Home » Uncategorized (Page 103)
Category Archives: Uncategorized
Honeysuckle soup directly targets influenza A viruses (IAV) including H1N1, H5N1 and H7N9 and can prevent IAV infection and reduce H5N1-induced mice death
‘Virological penicillin’: Plant MIR2911 directly targets influenza A viruses
6-Oct-2014
In a new study, Chen-Yu Zhang’s group at Nanjing University present an extremely novel finding that a plant microRNA, MIR2911, which is enriched in honeysuckle, directly targets influenza A viruses (IAV) including H1N1, H5N1 and H7N9. Drinking of honeysuckle soup can prevent IAV infection and reduce H5N1-induced mice death.
MicroRNAs (miRNAs) are a class of 19-24 nucleotide non-coding RNAs that do not encode for proteins. MiRNAs bind to target messenger RNAs to inhibit protein translation. In previous studies, the same group has demonstrated that stable miRNAs in mammalian serum and plasma are actively secreted from tissues and cells and can serve as a novel class of biomarkers for disease and act as signaling molecules in intercellular communication. They have also reported that plant miRNAs can enter into the host blood and tissues via the route of food-intake…
View original post 985 more words
“OPERATION” AND “SURGICAL INTERVENTION” HOBBY ; MOST PEOPLE’S SEEKS ALWAYS FOR “OPERATION” METHODS IN ALL DISEASE CONDITIONS ; “आपरेशन” कराने का शौक ; बहुत से ऐसे लोग है जो हर बीमारी के इलाज के लिये “आपरेशन” कराना सबसे बेहतर समझते है
आपरेशन कराने का शौक भी दूसरे शौक की तरह से ही है /
दुनिया मे तरह तरह के लोग है जिनको किसी न किसी बात का शौक होता है , किसी को पतम्ग ऊड़ाने का शौक, किसी को सिगरेट पीने का शौक, किसी को मन्दिर जाने का शौक, किसी को ज्वेलरी का शौक, किसी को पेन्टिन्ग का शौक , किसी को पढने का शौक, किसी को बहस करने का शौक और किसी को कुछ और किसी को कुछ दूसरा शौक होता है /
इसी तरह से मुझे बहुत ऐसे मरीज मिले जिनको आप्रेशन कराने का शौक होता है / इन मरीजो को ऐसे ही आपरेशन करने वाले डाक्टर भी मिल जाते है / कहावत है जहां चाह है वही राह है / आप्रेशन कराने के लिये बेताब मरीज मौजूद है तो उनके लिये आपरेशन करने वाले डाक्टर और सर्जन भी मौजूद है / क्यो न हो ? आपरेशन कराने के…
View original post 42 more words
Vibegron ビベグロン
Vibegron, MK-4618, KRP 114V
update FDA APPROVED 12/23/2020, GEMTESA, To treat overactive bladder
Target-based Actions Beta 3 adrenoceptor agonist
Indications Overactive bladder; Urinary incontinence
UPDATE 2018/9/21 pmda Beova JAPAN 2018Kyorin Pharmaceutical, under license from Merck, is developing vibegron (phase II, September 2014) for the treating of overactive bladder. In July 2014, Merck has granted to Kyorin an exclusive license to develop, manufacture and commercialize vibegron in Japan.
MK-4618 is being developed in phase II clinical trials at Merck & Co. for the treatment of overactive bladder. The company had been developing the compound for the treatment of endocrine disorders and hypertension; however, recent progress reports are not available at present.
In 2014, Merck licensed the product to Kyorin for development and commercialization in Japan.
The function of the lower urinary tract is to store and periodically release urine. This requires the orchestration of storage and micturition reflexes which involve a variety of afferent and efferent neural pathways, leading to modulation of central and peripheral neuroeffector mechanisms, and resultant coordinated regulation of sympathetic and parasympathetic components of the autonomic nervous system as well as somatic motor pathways. These proximally regulate the contractile state of bladder (detrusor) and urethral smooth muscle, and urethral sphincter striated muscle.
β Adrenergic receptors (βAR) are present in detrusor smooth muscle of various species, including human, rat, guinea pig, rabbit, ferret, dog, cat, pig and non-human primate. However, pharmacological studies indicate there are marked species differences in the receptor subtypes mediating relaxation of the isolated detrusor; β1AR predominate in cats and guinea pig, β2AR predominate in rabbit, and β3AR contribute or predominate in dog, rat, ferret, pig, cynomolgus and human detrusor. Expression of βAR subtypes in the human and rat detrusor has been examined by a variety of techniques, and the presence of β3AR was confirmed using in situ hybridization and/or reverse transcription-polymerase chain reaction (RT-PCR). Real time quantitative PCR analyses of β1AR, β2AR and β3AR mRNAs in bladder tissue from patients undergoing radical cystectomy revealed a preponderance of β3AR mRNA (97%, cf 1.5% for β1AR mRNA and 1.4% for β2AR mRNA). Moreover, β3AR mRNA expression was equivalent in control and obstructed human bladders. These data suggest that bladder outlet obstruction does not result in downregulation of β3AR, or in alteration of β3AR-mediated detrusor relaxation. β3AR responsiveness also has been compared in bladder strips obtained during cystectomy or enterocystoplasty from patients judged to have normal bladder function, and from patients with detrusor hyporeflexia or hyperreflexia. No differences in the extent or potency of β3AR agonist mediated relaxation were observed, consistent with the concept that the β3AR activation is an effective way of relaxing the detrusor in normal and pathogenic states.
Functional evidence in support of an important role for the β3AR in urine storage emanates from studies in vivo. Following intravenous administration to rats, the rodent selective β3AR agonist CL316243 reduces bladder pressure and in cystomeric studies increases bladder capacity leading to prolongation of micturition interval without increasing residual urine volume.
Overactive bladder is characterized by the symptoms of urinary urgency, with or without urgency urinary incontinence, usually associated with frequency and nocturia. The prevalence of OAB in the United States and Europe has been estimated at 16 to 17% in both women and men over the age of 18 years. Overactive bladder is most often classified as idiopathic, but can also be secondary to neurological condition, bladder outlet obstruction, and other causes. From a pathophysiologic perspective, the overactive bladder symptom complex, especially when associated with urge incontinence, is suggestive of detrusor overactivity. Urgency with or without incontinence has been shown to negatively impact both social and medical well-being, and represents a significant burden in terms of annual direct and indirect healthcare expenditures. Importantly, current medical therapy for urgency (with or without incontinence) is suboptimal, as many patients either do not demonstrate an adequate response to current treatments, and/or are unable to tolerate current treatments (for example, dry mouth associated with anticholinergic therapy). Therefore, there is need for new, well-tolerated therapies that effectively treat urinary frequency, urgency and incontinence, either as monotherapy or in combination with available therapies. Agents that relax bladder smooth muscle, such as β3AR agonists, are expected to be effective for treating such urinary disorders.
