 2, is described. A total of 22 synthetic steps are required to provide the single enantiomer prostanoid, with the longest linear sequence being 16 steps from 3-hydroxybenzotrifluoride. The route is based upon a cuprate-mediated coupling of the single enantiomer vinyl iodide 13 and the tricyclic ketone 5, of high stereochemical purity, to yield the single isomer bicyclic ketone 15. A Baeyer−Villiger oxidation provides the lactone 16 as a crystalline solid, thus limiting the need for chromatographic purification. DIBAL-H reduction, Wittig reaction, esterification, and silyl group deprotection complete the synthesis of travoprost.
(5Z,13E)–(9S,11R,15R)-9,11,15-Trihydroxy-16-(m-trifluoromethylphenoxy-17,18,19,20-tetranor-5,13-prostadienoic Acid, Isopropyl Ester (2).
The silyl-protected compound (20a+b) (202 g, 277 mmol) ………..DELETED……………………………………… All relevant fractions were combined and concentrated to give the title compound 2 (97 g, 70%) as a colourless oil,  +14.6 (c 1.0, CH2Cl2); IR νmax (film) 3374 and 1727 cm–1; 1H NMR (400 MHz, CDCl3) δ 7.39 (1H, t, J = 8), 7.22 (1H, d, J = 8), 7.15 (1H, s), 7.08 (1H, d, J = 8), 5.70 (2H, m), 5.40 (2H, m), 4.98 (1H, heptet, J = 6.5), 4.52 (1H, m), 4.18 (1H, m), 3.97 (3H, m), 3.25 (2H, br s), 2.60 (1H, br s), 2.38 (1H, m), 2.30−1.96 (7H, m), 1.76 (1H, dd, J = 16, 4), 1.65 (2H, quintet, J = 7), 1.55 (1H, m), and 1.20 (6H, d, J = 6); 13C NMR (100 MHz, CDCl3) δ 173.57, 158.67, 135.45, 131.87 (q, J = 32), 130.02, 129.85, 129.75, 128.93, 123.89 (q, J = 270), 118.06, 117.82, 111.48, 77.77, 72.70, 71.99, 70.86, 67.72, 55.82, 50.24, 42.84, 34.00, 26.60, 25.48, 24.83, and 21.81; m/z (CI) 501 (MH+, 21), 321 (34), 303 (44), and 249 (100).
+14.6 (c 1.0, CH2Cl2); IR νmax (film) 3374 and 1727 cm–1; 1H NMR (400 MHz, CDCl3) δ 7.39 (1H, t, J = 8), 7.22 (1H, d, J = 8), 7.15 (1H, s), 7.08 (1H, d, J = 8), 5.70 (2H, m), 5.40 (2H, m), 4.98 (1H, heptet, J = 6.5), 4.52 (1H, m), 4.18 (1H, m), 3.97 (3H, m), 3.25 (2H, br s), 2.60 (1H, br s), 2.38 (1H, m), 2.30−1.96 (7H, m), 1.76 (1H, dd, J = 16, 4), 1.65 (2H, quintet, J = 7), 1.55 (1H, m), and 1.20 (6H, d, J = 6); 13C NMR (100 MHz, CDCl3) δ 173.57, 158.67, 135.45, 131.87 (q, J = 32), 130.02, 129.85, 129.75, 128.93, 123.89 (q, J = 270), 118.06, 117.82, 111.48, 77.77, 72.70, 71.99, 70.86, 67.72, 55.82, 50.24, 42.84, 34.00, 26.60, 25.48, 24.83, and 21.81; m/z (CI) 501 (MH+, 21), 321 (34), 303 (44), and 249 (100).
…………………………………………
http://www.google.com/patents/EP2495235A1?cl=en
……………………………
http://www.google.com/patents/EP2454227A1?cl=en

Example 2
Synthesis of Travoprost MTBE MTBE
C
7b
8b
9b-iso
Travoprost Scheme 4. Synthesis of Travoprost
References
- Alcon Laboratories, Inc. (September 2011). “TRAVATAN – travoprost solution”. DailyMed. Bethesda, MD: U.S. National Library of Medicine. Retrieved 2011-09-30.
- Alcon Laboratories, Inc. (September 2011). “TRAVATAN Z (travoprost) solution”. DailyMed. Bethesda, MD: U.S. National Library of Medicine. Retrieved 2011-09-30.
- AHFS Consumer Medication Information (2011-01-01). “Travoprost Ophthalmic”. MedlinePlus. Bethesda, MD: U.S. National Library of Medicine. Retrieved 2011-09-30.
More References:
Selective FP prostaglandin receptor agonist. Isopropyl ester of (+)-fluprostenol, q.v. General prepn (not claimed): J. W. Stjernschantz, EP 364417 (1989 to Pharmacia).
Large scale synthesis: L. T. Boulton et al., Org. Process Res. Dev. 6, 138 (2002).
Pharmacology: M. R. Hellberg et al., J. Ocul. Pharmacol. Ther. 17, 421 (2001).
LC/MS/MS determn in plasma: B. A. McCue et al., J. Pharm. Biomed. Anal. 28, 199 (2002). Ocular hypotensive effects in dogs: A. B. Carvalho et al., Vet. Ophthalmol. 9, 121 (2006).
Clinical trial in glaucoma or ocular hypertension: R. L. Fellman et al., Ophthalmology 109, 998 (2002); in combination with timolol: J. S. Schuman et al., Am. J. Ophthalmol. 140, 242-250 (2005).
- Ota T, Aihara M, Narumiya S, Araie M: The effects of prostaglandin analogues on IOP in prostanoid FP-receptor-deficient mice. Invest Ophthalmol Vis Sci. 2005 Nov;46(11):4159-63. PubMed: 16249494
- Thieme H, Schimmat C, Munzer G, Boxberger M, Fromm M, Pfeiffer N, Rosenthal R: Endothelin antagonism: effects of FP receptor agonists prostaglandin F2alpha and fluprostenol on trabecular meshwork contractility. Invest Ophthalmol Vis Sci. 2006 Mar;47(3):938-45. PubMed: 16505027
- Lim KS, Nau CB, O’Byrne MM, Hodge DO, Toris CB, McLaren JW, Johnson DH: Mechanism of action of bimatoprost, latanoprost, and travoprost in healthy subjects. A crossover study. Ophthalmology. 2008 May;115(5):790-795.e4. PubMed: 18452763
- Neacsu AM: [Receptors involved in the mechanism of action of topical prostaglandines] Oftalmologia. 2009;53(2):3-7. PubMed: 19697832
- Costagliola C, dell’Omo R, Romano MR, Rinaldi M, Zeppa L, Parmeggiani F: Pharmacotherapy of intraocular pressure – part II. Carbonic anhydrase inhibitors, prostaglandin analogues and prostamides. Expert Opin Pharmacother. 2009 Dec;10(17):2859-70. PubMed: 19929706
- Ferrari G, Scagliotti GV: Serum and urinary vascular endothelial growth factor levels in non-small cell lung cancer patients. Eur J Cancer. 1996 Dec;32A(13):2368-9. PubMed: 9038626
- Toris CB, Gabelt BT, Kaufman PL: Update on the mechanism of action of topical prostaglandins for intraocular pressure reduction. Surv Ophthalmol. 2008 Nov;53 Suppl1:S107-20. PubMed: 19038618
- Arranz-Marquez E, Teus MA: Prostanoids for the management of glaucoma. Expert Opin Drug Saf. 2008 Nov;7(6):801-8. PubMed: 18983226
- Chen X, Ji ZL, Chen YZ: TTD: Therapeutic Target Database. Nucleic Acids Res. 2002 Jan 1;30(1):412-5. PubMed: 11752352
Common drugs adversely impair older adults ability to feed and dress oneself
Public Release: 31-Jul-2014
Common drugs adversely impair older adults’ physical as well as cognitive functioning
INDIANAPOLIS — A class of medications previously linked to cognitive impairment in older adults also appears to negatively affect their physical functioning according to investigators from the Regenstrief Institute, the Indiana University Center for Aging Research, the University of East Anglia and several other United Kingdom institutions.
In a systemic review of more than a decade of studies on the effects of drugs with anticholinergic properties, they report that these drugs have a significant adverse effect on both cognitive and physical functioning, including the ability to feed and dress oneself. Anticholinergic medications affect the brain by blocking acetylcholine, a nervous system neurotransmitter. They are sold over the counter as sleep aids and bladder leakage preventives and prescribed for many diseases including hypertension and congestive heart failure.
View original post 575 more words
Bimatoprost

BIMATOPROST
155206-00-1
Bimatoprost ophthalmic solution is a topical medication used for controlling the progression of glaucoma or ocular hypertension, by reducing intraocular pressure. It is a prostaglandin analogue that works by increasing the outflow of aqueous fluid from the eyes. It binds to the prostanoid FP receptor.
Allergan reported Lumigan® sales of US$625 million and Latisse® sales of US$100 million in 2013
| Systematic (IUPAC) name | |
|---|---|
| 7-[3,5-dihydroxy-2- (3-hydroxy-5-phenyl-pent-1-enyl)- cyclopentyl]-N-ethyl-hept-5-enamide | |
| Clinical data | |
| Trade names | Lumigan |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a602030 |
| Licence data | US Daily Med:link |
| Pregnancy cat. | C (US) |
| Legal status | ℞-only (US) |
| Routes | Topical (eye drops) |
| Identifiers | |
| CAS number | 155206-00-1 |
| ATC code | S01EE03 |
| PubChem | CID 5311027 |
| IUPHAR ligand | 1958 |
| DrugBank | DB00905 |
| ChemSpider | 4470565 |
| UNII | QXS94885MZ |
| Chemical data | |
| Formula | C25H37NO4 |
| Mol. mass | 415.566 g/mol |
Bimatoprost (marketed in the U.S., Canada and Europe by Allergan, under the trade name Lumigan) is a prostaglandinanalog/prodrug used topically (as eye drops) to control the progression of glaucoma and in the management of ocular hypertension. It reduces intraocular pressure (IOP) by increasing the outflow of aqueous fluid from the eyes.[1] In December 2008, the indication to lengthen eyelashes was approved by the U.S. Food and Drug Administration (FDA); the cosmetic formulation of bimatoprost is sold as Latisse /ləˈtiːs/.[2] In 2008-2011, at least three case series suggested that bimatoprost has the ability to reduce adipose (fat) tissue.[3][4][5]
Allergen originally developed Bimatoprost and marketed it as Lumigan® for the treatment of elevated intraocular pressure (IOP), with open-angle glaucoma or ocular hypertension. Bimatoprost was later reformulated as a topical formulation and marketed as Latisse® for use in the treatment of hypotrichosis of the eyelashes.
Cosmetic use
In patients using ophthalmic prostaglandins such as travoprost and latanoprost, it has been anecdotally noted[by whom?] that there had been an increase in diameter, density and length of eyelashes. Allergan has initiatedclinical trials investigating the usage of Lumigan as a cosmetic drug.[6] On December 5, 2008, the FDA Dermatologic and Ophthalmic Drugs Advisory Committee voted to approve bimatoprost for the cosmetic use of darkening and lengthening eyelashes.The medical term for this is treatment of hypotrichosis, however, the FDA approval is for purely cosmetic purposes.[7]
For cosmetic purposes, it is administered once daily by applying the solution to the skin at the base of the eyelash using an applicator device “Application Guide”, where it has its effect upon the hair follicle.
Bimatoprost activates prostamide alpha F2 receptors found in the hair follicle to stimulate its growth rate. Research led by Professor Randall and the University of Bradford found that it may also offer a treatment for scalp hair regrowth in trials conducted on samples taken from men undergoing hair transplants.[8]
According to Allergan’s package labeling, users of its Latisse cosmetic product didn’t develop darker irises in clinical studies; however, “patients should be advised about the potential for increased brown iris pigmentation which is likely to be permanent.”[9]
Several cosmetics companies have released products based on prostaglandin analogs, as non-drug cosmetics.
- Age Intervention Eyelash by Jan Marini Skin Research
- RevitaLash by Athena Cosmetics Corp.
These companies have been sued by Allergan for patent infringement.[6] The FDA has seized Age Intervention Eyelash as an “unapproved and misbranded drug” because Jan Marini Skin Research promoted it as something that increases eyelash growth[10] and because it is “adulterated” with bimatoprost.[11]
Fat-reducing properties
Reductions in orbital fat (i.e., fat around the eye) have been observed in patients using bimatoprost as glaucoma therapy.[12] Of particular interest, the loss of orbital fat was unilateral in patients who used bimatoprost on only one eye.[13] The effect appears reversible upon cessation of bimatoprost use. The effect is likely to explain deepening of the lid sulcus described in a series of three patients on bimatoprost.[14] The mechanism for the apparent fat reduction remains unclear. However, bimatoprost is chemically analogous to prostaglandin F2α (PGF2α), a compound which is known to reduce fat by inhibition of adipocyte differentiation and survival.[15]
Formulations
Lumigan is a 0.03% solution of bimatoprost, and contains benzalkonium chloride as a preservative. Contact lenses should therefore be removed before use, and replaced no less than 15 minutes later;[1] other eye drops or ointments should be given no less than five minutes before or after bimatoprost.[1]
Efficacy
Studies have shown once-daily bimatoprost 0.03% ophthalmic solution to be more effective than timolol twice daily in reduction of intraocular pressure (IOP) and as effective as or more effective than the prostaglandin analogues latanoprost and travoprost in reducing IOP.[16]
Side effects
Possible side effects of this medication are:
- May cause blurred vision.
- May cause eyelid redness.
- May permanently darken eyelashes.
- May cause eye discomfort.
- May eventually cause permanent darkening of the iris to brown.
- May cause a temporary burning sensation during use.
- May cause thickening of the eyelashes.
- It may cause unexpected growth of hair if applied inappropriately, on the cheek, for example.
- It may cause infection if the one-time applicators which come with the genuine product are reused.
- Lashes may grow so long that they become ingrown and scratch the cornea.
- May cause darkening of the eyelid or of the area beneath the eye.[17]
On November 19, 2007, the FDA issued a warning during the seizure of a bimatoprost-containing cosmetic.[18] The warning stated that “the extra dose of bimatoprost may decrease the prescription drug’s effectiveness. Damage to the optic nerve may lead to decreased vision and possibly blindness.”
PATENTS
| Country | Patent Number | Approved | Expires (estimated) |
|---|---|---|---|
| Canada | 2585691 | 2009-05-19 | 2026-03-14 |
| Canada | 2144967 | 2003-11-11 | 2013-09-09 |
| United States | 7351404 | 2004-05-25 | 2024-05-25 |
| United States | 6403649 | 1992-09-21 | 2012-09-21 |
Jiang Xing Chen, “Process for the production of intermediates for making prostaglandin derivatives such as latanaprost, travaprost, and bimatoprost.” U.S. Patent US20090287003, issued November 19, 2009.
US20090287003
LUMIGAN® 0.01% and 0.03% (bimatoprost ophthalmic solution) is a synthetic prostamide analog with ocular hypotensive activity. Its chemical name is (Z)-7-[(1R,2R,3R,5S)-3,5Dihydroxy-2-[(1E,3S)-3-hydroxy-5-phenyl-1-pentenyl]cyclopentyl]-5-N-ethylheptenamide, and Its molecular weight is 415.58. Its molecular formula is C24H37NO4. Its chemical structure is:
 |
Bimatoprost is a powder, which is very soluble in ethyl alcohol and methyl alcohol and slightly soluble in water. LUMIGAN® 0.01% and 0.03% is a clear, isotonic, colorless, sterile ophthalmic solution with an osmolality of approximately 290 mOsmol/kg.
LUMIGAN® 0.01% contains Active: bimatoprost 0.1 mg/mL; Preservative: benzalkonium chloride 0.2 mg/mL; Inactives: sodium chloride; sodium phosphate, dibasic; citric acid; and purified water. Sodium hydroxide and/or hydrochloric acid may be added to adjust pH. The pH during its shelf life ranges from 6.8-7.8.
LUMIGAN® 0.03% contains Active: bimatoprost 0.3 mg/mL; Preservative: benzalkonium chloride 0.05 mg/mL; Inactives: sodium chloride; sodium phosphate, dibasic; citric acid; and purified water. Sodium hydroxide and/or hydrochloric acid may be added to adjust pH. The pH during its shelf life ranges from 6.8-7.8.

