Home » Uncategorized (Page 104)
Category Archives: Uncategorized
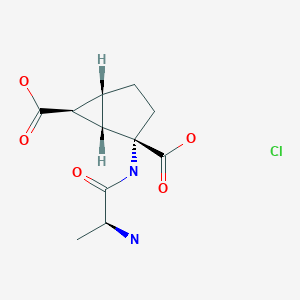
LEXANOPADOL, For Treatment of acute and chronic pain requiring opioid analgesia
LEXANOPADOL
trans-6′-Fluoro-N-methyl-4-phenyl-4′,9′-dihydro-3’H-spiro(cyclohexane-1,1′-pyrano(3,4-b)indol)-4-amine
PRONUNCIATION lex” an oh’ pa dol
THERAPEUTIC CLAIM Treatment of acute and chronic pain requiring opioid analgesia
CHEMICAL NAMES
1. Spiro[cyclohexane-1,1′(3’H)-pyrano[3,4-b]indol]-4-amine, 6′-fluoro-4′,9′-dihydro-N-methyl-4-phenyl-, trans-
2. Trans-6′-fluoro-N-methyl-4-phenyl-4′,9′-dihydro-3’H-spiro[cyclohexane-1,1′-pyrano[3,4-b]indol]-4-amine
3. Trans -6’-fluoro-4’,9’-dihydro-N-methyl-4-phenyl-spiro[cyclohexane-1,1’(3’H)-pyrano[3,4-b]indol]-4-amine
MOLECULAR FORMULA C23H25FN2O
MOLECULAR WEIGHT 364.5
SPONSOR Grűnenthal GmbH
CODE DESIGNATIONS GRT6006, GRT13106G
CAS REGISTRY NUMBER 1357348-09-4
UNIIDZ4NDW1LZX
WHO NUMBER 9765
gbk
The heptadecapeptide nociceptin is an endogenous ligand of the ORL1 (opioid receptor-like) receptor (Meunier et al., Nature 377, 1995, p. 532-535), which belongs to the family of opioid receptors and is to be found in many regions of the brain and spinal cord, and has a high affinity for the ORL1 receptor. The ORL1 receptor is homologous to the μ, κ and δ opioid receptors and the amino acid sequence of the nociceptin peptide has a marked similarity to those of the known opioid peptides. The receptor activation induced by nociceptin leads, via coupling with Gi/o proteins, to an inhibition of adenylate cyclase (Meunier et al., Nature 377, 1995, p. 532-535).
The nociceptin peptide shows a pronociceptive and hyperalgesic activity after intercerebroventicular administration in various animal models (Reinscheid et al., Science 270, 1995, p. 792-794). These findings can be explained as an inhibition of stress-induced analgesia (Mogil et al., Neuroscience 75, 1996, p. 333-337). In this connection, it has also been possible to demonstrate an anxiolytic activity of nociceptin (Jenck et al., Proc. Natl. Acad. Sci. USA 94, 1997, 14854-14858).
On the other hand, it has also been possible to demonstrate an antinociceptive effect of nociceptin in various animal models, in particular after intrathecal administration. Nociceptin has an antinociceptive action in various pain models, for example in the tail flick test in the mouse (King et al., Neurosci. Lett., 223, 1997, 113-116. It has likewise been possible to demonstrate an antinociceptive action of nociceptin in models for neuropathic pain, which is of particular interest inasmuch as the activity of nociceptin increases after axotomy of spinal nerves. This is in contrast to conventional opioids, the activity of which decreases under these conditions (Abdulla and Smith, J. Neurosci., 18, 1998, p. 9685-9694).
The ORL1 receptor is moreover also involved in regulation of further physiological and pathophysiological processes. These include, inter alia, learning and memory development (Manabe et al., Nature, 394, 1997, p. 577-581), audition (Nishi et al., EMBO J., 16, 1997, p. 1858-1864) and numerous further processes. A review article by Cabo et al. (Br. J. Pharmacol., 129, 2000, 1261-1283) gives an overview of the indications or biological processes in which the ORL1 receptor plays a role or with high probability could play a role. This mentions, inter alia: analgesia, stimulation and regulation of food intake, influence on μ-agonists, such as morphine, treatment of withdrawal symptoms, reduction in the addiction potential of opioids, anxiolysis, modulation of motor activity, impaired memory, epilepsy; modulation of neurotransmitter secretion, in particular glutamate, serotonin and dopamine, and therefore neurodegenerative diseases; influencing of the cardiovascular system, initiation of an erection, diuresis, anti-natriuresis, electrolyte balance, arterial blood pressure, water retention diseases, intestinal motility (diarrhea), relaxing effects on the respiratory tract, micturation reflex (urinary incontinence). The use of agonists and antagonists as anoretics, analgesics (also in co-administration with opioids) or nootropics is furthermore discussed.