PATENT
http://www.google.com/patents/WO2013062881A1?cl=en

EXAMPLE 3
To a three neck flask equipped with a N2 inlet, a thermo couple probe was charged pyrrolidine i-11 (10.0 g), sodium salt i-12 (7.87 g), followed by IPA (40 mL) and water (24 mL). 5 N HC1 (14.9 mL) was then slowly added over a period of 20 min to adjust pH = 3.3- 3.5, maintaining the batch temperature below 35 °C. Solid EDC hydrochloride (7.47 g) was charged in portions over 30 min. The reaction mixture was aged at RT for additional 0.5 – 1 h, aqueous ammonia (14%) was added dropwise to pH ~8.6. The batch was seeded and aged for additional 1 h to form a slurry bed. The rest aqueous ammonia (14%, 53.2 ml total) was added dropwise over 6 h. The resulting thick slurry was aged 2-3 h before filtration. The wet-cake was displacement washed with 30% IPA (30 mL), followed by 15% IPA (2 x 20mL) and water (2 X 20mL). The cake was suction dried under N2 overnight to afford 14.3 g of compound of Formula (I)-
1H NMR (DMSO) δ 10.40 (s, NH), 7.92 (d, J = 6.8, 1H), 7.50 (m, 2H), 7.32 (m, 2H), 7.29 (m, 2H), 7.21 (m, 1H), 7.16 (m, 2H), 6.24 (d, J = 6.8, 1H), 5.13 (dd, J = 9.6, 3.1, 1H), 5.08 (br s, OH), 4.22 (d, J = 7.2, 1H), 3.19 (p, J = 7.0, 1H), 3.16-3.01 (m, 3H), 2.65 (m, 1H), 2.59-2.49 (m, 2H), 2.45 (br s, NH), 2.16 (ddt, J = 13.0, 9.6, 3.1, 1H), 1.58 (m, 1H), 1.39 (m, 1H), 1.31-1.24 (m, 2H).
13C NMR (DMSO) δ 167.52, 165.85, 159.83, 154.56, 144.19, 136.48, 135.66, 129.16, 127.71, 126.78, 126.62, 119.07, 112.00, 76.71, 64.34, 61.05, 59.60, 42.22, 31.26, 30.12, 27.09, 23.82.
HPLC method – For monitoring conversion
Column: XBridge C18 cm 15 cm x 4.6 mm, 3.5 μιη particle size;
Column Temp. : 35 °C; Flow rate: 1.5 mL/min; Detection: 220 nm;
Mobile phase: A. 5 mM Na2B407.10 H20 B: Acetonitrile
Gradient:
HPLC method – For level of amide epimer detection
Column: Chiralpak AD-H 5 μηι, 250 mm x 4.6 mm.
Column Temp: 35 °C; Flow rate: 1.0 mL/min; Detection: 250 nm;
Mobile phase: Isocratic 30% Ethanol in hexanes + 0.1% isobutylamine
PATENT
WO 2009124167
http://www.google.com/patents/WO2009124167A1?cl=en
EXAMPLE 103
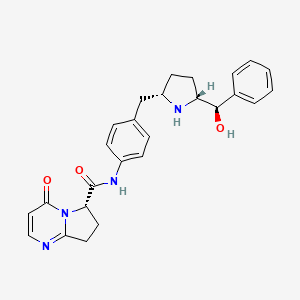
(6y)-N-r4-({(‘25′. 5R)-5-r(‘R)-hvdroxy(‘phenvnmethyl1pyrrolidin-2-yl}methvnphenyl1-4-oxo- 4,6J,8-tetrahydropyiτolori,2-α1pyrimidine-6-carboxamide
ter?-butyl(2R. 55f)-2-rCR)-hvdroxy(‘phenvnmethyl1-5-r4-(‘{r(‘65f)-4-oxo-4.6.7.8-
tetrahydropyrrolof 1.2-alpyrimidin-6- yl]carbonyl} amino)benzyl]pyrrolidine- 1 – carboxylate
To a solution of i-13a (21.4 g, 55.9 mmol) in N,N-dimethylformamide (100 ml) at O0C was added [(65)-4-oxo-4,6,7,8-tetrahydropyrrolo[l,2-α]pyrimidine-6-carboxylic acid (11.1 g, 61.5 mmol), followed by 1 -hydroxybenzotriazole (i-44, 7.55 g, 55.9 mmol), N-(3- dimethylaminopropyl)-Nl-ethylcarbodiimide hydrochloride (16.1 g, 84.0 mmol) and N,N- diisopropylethylamine (29.2 ml, 168 mmol). The reaction mixture was stirred from O0C to ambient temperature for 2 h. Water (600 ml) was added and it was extracted with dichloromethane (600 ml x 2). The combined organic layers were dried over Na2SO4. After removal of the volatiles, the residue was purified by using a Biotage Horizon® system (0-5% then 5% methanol with 10% ammonia/dichloromethane mixture) to afford the title compound which contained 8% of the minor diastereomer. It was further purified by supercritical fluid chromatography (chiral AS column, 40% methanol) to afford the title compound as a pale yellow solid (22.0 g, 72%). 1H NMR (CDCl3): δ 9.61 (s, IH), 7.93 (d, J = 6.6 Hz, IH), 7.49 (d, J = 8.4 Hz, 2H), 7.35-7.28 (m, 5H), 7.13 (d, J = 8.5 Hz, 2H), 6.40 (d, J = 6.7 Hz, IH), 5.36 (d, J = 8.6 Hz, IH), 4.38 (m, IH), 4.12-4.04 (m, 2H), 3.46 (m,lH), 3.15-3.06 (m, 2H), 2.91 (dd, J = 13.1, 9.0 Hz, IH), 2.55 (m, IH), 2.38 (m, IH), 1.71-1.49 (m, 13H). LC-MS 567.4 (M+23).
(6S)-N-\4-( U2S. 5R)-5-r(R)-hvdroxy(phenyl)methyl1pyrrolidin-2-
yl}methyl)phenyl1-4-oxo-4,6J,8-tetrahvdropyrrolori,2-α1pyrimidine-6- carboxamide
To a solution of the intermediate from Step A (2.50 g, 4.59 mmol) in dichloromethane (40 ml) was added trifluoroacetic acid (15 ml). The reaction mixture was stirred at ambient temperature for 1.5 h. After removal of the volatiles, saturated NaHCCh was added to make the PH value to 8-9. The mixture was then extracted with dichloromethane. The combined organic layers were dried over Na2SO4. After concentration, crystallization from methanol/acetonitrile afforded the title compound as a white solid (1.23g, 60%). 1H NMR (DMSO-Cl6): δ 10.40 (s, IH), 7.91 (d, J = 6.7 Hz, IH), 7.49 (d, J = 8.3 Hz, 2H), 7.32-7.26 (m, 4H), 7.21 (m, IH), 7.15 (d, J = 8.4 Hz, 2H), 6.23 (d, J = 6.7 Hz, IH), 5.11 (dd, J = 9.6, 2.9 Hz, IH), 5.10 (br, IH), 4.21 (d, J = 7.1 Hz, IH), 3.20-3.00 (m, 4H), 2.66-2.51 (m, 3H), 2.16 (m, IH), 1.57 (m, IH), 1.38 (m, IH), 1.29-1.23 (m, 2H). LC-MS 445.3 (M+l).
Using the Biological Assays described above, the human β3 functional activity of Example 103 was determined to be between 11 to 100 nM.
PATENT
CHECK STRUCTURE…………….CAUTION
http://www.google.com/patents/US8247415


CAUTION…………….