SYNTHESIS

……………………………….
http://www.google.com/patents/EP2495235A1?cl=en

-
For example, EP0364417B1 (Kabi Pharmacia AB) describes the following synthetic sequence:
-
The product is obtained as a mixture of epimers where the 15-OH can be in α-position or β-position. The removal of unwanted β-isomer and of other impurities represents one of the main difficulties in the preparation of prostaglandins and many methods have been proposed to reduce the amount of the undesired diastereoisomers formed during the preparation.
-
In EP0544899B1 (Pharmacia AB) the reduction of the α,β-unsaturated ketone is performed with lithium-tri(sec-butyl)borohydride at ―130°C.
-
In this kind of processes the low selectivity in the reduction of the keto group leads to a tedious separation of the diastereomers and generally decreases the global yield of the synthesis.
-
A different method of stereoselective reduction of the ketone is proposed in US7674921 B1 (Cayman Chemical Co.) where the reaction with lithium aluminium hydride in the presence of (S)-binaphtol in tetrahydrofuran at -78°C is reported.
-
An alternative is presented in US6927300B2 and in US 7157590B2(Finetech Lab. Ltd.) where the critical step of the reduction of the α,β-unsaturated keto-group is performed on the compound reported below
where R1 is an aryl carbonyl group in the presence of (-)β-chlorodiisopinocampheylborane in tetrahydrofuran at ―25°C which allows to obtain a ratio of 95/5 of the two diastereoisomers (R)-(IV)/(S)-(IV).
-
A further improvement in the preparation of PGF2α is directed to the reduction of lactone to lactol in order to introduce the α-chain by subsequent Wittig reaction with 4-carboxybutyltriphenylphosphonium bromide.
-
The reaction was usually carried out using diisobutylaluminum hydride at very low temperatures, namely between -80°C and ―40°C. Unfortunately, in these conditions the partial removal of the protecting group PPB or benzoyl (Bz) is possible.
-
In this way two additional steps of protection and deprotection are introduced possibly leading to a decrease of the global yield of the process at an advanced stage of the synthesis.
-
Moreover when P’ is a silylated group as reported in US7268239B2(Resolution Chemical Ltd.), a mixture of products is obtained because of the migration of the silyl group as shown in scheme 3
-
The same approach of hydrolysis of the aroyl group R followed by diprotection of the two hydroxyl groups with trialkylsilyl group or triaryl silyl group or tetrahydropiranyl group is disclosed in US7642370B2(Daichii Fine Chemical Co.). The protection of the two hydroxyl groups coming before the reduction of the lactone ring to lactol is described also in US7674921B1 (Cayman Chemical Co.) and in US6689901B2(Pharmacia and Upjohn Company).
-
It follows that one important issue in the synthesis of PGF2α is the involvement of intermediates with the most appropriate hydroxyl protecting group. As a consequence the use of the starting Corey lactone carrying a protection able to survive to the conditions of the subsequent reactions allows to form intermediates easy to isolate and to purify. Moreover the protection should be selectively removed in mild conditions.
-
In US5359095 (Pharmacia AB) the preparation of PGF2α prostaglandins is described starting from the commercially available (-)-Corey lactone (Ia) which is oxidized to the corresponding aldehyde (IIa)
-
The reaction is carried out in the presence of dicyclohexylcarbodiimide in dimethylsulfoxide and 1,2-dimethoxyethane; after quenching with orthophosphoric acid the aldehyde is obtained. In the same application it is reported that the crude aldehyde (II) is particularly unstable and must be used within a short period after preparation.
-
In US2010/0010239A1 (Sandoz AG) compound (I) is preferably oxidized in dichloromethane with oxalyl chloride and DMSO; the aldehyde (II) is not isolated and is processed directly in the solution where it is obtained or, when necessary, stored in solution at a temperature between ―20 and 0°C.
-
In US7268239B2 (Resolution Chem. Ltd.) the aldehyde (II)
where Z is (C6-C10)-aryl optionally substituted with one to three substituents independently selected from the group consisting of halo, C1 to C6 alkyl and unsubstituted C6 to C10 aryl, is formed by oxidation of the alcohol with sodium hypochlorite and 2,2,6,6-tetramethtyl-1-piperidinyloxy free radical (TEMPO); in order to avoid the risk of degradation, the aldehyde is directly used in the organic solution where it is synthesized.
-
According to US2009/0287003A1 (Eastar Chem. Corp.) five steps can be performed to prepare the aldehyde to be used as starting material for the synthesis of latanoprost. The five steps are reported in the scheme below:
-
In WO2010/097672 (Sifavitor) the starting material is the Corey lactone where the hydroxyl group is protected astert-butyldimethyl silyl derivative and during the synthesis a second protection is introduced according the scheme reported below referred to the preparation of Bimatoprost:
-
In a preferred embodiment, the conversion of compound 10a to Bimatoprost is performed by reaction with ethylamine in an organic solvent, which is chosen among amides, ethers, ketones and chlorinated solvents, more preferably N,N-dimethylformamide, at a temperature between ―35 and 25°C, more preferably between -20°C and -10°C, in the presence of triethylamine and a suitable coupling reagent, which is preferably 1-(methylsulfonyloxy)benzotriazole. The molar ratio between compound 10a and ethylamine is comprised between 1 and 5, more preferably is 3.5, while the molar ratio between compound 10a and the coupling reagent is comprised between 1 and 2.5, more preferably is 1.8.
-
When Latanoprost is the desired product the double bond on the side chain of compound 9a is hydrogenated to form compound 11, then by Wittig reaction with 4-carboxybutyltriphenylphosphonium bromide compound 11 is converted into Latanoprost acid 12. By conversion of the carboxylic acid into isopropyl ester, the final product Latanoprost is obtained:
EXAMPLE 13
(Z)-7-((1R,2R,3R,5S)-3,5-dihydroxy-2-((S,E)-3-hydroxy-5-phenylpent-1-enyl)cyclopentyl)hept-5-enoic acid (Bimatoprost free acid)
-
4-Carboxybutyltriphenylphosphonium bromide 15 (65.4 g, 0.148 mol) was suspended in tetrahydrofuran (150.0 mL) at 0°C under nitrogen atmosphere. A solution of potassium tert-butoxide in tetrahydrofuran (296.0 mL, 0.296 mol) was added dropwise and the mixture turned into orange. After stirring for 45 minutes at 0°C the system was cooled to ―15°C. A solution of (3aR,4R,5R,6aS)-4-((S,E)-3-hydroxy-5-phenylpent-1-enyl)hexahydro-2H-cyclopenta[b]furan-2,5-diol (10.0 g, 0.033 mol) in tetrahydrofuran (46.0 mL) was added dropwise at a temperature lower than – 10°C. After three hours at -15°C no more starting material was visible on TLC and water (200 mL) was added. The mixture was extracted with diisopropyl ether (144 mL) and the layers were separated. The aqueous phase was treated with 0.6 N HCl to pH 6.0. Two extractions with ethyl acetate (2x 250 mL) were then performed and the combined organic layers were concentrated under vacuum at 40°C. An oil (26.80 g) was obtained which was purified on silica gel (eluent: dichloromethane:methanol from 97.5:2.5 to 85:15). The fractions of interest were combined and concentrated at 35°C under reduced pressure affording a colorless oil (11.7 g, 0.030 mol, 91%).
-
1H-NMR {400 MHz, CDCl3, δ (ppm)}: 7.29-7.25 (m, 2H, Ph), 7.19-7.15 (m, 3H, Ph), 5.60 (dd, J=7.2, 15.2 Hz, 1H, vinyl), 5.51-5.41 (m, 2H, H vinyl), 5.38-5.31 (m, 1H, vinyl), 4.5-3.8 (m, 7H), 2.67 (m, 2H, -CH 2-Ph), 2.37-1.43 (m, 14H).
-
13C-NMR {400 MHz, CDCl3, δ (ppm)}: 177.2 (C), 141.9 (C), 134.9 (CH), 133.0 (CH), 129.6 (CH), 129.1 (CH), 128.4 (2xCH, arom.), 128.3 (2xCH arom.),125.8 (CH), 77.4 (CH), 72.3 (CH), 72.2 (CH), 55.1 (CH), 50.5 (CH), 42.7 (CH2), 38.5 (CH2), 32.9 (CH2), 31.8 (CH2), 26.3 (CH2), 25.2 (CH2), 24.4 (CH2).
-
HPLC-MS (ESI): [M-H2O+1]+= 371; [M+Na]+ = 411; [2M+Na]+ = 799.
EXAMPLE 14
(5Z)-7-[(2R)-3,5-Dihydroxy-2-[(1E)-3-hydroxy-5-phenylpent-1-en-1-yl]cyclopentyl]-N-ethylhept-5-enamide (Bimatoprost)
-
Bimatoprost acid (11.50 g, 0.030 mol) was dissolved in dimethylformamide (92.0 mL), and stirred at -15°C. Triethylamine (7.25 mL, 5.26 g, 0.052 mol) was then added over 5 minutes followed by the portionwise addition of 1-(methylsulfonyloxy)benzotriazole 16 (prepared according to Bulletin of the Chemical Society of Japan, 1978, 51(11), 3320-3329) (11.27 g, 0.053 mol). The mixture was then stirred for one hour at -15°C and an aqueous solution of ethylamine (70% weight, 8.4 mL, 0.104 mol) was added dropwise over 5 minutes. The temperature was allowed to reach 0°C and the reaction was checked by TLC. The mixture was washed with water (172.0 mL) and extracted four times with ethyl acetate (4x 230.0 mL). The combined organic layers were washed with 5% sodium bisulfate solution (100 mL, 50 mL, 50 mL). The bisulfate aqueous phases were extracted with ethyl acetate (50.0 mL). The organic layers were concentrated at 40°C under reduced pressure affording the crude product as an oil (16.98 g). Treatment with dichloromethane and diisopropylether at 0°C for one hour followed by filtration afforded a solid which was then recrystallized from ethyl acetate (6.79 g, 0.016 mol, 55%).
-
1H-NMR {400 MHz, CDCl3, δ (ppm)}: 7.26 (m, 2H, Ph), 7.17 (m, 3H, Ph), 6.07 (t, J=5.6Hz, 1H, -NH-), 5.57 (dd, J=7.6, 15.2 Hz, 1H, H-14), 5.45 (dd, J=8.8, 15.2 Hz, 1H, H-13), 5.35 (m, 2H, H-5+H-6), 4.32 (d, J=4.8 Hz, 1H, OH-11), 4.08 (m, 2H, H-9+H-15), 3.90 (m, 1H, H-11), 3.73 (m, 2H, OH-9+OH-15), 3.20 (m, 2H, -N-CH 2-CH3), 2.64 (m, 2H, -CH2-17), 2.30 (m, 1H, H-12), 2.18 (m, 2H, H-7+H-10), 2.11 (m, 3H, CH2-2+H-7), 2.03 (m, 2H, H-4), 1.89 (m, 1H, H-16), 1.77 (m, 2H, H-10+H-16), 1.64 (m, 2H, H-3), 1.45 (m, 1H, H-8), 1.09 (t, J=6.8Hz, 3H, -N-CH2-CH3).
-
13C-NMR {400 MHz, CDCl3, δ (ppm)}: 173.4 (C), 142.0 (C), 135.0 (CH), 133.2 (CH), 129.6 (CH), 129.1 (CH), 128.4 (2xCH arom), 128.3 (2xCH arom), 125.7 (CH arom), 77.5 (CH), 72.22 (CH), 72.20 (CH), 55.4 (CH), 50.1 (CH), 42.8 (CH2), 38.7 (CH2), 35.8 (CH2), 34.3 (-N-CH2), 31.8 (CH2), 26.6 (CH2), 25.6 (CH2), 25.3 (CH2), 14.7 (CH3).
-
HPLC-MS (ESI): [M+Na]+ = 438, [(M-H2O) +H]+=398, [(M-2H2O) +H]+=380.
………………………………..
http://www.google.com.ar/patents/US20090287003
Bimatoprost refers to (Z)-7-[(1R,2R,3R,5S)-3,5-Dihydroxy-2-[1E,3S)-3-hydroxy-5-phenyl-1-pentenyl]cyclopentyl]-5-N-ethylheptenamide, and its molecular weight is 415.58. Its molecular formula is C25H37NO4. Its chemical structure is:

……………………………
http://www.google.com/patents/EP2454227A1?cl=en
Example 1
Synthesis of Bimatoprost
9a-iso
Bimatoprost
Scheme 3. Synthesis of Bimatoprost.
An exemplary synthesis of bimatoprost, a prostaglandin analog, is shown in Scheme 3. The synthesis is scalable, highly convergent and includes a conjugate addition between two chiral synthons, cyclopentenone derivative 6 and vinyl iodide 7a to form ketone 8a. 7a and similar vinyl halides can be prepared in a manner analogous to that shown for the corresponding THP- protected vinyl iodide in U.S. Patent No. 7,109,371 to Clissold et al., or by other methods known in the art. Ketone 8a is reduced to the corresponding isomeric alcohols 9a and 9a-iso, followed by esterification with 5-hexenoic acid to produce ester intermediate 10a. A single isomer of the alcohol can be produced, if desired, by using a stereoselective reducing agent, such as a SELECTRIDE™ (Sigma-Aldrich, St. Louis, Missouri, United States of America) reducing agent. Ring-closure metathesis (RCM) of 10a produced 10- membered ring lactone 11a, which was subsequently deprotected to form lactone 12a. Ring-opening of lactone 12a with ethylamine produced Bimatoprost. The overall yield of Bimatoprost starting from 6 and 7a was good, with each step having a yield of about 60% or greater.
Individual steps in the synthesis of Bimatoprost are described further hereinbelow in Examples 5-10. An alternative step for the synthesis of ketone 8a, using an alkyne reagent, is also shown hereinbelow in Example 13. Ring- opening of 11a prior to deprotection to form a hydroxy-protected Bimatoprost and its subsequent deprotection are described in Examples 11 and 12. As shown below in Schemes 4-6 of Examples 2-4, other exemplary prostanoids were prepared via analogous routes as that shown in Scheme 3. Details regarding individual steps in these syntheses are also shown hereinbelow in Examples 5-10.
Synthesis of Bimatoprost from 12a:
As shown in Scheme 3 in Example 1 , a 250 ml_ 3-necked round-bottom flask equipped with a magnetic bar, a temperature probe, rubber septa, and a nitrogen gas inlet was charged at room temperature with 4.1 g (11.1 mmol) of deprotected lactone 12a in 20 ml_ of THF, 22.2 mi_( 44.3 mmol) of 2 M trimethylaluminum in THF, and 67 ml_ (133 mmol) of 2 M ethylamine in THF. The reaction mixture was heated at 40 °C for 18 h and TLC analysis indicated complete reaction. The mixture was diluted with 50 mL of water and the pH was adjusted to 6 with 1 N HCI. The layers were separated and the aqueous layer was back extracted with 20 mL of ethyl acetate for two times. The combined organic layers were washed with 40 mL of brine, dried over sodium sulfate, filtered, and concentrated.
The crude product was triturated with 20 mL of MTBE at 35 0C for 3 h, cooled to room temperature, and filtered to obtain 3.1 g (67.3% yield) of Bimatoprost, confirmed by 1H NMR.
Example 11
Synthesis of Protected Bimatoprost
Protected Bimatoprost
Scheme 7. Synthesis of Protected Bimatoprost.
As shown in Scheme 7 above, a 250 mL 3-necked round-bottom flask, equipped with a magnetic stirring bar, a temperature probe, rubber septa, and nitrogen inlet, was charged at room temperature, under nitrogen, with 3.0 g (5.01 mmol) of compound 11a in 3O mL of THF, and 15mL ethylamine, 2.0 M in THF. The mixture heated at 40 °C for 24 h and then reflux for another 3 h. TLC analysis (hexanes/ethyl acetate, 10:1) indicated complete reaction. The mixture was cooled to room temperature and diluted with 30 mL of MTBE and 25 mL of water. The layers were separated and the aqueous layer was washed with 15 mL of MTBE. The combined organic extracts were washed with 25 mL of brine, dried over sodium sulfate, filtered, concentrated, and chromatographically purified to afford 2.90 g (90.0% yield) of bis-silylated bimatoprost, confirmed by 1H NMR.
Example 12
Deprotection of Protected Bimatoprost
(8)
Protected Bimatoprost
Bimatoprost Scheme 8. Deprotection of Protected Bimatoprost.
As shown in Scheme 8 above, a 250 ml_ 3-necked round-bottom flask, equipped with a magnetic stirring bar, a temperature probe, rubber septa, and nitrogen inlet, was charged at room temperature, under nitrogen, with 3.Og (3.11 mmol) of protected bimatoprost from Example 12, 30 ml. of THF1 and 0.9 g (90.8 mmol) of ammonium hydrogen difluoride. The reaction mixture was heated at 40 0C for 24 h and TLC analysis (hexanes/ethyl acetate, 1 :1) indicated complete reaction. The mixture was then diluted with 30 ml. of MTBE and 25 ml_ of water followed by layer separation. The aqueous layer was back extracted with 15 ml_ of MTBE. The combined organic extracts were washed with 25 mL of brine, dried over sodium sulfate, filtered, concentrated, and chromatographically purified to afford 1.02 g (80.0% yield) of bimatoprost confirmed by 1H NMR.
………………………………
http://www.google.com/patents/WO2013186550A1?cl=en