The possible uses of compounds which bind to the ORL1 receptor and activate or inhibit this are correspondingly diverse. Alongside this, however, opioid receptors, such as the μ-receptor, but also the other sub-types of these opioid receptors, namely δ and κ, play a large role precisely in the area of pain therapy, but also in that of other indications of those mentioned. Accordingly, it is favourable if the compound also show an action on these opioid receptors.
SYN
http://www.google.com/patents/US20110319440

SYNTHESIS ……………..ON THE WAY ….. WATCH OUT
The dimethyl analogue is

Jirkovsky et al., J. Heterocycl. Chem., 12, 1975, 937-940;
Campaigne et al., J. Heterocycl. Chem., 2, 1965, 231-235;
Efange et al., J. Med. Chem., 41, 1998, 4486-4491;
Ellingboe et al., J. Med. Chem., 35, 1992, 1176-1183;
Pearson et al., Aust. J. Chem., 44, 1991, 907-917;
Yokohama et al., Chem. Pharm. Bull., 40, 1992, 2391-2398;
Beck et al., J. Chem. Soc. Perkin 1, 1992, 813-822;
Shinada et al., Tetrahedron Lett., 39, 1996, 7099-7102;
Garden et al., Tetrahedron, 58, 2002, 8399-8412;
Lednicer et al., J. Med. Chem., 23, 1980, 424-430.
|
2-10-2012
|
Pharmaceutical dosage forms comprising 6′-fluoro-(N-methyl- or N,N-dimethyl-)-4-phenyl-4′,9′-dihydro-3’H-spiro[cyclohexane-1,1′-pyrano[3,4,b]indol]-4-amine
|
|
|
11-9-2011
|
Compositions containing spirocyclic cyclohexane compounds
|
|
|
10-12-2011
|
Spirocyclic Cyclohexane Compounds Useful To Treat Substance Dependency
|
|
|
5-32-2011
|
Spirocyclic Cyclohexane Compounds
|
|
|
1-21-2011
|
MIXED ORL1/MU-AGONISTS FOR THE TREATMENT OF PAIN
|
|
|
9-22-2010
|
SPIROCYCLIC CYCLOHEXANE COMPOUNDS
|
|
|
6-17-2009
|
Spirocyclic cyclohexane compounds
|
|
|
9-12-2008
|
Spirocyclic Cyclohexane Compounds Useful To Treat Substance Dependency
|
|
|
5-30-2008
|
Mixed ORL1/mu-agonists for the treatment of pain
|
| US8614245 | Jan 8, 2013 | Dec 24, 2013 | Gruenenthal Gmbh | Crystalline (1r,4r)-6′-fluoro-N,N-dimethyl-4-phenyl-4′,9′-dihydro-3′H-spiro[cyclohexane-1,1′-pyrano[3,4,b]indol]-4-amine |
| US8618156 * | Jul 6, 2012 | Dec 31, 2013 | Gruenenthal Gmbh | Crystalline (1r,4r)-6′-fluoro-N,N-dimethyl-4-phenyl-4′,9′-dihydro-3’H-spiro[cyclohexane-1,1′-pyrano[3,4,b]indol]-4-amine |
| US8765800 | Mar 15, 2013 | Jul 1, 2014 | Gruenenthal Gmbh | Crystalline (1r,4r)-6′-fluoro-N,N-dimethyl-4-phenyl-4′,9′-dihydro-3′H-spiro[cyclohexane-1,1′-pyrano[3,4,b]indol]-4-amine |
| US20130231381 * | Mar 15, 2013 | Sep 5, 2013 | Gruenenthal Gmbh | Crystalline (1r,4r)-6′-fluoro-N,N-dimethyl-4-phenyl-4′,9′-dihydro-3’H-spiro[cyclohexane-1,1′-pyrano[3,4,b]indol]-4-amine |
| US5356896 * | Dec 22, 1992 | Oct 18, 1994 | Sandoz Ltd. | Alkaline stabiling medium |
| US20060004034 * | May 11, 2005 | Jan 5, 2006 | Gruenenthal Gmbh | Treating conditions associated with the nociceptin/ORL1 receptor system, e.g. pain, drug withdrawal, anxiety, muscle relaxants, anxiolytic agents; e.g. 1,1-[3-dimethylamino-3-(pyridin-2-yl)pentamethylene]-3,4-dihydro-1H-2,9-diazafluorene |
MAKE IN INDIA
http://makeinindia.com/sector/pharmaceuticals/
Read all about Organic Spectroscopy on ORGANIC SPECTROSCOPY INTERNATIONAL 
keep watching for synthesis update on this drug
QP Declaration: EMA publishes Comments
DRUG REGULATORY AFFAIRS INTERNATIONAL
QP Declaration: EMA publishes Comments
More than three years ago, the EMA has published two draft documents for a template for the QP’s declaration concerning GMP compliance of the API used as starting material and verification of its supply chain called “The QP declaration template“:
1. The draft template for the Qualified Person’s declaration
and
2. the respective draft Q&A on the template for the Qualified Person’s declaration
The QP Declaration should be provided in support of an application for a new marketing authorisation, variation or renewal of a medicinal product(s) authorised in the Community, using EU or national procedures within the scope of the respective Directives.