Example 103(6S)-N-[4-({(2S,5R)-5-[(R)-hydroxy(phenyl)methyl]pyrrolidin-2-yl}methyl)phenyl]-4-oxo-4,6,7,8-tetrahydropyrrolo[1,2-α]pyrimidine-6-carboxamide
Step A: tert-butyl(2R,5S)-2-[(R)-hydroxy(phenyl)methyl]-5-[4-({[(6S)-4-oxo-4,6,7,8-tetrahydropyrrolo[1,2-α]pyrimidin-6-yl]carbonyl}amino)benzyl]pyrrolidine-1-carboxylate
To a solution of i-13a (21.4 g, 55.9 mmol) in N,N-dimethylformamide (100 ml) at 0° C. was added [(6S)-4-oxo-4,6,7,8-tetrahydropyrrolo[1,2-α]pyrimidine-6-carboxylic acid (11.1 g, 61.5 mmol), followed by 1-hydroxybenzotriazole (i-44, 7.55 g, 55.9 mmol), N-(3-dimethylaminopropyl)-N′-ethylcarbodiimide hydrochloride (16.1 g, 84.0 mmol) and N,N-diisopropylethylamine (29.2 ml, 168 mmol). The reaction mixture was stirred from 0° C. to ambient temperature for 2 h. Water (600 ml) was added and it was extracted with dichloromethane (600 ml×2). The combined organic layers were dried over Na2SO4. After removal of the volatiles, the residue was purified by using a Biotage Horizon® system (0-5% then 5% methanol with 10% ammonia/dichloromethane mixture) to afford the title compound which contained 8% of the minor diastereomer. It was further purified by supercritical fluid chromatography (chiral AS column, 40% methanol) to afford the title compound as a pale yellow solid (22.0 g, 72%). 1H NMR (CDCl3): δ 9.61 (s, 1H), 7.93 (d, J=6.6 Hz, 1H), 7.49 (d, J=8.4 Hz, 2H), 7.35-7.28 (m, 5H), 7.13 (d, J=8.5 Hz, 2H), 6.40 (d, J=6.7 Hz, 1H), 5.36 (d, J=8.6 Hz, 1H), 4.38 (m, 1H), 4.12-4.04 (m, 2H), 3.46 (m, 1H), 3.15-3.06 (m, 2H), 2.91 (dd, J=13.1, 9.0 Hz, 1H), 2.55 (m, 1H), 2.38 (m, 1H), 1.71-1.49 (m, 13H). LC-MS 567.4 (M+23).
Step B: (6S)-N-[4-({(2S,5R)-5-[(R)-hydroxy(phenyl)methyl]pyrrolidin-2-yl}methyl)phenyl]-4-oxo-4,6,7,8-tetrahydropyrrolo[1,2-α]pyrimidine-6-carboxamide
To a solution of the intermediate from Step A (2.50 g, 4.59 mmol) in dichloromethane (40 ml) was added trifluoroacetic acid (15 ml). The reaction mixture was stirred at ambient temperature for 1.5 h. After removal of the volatiles, saturated NaHCO3 was added to make the PH value to 8-9. The mixture was then extracted with dichloromethane. The combined organic layers were dried over Na2SO4. After concentration, crystallization from methanol/acetonitrile afforded the title compound as a white solid (1.23 g, 60%). 1H NMR (DMSO-d6): δ 10.40 (s, 1H), 7.91 (d, J=6.7 Hz, 1H), 7.49 (d, J=8.3 Hz, 2H), 7.32-7.26 (m, 4H), 7.21 (m, 1H), 7.15 (d, J=8.4 Hz, 2H), 6.23 (d, J=6.7 Hz, 1H), 5.11 (dd, J=9.6, 2.9 Hz, 1H), 5.10 (br, 1H), 4.21 (d, J=7.1 Hz, 1H), 3.20-3.00 (m, 4H), 2.66-2.51 (m, 3H), 2.16 (m, 1H), 1.57 (m, 1H), 1.38 (m, 1H), 1.29-1.23 (m, 2H). LC-MS 445.3 (M+1).
Using the Biological Assays described above, the human β3 functional activity of Example 103 was determined to be between 11 to 100 nM.
PATENT
WO2014150639
Step 6. Preparation of Compound 1-7 from Compound 1-6 and Compound A-2

To a three neck flask equipped with a N2 inlet, a thermo couple probe was charged pyrrolidine hemihydrate 1-6 (10.3 g), sodium salt A-2 (7.87 g), followed by IPA (40 mL) and water (24 mL). 5 N HC1 (14.9 mL) was then slowly added over a period of 20 minutes to adjust pH = 3.3-3.5, maintaining the batch temperature below 35°C. Solid EDC hydrochloride (7.47 g) was charged in portions over 30 minutes. The reaction mixture was aged at RT for additional 0.5 – 1 hour, aqueous ammonia (14%) was added dropwise to pH -8.6. The batch was seeded and aged for additional 1 hour to form a slurry bed. The rest aqueous ammonia (14%, 53.2 ml total) was added dropwise over 6 hours. The resulting thick slurry was aged 2-3 hours before filtration. The wet-cake was displacement washed with 30% IPA (30 mL), followed by 15% IPA (2 x 20mL) and water (2 X 20mL). The cake was suction dried under N2 overnight to afford 14.3 g of compound 1-7.
1H NMR (DMSO) δ 10.40 (s, NH), 7.92 (d, J = 6.8, 1H), 7.50 (m, 2H), 7.32 (m, 2H), 7.29 (m, 2H), 7.21 (m, 1H), 7.16 (m, 2H), 6.24 (d, J = 6.8, 1H), 5.13 (dd, J = 9.6, 3.1, 1H), 5.08 (br s, OH), 4.22 (d, J = 7.2, 1H), 3.19 (p, J = 7.0, 1H), 3.16-3.01 (m, 3H), 2.65 (m, 1H), 2.59-2.49 (m, 2H), 2.45 (br s, NH), 2.16 (ddt, J = 13.0, 9.6, 3.1, 1H), 1.58 (m, 1H), 1.39 (m, 1H), 1.31-1.24 (m, 2H).
13C NMR (DMSO) δ 167.52, 165.85, 159.83, 154.56, 144.19, 136.48, 135.66, 129.16, 127.71, 126.78, 126.62, 119.07, 112.00, 76.71, 64.34, 61.05, 59.60, 42.22, 31.26, 30.12, 27.09, 23.82.
The crystalline freebase anhydrous form I of Compound 1-7 can be characterized by XRPD by

PATENT
WO-2014150633
Merck Sharp & Dohme Corp
Process for preparing stable immobilized ketoreductase comprises bonding of recombinant ketoreductase to the resin in a solvent. Useful for synthesis of vibegron intermediates. For a concurrent filling see WO2014150639, claiming the method for immobilization of ketoreductase. Picks up from WO2013062881, claiming the non enzymatic synthesis of vibegron and intermediates.