Example 7 – Experimental procedures for the synthesis of bimatoprost
A synthesis of bimatoprost is shown and described below.
bimatoprost (97) 7A. (±)-5-Phenyl-l-(trimethylsilyl)pent-l-yn-3-ol, 81
Following a modified procedure of Matsuda (Matsuda, F. Et al., C em. Eur. J. 5, 3252-3259 (1999)); n-butyllithium (1.6 M in hexanes, 8.8 ml, 14.1 mmol, 1.0 eq.) was added dropwise to a solution of ethynyltrimethylsilane (2.0 ml, 14.1 mmol, 1.0 eq.) in THF (6 ml) at -78 °C. After addition, the mixture was allowed to warm slowly to 0 °C and stirred for 1 h. The mixture was cooled to -78 °C and a solution of hydrocinnamaldehyde (2.2 ml, 17.0 mmol, 1.2 eq.) in THF (3 ml) was added dropwise. The mixture was then allowed to slowly warm to -10 °C and stirred for 1 h before being quenched by the addition of saturated NH4CI solution (10 ml) followed by EtOAc (10 ml). The aqueous layer was extracted with EtOAc (3 x 10 ml), the combined organic phases were washed with brine (15 ml) before being dried (MgS04), filtered and concentrated to give the crude product. This was purified by column chromatography on silica, eluting with petrol/EtOAc (100:5), giving the title product 81 as a clear, colourless oil (3.1 g, 94 %). Analytical data consistent with the literature (Matsuda, F. Et al., Chem. Eur. J. 5, 3252-3259 (1999)). max (filmVcm-1 3326 (broad), 2951, 2172, 1604, 1496, 1454, 1249, 1045, 838
*H NMR (400 MHz; CDCI3) δΗ = 0.23 (s, 9H, 3 x CH3), 2.07 (m, 3H, CH2, OH), 2.84 (t, J = 7.8 Hz, 2H, CH2), 4.40 (t, J = 6.6 Hz, 1H, CHO ), 7.25 (m, 3H, ArCH’s), 7.33 (app t, J = 7.3 Hz, 2H, ArCH’s)
13C NMR (100 MHz; CDCI3) 5C = 0.0 (3 x CH3), 31.6 (CH2), 39.3 (CH2), 62.3 (CH), 90.0 (C), 106.7 (C), 126.1 (ArCH), 128.6 (2 x ArCH), 128.7 (2 x ArCH), 141.4 (ArC)
m/z (ESI+) 255.1 [MNa]+, 215.1
7B. Methyl 3-phenylpropanoate, 82
82 Hydrocinnamic acid (10.0 g, 66.6 mmol, 1 eq.) was dissolved in methanol (90 ml), cone. H2S04 (1 ml) added dropwise with stirring and the reaction mixture was stirred under reflux for 5 h. The reaction mixture was cooled to room temperature and concentrated under reduced pressure. The residue was dissolved in water (100 ml) and extracted with EtOAc (3 x 50 ml). The combined organic phases were washed with 10 % NaHC03 aq. (2 x 50 ml), brine (50 ml) before being dried (MgS04), filtered and concentrated to give the title compound 82 (10.8 g, 99 %) as a clear, colourless oil. Analytical data consistent with the literature (Black, P. J. et al., Eur. J. Org. Chem. 4367-4378 (2006)). vmax (filmVcm-1 3028, 2952, 1734, 1436, 1194, 1160, 749, 697
*H NMR (400 MHz; CDCI3) δΗ = 2.68 (t, J = 7.8 Hz, 2H, CH2), 3.00 (t, J = 7.8 Hz, 2H,
CH2), 3.71 (s, 3H, CH3), 7.25 (m, 3H, ArCH’s), 7.33 (m, 2H, ArCH’s)
13C NMR (100 MHz; CDCI3) 5C = 31.1 (CH2), 35.8 (CH2), 51.7 (CH3), 126.4 (2 x ArCH),
128.4 (ArCH), 128.6 (2 x ArCH), 140.6 (ArC), 173.5 (C=0)
m/z (CI+) 165.1 [MH]+ (20%), 133.1 (100%), 105.1 (55%), 93.1 (51%), 85.0 (57%)
7C. /V-Methoxy-/V-methyl-3-phenylpropanamide, 83
Following a procedure of Trost (Trost, B. M. et al., J. Am. Chem. Soc. 128, 6745-6754
(2006)); to a slurry of Λ/,Ο-dimethylhydroxylamine hydrochloride (4.91 g, 50.4 mmol, 2.1 eq.) in toluene (50 ml) at -10 °C was added AIMe3 (2 M in hexanes, 25.2 ml, 50.4 mmol, 2.1 eq.) dropwise. After addition, the mixture was allowed to warm to r.t. and stirred for 1 h. The mixture was cooled to -5 °C and a solution of methyl 3-phenylpropanoate 82 (3.94 g, 24.0 mmol, 1 eq.) in toluene (40 ml) was added dropwise. The reaction mixture was then allowed to warm slowly to r.t. and stirred for 3 h. The solution was cooled to 0 °C and quenched carefully by dropwise addition of HCI and the reaction mixture was extracted with EtOAc (4 x 70 ml). The combined organic phases were washed with brine (50 ml) before being dried (MgS04), filtered and concentrated to give the crude product. This was purified by column chromatography on silica, eluting with petrol/EtOAc (80:20 to 75:25), giving the Weinreb amide 83 as a clear, colourless oil (4.45 g, 97 %). Analytical data consistent with the literature (Trost, B. M. et al., J. Am. Chem. Soc. 128, 6745-6754 (2006); Murphy, J. A. et al., Org. Lett. 7, 1427-1429 (2005)). max (film cm-1 3017, 2937, 1659, 1453, 1414, 1383, 1176, 988, 750
*H NMR (400 MHz; CDCI3) δΗ = 2.74 (t, J = 7.8 Hz, 2H, CH2), 2.97 (t, J = 7.8 Hz, 2H, CH2), 3.18 (s, 3H, CH3), 3.60 (s, 3H, CH30), 7.25 (m, 5H, ArCH’s)
13C NMR (100 MHz; CDCI3) 5C = 30.7 (CH3), 32.2 (CH2), 33.8 (CH2), 61.2 (CH30), 126.1
(ArCH), 128.4 (4 x ArCH), 141.3 (ArC), 173.7 (C=0)
m/z (CI+) 194.1 [MH]+ (100%), 164.1 (20%), 133.1 (12%)
7D. 5-Phenyl-l-(tri/sopropylsilyl)pent-l-yn-3-one, 84
Following a procedure of Trost (Trost, B. M. et al., J. Am. Chem. Soc. 128, 6745-6754
(2006)); /7-Butyllithium (2.5 M in hexanes, 10.4 ml, 26 mmol, 1.7 eq.) was added dropwise to a solution of tri/sopropylsilyl acetylene (5.8 ml, 26 mmol, 1.7 eq.) in THF (53 ml) at -78 °C. After addition, the mixture was allowed to warm slowly to 0 °C and stirred for 1 h. The mixture was cooled to -78 °C and a solution of 83 (3.0 g, 15.3 mmol, 1 eq.) in THF (20 ml) was added dropwise. The mixture was then allowed to slowly warm to -10 °C and stirred for 1 h before being quenched by the addition of saturated aq. NH4CI (50 ml). The mixture was extracted with EtOAc (3 x 30 ml) and the combined organic phases were washed with brine (50 ml) before being dried (MgS04), filtered and concentrated to give the crude product. This was purified by column chromatography on silica, eluting with petrol/Et20 (100:0 to 98:2), giving the title product 84 as a clear, colourless oil (4.61 g, 96 %). max (filmycnr1 2944, 2866, 1675, 1462, 1445, 1103, 881, 696
*H NMR (400 MHz; CDCI3) δΗ = 1.12 (m, 21 H, 6 x CH3, 3 x CH), 2.92 (m, 2H, CH2), 3.03 (m, 2H, CH2), 7.22 (m, 3H, ArCH’s), 7.31 (m, 2H, ArCH’s)
13C NMR (100 MHz; CDCI3) 5C = 11.1 (3 x CH), 18.6 (6 x CH3), 30.2 (CH2), 47.4 (CH2), 96.2 (C), 104.2 (C), 126.4 (ArCH), 128.4 (2 x ArCH), 128.7 (2 x ArCH), 140.4 (ArC), 186.7 (C=0) m/z (CI+) 315.3 [MH]+ (100%), 271.2 52%), 157.2 (27%)
HRMS (CI+) calcd for C20H3iOSi [MH]+ 315.2144, found 315.2139
7E. (S)-5-Phenyl-l-(tri/sopropylsilyl)pent-l-yn-3-ol, 85
Following a modified procedure of Trost (Trost, B. M. et al., J. Am. C em. Soc. 128, 6745- 6754 (2006)); Potassium hydroxide (8.5 mg, 0.15 mmol, 1.2 mol%) and RuCI(p- cymene)[(S,S)-Ts-DPEN] (80.7 mg, 0.127 mmol, 1 mol%) were added to /PrOH (110 ml) and the resultant mixture was stirred for 2 min at r.t.. 5-Phenyl-l-(tri/sopropylsilyl)pent-l-yn-3-one 84 (4.0 g, 12.7 mmol, 1 eq.) was added via syringe and the mixture was stirred at r.t. for 45 min. The mixture was concentrated in vacuo to give the crude product. This was purified by column chromatography on silica, eluting with petrol/Et20 (20:1 to 10:1), giving the title product 85 as a clear, colourless oil (4.0 g, 99 %). The enantiomeric excess was determined to be 99 % via HPLC analysis of its derivative 87. Analytical data consistent with the literature (Trost, B. M. et al., J. Am. Chem. Soc. 128, 6745-6754 (2006)). max (filmVcm-1 3322 (broad), 2942, 2864, 1462, 1045, 1011, 996, 882, 697, 675
*H NMR (300 MHz; CDCI3) δΗ = 1.07-1.11 (m, 21 H, 6 x CH3, 3 x CH), 1.83 (d, J = 5.5 Hz, 1H, OH), 2,03 (m, 2H, CH2), 2.82 (t, J = 7.9 Hz, 2H, CH2), 4.40 (dd, J = 6.4, 5.5 Hz, 1H, CHOH), 7.17-7.32 (m, 5H, ArCH’s)
13C NMR (75 MHz; CDCfe) 5C = 11.2 (3 x CH), 18.7 (6 x CH3), 31.6 (CH2), 39.7 (CH2), 62.4 (CH), 86.1 (C), 108.5 (C), 126.1 (ArCH), 128.6 (4 x ArCH), 141.5 (ArC)
m/z (ESI+) 339.2 [MNa]+, 299.2., 225.0
HRMS (ESI+) calcd for C20H32OSiNa [MNa]+ 339.2114, found 339.2127
[a]D 22 + 28.5 (c. 2.0, CHCI3) (lit., +27.17 (c. 2.14, CH2CI2)) 7F. (S)-5-Phenylpent-l-yn-3-ol, 86
Following a modified procedure of Trost (Trost, B. M. et al., J. Am. C em. Soc. 128, 6745- 6754 (2006)); Tetrabutylammonium fluoride (1.0 M in THF, 25 ml, 25 mmol, 2.5 eq.) was added to a solution of (S)-5-phenyl-l-(tri/sopropylsilyl)pent-l-yn-3-ol 85 (3.165 g, 10 mmol, 1 eq.) in THF (95 ml). The reaction mixture was stirred for 1 h at r.t. and then quenched by addition of saturated aq. NH4CI (50 ml). The mixture was extracted with Et20 (3 x 40 ml), the combined organic phases were washed with brine (50 ml) before being dried (MgS04), filtered and concentrated to give the crude product. This was purified by column chromatography on silica, eluting with petrol/Et20 (10: 1 to 9:1), giving the title product 86 as a clear, colourless oil (1.6 g, 99 %). max (filmVcm-1 3289 (broad), 1603, 1496, 1454, 1300 (broad), 1040, 1010, 744
*H NMR (400 MHz; CDCI3) δΗ = 2.10 (m, 3H, CH2, OH), 2.55 (d, J = 2.2 Hz, 1H, C≡CH), 2.85 (t, J = 7.8 Hz, 2H, CH2), 4.40 (m, 1H, CHOH), 7.25 (m, 3H, ArCH’s), 7.33 (m, 2H, ArCH’s)
13C NMR (100 MHz; CDCI3) 5C = 31.4 (CH2), 39.2 (CH2), 61.7 (CHOH), 73.5 (C≡CH), 84.8 (C≡CH), 126.2 (2 x ArCH), 128.6 (3 x ArCH), 141.2 (ArC)
m/z (CI+) 161.1 [MH]+ (15%), 155.1 (37%), 143.1 (88%), 119.1 (42%), 91.1 (100%) HRMS (CI+) calcd for CnH130 [MH]+161.0966, found 161.0972
[a]D 22 + 20.0 (c. 2.0, CHC )
7G. (S)-5-Phenyl-l-(trimethylsilyl)pent-l-yn-3-ol, 87
1) nBuLi, THF
-78 °C to 0 °C
OH 2) Me3SiCI OH
3) Citric acid
86 87 Following a procedure of Trost (Trost, B. M. et al., J. Am. C em. Soc. 128, 6745-6754 (2006)); /7-butyllithium (2.5 M in hexanes, 474 pL, 1.18 mmol, 3 eq.) was added dropwise to a solution of (S)-5-phenylpent-l-yn-3-ol 86 (63.3 mg, 0.39 mmol, 1 eq.) in THF (1 ml) at -78 °C. The mixture was allowed to warm to 0 °C and stirred 30 min before being cooled to -78 °C. TMSCI (148.2 pL, 1.18 mmol, 3 eq.) was added dropwise and the mixture was allowed to warm to r.t. and stirred for 2 h. Citric acid (65 mg) in methanol (0.7 ml) was added and the mixture stirred for 1 h. The mixture was poured into a mixture of brine (3 ml) and Et20 (5 ml). The aqueous layer was extracted with Et20 (3 x 10 ml) and the combined organic phases were washed with brine (10 ml) before being dried (MgS04), filtered and concentrated to give the crude product. This was purified by column chromatography on silica, eluting with petrol/Et20 (9:1), giving the title product 87 as a clear, colourless oil (18.5 mg, 15.5 %). Analytical data consistent with the literature (Trost, B. M. et al., J. Am. Chem. Soc. 128, 6745-6754 (2006)). vmax(filmVcm-1 3344 (broad), 2956, 2172, 1496, 1454, 1249, 1046, 838
*H NMR (400 MHz; CDCI3) δΗ = 0.19 (s, 9H, 3 x CH3), 1.80 (d, J = 5.6 Hz, 1H, OH), 2.02 (m, 2H, CH2), 2.80 (t, J = 7.8 Hz, 2H, CH2), 4.36 (dd, J = 6.3, 5.6 Hz, 1H, CHOH), 7.20 (m, 3H, ArCH’s), 7.29 (m, 2H, ArCH’s)
13C NMR (100 MHz; CDCI3) 5C = -0.1 (3 x CH3), 31.4 (CH2), 39.2 (CH2), 62.2 (CHOH), 89.9 (C), 106.4 (C), 125.9 (ArCH), 128.4 (2 x ArCH), 128.5 (2 x ArCH), 141.3 (ArC)
m/z (ESI+) 255.1 [MNa]+
HRMS (ESI+) calcd for Ci4H20OSiNa [MNa]+ 255.1175, found 255.1185
[a]D 22 + 28.6 (c. 0.735, CHCI3) (lit., +32.91 (c. 2.37, CH2CI2))
Chiral-HPLC data: ee = >99 % (Chiralcel OD column, 210 nm, hexane/2-propanol: 90/10, flow rate: 0.7 mlVmin, room temperature; ¾: minor 15.5 min, major 10.7 min)
7H. (S)-tert-Butyldimethyl(5-phenylpent-l-yn-3-yloxy)silane, 88
Following a procedure of Noyori (Suzuki, M. et al., J. Med. Chem. 41, 3084-3090 (1998)); Imidazole (919.1 mg, 13.5 mmol, 1.8 eq.) and t-butylchlorodimethylsilane (1.35 g, 9.0 mmol) were added to a solution of (S)-5-phenylpent-l-yn-3-ol 86 (1.2 g, 7.5 mmol, 1 eq.) in CH2CI2 (18 ml), cooled to 0 °C. The reaction mixture was then stirred at room temperature for 14 h before being poured into 1 M HCI (50 ml). The mixture was extracted with 40/60 petroleum ether (3 x 50 ml). The combined organic phases were washed with brine (50 ml) before being dried (MgS04), filtered and concentrated to give the crude product. This was purified by column chromatography on silica, eluting with petrol/Et20 (99:1), giving the title product 88 as a clear, colourless oil (1.77 g, 86 %). Analytical data consistent with the literature (Kiyotsuka, Y. et al., Org. Lett. 10, 1719-1722 (2008).; Sato, F. et al., EP 1211241 Al, Taisho Pharmaceutical co., LTD (2002)). max (filmVcm-1 3309, 2954, 2929, 2886, 2857, 1251, 1091, 834, 776
*H NMR (400 MHz; CDCI3) δΗ = 0.15 (s, 3H, CH3), 0.17 (s, 3H, CH3), 0.95 (s, 9H, 3 x CH3), 2.04 (m, 2H, CH2), 2.46 (d, J = 2.1 Hz, C≡CH), 2.81 (m, 2H, CH2), 4.41 (dt, J = 6.3, 2.1 Hz, CHOTBDMS), 7.24 (m, 3H, ArCH’s), 7.32 (m, 2H, ArCH’s)
13C NMR (100 MHz; CDCI3) 5C = -4.9 (CH3), -4.4 (CH3), 18.3 (C), 25.9 (3 x CH3), 31.4
(CH2), 40.3 (CH2), 62.2 (CHOTBDMS), 72.5 (C≡CH), 85.4 (C≡CH), 125.9 (ArCH), 128.5 (2 x ArCH), 128.6 (2 x ArCH), 141.7 (ArC)
m/z (CI+) 275.2 [MH]+ (12%), 259.1 (37%), 217.1 (50%), 189.1 (38%), 143.1 (100%) HRMS (CI+) calcd for Ci7H27OSi [MH]+275.1831, found 275.1825
[a]D 22 -17.0 (c. 2.0, CHCI3)
71. (S,£)-teif-Butyl(l-iodo-5-phenylpent-l-en-3-yloxy)dimethylsila
A flame dried Schlenk flask, evacuated and purged with nitrogen, was charged with alkyne 88 (1.50 g, 5.46 mmol, 1 eq.). Anhydrous CH2CI2 (35 ml) was added and the reaction stirred at r.t. Zr(Cp)2HCI (2.82 g, 10.9 mmol, 2 eq.) was added as a solid, in portions. The yellow suspension was stirred at r.t. for 1 h. The resulting yellow solution was cooled to 0 °C and iodine (1.52 g, 6.01 mmol, 1.1 eq.) added as a solid, in one portion. The cooling bath was removed and the reaction mixture stirred at room temperature for 1 h. The reaction mixture was poured into water (100 ml) and extracted with 40/60 petroleum ether (4 x 50 ml). The combined organic phases were washed with water (100 ml), saturated Na2S203 solution (2 x 100 ml) and brine (100 ml) before being dried (MgS04), filtered, and concentrated to give the crude material. This was purified by flash chromatography, eluting with 40/60 petroleum ether. The fractions containing product were combined and washed with saturated Na2S203 solution (20 ml), dried (MgS04), filtered, and concentrated to give the title compound 89 (1.98 g, 90%) as a clear, colourless oil. Analytical data consistent with the literature (Sato, F. et al., EP 1211241 Al, Taisho Pharmaceutical co., LTD (2002)). max (filmVcm-1 2952, 2928, 2856, 1604, 1360, 1251, 1086, 942, 833, 774
*H NMR (400 MHz; CDCI3) δΗ = 0.04 (s, 3H, CH3), 0.06 (s, 3H, CH3), 0.92 (s, 9H, 3 x CH3), 1.82 (m, 2H, CH2), 2.65 (m, 2H, CH2), 4.15 (dq, J = 6.0, 1.2 Hz, 1H, C /-/OTBDMS), 6.25 (dd, J = 14.3, 1.2 Hz, 1H, CH=CHI), 6.57 (dd, J = 14.3, 6.0 Hz, 1H, CH=CHI), 7.18 (m, 3H, ArCH’s), 7.29 (m, 2H, ArCH’s)
13C NMR (100 MHz; CDCI3) 5C = -4.9 (CH3), -4.5 (CH3), 18.2 (C), 25.8 (3 x CH3), 31.0 (CH2), 39.1 (CH2), 74.6 (CHOTBDMS), 76.1 (CH=CHI), 125.8 (ArCH), 128.3 (2 x ArCH), 128.4 (2 x ArCH), 141.8 (ArC), 148.8 (CH=CHI)
m/z (ESI+) 425.1 [MNa]+
HRMS (ESI+) calcd for Ci7H27IOSiNa [MNa]+ 425.0768, found 425.0754
[a]D 22 -4.5 (c. 2.0, CHCI3)
73. tert-Butyl((S,l£)-l-((3aR,4R,6aS)-2-methoxy-5- ((trimethylsilyloxy)methylene)hexahydro-2H-cyclopenta[d]furan-4-yl)-5- phenylpent-l-en-3-yloxy)dimethylsilane, 92
90 92 Vinyl iodide 89 (1.45 g, 3.6 mmol, 1.2 eq.) was added via syringe to a flame dried Schlenk flask (evacuated and purged with nitrogen several times and allowed to cool). Anhydrous Et20 (14.5 ml) was added via syringe and the resulting solution cooled to -78 °C. t-BuLi (1.6 M, 4.5 ml, 7.2 mmol, 2.4 eq.) was added dropwise and the reaction mixture stirred at -78 °C for 2 h and -40 °C for 2 h before being cooled back to -78 °C. Meanwhile, thiophene (303 mg, 288 μΙ, 3.6 mmol, 1.2 eq.) was added via syringe to a flame dried Schlenk flask (evacuated and purged with nitrogen several times and allowed to cool). Anhydrous THF (14.5 ml) was added via syringe and the resulting solution cooled to -30 °C. n-BuLi (1.6 M, 2.25 ml, 3.6 mmol, 1.2 eq.) was added dropwise and the solution stirred at -30 °C for 30 min. The solution was then cooled to -78 °C and CuCN (322.4 mg, 3.6 mmol, 1.2 eq.) added as a solid, in one portion. The cooling bath was removed and the suspension allowed to warm to r.t. The resulting tan/brown solution of cuprate was added dropwise via syringe to the Schlenk flask containing the vinyl lithium and anhydrous THF (14.5 ml) added. The mixture was stirred at -20 °C for 1 h to allow formation of mixed cuprate 90. This was cooled to -78 °C and a solution of enal 24 (504.6 mg, 3.0 mmol, 1.0 eq.) in anhydrous THF (14.5 ml) was added dropwise. The mixture was stirred at -78 °C for 1 h and then allowed to warm slowly to -20 °C. TMSCI (2.2 ml) was added via syringe followed by NEt3 (2.8 ml). The reaction was quenched by the addition of saturated NH4CI solution (80 ml) and extracted with Et20 (3 x 80 ml). The combined organic phases were washed with saturated NH4CI solution (40 ml) before being dried (MgS04), filtered, and concentrated to give the crude material as a yellow oil. This was used directly in the next step.
7K. (3aR 4R,5R,6aS)-4-((S,£)-3-(tert-Butyldimethylsilyloxy)-5-phenylpent-l- enyl)-2-methoxyhexahydro-2H-cyclopenta[d]furan-5-ol, 93
92 93
The crude material from the conjugate addition / trapping experiment, containing 92, was dissolved in C^Cb/MeOH (3:1) (32 ml) and cooled to -78 °C. A stream of ozone was passed through the stirred solution. The reaction was monitored periodically by TLC in order to judge completion of the ozonolysis (judged by consumption of silyl enol ether). At this point, the flask was purged with a stream of nitrogen during 10 min and NaBH4 (204 mg, 5.4 mmol) was added in one portion. The reaction mixture was stirred at -78 °C for 2 h before the cooling bath was removed and the reaction allowed to warm to r.t.. The reaction was stirred at r.t. for 1 h. The reaction mixture was poured into saturated NaCI solution (20 ml) and extracted with EtOAc (3 x 40 ml). The combined organic phases were dried (MgS04), filtered, and concentrated to give the crude product as a pale yellow oil. This was purified by column chromatography on silica, eluting with petrol/EtOAc (9:1 to 8:2), giving the alcohol 93 (as a mixture of diastereoisomers) as a clear, colourless oil (731.0 mg, 56.0% (2 steps from enal 24)). max (filmVcm-1 3434 (broad), 2928, 1496, 1471, 1454, 1360, 1250, 1098, 1044, 1003, 970, 834, 774
*H NMR (400 MHz; CDCI3)
δΗ = (mixture of 2 diastereoisomers, signals of minor diastereoisomer indicated by *) 0.05 (s, 3H, CH3), 0.06* (s, 3H, CH3), 0.07 (s, 3H, CH3), 0.08* (s, 3H, CH3), 0.93 (s, 9H, 3 x CH3), 0.94* (s, 9H, 3 x CH3), 1.74-2.52* (m, 8H, 3 x CH2, 2 x CH), 1.74-2.52 (m, 7H, 3 x CH2, CH), 2.56-2.76* (m, 2H, CH2), 2.56-2.76 (m, 3H, CH2, CH), 3.35 (s, 3H, OCH3), 3.39* (s, 3H, OCH3), 3.81* (m, 1H, CHOH), 3.94 (m, 1H, CHOH), 4.15* (m, 1H, C/-OTBDMS), 4.15 (m, 1H, CHOTBDMS), 4.53 (app td, J = 6.6, 3.2 Hz, 1H, CH), 4.63* (app td, J = 7.5, 4.6 Hz, 1H, CH), 5.09* (app d, J = 5.6 Hz, 1H, CH), 5.14 (app d, J = 4.4 Hz, 1H, CH), 5.48* (m, 1H, =CH), 5.48 (m, 1H, =CH), 5.60* (m, 1H, =CH), 5.60 (m, 1H, =CH), 7.20* (m, 3H, ArCH’s), 7.20 (m, 3H ArCH’s), 7.30* (m, 2H, ArCH’s), 7.30 (m, 2H, ArCH’s)