The consultation for the template ended on 30 April 2011. In June 2014, the final version was published together with a template guidance. Now, three months after publication of the final document, the comments from 2011 have been published.
The…
View original post 559 more words
WHO publishes New Version of the Draft on “Hold-Time” Studies
DRUG REGULATORY AFFAIRS INTERNATIONAL
WHO publishes New Version of the Draft on “Hold-Time” Studies
The 2nd revision for comment was published already in February this year (we reported). Now, a 3rd version is available – also for comment. The document describes the design of hold-time studies for the determination of time limits which have to be determined according to the generally applicable intermediate and bulk products. This should avoid that the storage of intermediate or bulk products from having any negative influence on their quality or the quality of a finished before processing to the next stage.
Chapter 2 which defines what intermediate and bulk products are has been added. It is now explicitly pointed out that hold-time investigations are part of the process validation. In turn, the reference to retrospective observation has been removed from the current version as well as – fortunately – the incomprehensible paragraph on the ‚most probable /…
View original post 113 more words
How to identify Out-of-Trend Results in Stability Studies?
DRUG REGULATORY AFFAIRS INTERNATIONAL

How to identify Out-of-Trend Results in Stability Studies?
http://www.gmp-compliance.org/enews_4522_How-to-identify-Out-of-Trend-Results-in-Stability-Studies_8360,8348,8430,Z-QCM_n.html
An article in PharmTech from June 2013 (by Trajkovic-Jolevska et. al) deals with the methods to identify Out-of-Trend (OOT) results in ongoing stability studies.
With regard to stability studies, it is important to make the difference between Out-of-Specification (OOS) and Out-of-Trend (OOT). Both the pharmaceutical industry and authorities often misuse these two terms.
The article defines OOT results as those results which don’t follow the expected trend, either in comparison with other stability batches or compared to previous results collected during a stability study. OOT results aren’t necessarily OOS, but they don’t look like a typical data point.
Although OOT results are a serious problem, neither the scientific literature nor regulatory guidelines fully address them.
The aim of the study described in this Pharmtech article by Trajkovic-Jolevska et. al was to perform a statistical evaluation of the statistical methods used in…
View original post 112 more words
Pirenperone, R 47465

ON THE LEFT OR ABOVE
IS
3-(2- {4-[(4-fluorophenyl)carbonyl]piperidin- 1 -yl} ethyl)-2-methyl-4H-pyrido[ 1 ,2- α]pyrimidin-4-one
3-(2-{4-[(4-fluorophenyl)carbonyl]piperidin-1-yl}ethyl)-2-methyl-4H-pyrido[1,2-α]pyrimidin-4-one
3-[2-[4-(4-fluorobenzoyl)-1-piperidinyl]ethyl]-2-methyl-4H-pyrido[1,2-a]pyri- midin-4-one
pirenperone CAS : 75444-65-4
- C23 H24 F N3 O2
- 4H-Pyrido[1,2-a]pyrimidin-4-one, 3-[2-[4-(4-fluorobenzoyl)-1-piperidinyl]ethyl]-2-methyl-
- R 47465
Cardiovascular disease; Inflammatory disease; Neoplasm; Pain
Calcium channel modulator T-type
……………………………..
http://www.google.co.in/patents/US4342870
http://www.google.co.in/patents/EP0037265A1
Example XXIV
-
A solution of 2 parts of 3-[2-[4-(4-fluorobenzoyl)-1 -piperidinyl]ethyl]-2 -methyl-4H -pyricio[1, 2 -a]pyrimidin-4-one in 64 parts of 2-propanol is warm acidified with 2-propanol saturated with hydrogen chloride. The formed hydrochloride salt is allowed to crystallize. It is filtered off and dried, yielding 2 parts (85. 5%) of 3-[2-[4-(4-fluorobenzoyl)-1-piperidinyl]ethyl]-2-methyl-4H-pyrido[1,2-a]pyri- midin-4-one dihydrochloride; mp. + 300°C.
-
In a similar manner there are also prepared:
- 3-[2 -[4-(4-fluorobenzoyl)-1-piperidiny]yethyl7-2 -methyl-4H-pyrido-[1, 2 -a]pyrimidin-4-one sulfate (1 : 2); mp. 254. 7°C; and
- 3-[2-[4-(4-fluorobenzoyl)-1 -piperidinyl]ethyl]-2-methyl-4H-pyrido-[1, 2-a]pyrimidin-4-one phosphate (1 : 2) ; mp. 243.8°C.