PAPER
Discovery of Vibegron: A Potent and Selective β3 Adrenergic Receptor Agonist for the Treatment of Overactive Bladder
http://pubs.acs.org/doi/abs/10.1021/acs.jmedchem.5b01372
http://pubs.acs.org/doi/suppl/10.1021/acs.jmedchem.5b01372/suppl_file/jm5b01372_si_001.pdf

The discovery of vibegron, a potent and selective human β3-AR agonist for the treatment of overactive bladder (OAB), is described. An early-generation clinical β3-AR agonist MK-0634 (3) exhibited efficacy in humans for the treatment of OAB, but development was discontinued due to unacceptable structure-based toxicity in preclinical species. Optimization of a series of second-generation pyrrolidine-derived β3-AR agonists included reducing the risk for phospholipidosis, the risk of formation of disproportionate human metabolites, and the risk of formation of high levels of circulating metabolites in preclinical species. These efforts resulted in the discovery of vibegron, which possesses improved druglike properties and an overall superior preclinical profile compared to MK-0634. Structure–activity relationships leading to the discovery of vibegron and a summary of its preclinical profile are described.
| Reference | ||
|---|---|---|
| 1 | H.P. Kaiser, et al., “Catalytic Hydrogenation of Pyrroles at Atmospheric Pressure“, J. Org. Chem., vol. 49, No. 22, p. 4203-4209 (1984). | |
ClinicalTrials.gov Web Site 2011, April 28
| WO2011043942A1 * | Sep 27, 2010 | Apr 14, 2011 | Merck Sharp & Dohme Corp. | Combination therapy using a beta 3 adrenergic receptor agonist and an antimuscarinic agent |
| US20090253705 * | Apr 2, 2009 | Oct 8, 2009 | Richard Berger | Hydroxymethyl pyrrolidines as beta 3 adrenergic receptor agonists |
| US20110028481 * | Apr 2, 2009 | Feb 3, 2011 | Richard Berger | Hydroxymethyl pyrrolidines as beta 3 adrenergic receptor agonists |
| Citing Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| US8642661 | Aug 2, 2011 | Feb 4, 2014 | Altherx, Inc. | Pharmaceutical combinations of beta-3 adrenergic receptor agonists and muscarinic receptor antagonists |
| US8653260 | Jun 20, 2012 | Feb 18, 2014 | Merck Sharp & Dohme Corp. | Hydroxymethyl pyrrolidines as beta 3 adrenergic receptor agonists |
| US20120202819 * | Sep 27, 2010 | Aug 9, 2012 | Merck Sharp & Dohme Corporation | Combination therapy using a beta 3 adrenergic receptor agonists and an antimuscarinic agent |
| US20020028835 | Jul 12, 2001 | Mar 7, 2002 | Baihua Hu | Cyclic amine phenyl beta-3 adrenergic receptor agonists |
| US20070185136 | Feb 2, 2007 | Aug 9, 2007 | Sanofi-Aventis | Sulphonamide derivatives, their preparation and their therapeutic application |
| US20110028481 | Apr 2, 2009 | Feb 3, 2011 | Richard Berger | Hydroxymethyl pyrrolidines as beta 3 adrenergic receptor agonists |
| WO2003072572A1 | Feb 17, 2003 | Sep 4, 2003 | Jennifer Anne Lafontaine | Beta3-adrenergic receptor agonists |
|
8-22-2012
|
Hydroxymethyl pyrrolidines as [beta]3 adrenergic receptor agonists
|
////////////C1CC(NC1CC2=CC=C(C=C2)NC(=O)C3CCC4=NC=CC(=O)N34)C(C5=CC=CC=C5)O
LANOCONAZOLE
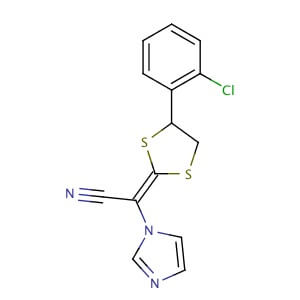
Lanoconazole
- Latoconazole, Lanoconazole, TJN-318, NND-318, Astat,
Nihon Nohyaku (Originator), Tsumura (Licensee)
| Synonym: | 2-[4-(2-Chlorophenyl)-1,3-dithiolan-2-ylidene]-2-imidazol-1-yl-acetonitrile |
| Application: | An antifungal compound |
| CAS Number: | 101530-10-3 |
| Molecular Weight: | 319.83 |
| Molecular Formula: | C14H10ClN3S2 |
Brief background information
| Appearance: | Crystalline |
| Physical State: | Solid |
| Solubility: | Soluble in chloroform, and methanol. Insoluble in water. |
| Storage: | Store at -20° C |
| Melting Point: | 129-132 °C |
| Boiling Point: | ~477.6 °C at 760 mmHg (Predicted) |
| Density: | ~1.4 g/cm3 (Predicted) |
| Refractive Index: | n20D 1.73 (Predicted) |
| pK Values: | pKb: 3.76 (Predicted) |
| WGK Germany: | 3 |
| RTECS: | NI3393500 |
| PubChem CID: | 3002820 |
| Merck Index: | 14: 5357 |
| MDL Number: | MFCD00865590 |
| Beilstein Registry: | 4819111 |
| Salt | ATC | Formula | MM | CAS |
|---|---|---|---|---|
| – | D01 | C 14 H 10 ClN 3 S 2 | 319.84 g / mol | 101530-10-3 |

Application
-
antifungal
Synthesis pathway
| Synthesis a) |
|---|
  |
Trade Names
| Country | Trade name | Manufacturer |
|---|---|---|
| Japan | Astatine | Tsumura |
| Ukraine | No | No |
Formulations
-
1% cream;
-
1% ointment;
-
1% solution
Links
-
EP 218 736 (Nihon Nohyaku; EP-prior. 9.10.1985).
1. Oka, H., et al., 1992. Therapeutic efficacy of latoconazole in formulations of clinical use on experimental dermatophytosis in guinea pigs. Arzneimittel-Forschung. 42(3): 345-9. PMID: 1497697
2. Niwano, Y., et al., 1994. Therapeutic efficacy of lanoconazole, a new imidazole antimycotic agent, for experimental cutaneous candidiasis in guinea pigs. Antimicrobial agents and chemotherapy. 38(9): 2204-6. PMID: 7811048
3 http://aac.asm.org/content/38/9/2204.full.pdf
References 1. Seo, A., Kanno, H., Hasegawa, N. et al. (Nihon Nohyaku Co., Ltd.). Antimycotic agent and fungicidal agent. US 4738976. 2. Seo, A ., Sugano, H., Hasegawa, C., Ikeda, K., Munechica, Y., Konoe, T., Konaka, M. (Nihon Nohyaku Co., Ltd.). Antifungal agent. JP 87093227. 3. Seo , A., Sugano, H., Hasegawa, C., Ikeda, K., Nishimura, A., Miyashiro, Y. (Nihon Nohyaku Co., Ltd.). Non-medicinal bactericidal agents and method for their preparation. JP 87093204. 4. Seo, A., Sugano, H., Hasegawa, C., Miyashiro, Y., Nishimura, A., Ikeda, K. (Nihon Nohyaku Co., Ltd.). Ketene S, S-acetals. JP 85218387. 5. Seo, A., Kanno, H., Hasegawa, N. et al. (Nihon Nohyaku Co., Ltd.). A novel ketene S, S-acetal deriv., a process for manufacturing thereof and a method for curing mycosis by administering it. EP 218736.
MAKE IN INDIA
http://makeinindia.com/sector/pharmaceuticals/
Read all about Organic Spectroscopy on ORGANIC SPECTROSCOPY INTERNATIONAL 



Metformin, thyroid-pituitary axis, diabetes mellitus, and metabolism
Leaders in Pharmaceutical Business Intelligence Group, LLC, Doing Business As LPBI Group, Newton, MA
Metformin, thyroid-pituitary axis, diabetes mellitus, and metabolism
Larry H, Bernstein, MD, FCAP, Author and Curator
and Aviva Lev-Ari, PhD, RN
The following article is a review of the central relationship between the action of
metformin as a diabetic medication and its relationship to AMPK, the important and
essential regulator of glucose and lipid metabolism under normal activity, stress, with
its effects on skeletal muscle, the liver, the action of T3 and more.