13C NMR (100 MHz; CDCI3) 5C = (mixture of 2 diastereoisomers, signals of minor diastereoisomer indicated by *) -4.6 (CH3), -4.6* (CH3), -4.0* (CH3), -3.9 (CH3), 18.4 (C), 18.4* (C), 26.0 (3 x CH3), 26.0* (3 x CH3), 31.8 (CH2), 31.9 (CH2), 38.0 (CH2), 39.3 (CH2), 39.7 (CH2), 40.2 (CH2), 40.3 (CH2), 42.5 (CH2), 45.8 (CH), 46.1* (CH), 54.6* (CH30), 55.0 (CH30), 56.7* (CH), 57.6 (CH), 72.8 (CHOTBDMS), 72.9* (CHOTBDMS), 77.9* (CHOH), 78.9 (CHOH), 81.2 (CH), 83.3* (CH), 106.6 (CH), 107.7* (CH), 125.7 (ArCH), 125.8 (ArCH), 126.0 (ArCH), 128.4 (ArCH), 128.5 (ArCH), 128.6 (ArCH), 130.3* (=CH), 131.1 (=CH), 135.1* (=CH), 135.2 (=CH), 142.2* (ArC), 142.5 (ArC)
m/z (ESI+) 455.1 [MNa]+
HRMS (ESI+) calcd for C25H4o04SiNa [MNa]+ 455.2588, found 455.2587 7L. (3a ?,4 ?,5 ?,6aS)-4-((S,E)-3-Hydroxy-5-phenylpent-l-enyl)hexahyd cyclopenta[b]furan-2,5-diol, 94
93 94
Alcohol 93 (210 mg, 0.485 mmol) was stirred with 1.5% aqueous HCI / THF (3:2) (10 ml) at r.t. for 16 h. The mixture was extracted with CH2CI2 (5 x 15 ml) and the combined organic phases were dried (MgS04), filtered, and concentrated to give the triol 94 and silanol byproduct as a clear, colourless oil. This material was taken forward for the subsequent transformation without purification.
7M. 5-Bromo-/V-ethylpentanamide, 95
1) SOCI2, DMF cat.
Toluene, 55 °C
95
Dimethylformamide (116 μΙ_, 1.5 mmol, 0.1 eq.) and thionyl chloride (1.63 ml, 22.5 mmol, 1.5 eq.) were added to a solution of 5-bromovaleric acid (2.71 g, 15 mmol, 1 eq.) in toluene (20 ml) and the reaction mixture was stirred at 50 °C for 4 h. The volatiles were removed under high vacuum, THF (130 ml) was added and the mixture was cooled to 0 °C. N,N- Diisopropylethylamine (4.2 ml, 24 mmol, 1.6 eq.) and ethylamine (2 M solution in THF, 9.0 ml, 18 mmol, 1.2 eq.) were added dropwise. The reaction mixture was stirred for 1 h at 0 °C before being quenched by the addition of saturated aq. NH4CI (100 ml). The reaction mixture was extracted with Et20 (4 x 75 ml) and the combined organic phases were washed with brine (100 ml) before being dried (MgS04), filtered, and concentrated to give the crude material. This was purified by flash chromatography, eluting with petrol/EtOAc (4:6), giving the title product 95 as a clear, colourless oil (2.75 g, 88 %). Analytical data consistent with the literature (Halazy, S. et al., WO 9612713, Pierre Fabre Medicament (1996)). max (filmVcm-1 3275 (broad), 2970, 2933, 1640, 1543, 1439, 1276, 1150, 643
*H NMR (400 MHz; CDCI3) δΗ = 1.13 (t, J = 7.2 Hz, 3H, CH3), 1.78 (m, 2H, CH2), 1.89 (m, 2H, CH2), 2.19 (t, J = 7.3 Hz, CH2CO), 3.27 (dt, J = 7.2, 5.5 Hz, 2H, NHCH2), 3.41 (t, J = 6.5 Hz, 2H, BrCH2), 5.79 (broad s, 1H, NH)
13C NMR (100 MHz; CDCI3) 5C = 15.0 (CH3), 24.4 (CH2), 32.2 (CH2Br), 33.4 (CH2), 34.4 (NHCH2), 35.7 (CH2C=0), 172.3 (C=0)
m/z (CI+) 210.0 [M81BrH]+ (80%), 208.0 [M79BrH]+ (80%), 128.1 (100%), 107.1 (60%) HRMS (CI+) calcd for C7Hi5 81BrNO [M81BrH]+ 210.0317, found 210.0320; calcd for
C7H15 79BrNO [M79BrH]+ 208.0337, found 208.0341
(5-(Ethylamino)-5-oxopentyl)triphenylphosphonium bromide, 96
Triphenylphosphine (2.88 g, 11 mmol, 1.1 eq.) was added to a solution of 5-bromo-A/- ethylpentanamide 95 (2.08 g, 10 mmol, 1 eq.) in MeCN (5 ml) and the mixture was stirred for 14 h at 80 °C. The mixture was allowed to cool to r.t. and concentrated under vacuum. The residue was added dropwise into Et20 (100 ml) and stirred vigourously for 10 min. The resulting solid was filtered, washed with Et20 (2 x 10 ml) and dissolved in CH2CI2 (15 ml). This solution was then added dropwise into Et20 (200 ml) and stirred for 10 min. Solids were filtered, washed with Et20 (2 x 10 ml) and dried under high vacuum to give the title product 96 as a white powder (3.87 g, 82 %).
Mp 177-179 °C
max (filmVcm“1 3275 (broad), 2970, 2933, 1640, 1543, 1439, 1276, 1150, 643
*H NMR (400 MHz; CDCI3) δΗ = 1.06 (t, J = 7.3 Hz, 3H, CH3), 1.63 (m, 2H, CH2), 1.91 (quin., J = 6.5 Hz, CH2), 2.57 (t, J = 6.5 Hz, 2H, CH2C=0), 3.15 (q, J = 7.3, 5.6 Hz, 2H, NHCH2), 3.66 (m, 2H, CH2P+), 7.66 (m, 6H, ArCH’s), 7.75-7.82 (m, 9H, ArCH’s), 8.26 (t, J = 5.6 Hz, 1H, NH)
13C NMR (100 MHz; CDCI3) 5C = 14.8 (s, CH3), 21.1 (d, J = 4.6 Hz, CH2), 22.4 (d, J = 49.9 Hz, CH2P+), 26.1 (d, J = 17.7 Hz, CH2), 34.1 (s, CH2), 34.2 (s, CH2NH), 118.2 (d, J = 86.1 Hz, 3 x ArC), 130.6 (d, J = 12.3 Hz, 6 x ArCH), 133.8 (d, J = 10.0 Hz, 6 x ArCH), 135.1 (d, J = 3.1 Hz, 3 x ArCH), 172.7 (C=0) m/z (ESI+) 390.2 [M-Br]+
HRMS (ESI+) calcd for C25H29NOP [M-Br]+ 390.1981, found 390.1971
70. (Z)-7-((lR,2R,3R,5S)-3,5-Dihydroxy-2-((S,£)-3-hydroxy-5-phenylpent-l- enyl)cyclopentyl)-/V-ethylhept-5-enamide, bimatoprost, 97
(5-(Ethylamino)-5-oxopentyl)triphenylphosphonium bromide 96 (1.37 g, 2.91 mmol, 6 eq.) was added to a flame dried Schlenk flask, under N2, and anhydrous THF (10 ml) added. The resulting suspension was cooled to 0 °C. KOt-Bu (653.0 mg, 5.82 mmol, 12 eq.) was added in one portion and the resulting orange mixture stirred at 0 °C for 40 min. A solution of crude triol 94 (0.485 mmol, 1 eq.) in anhydrous THF (2.5 ml) was added dropwise via syringe. After complete addition the mixture was stirred at r.t. for 1 h. The reaction was quenched with saturated aq. NH4CI (10 ml) and extracted with EtOAc (5 x 10 ml). The combined organic phases were dried (MgS04), filtered, and concentrated to give the crude material as solids. These were triturated with EtOAc (10 ml) and the solids filtered and washed with EtOAc (4 x 10 ml). The filtrate was concentrated under vacuum and purified by column chromatography on silica, eluting with EtOAc/MeOH (97.5:2.5 to 95:5) to give 97 (99.2 mg) as a yellowish oil which was further purified by preparative TLC (EtOAc/MeOH 5%) to give BIMATOPROST97 (82.6 mg, 41% over 2 steps) as a clear, colourless oil. Analytical data consistent with the literature (Zanoni, G. et al., Tetrahedron 66, 7472-7478 (2010); Gutman, A. et al., US 20090163596 (2009)).
max (filmVcm-1 3300 (broad), 2930, 1643, 1550, 1453, 1332, 1293, 1048, 1029, 968, 729, 698
*H NMR (400 MHz; CDCI3) δΗ = 1.09 (t, J = 7.1 Hz, 3H, CH3), 1.42-2.40 (m, 14H, 6 x CH2, 2 x CH), 2.67 (m, 2H, CH2), 3.22 (dq, J = 7.1, 6.3 Hz, 2H, CH2NH), 3.41 (broad s, 3H, 3 x OH), 3.80-4.30 (broad m, 3H, 3 x CHOH), 5.37 (m, 2H, 2 x =CH), 5.47 (dd, J = 15.2, 7.9 Hz, 1H, =CH), 5.59 (dd, J = 15.2, 7.9 Hz, 1H, =CH), 5.90 (broad s, 1H, NH), 7.17 (m, 3H, ArCH’s), 7.26 (m, 2H, ArCH’s)
13C NMR ( 100 MHz; CDCI3) 5C = 14.8 (CH3), 25.4 (CH2), 25.6 (CH2), 26.7 (CH2), 31.9 (CH2), 34.4 (CH2NH), 35.8 (CH2C=0), 38.8 (CH2), 42.9 (CH2), 50.2 (CH), 55.5 (CH), 72.3 (CHOH), 72.4 (CHOH), 77.7 (CHOH), 125.8 (ArCH), 128.4 (2 x ArCH), 128.5 (2 x ArCH), 129.1 (=CH), 129.7 (=CH), 133.7 (=CH), 135.1 (=CH), 142.0 (ArC), 173.4 (C=0)
m/z (ESI+) 438.2 [MNa]+
HRMS (ESI+) calcd for Q^IV^Na [MNa]+ 438.2614, found 438.2615
[a]D 22 +41.1 (c. 0.35, CH2CI2) (lit. – Zanoni, G. et al., Tetrahedron 66, 7472-7478 (2010), +32.7 (c. 0.33, CH2CI2)) (lit. – Gutman, A. et al., US 20090163596 (2009), +36 (c. 1, MeOH))
References
- “Bimatoprost Ophthalmic”. MedlinePlus. January 1, 2003. Archived from the original on 2007-10-05. Retrieved 2007-11-19.
- “Allergan gets FDA approval for eyelash treatment”. BusinessWeek. Associated Press. December 26, 2008. Retrieved December 26, 2008.
- Park J, Cho HK, Moon JI (2011). “Changes to upper eyelid orbital fat from use of topical bimatoprost, travoprost, and latanoprost.”. Japanese Ophthalmological Society 55 (1): 22–27.doi:10.1007/s10384-010-0904-z. PMID 21331688.
- Jayaprakasam A, Ghazi-Nouri S. (2010). “Periorbital fat atrophy – an unfamiliar side effect of prostaglandin analogues.”. Orbit 29 (6): 357–359. doi:10.3109/01676830.2010.527028.PMID 21158579.
- Filippopoulos T, Paula JS, Torun N, Hatton MP, Pasquale LR, Grosskreutz CL. (2008). “Periorbital changes associated with topical bimatoprost.”. Ophthalmology Plastic and Reconstructive Surgery 24 (4): 302–307. doi:10.1097/IOP.0b013e31817d81df. PMID 18645437.
- Rundle, Rhonda L. (2007-11-19). “Drug That Lengthens Eyelashes Sets Off Flutter”. The Wall Street Journal. Retrieved 2007-11-19.
- The Pink Sheet: [1] Lauren Smith December 15, 2008; Volume 70, Number 050,Page[verification needed]
- Federation of American Societies for Experimental Biology Journal “The prostamide-related glaucoma therapy, bimatoprost, offers a novel approach for treating scalp alopecias” Randall et all. October 26, 2012.
- Latisse prescribing information: “Important Safety Information”
- MSNBC: FDA Seizes $2 Million Of Potentially Harmful SJ Eye Product KNTV-TV November 17, 2007[dead link]
- Reuters: “U.S. seizes discontinued eyelash product”. Jim Wolf. November 16, 2007.
- Tappeiner C, Perren B, Iliev ME, Frueh BE, Goldblum D (May 2008). “Orbitale Fettgewebsatrophie bei lokaler Bimatoprost-Therapie – Kann Bimatoprost einen Enophthalmus verursachen?” [Orbital fat atrophy in glaucoma patients treated with topical bimatoprost–can bimatoprost cause enophthalmos?]. Klinische Monatsblätter für Augenheilkunde (in German)225 (5): 443–5. doi:10.1055/s-2008-1027362. PMID 18454393.
- Filippopoulos T, Paula JS, Torun N, Hatton MP, Pasquale LR, Grosskreutz CL (2008). “Periorbital changes associated with topical bimatoprost”. Ophthalmic Plastic and Reconstructive Surgery 24 (4): 302–7. doi:10.1097/IOP.0b013e31817d81df. PMID 18645437.
- Peplinski LS, Albiani Smith K (August 2004). “Deepening of lid sulcus from topical bimatoprost therapy”. Optometry and Vision Science 81 (8): 574–7.doi:10.1097/01.opx.0000141791.16683.4a. PMID 15300114.
- Serrero G, Lepak NM (April 1997). “Prostaglandin F2alpha receptor (FP receptor) agonists are potent adipose differentiation inhibitors for primary culture of adipocyte precursors in defined medium”. Biochemical and Biophysical Research Communications 233 (1): 200–2. doi:10.1006/bbrc.1997.6433. PMID 9144422.
- Curran MP (2009). “Bimatoprost: a review of its use in open-angle glaucoma and ocular hypertension”. Drugs Aging 26 (12): 1049–71. doi:10.2165/11203210-000000000-00000.PMID 19929032.
- “Long Lashes Without Prescription, but With Risks”. Catherine Saint Louis. The New York Times. May 1, 2010
- “Potentially Harmful “Cosmetic” Eye Product Seized” (Press release). U.S. Food and Drug Administration (FDA). November 19, 2007. Retrieved 2007-12-05.
Citations
- Chen M, Cheng C, Chen Y, Chou C, Hsu W (2006). “Effects of bimatoprost 0.03% on ocular hemodynamics in normal tension glaucoma.”. J Ocul Pharmacol Ther 22 (3): 188–93. doi:10.1089/jop.2006.22.188. PMID 16808680.
- Kruse P, Rieck P, Sherif Z, Liekfeld A (2006). “Cystoid macular edema in a pseudophakic patient after several glaucoma procedures. Is local therapy with bimatoprost the reason?”. Klinische Monatsblätter für Augenheilkunde 223 (6): 534–7. doi:10.1055/s-2005-858992. PMID 16804825.
- Steinhäuser S (2006). “Decreased high-density lipoprotein serum levels associated with topical bimatoprost therapy.”. Optometry 77 (4): 177–9.doi:10.1016/j.optm.2006.02.001. PMID 16567279.
- Park J, Cho HK, Moon JI (2011). “Changes to upper eyelid orbital fat from use of topical bimatoprost, travoprost, and latanoprost.”. Japanese Ophthalmological Society 55 (1): 22–27. doi:10.1007/s10384-010-0904-z. PMID 21331688.
- Jayaprakasam A, Ghazi-Nouri S. (2010). “Periorbital fat atrophy – an unfamiliar side effect of prostaglandin analogues.”. Orbit 29 (6): 357–359.doi:10.3109/01676830.2010.527028. PMID 21158579.
- Filippopoulos T, Paula JS, Torun N, Hatton MP, Pasquale LR, Grosskreutz CL. (2008). “Periorbital changes associated with topical bimatoprost.”. Ophthalmology Plastic and Reconstructive Surgery 24 (4): 302–307. doi:10.1097/IOP.0b013e31817d81df. PMID 18645437.
External links
- Medical News Today: FDA Seizes $2 Million Of Cosmetic Eye Product Which Contains Drug Ingredient And Makes Unapproved Drug Claims. Christian Nordqvist. 18 November 2007
- Wired Science: FDA Seizes Cosmetic That Can Blind. Brandon Keim. November 19, 2007
- Eye Drops: [The generic name of the Latisse eye drop is Bimatoprost Ophthalmic Solution 0.03%]. Crazzy Paul. Aug 01, 2013
|
1-25-2012
|
Method of enhancing hair growth
|
|
|
1-25-2012
|
NITRIC OXIDE DONATING PROSTAMIDES
|
|
|
11-25-2011
|
BIMATOPROST CRYSTALLINE FORM I
|
|
|
10-19-2011
|
Method of Enhancing Hair Growth
|
|
|
7-22-2011
|
IMPROVED PROCESS FOR THE PRODUCTION OF BIMATOPROST
|
|
|
6-3-2011
|
Process for the Preparation of Prostaglandin Analogues and Intermediates Thereof
|
|
|
5-27-2011
|
COMPOSITIONS AND METHODS FOR STIMULATING HAIR GROWTH
|
|
|
5-27-2011
|
ENHANCED BIMATOPROST OPHTHALMIC SOLUTION
|
|
|
5-25-2011
|
Bimatoprost crystalline form I
|
|
|
5-13-2011
|
COMPOSITIONS FOR ENHANCING HAIR GROWTH
|
|
3-2-2011
|
Process for the Preparation of Prostaglandin Analogues and Intermediates Thereof
|
|
|
12-24-2010
|
METHOD FOR THE PURIFICATION OF PROSTAGLANDINS
|
|
|
12-15-2010
|
Enhanced bimatoprost ophthalmic solution
|
|
|
9-17-2010
|
Compositions and Methods for Reducing Body Fat
|
|
|
5-28-2010
|
COMPLEXES OF PROSTAGLANDIN DERIVATIVES AND MONOSUBSTITUTED, CHARGED BETA-CYCLODEXTRINS
|
|
|
4-30-2010
|
AMINO ACID SALTS OF PROSTAGLANDINS
|
|
|
4-30-2010
|
AMINO ACID SALTS OF PROSTAGLANDINS
|
|
|
2-24-2010
|
Compositions and methods for reducing body fat
|
|
|
1-27-2010
|
10-HYDROXY-11-DIHYDROPROSTAGLANDIN ANALOGS AS SELECTIVE EP4 AGONISTS
|
|
|
1-15-2010
|
Process for the Production of Prostaglandins and Prostaglandin Analogs
|
|
11-20-2009
|
Process for the production of intermediates for making prostaglandin derivatives such as latanaprost, travaprost, and bimatoprost
|
|
|
11-4-2009
|
Enzymatic transformation of a prostaglandin (bimatoprost) intermediate
|
|
|
10-16-2009
|
Method for preparing prostaglandin F analogue
|
|
|
8-14-2009
|
METHOD OF ENHANCING HAIR GROWTH
|
|
|
6-12-2009
|
Enhanced Bimatoprost Ophthalmic Solution
|
|
|
4-3-2009
|
METHOD FOR SCREENING OF PROSTAGLANDIN COMPOUNDS COMPRISING AN OPTIMAL FORMULATION FOR THE ENHANCEMENT OF HAIR GROWTH AND THE STIMULATION OF FOLLICULAR ANAGEN AND FORMULATIONS RESULTING THEREFROM
|
|
|
10-15-2008
|
5-Thiopiperdinyl prostaglandin e analogs
|
|
|
2-20-2008
|
COMPOSITIONS AND METHODS COMPRISING PROSTAGLANDIN-RELATED COMPOUNDS AND TREFOIL FACTOR FAMILY PEPTIDES FOR THE TREATMENT OF GLAUCOMA WITH REDUCED HYPEREMIA
|
|
|
2-15-2008
|
Novel Prostamides For The Treatment Of Glaucoma And Related Diseases
|
|
|
11-23-2007
|
COMPOSITIONS AND METHODS COMPRISING PROSTAGLANDIN-RELATED COMPOUNDS AND TREFOIL FACTOR FAMILY PEPTIDES FOR THE TREATMENT OF GLAUCOMA WITH REDUCED HYPEREMIA
|
|
7-18-2007
|
Compositions and methods comprising prostaglandin related compounds and trefoil factor family peptides for the treatment of glaucoma with reduced hyperemia
|
|
|
5-18-2007
|
NOVEL PROSTAMIDES FOR THE TREATMENT OF GLAUCOMA AND RELATED DISEASES
|
|
|
4-27-2007
|
Compositions comprising benzo (g) quinoline derivatives and prostaglandin derivatives
|
|
|
3-7-2007
|
Prostamides for the treatment of glaucoma and related diseases
|
|
|
1-31-2007
|
10-Hydroxy-11-dihydroprostaglandin analogs as selective EP4 agonists
|
|
|
1-24-2007
|
Process for the preparation of prostaglandin derivatives
|
|
|
1-10-2007
|
Protected diols for prostaglandin synthesis
|
|
|
1-3-2007
|
Process for the preparation of 17-phenyl-18,19,20-thinor-pgf 2a and its derivatives
|
|
|
11-29-2006
|
Protected and unprotected triols for prostaglandin synthesis
|
|
|
9-20-2006
|
Prostaglandin synthesis
|
|
9-6-2006
|
Cyclopentane heptan(ENE)OIC acid, 2-heteroarylalkenyl derivatives as therapeutic agents
|
|
|
7-5-2006
|
Method for imparting artificial tan to human skin
|
|
|
8-24-2005
|
Inhibition of irritating side effects associated with use of a topical ophthalmic medication
|
|
|
3-18-2005
|
Methods for the treatment of gray hair using cyclopentane(ene) heptan(en)oic acid amides
|
|
|
3-9-2005
|
9,11-cycloendoperoxide pro-drugs of prostaglandin analogues for treatment of ocular hypertension and glaucoma
|
|
|
2-11-2005
|
Compositions for delivery of therapeutics into the eyes and methods for making and using same
|
|
|
5-29-2002
|
Ocular hypotensive lipids
|
| EP0364417A1 | Sep 6, 1989 | Apr 18, 1990 | Pharmacia AB | Prostaglandin derivatives for the treatment of glaucoma or ocular hypertension |
| EP0364417B1 | Sep 6, 1989 | Feb 9, 1994 | Pharmacia AB | Prostaglandin derivatives for the treatment of glaucoma or ocular hypertension |
| EP0544899B1 | Jun 19, 1992 | Sep 6, 1995 | CHINOIN Gyogyszer és Vegyészeti Termékek Gyára RT. | Prostaglandins |
| US5359095 | Feb 8, 1994 | Oct 25, 1994 | Pharmacia Ab | Hydrogenation of doulbe bond in intermediate compound without deoxygenatio of allytic alcohol |
| US6689901 | Jun 25, 2002 | Feb 10, 2004 | Pharmacia & Upjohn Company | Process and intermediates to prepare latanoprost |
| US6927300 | Jan 26, 2001 | Aug 9, 2005 | Finetech Laboratories Ltd | Process for the preparation of Latanoprost |
| US7157590 | May 3, 2002 | Jan 2, 2007 | Finetech Laboratories Ltd. | Chemical intermediate for vision defect drug |
| US7268239 | Jul 27, 2005 | Sep 11, 2007 | Resolution Chemicals Limited | Process for the preparation of prostaglandins and analogues thereof |
| US7642370 | Mar 20, 2007 | Jan 5, 2010 | Daiichi Fine Chemical Co., Ltd. | Method for preparing prostaglandin derivative |
| US7674921 | Feb 25, 2008 | Mar 9, 2010 | Cayman Chemical Company, Inc. | cloprostenol or latanoprost; topical use; highly crystalline structures that are easy to formulate into ophthalmic solutions; hydrolysis of these analogs releases only the active PGF2 alpha analog free acid, without the production of toxic and irritant small aliphatic alcohol coproducts |
| US20090259066 * | Jul 3, 2008 | Oct 15, 2009 | Everlight Usa, Inc. | Method for preparing prostaglandin F analogue |
| US20090287003 | Sep 29, 2006 | Nov 19, 2009 | Jiang Xing Chen | Process for the production of intermediates for making prostaglandin derivatives such as latanaprost, travaprost, and bimatoprost |
| US20100010239 | Jul 10, 2009 | Jan 14, 2010 | Sandoz Ag | Process for the Production of Prostaglandins and Prostaglandin Analogs |
| WO2010097672A1 | Feb 18, 2010 | Sep 2, 2010 | Sifavitor S.R.L. | Process for the preparation of prostaglandin derivatives |
Organic spectroscopy should be brushed up and you get confidence
read my blog
ORGANIC SPECTROSCOPY INTERNATIONAL is the blog
Organic chemists from Industry and academics to interact on Spectroscopy techniques for Organic compounds ie NMR, MASS, IR, UV Etc. email me ……….. amcrasto@gmail.com
http://orgspectroscopyint.blogspot.in/ is the link
 amcrasto@gmail.com
amcrasto@gmail.com
Lumiracoxib…Selective cyclooxygenase-2-(COX-2) inhibitor. Anti-inflammatory.