WO-2014143915
http://www.google.com/patents/WO2014143915A1?cl=en
Novelcrystalline polymorphic forms of pirenperone, useful for treating disorders associated with T-type calcium ion channels such as pain syndrome, neoplasm, cardiovascular disease or inflammation. VM Discovery, from which VM Therapeutics was spun out, was investigating the VMD-C300 series of compounds which act as T-type calcium channel modulators, including VMD-3816 and VMD-3222, for treating cancer, pain, neurological diseases and cardiovascular diseases; but as of September 2014, this program was assumed to be discontinued. See WO2009108798, (by the inventor, assigned to VM Discovery) claiming use of the same compound for treating same indications.
It was first disclosed in the now-expired US Patent No. 4,342,870 (Claim 5), and intended to be used as potential anti-anxiety drug. However, the early human clinical studies has shown that the compound did not show any dose-related anti-anxiety effects as hoped, but otherwise the compound was safe in human (ref. Ansseau M, Doumo t A, Thlry D, Gelders Y. “Pilot study of a specific serotonergic antagonist, pirenperone, in the treatment of anxiety disorders”, Acta Psychiatr Belg, 1983 Sep-Oct;83(5):517-24). in the US Patent No. 4,342,870, there is no crystalline polymorphic form disclosed, nor disclosure of potential uses for management of pain and treatment of other related diseases or disorders.
It was further disclosed in the PCT patent application WO/2009/108798 as “Compound 10 (pirenperone)‘” to be used for novel T-type calcium ion channel antagonist for management of pain and treatment of other diseases or disorders associated to the T-type calcium ion channels.
Surprisingly, we have found that there are many crystalline polymorphic forms of this compound which may affect the compound’s pharmaceutical safety and pharmacology properties.
MAKE IN INDIA
http://makeinindia.com/sector/pharmaceuticals/
A CASE OF ICHCHTHYOSIS ; A TYPICAL SKIN DISORDER ; E.T.G AYURVEDASCAN TEST EVALUATION ; “इक्थियासिस” जैसे लाइलाज चर्म रोग का ई०टी०जी० आयुर्वेदास्कैन आधारित आन्कलन
चर्म रोग ICHCHTHYOSIS या इख्तोयासिस एक तरह की ऐसी तकलीफ है जो त्वचा के टिश्यूज से जुड़ी हुयी बीमारी है / इस बीमारी मे त्वचा का रन्ग काला पड़ जाता है और त्वचा मोटी हो जाती है / इसके अलावा त्वचा का रन्ग काला और वर्ण cracks यानी फटी हुयी और आकार मछली की खाल जैसा हो जाता है /
जिस मरीज का नीचे दिया गया चित्र है उसे लगभग चार साल से यह तकलीफ रही है / अन्ग्रेजी और देशी और होम्योपैथी का इलाज कराने के बाद इसे आराम नही मिला / हमारे यहा से इलाज करा चुके एक मरीज द्वारा हमारे सन्स्थान मे इलाज कराने के लिये प्रोत्साहित किये जाने के बाद यह मरीज इलाज के लिये हमारे यहां आया है /
मरीज के दोनो पैरो और शरीर के लगभग सभी हिस्सो मे इसी तरह के scabs मौजूद है /.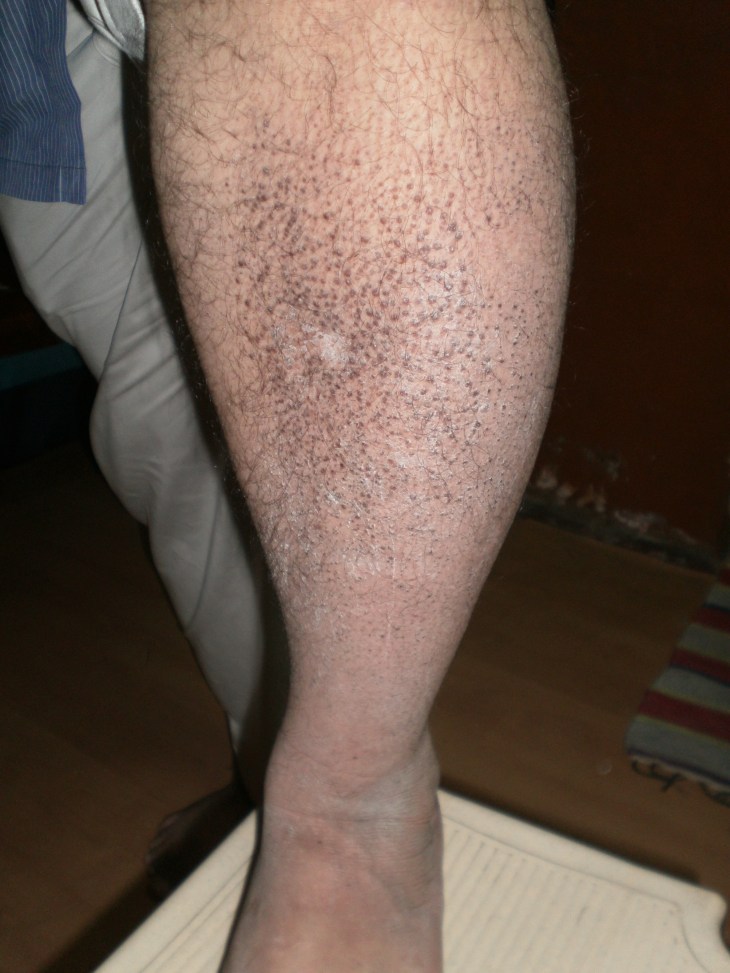
नीचे दिये गये चित्र मे यह चर्म रोग…
View original post 339 more words
Leflunomide

Leflunomide
RS-34821, SU-101, HWA-486, Arava,75706-12-6,
- Arava
- HSDB 7289
- HWA 486
- HWA-486
- Leflunomida
- Leflunomida [INN-Spanish]
- Leflunomide
- Leflunomidum
- Leflunomidum [INN-Latin]
- SU 101 (pharmaceutical)
- SU101
- UNII-G162GK9U4W
Leflunomide (brand names: Arabloc, Arava, Lunava, Repso) is an immunosuppressive disease-modifying antirheumatic drug (DMARD),[2] used in active moderate to severe rheumatoid arthritis and psoriatic arthritis. It is a pyrimidine synthesis inhibitor.[3]
Medical use
Rheumatoid arthritis and psoriatic arthritis are the only indications that have received regulatory approval.[1][4] Clinical studies regarding the following diseases have been conducted:[5]
- Polyoma BK Virus Nephropathy[6]
- Kimura’s disease[7]
- Systemic lupus erythematosus[8]
- Felty’s syndrome [9]
- Takayasu arteritis[10]
- Wegener’s granulomatosis[9]