We start with a case study and a publication in the J Can Med Assoc. Then we shall look
into key literature on these metabolic relationships.
Part I. Metformin , Diabetes Mellitus, and Thyroid Function
Hypothyroidism, Insulin resistance and Metformin
May 30, 2012 By Janie Bowthorpe
The following was written by a UK hypothyroid patient’s mother –
Sarah Wilson.
My daughter’s epilepsy is triggered by unstable blood sugars. And since taking
Metformin to control her blood sugar, she has significantly reduced the number of
seizures. I have been doing research and read numerous academic medical journals,
which got me thinking about natural thyroid hormone and Hypothyroidism. My hunch
was that when patients develop hypothyroid symptoms, they are actually becoming
insulin resistant (IR). There are many symptoms in common between women with
polycystic ovaries and hypothyroidism–the hair loss, the weight gain, etc.
(http://insulinhub.hubpages.com/hub/PCOS-and-Hypothyroidism).
A hypothyroid person’s body behaves as if it’s going into starvation mode and so, to
preserve resources and prolong life, the metabolism changes. If hypothyroid is prolonged
or pronounced, then perhaps, chemical preservation mode becomes permanent even
with the reintroduction of thyroid hormones. To get back to normal, they need
a “jump-start” reinitiate a higher rate of metabolism. The kick start is initiated through
AMPK, which is known as the “master metabolic regulating enzyme.”
(http://en.wikipedia.org/wiki/AMP-activated protein kinase).
Guess what? This is exactly what happens to Diabetes patients when Metformin is
introduced. http://en.wikipedia.org/wiki/Metformin
Suggested articles: http://www.springerlink.com/content/r81606gl3r603167/ and
http://onlinelibrary.wiley.com/doi/10.1111/j.1365-2265.2011.04029.x/pdf
Note the following comments/partial statements:
“Hypothyroidism is characterized by decreased insulin responsiveness”;
“the pivotal regulatory role of T3 in major metabolic pathways”.
The community knows that T3/NTH (natural thyroid hormone [Armour]) makes
hypothyroid patients feel better – but the medical establishment is averse to T3/NTH
(treating subclinical hypoT (T3/T4 euthyroid) with natural dessicated thyroid (NDT).
The medical establishment might find an alternative view about impaired metabolism
more if shown real proof that the old NDT **was/is** having the right result –i.e., the
T3 is jump-starting the metabolism by re-activating AMPK.
If NDT also can be used for hypothyroidism without the surmised “dangers” of NTH,
then they should consider it. [The reality in the choice is actually recombinant TH
(Synthroid)]. Metformin is cheap, stable and has very few serious side effects. I use the
car engine metaphor, and refer to glucose as our petrol, AMPK as the spark plug and
both T3 and Metformin as the ignition switches. Sometimes if you have flat batteries in
the car, it doesn’t matter how much you turn the ignition switch or pump the petrol
pedal, all it does is flatten the battery and flood the engine.
Dr. Skinner in the UK has been treating “pre-hypothyroidism” the way that some
doctors treat “pre-diabetes”. Those hypothyroid patients who get treated early
might not have had their AMPK pathways altered and the T4-T3 conversion still works.
There seems to be no reason why thyroid hormone replacement therapy shouldn’t
logically be given to ward off a greater problem down the line.
It’s my belief that there is clear and abundant academic evidence that the AMPK/
Metformin research should branch out to also look at thyroid disease.
Point – direct T3 is kicking the closed -down metabolic process back into life,
just like Metformin does for insulin resistance.
http://www.hotthyroidology.com/editorial_79.html
There is serotonin resistance! http://www.ncbi.nlm.nih.gov/pubmed/17250776
Metformin Linked to Risk of Low Levels of Thyroid Hormone
CMAJ (Canadian Medical Association Journal) 09/22/2014
Metformin, the drug commonly for treating type 2 diabetes,
- is linked to an increased risk of low thyroid-stimulating hormone
(TSH) levels - in patients with underactive thyroids (hypothyroidism),
according to a study in CMAJ (Canadian Medical Association Journal).
Metformin is used to lower blood glucose levels
- by reducing glucose production in the liver.
previous studies have raised concerns that
- metformin may lower thyroid-stimulating hormone levels.
Study characteristics:
- Retrospective long-term
- 74 300 patient who received metformin and sulfonylurea
- 25-year study period.
- 5689 had treated hypothyroidism
- 59 937 had normal thyroid function.
Metformin and low levels of thyroid-stimulating hormone in
patients with type 2 diabetes mellitus
Jean-Pascal Fournier, Hui Yin, Oriana Hoi Yun Yu, Laurent Azoulay +
Centre for Clinical Epidemiology (Fournier, Yin, Yu, Azoulay), Lady Davis Institute,
Jewish General Hospital; Department of Epidemiology, Biostatistics and Occupational
Health (Fournier), McGill University; Division of Endocrinology (Yu), Jewish General
Hospital; Department of Oncology (Azoulay), McGill University, Montréal, Que., Cananda
CMAJ Sep 22, 2014, http://dx.doi.org:/10.1503/cmaj.140688
Background:
- metformin may lower thyroid-stimulating hormone (TSH) levels.
Objective:
- determine whether the use of metformin monotherapy, when compared with
sulfonylurea monotherapy, - is associated with an increased risk of low TSH levels(< 0.4 mIU/L)
- in patients with type 2 diabetes mellitus.
Methods:
- Used the Clinical Practice Research Datalink,
- identified patients who began receiving metformin or sulfonylurea monotherapy
between Jan. 1, 1988, and Dec. 31, 2012. - 2 subcohorts of patients with treated hypothyroidism or euthyroidism,
followed them until Mar. 31, 2013.
- Used Cox proportional hazards models to evaluate the association of low TSH
levels with metformin monotherapy, compared with sulfonylurea monotherapy,
in each subcohort.
Results:
- 5689 patients with treated hypothyroidism and 59 937 euthyroid patients were
included in the subcohorts.
For patients with treated hypothyroidism:
- 495 events of low TSH levels were observed (incidence rate 0.1197/person-years).
- 322 events of low TSH levels were observed (incidence rate 0.0045/person-years)
in the euthyroid group.
- metformin monotherapy was associated with a 55% increased risk of low TSH
levels in patients with treated hypothyroidism (incidence rate 0.0795/person-years
vs.0.1252/ person-years, adjusted hazard ratio [HR] 1.55, 95% confidence
interval [CI] 1.09– 1.20), compared with sulfonylurea monotherapy, - the highest risk in the 90–180 days after initiation (adjusted HR 2.30, 95% CI
1.00–5.29). - No association was observed in euthyroid patients (adjusted HR 0.97, 95% CI 0.69–1.36).