Lumiracoxib
2-[(2-Chloro-6-fluorophenyl)amino]-5-methylbenzeneacetic Acid;
2-[2-(2-Chloro-6-fluorophenylamino)-5-methylphenyl]acetic Acid;
CGS 35189; COX 189; Prexige;
Applications: Selective cyclooxygenase-2-(COX-2) inhibitor. Anti-inflammatory.
| Systematic (IUPAC) name | |
|---|---|
| {2-[(2-chloro-6-fluorophenyl)amino]-5-methylphenyl} acetic acid |
|
| Clinical data | |
| Trade names | Prexige |
| AHFS/Drugs.com | International Drug Names |
| Pregnancy cat. | C (AU) |
| Legal status | ℞-only Withdrawn (Australia, New Zealand, Canada, UK, Germany,Austria, Belgium, Cyprus, Brazil) |
| Routes | Oral |
| Pharmacokinetic data | |
| Bioavailability | 74-90%[1] |
| Protein binding | >98%[1] |
| Metabolism | Predominantly in the liver viaoxidation and hydroxylation(CYP2C9)[1] |
| Half-life | 5-8 hours[1] |
| Excretion | Urine (54%) and faeces (43%)[1] |
| Identifiers | |
| CAS number | 220991-20-8 |
| ATC code | M01AH06 |
| PubChem | CID 151166 |
| DrugBank | DB01283 |
| ChemSpider | 133236 |
| UNII | |
| PDB ligand ID | LUR (PDBe, RCSB PDB) |
| Chemical data | |
| Formula | C15H13ClFNO2 |
| Mol. mass | 293.72 g/mol |
Lumiracoxib (rINN) is a carboxylic acid COX-2 selective inhibitor non-steroidal anti-inflammatory drug, manufactured by Novartis and still sold in few countries, including Mexico, Ecuador and the Dominican Republic, under the trade name Prexige (sometimes misquoted as “Prestige” by the media).[1]
Lumiracoxib has several distinctive features. Its structure is different from that of other COX-2 inhibitors, such as celecoxib: lumiracoxib is an analogue of diclofenac (one chlorine substituted by fluorine, the phenylacetic acid has another methyl group in meta position), making it a member of the arylalkanoic acid class of NSAIDs; it binds to a different site on the COX-2 enzyme than do other COX-2 inhibitors; it is the only acidic coxib and has the highest COX-2 selectivity of any NSAID.[2]
Since its original approval, lumiracoxib has been withdrawn from the market in several countries, mostly due to its potential for causing liver failure (sometimes requiring liver transplantation). It has never been approved for use in the United States.[1]
History
The TARGET study (Therapeutic Arthritis Research and Gastrointestinal Event Trial) was conducted with more than 18,000 patients to test its gastrointestinal and cardiovascular safety against naproxen and ibuprofen and also study its efficacy against these two NSAIDs.
In November 2006, Prexige received marketing approval for all European Union countries through a common procedure called MRP. However, in August 2007, Prexige was withdrawn from the market in Australia following 8 serious liver adverse events, including 2 deaths and 2 liver transplants.[3] On September 27, 2007, the US Food and Drug Administration issued a not approvable letter for lumiracoxib, requiring additional safety data.[4] Canada withdrew Prexige (approved at 100 mg dose only) in October 2007.[5] Several European Union countries followed suit in November 2007.[6]
The FDA rejected Prexige as a trade name for lumiracoxib in 2003. Prexede was suggested as an alternative, but the FDA Division of Medication Errors and Technical Support (DMETS) subsequently recommended against it as well.[7]
Withdrawal from market
On August 11, 2007, Australia’s Therapeutic Goods Administration (TGA, the national agency responsible for regulation of pharmaceuticals) cancelled the registration of lumiracoxib in Australia due to concerns that it may cause liver failure.[8]
According to the TGA’s Principal Medical Adviser, Dr Rohan Hammett, as of 10 August 2007 the TGA had received 8 reports of serious adverse liver reactions to the drug, including two deaths and two liver transplants.
“The TGA and its expert advisory committee, the Adverse Drug Reactions Advisory Committee (ADRAC), have urgently investigated these reports. ADRAC has today recommended the cancellation of the registration of Lumiracoxib due to the severity of the reported side effects associated with this drug,” Dr Hammett said.
“The TGA has taken this advice to cancel the registration of Lumiracoxib in order to prevent further cases of severe liver damage.
“It seems that the longer people are on the medicine, the greater the chance of liver injury. The TGA is, therefore, advising people to stop taking the Lumiracoxib immediately and to discuss alternative treatments with their doctor,” Dr Hammett said.[9]
New Zealand has followed suit with Australia in recalling Prexige.[10]
On October 3, 2007, Health Canada requested sales of Prexige to stop. Novartis has agreed to the request and has taken steps to do so.[11] On December 13, 2007, the European Medicines Agency recommended the withdrawal for Prexige from all EU markets.[12]
On January 17, 2008, the Philippines Department of Health ordered Novartis Healthcare Phils. Inc. (Novartis) to remove (recall) all lumiracoxib from local drug stores in 2 weeks due to the harmful effects of the drug (potential serious liver-related side effects, hepatotoxicity or malfunction of the lungs).[13]
On July 22, 2008, The Brazilian National Health Surveillance Agency ordered the withdrawal of 100 mg formulations of lumiracoxib and suspended marketing of the 400 mg formulation for 90 days,[14] after a three-year safety review found a marked increase in adverse event reports; 35% of lumiracoxib-associated adverse events reported worldwide between July 2005 and April 2008 were found to have occurred in Brazil.[15] Lumiracoxib was definitively withdrawn from the Brazilian market on October 3, 2008.[16]
On November 12, 2008, INVIMA, the Colombian National Institute for Food and Drug Surveillance ordered the withdrawal of all presentations of lumiracoxib (Prexige), due to the international reports on hepatotoxicity.
MECHANISM