- Ankylosing spondylitis[11]
- Crohn’s disease[12][13]
- Sarcoidosis[14]
- Uveitis[15]
- Still’s disease[16]
- Prostate cancer[17]
- Pemphigoid[18]
- Prevention of organ transplant rejection[19]
Side effects
Its principle dose-limiting side effects are liver damage, lung disease and immunosuppression.[19] The most common side effects (occurring in >1% of those treated with it) are, in approximately descending order of frequency:[1][4][20][21][22][23][24] diarrhoea, respiratory tract infections, hair loss, high blood pressure, rash, nausea, bronchitis, headache, abdominal pain, abnormal liver function tests, back pain, indigestion, urinary tract infection, dizziness, infection, joint disorder, itchiness, weight loss, loss of appetite, cough, gastroenteritis, pharyngitis, stomatitis, tenosynovitis, vomiting, weakness, allergic reaction, chest pain, dry skin, eczema,paraesthesia, pneumonia, rhinitis, synovitis, cholelithiasis and shortness of breath. Whereas uncommon side effects (occurring in 0.1-1% of those treated with the drug) include:[4] constipation, oral thrush, stomatitis, taste disturbance, thrombocytopenia and hives. Rarely (in 0.1% of those treated with it) it can cause:[4] anaphylaxis, angiooedema, anaemia, agranulocytosis, eosinophilia,leucopenia, pancytopenia, vasculitis, toxic epidermal necrolysis, Stevens-Johnson syndrome, cutaneous lupus erythematosus, severe infection, interstitial lung disease, cirrhosis and liver failure.
Contraindications
Contraindications include:[1]
- Pregnancy, women of childbearing potential (unless contraception used)
- Liver disease, hepatitis B/Cseropositive
- Active serious infections
- Hypersensitivity
Interactions
Other immunomodulatory treatments should be avoided due to the potential for additive immunosuppressant effects, or in the case of immunostimulants like echinacea or astragalus, reduced therapeutic effects.[1] Likewise live vaccines (like haemophilus influenzae type b vaccine and yellow fever vaccines) should be avoided due to the potential for severe infection due to the immunosuppressive nature of the treatment.[1]
The concomitant use of methotrexate, in particular, may lead to severe or even fatal liver- or hepatotoxicity. Seventy-five percent of all cases of severe liver damage reported until early 2001 were seen under combined drug therapy leflunomide plus methotrexate.[25]However, some studies have shown that the combination of methotrexate and leflunomide in patients with rheumatoid arthritis gave better results than either drug alone.[25]
Mechanism of action
Leflunomide is an immunomodulatory drug that achieves its effects by inhibiting the mitochondrial enzyme dihydroorotate dehydrogenase(an enzyme involved in de novo pyrimidine synthesis) (abbreviation DHODH), which plays a key role in the de novo (from scratch) synthesis of the uridine monophosphate (rUMP), which is required for the synthesis of DNA and RNA, hence leflunomide inhibits the reproduction of rapidly dividing cells, especially lymphocytes.[19] The inhibition of human DHODH by teriflunomide, the active metabolite of leflunomide, occurs at levels (approximately 600 nM) that are achieved during treatment of rheumatoid arthritis (RA).[26] Teriflunomide also inhibits several tyrosine kinases.[19] Teriflunomide prevents the expansion of activated and autoimmune lymphocytes by interfering with their cell cycle progression while nonlymphoid cells are able to use another pathway to make their ribonucleotides by use of salvage pyrimidine pathway, which makes them less dependent on de novo synthesis.[26] Teriflunomide also has antiviral effects against numerous viruses including CMV, HSV1 and the BK virus, which it achieves by inhibiting viral replication by interfering with nucleocapsid tegumentation and hence virion assembly.[19]