Interpretation: The clinical consequences of this needs further investigation.
| Crude and adjusted hazard ratios for suppressed thyroid-stimulating hormone levels (< 0.1 mIU/L) associated with the use metformin monotherapy, compared |
View original post 1,073 more words
More than 40 plant-based compounds can turn on genes that slow the spread of cancer
07 SEP 0212
WSU researcher documents links between nutrients, genes and cancer spread
More than 40 compounds turn on genes slowing metastasis
PULLMAN, Wash.—More than 40 plant-based compounds can turn on genes that slow the spread of cancer, according to a first-of-its-kind study by a Washington State University researcher.
Gary Meadows, WSU professor and associate dean for graduate education and scholarship in the College of Pharmacy, says he is encouraged by his findings because the spread of cancer is most often what makes the disease fatal. Moreover, says Meadows, diet, nutrients and plant-based chemicals appear to be opening many avenues of attack.
“We’re always looking for a magic bullet,” he says. “Well, there are lots of magic bullets out there in what we eat and associated with our lifestyle. We just need to take advantage of those. And they can work together.”
Meadows started the study, recently published online in…
View original post 523 more words
(S)-Atenolol

(S)-atenolol
- Description
Selective β1 adrenoceptor antagonist
- Biological descriptionSelective β1 adrenoceptor antagonist. Orally active. Limited ability to cross the blood-brain barrier. Antihypertensive activity in vivo.
Properties
- Chemical name(S)-(-)-4-[2-Hydroxy-3-[(1-methylethyl)amino]propoxy]benzeneacetamide
- Molecular Weight 266.34
- Molecular formula C14H22N2O3
- CAS Number 93379-54-5
1H NMR (DMSO-d6): δ 0.99 (d, J=7 Hz, 6H, 2×CH3), 2.60 (m, 1H, CH), 2.74 (m, 2H, CH2), 3.27 (s, 2H, CH2), 3.88 (m, 4H, CH2, CH, NH), 6.83 (d, J=8 Hz, 2H, Ar—H), 7.14 (d, J=8 Hz, 2H, Ar—H), 7.40 (bs, 1H).
22.01, 22.09,
41.26, 48.39, 49.38, 67.73, 70.58, 114.16, 128.41, 129.93, 157.17, 172.59 ppm.
The compound (R,S)-atenolol (4-[2-hydroxy-3-[(1-methylethyl)amino]propoxy]-benzeneacetamide) is useful as a β-adrenegic blocker for the treatment of angina pectoris, arrhythmia and hypertension. It is known that atenolol is a 1-aryloxy-3-aminopropane-2-ol derivative wherein the hydroxy bearing carbon is an asymmetric carbon and hence exists as R- and S-isomers. It is also known that the S-isomer is particularly useful as a β-adrenegic blocker in view of its superior pharmacological activities. It is reported that S-atenolol has hypotensive activity and activity on brachycardia (A. A. Pearson, T. E. Gaffney, T. Walle, P. J. Privitera; J. Pharmacol. Exp. Ther., 250(3), 759, 1989).
In prior art, the optical resolution of racemic atenolol has been studied to obtain the desired optically active atenolol, however, any practical method has not been reported so far. It is also reported that the diastereomers of atenolol having high purity is obtained from racemic mixture by using (R,R)-O,O-di-toluoyltartaric acid anhydride (M. J. Wilson et al., J. Chromatogr. (NLD) 431 (1), 222–227, 1988). However, this method is not suitable for large scale production of optically active atenolol as it requires a large volume of solvent and further it is technically very troublesome to recycle (R,R)-O,O-di-toluoyltartaric acid anhydride.
Another method of preparing optically active atenolol has been proposed in JP-A-50-77331 and DE-A-2453324:
Wherein Z is halogen atom or sulphonyloxy group, and * means asymmetric carbon.
However, this process has some disadvantages as this process requires several steps for obtaining optically active S-atenolol stating from D-manitol; moreover the yield of S-atenolol by this process is less than 50% and the optical purity is just about 80% ee.
Another method for the preparation of S-atenolol has been reported in U.S. Pat. No. 5,223,646 which consists of reacting sodium salt of 4-carbamoylmethylphenol with R-epichlorohydrin at 0° to 35° C. to obtain an intermediate—an optically active glycidyl ether and then reacting the optically active intermediate glycidyl ether with isopropylamine to obtain S-atenolol (see also EP-435068 A2; EP-605384; JP 03077856 A2).
It has also been reported that the above procedure gives optically active glycidyl ether and atenolol of 90–96% ee optical purity. According to this report, the optical purity of atenolol may be enhanced to 98% or higher, if the intermediate optically active glycidyl ether is repeatedly recrystallised from a suitable solvent.
It has also been reported that the optically active atenolol in an optical purity of 98% or higher can be produced from atenolol of lower optical purity by converting it to its salt with Bronsted’s acid (K. Kazuhiro; T. Yosikazu; F. Yoshiro; Y. Hiroshi; O. Junzo, Chem. Pharm. Bull., 46(3), 505–507, 1998).
The separation of the atenolol salt having higher optical purity (>98% ee) is carried out by dissolving the atenolol salt having lower optical purity in a solvent, precipitating solid materials having a high content of racemic atenolol salt, and then isolating the desired atenolol salt having higher optical purity (>98% ee) by solid-liquid separation method. The optically active salt having high optical purity is then subjected to removal of acid moiety to isolate the desired optically active atenolol in free form. Though this process yields atenolol of higher optical purity, it involves salt formation and tedious separation of racemic salt from an optically active salt, which leads to the lower yields of desired optically active atenolol. Further, the salt has to be converted to free atenolol either by neutralisation or using ion exchange resins. Thus, this process gives lower overall yield of the desired optically active atenolol is low.
There is therefore a need to provide a process whereby S-atenolol may be obtained in high yield and high optical purity.
Emcure Pharmaceuticals Limited
http://www.google.com/patents/US6982349
Satish Ramanlal Mehta, Baburao Manikroa Bhawal, Vishnu Hari Deshpande, Mukund Keshav Gurjar
Accordingly, the present invention provides a process for the preparation of (S)-atenolol (1), which comprises the steps of:
-
- a) reacting a phenol of formula 2:
with an (R)-epichlorohydrin of formula (3):
in presence of an alkali metal hydroxide and a quaternary ammonium salt as phase transfer catalyst (PTC) in an aqueous solution at a temperature in a range of −10° C. to 0° C. to obtain optically active intermediate glycidyl ether of formula 4:
- b) reacting the optically active intermediate glycidyl ether (4) with isopropylamine at 10° to 40° C. to obtain (S)-atenolol of the formula 1:
in good chemical yield and high optical purity (>99 ee).
- a) reacting a phenol of formula 2:
One major advantage of this process is that S-atenolol may be obtained directly without going through the cumbersome step of recrystallization or additional salt formation step, as in the prior art.
The aqueous alkali metal hydroxide used in the process is selected from sodium hydroxide or potassium hydroxide and is used as aqueous solution in 1 to 1.5 moles to 1 mole of the phenol 2. The (R)-epichlorohydrin (3) used in the process is preferably of high optical purity and used in an amount of 1 to 3 moles, more preferably 1 to 1.6 moles, to 1 mole of phenol (2).
The quaternary ammonium salt has the formula:
R1R2R3R4N+X−
Wherein R1, R2, R3 and R4 are same or different, each an alkyl group having 1 to 16 carbon atoms (e.g. methyl, ethyl, propyl butyl etc), phenyl or benzyl, X is chlorine, bromine, iodine, hydrogen sulphate or hydroxyl group. The amount of quaternary ammonium salt used is 0.001 to 2% by weight of phenol (2).