Synthesis







References
- Shi, S; Klotz, U (March 2008). “Clinical use and pharmacological properties of selective COX-2 inhibitors.”. European Journal of Clinical Pharmacology 64 (3): 233–52.doi:10.1007/s00228-007-0400-7. PMID 17999057.
- Tacconelli S, Capone ML, Patrignani P (2004). “Clinical pharmacology of novel selective COX-2 inhibitors”. Curr Pharm Des 10 (6): 589–601. doi:10.2174/1381612043453108.PMID 14965322.
- Urgent medicine recall – Lumiracoxib (PREXIGE)
- http://hugin.info/134323/R/1156327/223186.pdf
- Withdrawal of Market Authorization for Prexige
- Media releases
- http://www.fda.gov/ohrms/dockets/ac/05/briefing/2005-4090B1_33_GG-FDA-Tab-U.pdf
- Medicines Regulator cancels registration of anti inflammatory drug, Lumiracoxib, Therapeutic Goods Administration, 11 August 2007. Retrieved on 2007-08-11
- http://www.tga.gov.au/media/2007/070811-lumiracoxib.htm
- “NZ regulators ban arthritis drug”. The New Zealand Herald. 21 August 2007. Retrieved 12 September 2011.
- http://www.novartis.ca/downloads/en/letters/prexige_fact_20071003_e.pdf
- Press release: European Medicines Agency recommends withdrawal of the marketing authorisations for lumiracoxib-containing medicines, 13 December 2007
- Abs-Cbn Interactive, DOH recalls lumiracoxib, sets two-week deadline
- “Anvisa cancela registro do Prexige; consumidor deve substituir medicamento”. Folha de S. Paulo (in Portuguese). July 22, 2008. Retrieved 2008-07-22.
- “Anvisa cancela registro do antiinflamatório Prexige” (Press release) (in Portuguese). Anvisa. July 22, 2008. Retrieved 2008-07-22.
- “Anvisa suspende venda e uso de 2 antiinflamatórios” (in Portuguese). Terra. October 3, 2008. Retrieved 2008-10-03.
External links
- Prexige
- Forbes
- FDA request more information September 23, 2003
- NPS RADAR
- FDA – Background Document for Lumiracoxib 1/13/2005
- http://www.nzherald.co.nz/category/story.cfm?c_id=278&objectid=10459030