Pharmacokinetics
It has an oral bioavailability of 80%, protein binding of >99%, metabolism sites of the GI mucosa and liver, volume of distribution (Vd) of 0.13 L/kg, elimination half-life of 14-18 days and excretion routes of faeces (48%) and urine (43%).[19][1][20]
 |
|
| Systematic (IUPAC) name | |
|---|---|
| 5-methyl-N-[4-(trifluoromethyl) phenyl]-isoxazole-4-carboxamide | |
| Clinical data | |
| Trade names | Arabloc, Arava, Lunava, Repso |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a600032 |
| Licence data | EMA:Link, US FDA:link |
| Pregnancy cat. | |
| Legal status | |
| Routes | Oral (tablets) |
| Pharmacokinetic data | |
| Bioavailability | 80%[1] |
| Protein binding | >99%[1] |
| Metabolism | GI mucosa and liver[1] |
| Half-life | 14-18 days[1] |
| Excretion | Faeces (48%), urine (43%)[1] |
| Identifiers | |
| CAS number | 75706-12-6 |
| ATC code | L04AA13 |
| PubChem | CID 3899 |
| DrugBank | DB01097 |
| ChemSpider | 3762 |
| UNII | G162GK9U4W |
| KEGG | D00749 |
| ChEBI | CHEBI:6402 |
| ChEMBL | CHEMBL960 |
| Chemical data | |
| Formula | C12H9F3N2O2 |
| Mol. mass | 270.207 g/mol |
……………………………

…………………………..
http://www.beilstein-journals.org/bjoc/single/articleFullText.htm?publicId=1860-5397-7-57#S86
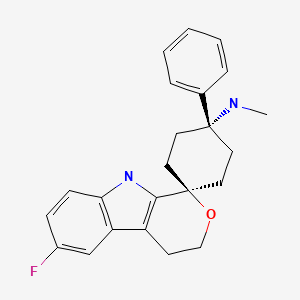
Leflunomide is a pyrimidine synthase inhibitor of the DMARD-type (disease-modifying anti-rheumatic drug) marketed by Sanofi-Aventis. Unlike NSAIDs, which only deal with symptoms of rheumatoid arthritis, DMARDs target the cause of it. DMARDs are not necessarily structurally or mechanistically related. The effect of leflunomide is possibly due to its regulation of the immune system via affecting lymphocytes. Its synthesis [134] is relatively straightforward starting with a Knoevenagel condensation of ethyl acetoacetate (39) and triethyl orthoformate in the presence of acetic anhydride. The resulting ethyl ethoxymethylene acetoacetate (448) is next condensed with hydroxylamine hydrate in methanol to yield ethyl 5-methylisoxazole-4-carboxylate (449). The ethyl ester is hydrolysed under acidic conditions and the carboxylic acid activated with thionyl chloride in DMF for amide formation with 4-trifluoromethylaniline (450) (Scheme 86).
US patent 5,494,911 discloses process for preparation of Teriflunomide in Example- 4 as shown in given below scheme-I.
References
- “Arava (leflunomide) dosing, indications, interactions, adverse effects, and more”. Medscape Reference. WebMD. Retrieved 11 March 2014.
- Dougados M, Emery P, Lemmel EM, Zerbini CA, Brin S, van Riel P (January 2005).“When a DMARD fails, should patients switch to sulfasalazine or add sulfasalazine to continuing leflunomide?”. Annals of the rheumatic diseases 64 (1): 44–51.doi:10.1136/ard.2003.016709. PMC 1755199. PMID 15271770.
- Pinto P, Dougados M (2006). “Leflunomide in clinical practice”. Acta reumatológica portuguesa 31 (3): 215–24. PMID 17094333.
- ^ Jump up to:a b c d Rossi, S, ed. (2013). Australian Medicines Handbook (2013 ed.). Adelaide: The Australian Medicines Handbook Unit Trust. ISBN 978-0-9805790-9-3.
- Jump up^ http://clinicaltrials.gov/ct2/results?term=Leflunomide
- Jump up^ Blanckaert, K; De Vriese, AS (23 September 2006). “Current recommendations for diagnosis and management of polyoma BK virus nephropathy in renal transplant recipients” (PDF). Nephrology Dialysis Transplantation 21 (12): 3364–3367.doi:10.1093/ndt/gfl404.
- Jump up^ Dai, L; Wei, XN; Zheng, DH; Mo, YQ; Pessler, F; Zhang, BY (June 2011). “Effective treatment of Kimura’s disease with leflunomide in combination with glucocorticoids.”.Clinical Rheumatology 30 (6): 859–65. doi:10.1007/s10067-011-1689-2.PMID 21286771.