The Applicant studied the reaction temperature extensively and found that it plays an important role in deciding optical purity of (S)-atenolol (1) formed via optically active glycidyl ether. When the reaction of phenol (2) and (R)-epichlorohydrin is carried out at 5° C. or at any other higher temperature, (S)-atenolol (1) of a lower optical purity was obtained via optically active glycidyl ether, as for example in EP 435068.
The Applicant, after studying the prior art processes found that during the course of these reactions, the phenoxide (or phenol) attacks the C-1 carbon atom of (R)-epichlorohydrin with the expulsion of chloride to yield (R)-glycidyl ether, which on reaction with isopropyl amine gives (R)-atenolol. The original epoxide ring remains unchanged in the reaction.
Thus, the reaction of phenol (2) at carbon centre C-1 of (R)-epichlorohydrin by nucleophilic displacement of chlorine leads to the formation of undesired (R)-atenolol via optically active (R)-glycidyl ether as a side product, which accounts for the low yield of optically active S-atenolol in the prior art.
The Applicant then conducted this reaction at a lower temperature and found to their surprise that S-atenolol could be obtained in high yield. The reason is that during the course of reaction, the phenoxide (or phenol) ion attacks the C-3 carbon atom of (R)-epichlorohydrin and opens the epoxide ring. The new epoxide ring formation takes place by the attack of O− on C-3 carbon with expulsion of chloride to give (S)-glycidyl ether, which on reaction with isopropyl amine gives (S)-atenolol. Thus, the reaction of phenol (2) at carbon centre C-3 of (R)-epichlorohydrin leads to the formation desired (S)-atenolol (1) as a major product via optically active glycidyl ether (4).
The lower optical purity in (S)-atenolol formation in the prior art may therefore be on account of the slow reaction rate at carbon atom 1 and the high yield of S-atenolol obtained by the process of the present invention may be due to the reaction at carbon atom 3 of (R)-epichlorohydrin (3). Both these reactions occurring on different atoms are shown as path ‘a’ and path ‘b’ in the following scheme herebelow.
Path ‘a’ is the process of the present invention whereas path ‘b’ is the process of the prior art.
EXAMPLE 1A mixture of (R)-epichlorohydrin ([α]D 25: −35.1 (neat), 138.75 g, 1.5 mole) and water (82 ml) was cooled to −7° C. and to this cold reaction mixture is added a solution of 4-hydroxyphenyl acetamide of formula 1 (151.00 g, 1 mole) and benzyltrimethylammonium chloride (1.3 g) in sodium hydroxide [40 g, 1 mole; dissolved in water (670 ml)] with stirring over a period of 3 hrs. maintaining the temperature at −7° C. to −5° C. The reaction mixture is then stirred further at −7° C. to −5° C. for 50 hrs. The precipitated solid is filtered, washed with water and dried at 60° C. to give 176 g of a mixture of S-glycidyl ether of formula 4 and S-chlorohydrin of formula 5 in about 3:2 ratio. m.p. 159–161° C.
EXAMPLE 2A mixture of isopropylamine (1.1 kg) and water (200 ml) is cooled to 10° C. and a mixture of S-glycidyl ether of formula 4 and S-chlorohydrin of formula 5 obtained in Example 1 (176 g) is added to it in lots maintaining temperature between 10 to 15° C. over a period of 3 hrs. The reaction is then stirred further for another 10 hr. The excess of isopropylamine is removed by distillation and the residue was treated with the water. The slurry so obtained is acidified with 5N HCl to pH 2.0. The resulting solution is then filtered, washed with water. The filtrate is basified with 2N NaOH to pH 11.7 and precipitated solid is filtered washed with water and dried to get (S)-atenolol (206 g, 91%) in 99.1% ee when analysed by using Chiracel OD column.
m.p. 152–153° C.
[α]D 25: −17.2 (c=1.0, 1N HCl).
IR: νmax 3352, 3168, 1635, 1242 cm−1.
1H NMR (DMSO-d6): δ 0.99 (d, J=7 Hz, 6H, 2×CH3), 2.60 (m, 1H, CH), 2.74 (m, 2H, CH2), 3.27 (s, 2H, CH2), 3.88 (m, 4H, CH2, CH, NH), 6.83 (d, J=8 Hz, 2H, Ar—H), 7.14 (d, J=8 Hz, 2H, Ar—H), 7.40 (bs, 1H).
13C NMR (DMSO-d6): 22.01, 22.09, 41.26, 48.39, 49.38, 67.73, 70.58, 114.16, 128.41, 129.93, 157.17, 172.59 ppm.
OTHER INFO
Despite both optical isomers being bioactive, as briefly mentioned in the Physical section, recent studies have shown that the S-ATENOLOL isomer was found to avoid the occasional side effect of an excessively lowered heart rate sometimes encountered with the racemate. The following steps are involved with the isolation of each enantiomer from 1-[p-[ (butoxy-carbonyl)methyl]phenoxy]-3-chloropropan-2-ol (7) using Lipase Catalysis.
The subsequent step highlights the production of S-ATENOLOL via (S-)1-[p-[ (butoxy-
carbonyl)methyl]phenoxy]-3-chloropropan-2-ol (8). This is fundamentally achieved using lipase from Pseudomonas Cepacia in a mixture of acetic anhydride and DIPE.
STEP 4 (b)

Whereas lipase from Pseudomonas Cepacia was used to isolate the S-ATENOLOL ISOMER, lipase from Candida Cylindracea is utilised in the production of the R + ATENOLOL ISOMER. The same principle applies in each isomeric scenario as demonstated below.
STEP 5 (a)

Acetic anhydride may also be used as a substitute for 1-butanol however, its inherent toxicity led to one opting for 1-butanol. Even though 1-butanol is harmful in its own right, on a relative scale is was the most suitable and effective alternative evolving an approximate 94 %conversion.
STEP 5 (b)

Yet again, greater than 95 % conversion is achieved after purifying the precipitate by treating(R) 1-[p-[(butoxy-carbonyl)methyl]phenoxy]-3-chloropropan-2-ol (10) with 2-methyl-ethanamine. This is then followed by addition of ammonium hydroxide in methanol and finally single recrystallisation in ethyl acetate.
References for (S)-(-)-Atenolol (ab120856)
This product has been referenced in:
- Agon P et al. Permeability of the blood-brain barrier for atenolol studied by positron emission tomography. J Pharm Pharmacol 43:597-600 (1991). Read more (PubMed: 1681079) »
- Tsuchihashi H et al. Characteristics of 125I-iodocyanopindolol binding to beta-adrenergic and serotonin-1B receptors of rat brain: selectivity of beta-adrenergic agents. Jpn J Pharmacol 52:195-200 (1990). Read more (PubMed: 1968985) »
| Cited Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| US4085136 | Jun 9, 1976 | Apr 18, 1978 | Imperial Chemical Industries Limited | Adrenergic blocking agents |
| US5223646 | Apr 21, 1992 | Jun 29, 1993 | Daiso Company, Ltd. | Process for producing optically active atenolol and intermediate thereof |
| JPH0377856A | Title not available | |||
| JPH01102072A * | Title not available | |||
| JPH04198175A * | Title not available |
| Cited Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| US4085136 | Jun 9, 1976 | Apr 18, 1978 | Imperial Chemical Industries Limited | Adrenergic blocking agents |
| US5223646 | Apr 21, 1992 | Jun 29, 1993 | Daiso Company, Ltd. | Process for producing optically active atenolol and intermediate thereof |
| JPH0377856A | Title not available | |||
| JPH01102072A * | Title not available | |||
| JPH04198175A * | Title not available |
Nicox stock leaps on positive Ph III glaucoma drug data , 英文名称

- 4- (nitrooxy) butyl (5Z) -7 – {(1R, 2R, 3R, 5S) -3,5-dihydroxy-2 – [(3R) -3-hydroxy-5-phenylpentyl] cyclopentyl} hept-5- enoate
- CAS No.860005-21-6
- Formula C 27 H 41 NO 8
The firms have published top-line results from the pivotal Phase 3 studies conducted with Vesneo (latanoprostene bunod) for the reduction of intraocular pressure in patients with glaucoma or ocular hypertension. The drug is a nitric oxide-donating prostaglandin F2-alpha analog licensed by Nicox to Bausch + Lomb.