ROFECOXIB

ROFECOXIB
MK-966, MK-0966, Vioxx
162011-90-7
Rofecoxib /ˌrɒfɨˈkɒksɪb/ is a nonsteroidal anti-inflammatory drug (NSAID) that has now been withdrawn over safety concerns. It was marketed by Merck & Co. to treat osteoarthritis, acute pain conditions, and dysmenorrhoea. Rofecoxib was approved by the Food and Drug Administration (FDA) on May 20, 1999, and was marketed under the brand names Vioxx, Ceoxx, and Ceeoxx.
 |
|
 |
|
| Systematic (IUPAC) name | |
|---|---|
| 4-(4-methylsulfonylphenyl)-3-phenyl-5H-furan-2-one | |
| Clinical data | |
| Pregnancy cat. | C (AU) |
| Legal status | Prescription Only (S4) (AU)withdrawn |
| Routes | oral |
| Pharmacokinetic data | |
| Bioavailability | 93% |
| Protein binding | 87% |
| Metabolism | hepatic |
| Half-life | 17 hours |
| Excretion | biliary/renal |
| Identifiers | |
| CAS number | 162011-90-7 |
| ATC code | M01AH02 |
| PubChem | CID 5090 |
| DrugBank | DB00533 |
| ChemSpider | 4911 |
| UNII | 0QTW8Z7MCR |
| Chemical data | |
| Formula | C17H14O4S |
| Mol. mass | 314.357 g/mol |
Rofecoxib gained widespread acceptance among physicians treating patients with arthritis and other conditions causing chronic or acute pain. Worldwide, over 80 million people were prescribed rofecoxib at some time.[1]
On September 30, 2004, Merck withdrew rofecoxib from the market because of concerns about increased risk of heart attack and stroke associated with long-term, high-dosage use. Merck withdrew the drug after disclosures that it withheld information about rofecoxib’s risks from doctors and patients for over five years, resulting in between 88,000 and 140,000 cases of serious heart disease.[2] Rofecoxib was one of the most widely used drugs ever to be withdrawn from the market. In the year before withdrawal, Merck had sales revenue of US$2.5 billion from Vioxx.[3] Merck reserved $970 million to pay for its Vioxx-related legal expenses through 2007, and have set aside $4.85bn for legal claims from US citizens.
Rofecoxib was available on prescription in both tablet-form and as an oral suspension. It was available by injection for hospital use.
Mode of action
Cyclooxygenase (COX) has two well-studied isoforms, called COX-1 and COX-2. COX-1 mediates the synthesis of prostaglandinsresponsible for protection of the stomach lining, while COX-2 mediates the synthesis of prostaglandins responsible for pain and inflammation. By creating “selective” NSAIDs that inhibit COX-2, but not COX-1, the same pain relief as traditional NSAIDs is offered, but with greatly reduced risk of fatal or debilitating peptic ulcers. Rofecoxib is a selective COX-2 inhibitor, or “coxib”.
Others include Merck’s etoricoxib (Arcoxia), Pfizer’s celecoxib (Celebrex) and valdecoxib (Bextra). Interestingly, at the time of its withdrawal, rofecoxib was the only coxib with clinical evidence of its superior gastrointestinal adverse effect profile over conventional NSAIDs. This was largely based on the VIGOR (Vioxx GI Outcomes Research) study, which compared the efficacy and adverse effect profiles of rofecoxib and naproxen.[4]
Pharmacokinetics
The therapeutic recommended dosages were 12.5, 25, and 50 mg with an approximate bioavailability of 93%.[5][6][7] Rofecoxib crossed the placenta and blood–brain barrier,[5][6][8]and took 1–3 hours to reach peak plasma concentration with an effective half-life (based on steady-state levels) of approximately 17 hours.[5][7][9] The metabolic products are cis-dihydro and trans-dihydro derivatives of rofecoxib[5][9] which are primarily excreted through urine.
Fabricated efficacy studies
On March 11, 2009, Scott S. Reuben, former chief of acute pain at Baystate Medical Center, Springfield, Mass., revealed that data for 21 studies he had authored for the efficacy of the drug (along with others such as celecoxib) had been fabricated in order to augment the analgesic effects of the drugs. There is no evidence that Reuben colluded with Merck in falsifying his data. Reuben was also a former paid spokesperson for the drug company Pfizer (which owns the intellectual property rights for marketing celecoxib in the United States). The retracted studies were not submitted to either the FDA or the European Union’s regulatory agencies prior to the drug’s approval. Drug manufacturer Merckhad no comment on the disclosure.[10]
Adverse drug reactions
Aside from the reduced incidence of gastric ulceration, rofecoxib exhibits a similar adverse effect profile to other NSAIDs.
Prostaglandin is a large family of lipids. Prostaglandin I2/PGI2/prostacyclin is just one member of it. Prostaglandins other than PGI2 (such as PGE2) also play important roles in vascular tone regulation. Prostacyclin/thromboxane are produced by both COX-1 and COX-2, and rofecoxib suppresses just COX-2 enzyme, so there is no reason to believe that prostacyclin levels are significantly reduced by the drug. And there is no reason to believe that only the balance between quantities of prostacyclin and thromboxane is the determinant factor for vascular tone.[11] Indeed Merck has stated that there was no effect on prostacyclin production in blood vessels in animal testing.[12] Other researchers have speculated that the cardiotoxicity may be associated with maleic anhydride metabolites formed when rofecoxib becomes ionized under physiological conditions. (Reddy & Corey, 2005)
Adverse cardiovascular events
VIGOR study and publishing controversy
The VIGOR (Vioxx GI Outcomes Research) study, conducted by Bombardier, et al., which compared the efficacy and adverse effect profiles of rofecoxib and naproxen, had indicated a significant 4-fold increased risk of acute myocardial infarction (heart attack) in rofecoxib patients when compared with naproxen patients (0.4% vs 0.1%, RR 0.25) over the 12 month span of the study. The elevated risk began during the second month on rofecoxib. There was no significant difference in the mortality from cardiovascular events between the two groups, nor was there any significant difference in the rate of myocardial infarction between the rofecoxib and naproxen treatment groups in patients without high cardiovascular risk. The difference in overall risk was by the patients at higher risk of heart attack, i.e. those meeting the criteria for low-dose aspirin prophylaxis of secondary cardiovascular events (previous myocardial infarction, angina, cerebrovascular accident, transient ischemic attack, or coronary artery bypass).
Merck’s scientists interpreted the finding as a protective effect of naproxen, telling the FDA that the difference in heart attacks “is primarily due to” this protective effect (Targum, 2001). Some commentators have noted that naproxen would have to be three times as effective as aspirin to account for all of the difference (Michaels 2005), and some outside scientists warned Merck that this claim was implausible before VIGOR was published.[13] No evidence has since emerged for such a large cardioprotective effect of naproxen, although a number of studies have found protective effects similar in size to those of aspirin.[14][15] Though Dr. Topol’s 2004 paper criticized Merck’s naproxen hypothesis, he himself co-authored a 2001 JAMA article stating “because of the evidence for an antiplatelet effect of naproxen, it is difficult to assess whether the difference in cardiovascular event rates in VIGOR was due to a benefit from naproxen or to a prothrombotic effect from rofecoxib.” (Mukherjee, Nissen and Topol, 2001.)
The results of the VIGOR study were submitted to the United States Food and Drug Administration (FDA) in February 2001. In September 2001, the FDA sent a warning letter to the CEO of Merck, stating, “Your promotional campaign discounts the fact that in the VIGOR study, patients on Vioxx were observed to have a four to five fold increase in myocardial infarctions (MIs) compared to patients on the comparator non-steroidal anti-inflammatory drug (NSAID), Naprosyn (naproxen).”[16] This led to the introduction, in April 2002, of warnings on Vioxx labeling concerning the increased risk of cardiovascular events (heart attack and stroke).
Months after the preliminary version of VIGOR was published in the New England Journal of Medicine, the journal editors learned that certain data reported to the FDA were not included in the NEJM article. Several years later, when they were shown a Merck memo during the depositions for the first federal Vioxx trial, they realized that these data had been available to the authors months before publication. The editors wrote an editorial accusing the authors of deliberately withholding the data.[17] They released the editorial to the media on December 8, 2005, before giving the authors a chance to respond. NEJM editor Gregory Curfman explained that the quick release was due to the imminent presentation of his deposition testimony, which he feared would be misinterpreted in the media. He had earlier denied any relationship between the timing of the editorial and the trial. Although his testimony was not actually used in the December trial, Curfman had testified well before the publication of the editorial.[18]
The editors charged that “more than four months before the article was published, at least two of its authors were aware of critical data on an array of adverse cardiovascular events that were not included in the VIGOR article.” These additional data included three additional heart attacks, and raised the relative risk of Vioxx from 4.25-fold to 5-fold. All the additional heart attacks occurred in the group at low risk of heart attack (the “aspirin not indicated” group) and the editors noted that the omission “resulted in the misleading conclusion that there was a difference in the risk of myocardial infarction between the aspirin indicated and aspirin not indicated groups.” The relative risk for myocardial infarctions among the aspirin not indicated patients increased from 2.25 to 3 (although it remained statitistically insignificant). The editors also noted a statistically significant (2-fold) increase in risk for serious thromboembolic events for this group, an outcome that Merck had not reported in the NEJM, though it had disclosed that information publicly in March 2000, eight months before publication.[19]
The authors of the study, including the non-Merck authors, responded by claiming that the three additional heart attacks had occurred after the prespecified cutoff date for data collection and thus were appropriately not included. (Utilizing the prespecified cutoff date also meant that an additional stroke in the naproxen population was not reported.) Furthermore, they said that the additional data did not qualitatively change any of the conclusions of the study, and the results of the full analyses were disclosed to the FDA and reflected on the Vioxx warning label. They further noted that all of the data in the “omitted” table were printed in the text of the article. The authors stood by the original article.[20]
NEJM stood by its editorial, noting that the cutoff date was never mentioned in the article, nor did the authors report that the cutoff for cardiovascular adverse events was before that for gastrointestinal adverse events. The different cutoffs increased the reported benefits of Vioxx (reduced stomach problems) relative to the risks (increased heart attacks).[19]
Some scientists have accused the NEJM editorial board of making unfounded accusations.[21][22] Others have applauded the editorial. Renowned research cardiologist Eric Topol,[23] a prominent Merck critic, accused Merck of “manipulation of data” and said “I think now the scientific misconduct trial is really fully backed up”.[24] Phil Fontanarosa, executive editor of the prestigious Journal of the American Medical Association, welcomed the editorial, saying “this is another in the long list of recent examples that have generated real concerns about trust and confidence in industry-sponsored studies”.[25]
On May 15, 2006, the Wall Street Journal reported that a late night email, written by an outside public relations specialist and sent to Journal staffers hours before the Expression of Concern was released, predicted that “the rebuke would divert attention to Merck and induce the media to ignore the New England Journal of Medicine‘s own role in aiding Vioxx sales.”[26]
“Internal emails show the New England Journal’s expression of concern was timed to divert attention from a deposition in which Executive Editor Gregory Curfman made potentially damaging admissions about the journal’s handling of the Vioxx study. In the deposition, part of the Vioxx litigation, Dr. Curfman acknowledged that lax editing might have helped the authors make misleading claims in the article.” The Journal stated that NEJM‘s “ambiguous” language misled reporters into incorrectly believing that Merck had deleted data regarding the three additional heart attacks, rather than a blank table that contained no statistical information; “the New England Journal says it didn’t attempt to have these mistakes corrected.”[26]
Alzheimer’s studies
In 2000 and 2001, Merck conducted several studies of rofecoxib aimed at determining if the drug slowed the onset of Alzheimer’s disease. Merck has placed great emphasis on these studies on the grounds that they are relatively large (almost 3000 patients) and compared rofecoxib to a placebo rather than to another pain reliever. These studies found an elevated death rate among rofecoxib patients, although the deaths were not generally heart-related. However, they did not find any elevated cardiovascular risk due to rofecoxib.[27] Before 2004, Merck cited these studies as providing evidence, contrary to VIGOR, of rofecoxib’s safety.
APPROVe study
In 2001, Merck commenced the APPROVe (Adenomatous Polyp PRevention On Vioxx) study, a three-year trial with the primary aim of evaluating the efficacy of rofecoxib for theprophylaxis of colorectal polyps. Celecoxib had already been approved for this indication, and it was hoped to add this to the indications for rofecoxib as well. An additional aim of the study was to further evaluate the cardiovascular safety of rofecoxib.
The APPROVe study was terminated early when the preliminary data from the study showed an increased relative risk of adverse thrombotic cardiovascular events (includingheart attack and stroke), beginning after 18 months of rofecoxib therapy. In patients taking rofecoxib, versus placebo, the relative risk of these events was 1.92 (rofecoxib 1.50 events vs placebo 0.78 events per 100 patient years). The results from the first 18 months of the APPROVe study did not show an increased relative risk of adverse cardiovascular events. Moreover, overall and cardiovascular mortality rates were similar between the rofecoxib and placebo populations.[28]
In summary, the APPROVe study suggested that long-term use of rofecoxib resulted in nearly twice the risk of suffering a heart attack or stroke compared to patients receiving a placebo.
Other studies
Pre-approval Phase III clinical trials, like the APPROVe study, showed no increased relative risk of adverse cardiovascular events for the first eighteen months of rofecoxib usage (Merck, 2004). Others have pointed out that “study 090,” a pre-approval trial, showed a 3-fold increase in cardiovascular events compared to placebo, a 7-fold increase compared to nabumetone (another [NSAID]), and an 8-fold increase in heart attacks and strokes combined compared to both control groups.[29][30] Although this was a relatively small study and only the last result was statistically significant, critics have charged that this early finding should have prompted Merck to quickly conduct larger studies of rofecoxib’s cardiovascular safety. Merck notes that it had already begun VIGOR at the time Study 090 was completed. Although VIGOR was primarily designed to demonstrate new uses for rofecoxib, it also collected data on adverse cardiovascular outcomes.
Several very large observational studies have also found elevated risk of heart attack from rofecoxib. For example, a recent retrospective study of 113,000 elderly Canadians suggested a borderline statistically significant increased relative risk of heart attacks of 1.24 from Vioxx usage, with a relative risk of 1.73 for higher-dose Vioxx usage. (Levesque, 2005). Another study, using Kaiser Permanente data, found a 1.47 relative risk for low-dose Vioxx usage and 3.58 for high-dose Vioxx usage compared to current use of celecoxib, though the smaller number was not statistically significant, and relative risk compared to other populations was not statistically significant. (Graham, 2005).
Furthermore, a more recent meta-study of 114 randomized trials with a total of 116,000+ participants, published in JAMA, showed that Vioxx uniquely increased risk of renal (kidney) disease, and heart arrhythmia.[31]
Other COX-2 inhibitors
Any increased risk of renal and arrhythmia pathologies associated with the class of COX-2 inhibitors, e.g. celecoxib (Celebrex), valdecoxib (Bextra), parecoxib (Dynastat),lumiracoxib, and etoricoxib is not evident,[31] although smaller studies[32][33] had demonstrated such effects earlier with the use of celecoxib, valdecoxib and parecoxib.
Nevertheless, it is likely that trials of newer drugs in the category will be extended in order to supply additional evidence of cardiovascular safety. Examples are some more specific COX-2 inhibitors, including etoricoxib (Arcoxia) and lumiracoxib (Prexige), which are currently (circa 2005) undergoing Phase III/IV clinical trials.
Besides, regulatory authorities worldwide now require warnings about cardiovascular risk of COX-2 inhibitors still on the market. For example, in 2005, EU regulators required the following changes to the product information and/or packaging of all COX-2 inhibitors:[34]
- Contraindications stating that COX-2 inhibitors must not be used in patients with established ischaemic heart disease and/or cerebrovascular disease (stroke), and also in patients with peripheral arterial disease
- Reinforced warnings to healthcare professionals to exercise caution when prescribing COX-2 inhibitors to patients with risk factors for heart disease, such as hypertension, hyperlipidaemia (high cholesterol levels), diabetes and smoking
- Given the association between cardiovascular risk and exposure to COX-2 inhibitors, doctors are advised to use the lowest effective dose for the shortest possible duration of treatment
Other NSAIDs
Since the withdrawal of Vioxx it has come to light that there may be negative cardiovascular effects with not only other COX-2 inhibitiors, but even the majority of other NSAIDs. It is only with the recent development of drugs like Vioxx that drug companies have carried out the kind of well executed trials that could establish such effects and these sort of trials have never been carried out in older “trusted” NSAIDs such as ibuprofen, diclofenac and others. The possible exceptions may be aspirin and naproxen due to their anti-platelet aggregation properties.
Withdrawal
Due to the findings of its own APPROVe study, Merck publicly announced its voluntary withdrawal of the drug from the market worldwide on September 30, 2004.[35]
In addition to its own studies, on September 23, 2004 Merck apparently received information about new research by the FDA that supported previous findings of increased risk of heart attack among rofecoxib users (Grassley, 2004). FDA analysts estimated that Vioxx caused between 88,000 and 139,000 heart attacks, 30 to 40 percent of which were probably fatal, in the five years the drug was on the market.[36]
On November 5, the medical journal The Lancet published a meta-analysis of the available studies on the safety of rofecoxib (Jüni et al., 2004). The authors concluded that, owing to the known cardiovascular risk, rofecoxib should have been withdrawn several years earlier. The Lancet published an editorial which condemned both Merck and the FDA for the continued availability of rofecoxib from 2000 until the recall. Merck responded by issuing a rebuttal of the Jüni et al. meta-analysis that noted that Jüni omitted several studies that showed no increased cardiovascular risk. (Merck & Co., 2004).
In 2005, advisory panels in both the U.S. and Canada encouraged the return of rofecoxib to the market, stating that rofecoxib’s benefits outweighed the risks for some patients. The FDA advisory panel voted 17-15 to allow the drug to return to the market despite being found to increase heart risk. The vote in Canada was 12-1, and the Canadian panel noted that the cardiovascular risks from rofecoxib seemed to be no worse than those from ibuprofen—though the panel recommended that further study was needed for all NSAIDs to fully understand their risk profiles. Notwithstanding these recommendations, Merck has not returned rofecoxib to the market.[37]
In 2005, Merck retained Debevoise & Plimpton LLP to investigate Vioxx study results and communications conducted by Merck. Through the report, it was found that Merck’s senior management acted in good faith, and that the confusion over the clinical safety of Vioxx was due to the sales team’s overzealous behavior. The report that was filed gave a timeline of the events surrounding Vioxx and showed that Merck intended to operate honestly throughout the process. Any mistakes that were made regarding the mishandling of clinical trial results and withholding of information was the result of oversight, not malicious behavior. The Martin Report did conclude that the Merck’s marketing team exaggerated the safety of Vioxx and replaced truthful information with sales tactics.[citation needed] The report was published in February 2006, and Merck was satisfied with the findings of the report and promised to consider the recommendations contained in the Martin Report. Advisers to the US Food and Drug Administration (FDA) have voted, by a narrow margin, that it should not ban Vioxx — the painkiller withdrawn by drug-maker Merck.
They also said that Pfizer’s Celebrex and Bextra, two other members of the family of painkillers known as COX-2 inhibitors, should remain available, despite the fact that they too boost patients’ risk of heart attack and stroke. url = http://www.nature.com/drugdisc/news/articles/433790b.html The recommendations of the arthritis and drug safety advisory panel offer some measure of relief to the pharmaceutical industry, which has faced a barrage of criticism for its promotion of the painkillers. But the advice of the panel, which met near Washington DC over 16–18 February, comes with several strings attached.
For example, most panel members said that manufacturers should be required to add a prominent warning about the drugs’ risks to their labels; to stop direct-to-consumer advertising of the drugs; and to include detailed, written risk information with each prescription. The panel also unanimously stated that all three painkillers “significantly increase the risk of cardiovascular events”.
The panel voted 17 to 15 against banning Vioxx (rofecoxib) entirely; the vote on Bextra (valdecoxib) was 17 to 13 with 2 abstentions; Celebrex (celecoxib) was endorsed 31 to 1. Shares of Merck, based in Whitehouse Station, New Jersey, and New York-based Pfizer closed up 13% and 7% respectively on 18 February, 2013, the day of the votes.
The FDA is expected to act on the recommendations within weeks. Although the agency usually follows the recommendations of its outside advisers, it is not bound to do so. A top official said that, in light of the closeness of some of the votes, the agency will examine the panel members’ comments in detail before deciding what to do.
An official from Merck said during the meeting that it would consider reintroducing Vioxx, which it withdrew in September 2004. On April 7, 2005, Pfizer withdrew Bextra from the U.S. market on recommendation by the FDA. Pfizer’s other painkiller, Celebrex, is still on the market.
Litigation
As of March 2006, there had been over 10,000 cases and 190 class actions filed against Merck[citation needed] over adverse cardiovascular events associated with rofecoxib and the adequacy of Merck’s warnings. The first wrongful death trial, Rogers v. Merck, was scheduled in Alabama in the spring of 2005, but was postponed after Merck argued that the plaintiff had falsified evidence of rofecoxib use.[1]
On August 19, 2005, a jury in Texas voted 10-2 to hold Merck liable for the death of Robert Ernst, a 59-year-old man who allegedly died of a rofecoxib-induced heart attack. The plaintiffs’ lead attorney was Mark Lanier. Merck argued that the death was due to cardiac arrhythmia, which had not been shown to be associated with rofecoxib use. The jury awarded Carol Ernst, widow of Robert Ernst, $253.4 million in damages. This award will almost certainly be capped at no more than US$26.1 million because of punitive damages limits under Texas law.[2] As of March 2006, the plaintiff had yet to ask the court to enter a judgment on the verdict; Merck has stated that it will appeal.
On November 3, 2005, Merck won the second case Humeston v. Merck, a personal injury case, in Atlantic City, New Jersey. The plaintiff experienced a mild myocardial infarction and claimed that rofecoxib was responsible, after having taken it for two months. Merck argued that there was no evidence that rofecoxib was the cause of Humeston’s injury and that there is no scientific evidence linking rofecoxib to cardiac events with short durations of use. The jury ruled that Merck had adequately warned doctors and patients of the drug’s risk.[3]
The first federal trial on rofecoxib, Plunkett v. Merck, began on November 29, 2005 in Houston. The trial ended in a hung jury and a mistrial was declared on December 12, 2005. According to the Wall Street Journal, the jury hung by an eight to one majority, favoring the defense. Upon retrial in February 2006 in New Orleans, where the Vioxx multidistrict litigation (MDL) is based, a jury found Merck not liable, even though the plaintiffs had the NEJM editor testify as to his objections to the VIGOR study.
On January 30, 2006, a New Jersey state court dismissed a case brought by Edgar Lee Boyd, who blamed Vioxx for gastrointestinal bleeding that he experienced after taking the drug. The judge said that Boyd failed to prove the drug caused his stomach pain and internal bleeding.
In January 2006, Garza v. Merck began trial in Rio Grande City, Texas. The plaintiff, a 71-year-old smoker with heart disease, had a fatal heart attack three weeks after finishing a one-week sample of rofecoxib. On April 21, 2006 the jury awarded the plaintiff $7 million compensatory and $25 million punitive. The Texas state court of appeals in San Antonio later rules Garza’s fatal heart attack probably resulted from pre-existing health conditions unrelated to his taking of Vioxx, thus reversing the $32 million jury award.[4]
On April 5, 2006, the jury held Merck liable for the heart attack of 77-year-old John McDarby, and awarded Mr McDarby $4.5 million in compensatory damages based on Merck’s failure to properly warn of Vioxx safety risks. After a hearing on April 11, 2006, the jury also awarded Mr McDarby an additional $9 million in punitive damages. The same jury found Merck not liable for the heart attack of 60-year-old Thomas Cona, a second plaintiff in the trial, but was liable for fraud in the sale of the drug to Cona.
Merck has reserved $970 million to pay for its Vioxx-related legal expenses through 2007, and have set aside $4.85bn for legal claims from US citizens. Patients who claim to have suffered as a result of taking Vioxx in countries outside the US are campaigning for this to be extended.
In March 2010, an Australian class-action lawsuit against Merck ruled that Vioxx doubled the risk of heart attacks, and that Merck had breached the Trade Practices Act by selling a drug which was unfit for sale.[38]
In November 2011, Merck announced a civil settlement with the US Attorney’s Office for the District of Massachusetts, and individually with 43 US states and the District of Columbia, to resolve civil claims relating to Vioxx.[5] Under the terms of the settlement, Merck agreed to pay two-thirds of a previously recorded $950 million reserve charge in exchange for release from civil liability. Litigation with seven additional states remains outstanding. Under separate criminal proceedings, Merck plead guilty to a federal misdemeanor charge relating to the marketing of the drug across state lines, incurring a fine of $321.6 million.[6]
Other effects
Rofecoxib was shown to improve premenstrual acne vulgaris in a placebo controlled study.[39]
Synthesis
Rofecoxib synthesis.[40]
,,,,,,,,,,,,,,,,,
The oxidation of 4- (methylsulfanyl) acetophenone (X) with monoperoxyphthalic acid (MMPP) in dichloro-methane / methanol gives the corresponding sulfone (XI), which is brominated with Br2 / AlCl3 in chloroform, yielding the expected phenacyl bromide ( XII). Finally, this compound is cyclocondensed with phenylacetic acid (I) by means of 1,8-diazabicyclo [5.4.0] undec-7-ene (DBU) and triethylamine in acetonitrile. 5) Reaction of [4- (methylsulfonyl ) phenyl] phenylacetyl-ene (XIII) with CO catalyzed by Rh4 (CO) 12 in THF at 100 C in a stainless steel autoclave at 100 Atm pressure, followed by a chromatographic separation in a silicagel column to eliminate the undesired regioisomer.