- Jump up^ Wu, GC; Xu, XD; Huang, Q; Wu, H (February 2013). “Leflunomide: friend or foe for systemic lupus erythematosus?”. Rheumatology International 33 (2): 273–6.doi:10.1007/s00296-012-2508-z. PMID 22961090.
- ^ Jump up to:a b Sanders, S; Harisdangkul, V (2002). “Leflunomide for the treatment of rheumatoid arthritis and autoimmunity”. American Journal of Medical Sciences 323 (4): 190–3.doi:10.1097/00000441-200204000-00004. PMID 12003373.
- Jump up^ Unizony, S; Stone, JH; Stone, JR (January 2013). “New treatment strategies in large-vessel vasculitis.”. Current Opinion in Rheumatology 25 (1): 3–9.doi:10.1097/BOR.0b013e32835b133a. PMID 23114585.
- Jump up^ Haibel, H; Rudwaleit, M; Braun, J; Sieper, J (January 2005). “Six months open label trial of leflunomide in active ankylosing spondylitis.” (PDF). Annals of the Rheumatic Diseases 64 (1): 124–6. doi:10.1136/ard.2003.019174. PMC 1755172.PMID 15608310.
- Jump up^ Prajapati, DN; Knox, JF; Emmons, J; Saeian, K; Csuka, ME; Binion, DG (August 2003). “Leflunomide treatment of Crohn’s disease patients intolerant to standard immunomodulator therapy.”. Journal of Clinical Gastroenterology 37 (2): 125–8.doi:10.1097/00004836-200308000-00006. PMID 12869881.
- Jump up^ Holtmann, MH; Gerts, AL; Weinman, A; Galle, PR; Neurath, MF (April 2008). “Treatment of Crohn’s disease with leflunomide as second-line immunosuppression : a phase 1 open-label trial on efficacy, tolerability and safety.”. Digestive Diseases and Sciences 53 (4): 1025–32. doi:10.1007/s10620-007-9953-7. PMID 17934840.
- Jump up^ Panselinas, E; Judson, MA (October 2012). “Acute pulmonary exacerbations of sarcoidosis.” (PDF). Chest 142 (4): 827–36. doi:10.1378/chest.12-1060.PMID 23032450.
- Jump up^ Roy, M (August 2007). “Early clinical experience with leflunomide in uveitis.”. Canadian Journal of Ophthalmology 42 (4): 634. doi:10.3129/canjophthalmol.i07-085.PMID 17641721.
- Jump up^ Pirildar, T (May 2003). “Treatment of adult-onset Still’s disease with leflunomide and chloroquine combination in two patients.”. Clinical Rheumatology 22 (2): 157.doi:10.1007/s10067-002-0667-0. PMID 12740686.
- Jump up^ “Mitoxantrone and Prednisone With or Without Leflunomide in Treating Patients With Stage IV Prostate Cancer”. ClinicalTrials.gov. National Institute of Health. September 2012. Retrieved 11 March 2014.
- Jump up^ “Leflunomide Associated With Topical Corticosteroids for Bullous Pemphigoid (ARABUL)”. ClinicalTrials.gov. National Institute of Health. December 2008. Retrieved 11 March 2014.
- ^ Jump up to:a b c d e f Teschner, S; Burst, V (September 2010). “Leflunomide: a drug with a potential beyond rheumatology.”. Immunotherapy 2 (5): 637–50. doi:10.2217/imt.10.52.PMID 20874647.
- ^ Jump up to:a b “PRODUCT INFORMATION ARAVA®” (PDF). TGA eBusiness Services. sanofi-aventis australia pty ltd. 7 August 2012. Retrieved 11 March 2014.
- Jump up^ “Arava : EPAR – Product Information” (PDF). European Medicines Agency. Sanofi-Aventis Deutschland GmbH. 21 November 2013. Retrieved 11 March 2014.
- Jump up^ “Data Sheet Arava®” (PDF). Medsafe. sanofi-aventis new zealand limited. 29 June 2012. Retrieved 11 March 2014.
- Jump up^ “ARAVA (leflunomide) tablet, film coated [sanofi-aventis U.S. LLC]”. DailyMed. sanofi-aventis U.S. LLC. November 2012. Retrieved 11 March 2014.
- Jump up^ “Arava 100mg Tablets – Summary of Product Characteristic”. electronic Medicines Compendium. SANOFI. 21 February 2014. Retrieved 11 March 2014.
- ^ Jump up to:a b Lee, S.; Park, Y.; Park, J.; Kang, Y.; Nam, E.; Kim, S.; Lee, J.; Yoo, W.; Lee, S. (2009). “Combination treatment with leflunomide and methotrexate for patients with active rheumatoid arthritis”. Scandinavian journal of rheumatology 38 (1): 11–14.doi:10.1080/03009740802360632. PMID 19191187.