……………….
The synthesis of rofecoxib can be performed by several different ways: 1) The condensation of phenylacetic acid (I) with ethyl bromoacetate (II) by means of triethylamine in THF yields 2- (phenylacetoxy) acetic acid ethyl ester (III), which is cyclized to the hydroxyfuranone (IV) by means of potassium tert-butoxide in tert-butanol. The reaction of (IV) with triflic anhydride and diisopropylethylamine in dichloro-methane affords the corresponding triflate (V), which by reaction with LiBr in hot acetone yields the bromofuranone (VI) The condensation of (VI) with 4- (methylsulfanyl) phenylboronic acid (VII) by means of Na2CO3 and Pd (Ph3P) 4 in hot toluene gives 4- [4- (methylsulfanyl) -phenyl]. – 3-phenylfuran-2 (5H) -one (VIII), which is finally oxidized with 2KHSO5.KHSO4.K2SO4 (oxone). 2) The intermediate (VIII) can also be obtained by condensation of triflate (V) with boronic acid ( VII) by means of Na2CO3 and Pd (Ph3P) 4 in hot toluene. 3) The intermediate (VIII) can also be synthesized by the reaction of triflate (V) with tetramethylammonium chloride, giving the chlorofuranone (IX), which is then condensed with boronic acid (VII) as before.

Footnotes
- Jump up^ http://www.npr.org/templates/story/story.php?storyId=4054991
- Jump up^ “Up to 140,000 heart attacks linked to Vioxx.”. New Scientist. 2005-01-25. p. 1.
- Jump up^ “Merck Sees Slightly Higher 2007 Earnings”. New York Times. Reuters. 2006-12-07. p. A1.
- Jump up^ Bombardier, C.; Laine, L.; Reicin, A.; Shapiro, D.; Burgos-Vargas, R.; Davis, B.; Day, R.; Ferraz, M. B.; Hawkey, C. J.; Hochberg, M. C.; Kvien, T. K.; Schnitzer, T. J.; Vigor Study, G. (2000). “Comparison of Upper Gastrointestinal Toxicity of Rofecoxib and Naproxen in Patients with Rheumatoid Arthritis”. New England Journal of Medicine 343 (21): 1520–1528, 2 1528 following 1528. doi:10.1056/NEJM200011233432103. PMID 11087881. edit
- ^ Jump up to:a b c d Merck & Co. VIOXX (rofecoxib tablets and oral suspension). Accessed at: http://www.merck.com/product/usa/pi_circulars/v/vioxx/vioxx_pi.pdf 01 Feb 2010
- ^ Jump up to:a b Gold Standard Inc. Rofecoxib Vioxx Accessed at: http://www.mdconsult.com/das/pharm/body/181267313-3/946823742/full/2399 01 Feb 2010
- ^ Jump up to:a b Davies, N. M.; Teng, X. W.; Skjodt, N. M. (2003). “Pharmacokinetics of rofecoxib: a specific cyclo-oxygenase-2 inhibitor”. Clinical pharmacokinetics 42 (6): 545–556.PMID 12793839. edit
- Jump up^ Padi, S.; Kulkarni, S. (2004). “Differential effects of naproxen and rofecoxib on the development of hypersensitivity following nerve injury in rats”. Pharmacology, Biochemistry, and Behavior 79 (2): 349–358. doi:10.1016/j.pbb.2004.08.005. PMID 15501312. edit
- ^ Jump up to:a b Scott, L. J.; Lamb, H. M. (1999). “Rofecoxib”. Drugs 58 (3): 499–505; discussion 506–7. doi:10.2165/00003495-199958030-00016. PMID 10493277. edit
- Jump up^ Winstein, Keith J. (March 11, 2009). “Top Pain Scientist Fabricated Data in Studies, Hospital Says”. The Wall Street Journal.
- Jump up^ Vane, J.; Bakhle, Y.; Botting, R. (1998). “Cyclooxygenases 1 and 2”. Annual review of pharmacology and toxicology 38: 97–120. doi:10.1146/annurev.pharmtox.38.1.97.PMID 9597150. edit
- Jump up^ sfgate.com
- Jump up^ www.saferdrugsnow.org
- Jump up^ Karha, J.; Topol, E. J. (2004). “The sad story of Vioxx, and what we should learn from it”. Cleveland Clinic journal of medicine 71 (12): 933–934, 936, 934–9.doi:10.3949/ccjm.71.12.933. PMID 15641522. edit
- Jump up^ Solomon, D. H.; Glynn, R. J.; Levin, R.; Avorn, J. (2002). “Nonsteroidal anti-inflammatory drug use and acute myocardial infarction”. Archives of Internal Medicine 162 (10): 1099–1104.doi:10.1001/archinte.162.10.1099. PMID 12020178. edit
- Jump up^http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/EnforcementActivitiesbyFDA/WarningLettersandNoticeofViolationLetterstoPharmaceuticalCompanies/UCM166383.pdf
- Jump up^ Curfman, G.; Morrissey, S.; Drazen, J. (2005). “Expression of concern: Bombardier et al., “Comparison of upper gastrointestinal toxicity of rofecoxib and naproxen in patients with rheumatoid arthritis,” N Engl J Med 2000;343:1520-8″. The New England Journal of Medicine 353 (26): 2813–2814. doi:10.1056/NEJMe058314. PMID 16339408. edit
- Jump up^ http://www.forbes.com/work/feeds/ap/2006/02/13/ap2523250.html. Missing or empty
|title=(help)[dead link] - ^ Jump up to:a b Curfman, G.; Morrissey, S.; Drazen, J. (2006). “Expression of concern reaffirmed”. The New England Journal of Medicine 354 (11): 1193. doi:10.1056/NEJMe068054.PMID 16495386. edit
- Jump up^ Bombardier, C.; Laine, L.; Burgos-Vargas, R.; Davis, B.; Day, R.; Ferraz, M.; Hawkey, C.; Hochberg, M.; Kvien, T.; Schnitzer, T. J.; Weaver, A. (2006). “Response to expression of concern regarding VIGOR study”. The New England Journal of Medicine 354 (11): 1196–1199. doi:10.1056/NEJMc066096. PMID 16495387. edit
- Jump up^ http://pipeline.corante.com/archives/2006/02/22/nejm_vs_its_contributors_round_two.php
- Jump up^ http://dimer.tamu.edu/simplog/archive.php?blogid=3&pid=3293
- Jump up^ http://genetics.case.edu/faculty2.php?fac=ejt9
- Jump up^ http://www.medicinenet.com/script/main/art.asp?articlekey=56384&page=2
- Jump up^ http://www.beasleyallen.com/news/vioxx-plaintiffs-seek-mistrial-after-allegation-on-merck-study/
- ^ Jump up to:a b David Armstrong (2006-05-15). “How the New England Journal Missed Warning Signs on Vioxx”. Wall Street Journal. p. A1.
- Jump up^ Konstam, M. A.; Weir, M. R.; Reicin, A.; Shapiro, D.; Sperling, R. S.; Barr, E.; Gertz, B. J. (2001). “Cardiovascular thrombotic events in controlled, clinical trials of rofecoxib”. Circulation104 (19): 2280–2288. doi:10.1161/hc4401.100078. PMID 11696466. edit
- Jump up^ Bresalier, R.; Sandler, R.; Quan, H.; Bolognese, J.; Oxenius, B.; Horgan, K.; Lines, C.; Riddell, R.; Morton, D.; Lanas, A.; Konstam, M. A.; Baron, J. A.; Adenomatous Polyp Prevention on Vioxx (APPROVe) Trial Investigators (2005). “Cardiovascular events associated with rofecoxib in a colorectal adenoma chemoprevention trial”. The New England Journal of Medicine 352(11): 1092–1102. doi:10.1056/NEJMoa050493. PMID 15713943. edit
- Jump up^ http://www.fda.gov/ohrms/dockets/ac/01/briefing/3677b2_06_cardio.pdf
- Jump up^ Wolfe, M. M. (2004). “Rofecoxib, Merck, and the FDA”. The New England Journal of Medicine 351 (27): 2875–2878; author 2878 2875–2878. doi:10.1056/NEJM200412303512719.PMID 15625749. edit
- ^ Jump up to:a b Zhang, J.; Ding, E.; Song, Y. (2006). “Adverse effects of cyclooxygenase 2 inhibitors on renal and arrhythmia events: meta-analysis of randomized trials”. Journal of the American Medical Association 296 (13): 1619–1632. doi:10.1001/jama.296.13.jrv60015. PMID 16968832. edit
- Jump up^ Solomon, S.; McMurray, J.; Pfeffer, M.; Wittes, J.; Fowler, R.; Finn, P.; Anderson, W.; Zauber, A.; Hawk, E.; Bertagnolli, M.; Adenoma Prevention with Celecoxib (APC) Study Investigators (2005). “Cardiovascular risk associated with celecoxib in a clinical trial for colorectal adenoma prevention”. The New England Journal of Medicine 352 (11): 1071–1080.doi:10.1056/NEJMoa050405. PMID 15713944. edit
- Jump up^ Nussmeier, N.; Whelton, A.; Brown, M.; Langford, R.; Hoeft, A.; Parlow, J.; Boyce, S.; Verburg, K. (2005). “Complications of the COX-2 inhibitors parecoxib and valdecoxib after cardiac surgery”. The New England Journal of Medicine 352 (11): 1081–1091. doi:10.1056/NEJMoa050330. PMID 15713945. edit
- Jump up^ “European Medicines Agency concludes action on COX-2 inhibitors” (pdf). European Medicines Agency. Retrieved 2008-04-16.
- Jump up^ “Merck Announces Voluntary Worldwide Withdrawal of VIOXX” (pdf). Retrieved 2008-04-16.
- Jump up^ “Congress Questions Vioxx, FDA”. PBS NewsHour. 2004-11-18. Retrieved 2013-06-03.
- Jump up^ “SUMMARY: Report of the Expert Advisory Panel on the Safety of Cox-2 Selective Non-steroidal Anti-Inflammatory Drugs (NSAIDs)”. Health Canada. 2005-07-06. Retrieved 2011-06-04.
- Jump up^ Drug unfit for sale, says judge in compo case The Age, March 6, 2010
- Jump up^ http://bioline.utsc.utoronto.ca/archive/00002693/01/dv04120.pdf#search=%22acne%20rofecoxib%22
- Jump up^ http://vioxxlawyer.org/rofecoxib-synthesis/
References
- FDA (2005). “Summary minutes for the February 16, 17 and 18, 2005, Joint meeting of the Arthritis Advisory Committee and the Drug Safety and Risk Management Advisory Committee.” Published on the internet, March 2005. Link
- Fitzgerald GA, Coxibs and Cardiovascular Disease, N Engl J Med 2004;351(17): 1709–1711. PMID 15470192.
- Grassley CE (15 Oct 2004). Grassley questions Merck about communication with the FDA on Vioxx. Press Release.
- Jüni P, Nartey L, Reichenbach S, Sterchi R, Dieppe PA, Egger M (2004). Risk of cardiovascular events and rofecoxib: cumulative meta-analysis. Lancet (published online; see also Merck response below)
- Karha J and Topol EJ. The sad story of Vioxx, and what we should learn from it Cleve Clin J Med 2004; 71(12):933-939. PMID 15641522
- Michaels, D. (June 2005) DOUBT Is Their Product, Scientific American, 292 (6).
- Merck & Co., (5 Nov 2004). Response to Article by Juni et al. Published in The Lancet on Nov. 5. Press Release.
- Merck & Co (30 Sep 2004) Merck Announces Voluntary Worldwide Withdrawal of VIOXX. Press release [7].
- D. M. Mukherjee, S. E. Nissen, and E. J. Topol, “Risk of Cardiovascular Events Associated with Selective COX-2 Inhibitors,” Journal of the American Medical Association 186 (2001): 954–959.
- Nussmeier NA, Whelton AA, Brown MT, Langford RM, Hoeft A, Parlow JL, et al. Complications of the COX-2 inhibitors parecoxib and valdecoxib after cardiac surgery. N Engl J Med 2005;352(11):1081-91. PMID 15713945
- Okie, S (2005) “Raising the safety bar–the FDA’s coxib meeting.” N Engl J Med. 2005 Mar 31;352(13):1283-5. PMID 15800221.
- Leleti Rajender Reddy, Corey EJ. Facile air oxidation of the conjugate base of rofecoxib (Vioxx), a possible contributor to chronic human toxicity Tetrahedron Lett 2005, 46: 927. doi:10.1016/j.tetlet.2004.12.055
- Swan SK et al., Effect of Cyclooxygenase-2 Inhibition on Renal Function in Elderly Persons Receiving a Low-Salt Diet. Annals of Int Med 2000; 133:1–9
- Targum, SL. (1 Feb. 2001) Review of cardiovascular safety database. FDA memorandum. [8]
- Wolfe, MM et al., Gastrointestinal Toxicity of Nonsteroidal Anti-anflamattory Drugs, New England Journal of Medicine. 1999; 340; 1888-98.
External links
- National Public Radio 2004 Q&A on the case, following withdrawal announcement
- Court TV’s full coverage of the Vioxx civil trials
- Merck website on Vioxx litigation
- FDA Public Health Advisory on Vioxx
- David Michaels. Doubt is Their Product Scientific American, June 2004, p. 96-101
- JURIST, Much Pain, Much Gain: Skeptical Ruminations on the Vioxx Litigation
- Ted Frank, American Enterprise Institute, The Vioxx Litigation, Part I and Part II, December 2005
- briandeer.com – Vioxx: the UK connection
- Campaign for compensation for Vioxx victims outside the US

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....