- ^ Jump up to:a b Fox, RI; Herrmann, ML; Frangou, CG; Wahl, GM; Morris, RE; Strand, V; Kirschbaum, BJ (December 1999). “Mechanism of action for leflunomide in rheumatoid arthritis.”. Clinical Immunology 93 (3): 198–208. doi:10.1006/clim.1999.4777.PMID 10600330.
External links
- National Rheumatoid Arthritis Society (NRAS) Information about Disease Modifying drugs such as Leflunomide
- http://www.arava.com/professional/home.do (full prescribing information)
- http://www.rheuma-online.de/medikamente/leflunomid-arava/studien-zu-leflunomid-arava/gibt-es-untersuchungen-zu-leflunomid-in-weiteren-einsatzgebieten.html (in German, regarding potential indications)
- http://www.arznei-telegramm.de/register/0204507.pdf (in German, regarding discontinuation of the drug)
- http://www.emea.europa.eu/pdfs/human/press/pus/561101en.pdf (warning as of 2001 regarding hepatotoxicity) (URL DEAD 16 Oct 2010)
- The safety of leflunomide Australian Prescriber
Dosages/Routes/Forms
by SANOFI AVENTIS US
| Strength | Form/Route | Marketing Status | RLD | TE Code |
|---|---|---|---|---|
| 10MG | TABLET;ORAL | 1 | 0 | AB |
| 20MG | TABLET;ORAL | 1 | 1 | AB |
| 100MG | TABLET;ORAL | 1 | 1 |
Approval History

Spectra
UV – spectrum
IR – spectrum
Links
-
UV and IR Spectra. H.-W. Dibbern, RM Muller, E. Wirbitzki, 2002 ECV
-
NIST / EPA / NIH Mass Spectral Library 2008
-
Handbook of Organic Compounds. NIR, IR, Raman, and UV-Vis Spectra Featuring Polymers and Surfactants, Jr., Jerry Workman.Academic Press, 2000.
-
Handbook of ultraviolet and visible absorption spectra of organic compounds, K. Hirayama. Plenum Press Data Division, 1967.
LEFLUNOMIDE IMPURITY C [EP
5-Methyl-N-(3-(trifluoromethyl)phenyl)isoxazole-4-carboxamide
LEFLUNOMIDE IMPURITY D [EP]
5-Methylisoxazole-4-carboxylic acid
LEFLUNOMIDE IMPURITY E [EP]
3-Methyl-N-(4-(trifluoromethyl)phenyl)isoxazole-4-carboxamide
LEFLUNOMIDE IMPURITY F [EP]
5-Methyl-N-(2-(trifluoromethyl)phenyl)isoxazole-4-carboxamide
LEFLUNOMIDE IMPURITY G [EP]
5-Methyl-N-(4-methylphenyl)isoxazole-4-carboxamide
LEFLUNOMIDE IMPURITY H [EP]
2-Cyano-N-(4-(trifluoromethyl)phenyl)acetamide
Wild berry extract may strengthen effectiveness of pancreatic cancer drug
Public Release: 17-Sep-2014
The findings prompt the researchers to suggest that adding ‘nutraceuticals’ to chemotherapy cycles may improve the effectiveness of conventional drugs, particularly in hard to treat cancers, such as pancreatic cancer.
They base their findings on the effectiveness of extract of chokeberry (Aronia melanocarpa) in killing off cancer cells—a process known as apoptosis.
Chokeberry is a wild berry that grows on the eastern side of North America in wetlands and swamp areas. The berry is high in vitamins and antioxidants, including various polyphenols—compounds that are believed to mop up the harmful by-products of normal cell activity.
The researchers chose to study the impact of the extract on pancreatic cancer, because of its persistently dismal prognosis: less than 5% of patients are alive five years after their diagnosis.
View original post 373 more words
Coconut water is an excellent sports drink — for light exercise
PHILADELPHIA, Aug. 20, 2012 — Coconut water really does deserve its popular reputation as Mother Nature’s own sports drink, a new scientific analysis of the much-hyped natural beverage concluded here today at the 244th National Meeting & Exposition of the American Chemical Society (ACS).
However, people who engage in strenuous exercise that involves a lot of sweat might want to take it all with a grain of salt ― literally ― or stick with a more traditional sports drink like Gatorade, said Chhandashri Bhattacharya, Ph.D. She presented a report on an analysis of coconut water to the ACS, the world’s largest scientific society, which is meeting here this week.
“Coconut water is a natural drink that has everything your average sports drink has and more,” said Bhattacharya. “It has five times more potassium than Gatorade or Powerade. Whenever you get cramps in your muscles, potassium will help you to get…
View original post 370 more words

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....













![[1860-5397-7-57-i86]](https://i0.wp.com/www.beilstein-journals.org/bjoc/content/inline/1860-5397-7-57-i86.png)