Read more at: http://www.pharmatimes.com/Article/14-09-25/Nicox_stock_leaps_on_positive_Ph_III_glaucoma_drug_data.aspx#ixzz3ETxo7SBd
prostaglandin nitrooxyderivatives, pharmaceutical compositions containing them and their use as drugs for treating glaucoma and ocular hypertension. Glaucoma is optic nerve damage, often associated with increased intraocular pressure (IOP), that leads to progressive, irreversible loss of vision. . Almost 3 million people in the United States and 14 million people worldwide have glaucoma; this is the third leading cause of blindness worldwide. Glaucoma occurs when an imbalance in production and drainage of fluid in the eye (aqueous humor) increases eye pressure to unhealthy levels. It is known that elevated IOP can be at least partially controlled by administering drugs which either‘ reduce the production of aqueous humor within the eye or increase the fluid drainage, such as beta-blockers, α- agonists, ■ ‘ cholinergic agents, carbonic anhydrase inhibitors, or prostaglandin analogs. . Several side effects are associated with the drugs conventionally used to treat glaucoma. . ■ Topical beta-blockers show serious pulmonary side effects, depression, fatigue,’ confusion, impotence, hair loss, heart failure and bradycardia. Topical -agonists have a fairly high incidence of allergic, .or toxic reactions; topical cholinergic agents (miotics) can cause visual side effects. The side effects associated with oral carbonic anhydrase inhibitors include fatigue, anorexia, depression, paresthesias and serum■ electrolyte abnormalities (The Merck Manual of Diagnosis and Therapy, Seventeenth Edition, M. H. Beers and R. Berkow Editors, Sec. 8, Ch. 100) . Finally, the topical prostaglandin analogs (bimatoprost, latanoprost, travoprost and unoprostone) ‘ used in the treatment of glaucoma, can produce ocular side effects, such as increased pigmentation of the iris, ocular irritation, conjunctival hyperaemia, iritis, uveitis and macular oedema (Martindale, Thirty-third edition, p. 1.445) U.S. Pat. No. 3,922,293 describes monocarboxyacylates of prostaglandins F-type and their 15β isomers, at the C-9 position, and processes for preparing them; U.S. Pat. No. 6,417,228 discloses 13-aza prostaglandins having functional PGF2α receptor agonist activity and their use in treating glaucoma and ocular hypertension. WO 90/02553 • discloses the use ‘ of prostaglandins derivatives of PGA, PGB, PGE and PGF, in which the omega chain contains a ring structure, for the treatment of glaucoma or ocular hypertension. WO 00/51978 describes novel nitrosated and/or nitrosylated prostaglandins, ‘ • in ‘ particular novel derivatives of PGEi, novel compositions and their use for treating sexual dysfunctions. • : U.S.- Pat. No. 5,625,083 • discloses” ‘diriitroglycerol esters of prostaglandins which may‘ be used as vasodilators, antihypertensive cardiovascular agents- or bronchodilators . U.S. Pat. No. 6,211,233 discloses compounds of the general formula A-Xι-N02,‘ wherein A contains ‘a ■■ – prostaglandin residue, .in ‘particular .‘PGEi, and Xi • is a bivalent connecting bridge; .’and their use fo ‘ treating impotence. It is an object of the present invention to provide new derivatives of prostaglandins able not only to eliminate or at least reduce the side ■ effects associated with these compounds, but also to possess an improved pharmacological activity. It has been surprisingly found that prostaglandin nitroderivatives have a significantly improved overall profile as compared to native, prostaglandins both in terms of -wider pharmacological .activity and enhanced tolerability. In particular, it has been recognized that the prostaglandin nitroderivatives of the present invention can be employed for treating glaucoma and ocular hypertension. The compounds of the present invention are indicated for the reduction of intraocular pressure in patients with open-angle glaucoma or with chronic angle- closure glaucoma who underwent peripheral iridotomy or laser iridoplasty.
Latanoprostene bunod
 Currently in Phase 3 clinical development with Nicox’s partner Bausch + Lomb
Currently in Phase 3 clinical development with Nicox’s partner Bausch + Lomb
Latanoprostene bunod is a nitric oxide-donating prostaglandin F2-alpha analog in Phase 3 clinical development for the reduction of intraocular pressure in patients with glaucoma and ocular hypertension. It was licensed to Bausch + Lomb by Nicox in March 2010
Bausch + Lomb initiated a global Phase 3 program for latanoprostene bunod (previously known as BOL-303259-X and NCX 116) in January 2013. This pivotal Phase 3 program includes two separate randomized, multicentre, double-masked, parallel-group clinical studies, APOLLO andLUNAR, designed to compare the efficacy and safety of latanoprostene bunod administered once daily (QD) with timolol maleate 0.5% administered twice daily (BID) in lowering intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension.
The primary endpoint of both studies, which will include a combined total of approximately 800 patients, is the reduction in mean IOP measured at specified time points during three months of treatment. The Phase 3 studies are pivotal for U.S. registration and will be conducted in North America and Europe.
In July 2013, Bausch + Lomb initiated two additional studies in Japan: JUPITER (Phase 3) and KRONUS (Phase 1). A confirmatory efficacy study is expected to be required for the Japanese registration of latanoprostene bunod.
Phase 2b top-line results
A phase 2b study conducted by Bausch + Lomb with latanoprostene bunod met its primary efficacy endpoint and showed positive results on a number of secondary endpoints, including responder rate.
No new class of drugs has come to market for treating glaucoma since 1996, when the FDA approved the first prostaglandin analogue, latanoprost (Xalatan). That could change soon: Experts who follow drug development are hopeful that we’re on the brink of reaping the benefits of years of research.
“It’s been a decade and a half and counting since we’ve had new class of drugs to treat glaucoma. We’ve had formulary improvements and fixed combinations, but no novel agents,” said Louis B. Cantor, MD, at Indiana University. “We’ve gone through a long dry spell but are just beginning to see, in the last couple of years, exploration by pharma of some new types of drugs.” But, he added, “We don t know how well those will pan out.
The uncertainty about “panning out” involves both drug efficacy and marketplace issues. As Dr. Cantor said, “Prostaglandin analogues are pretty effective. For a company to go into the investment of developing a new class of drugs for glaucoma, they have to be better than prostaglandin analogues.
Andrew G. Iwach, MD, at the University of California, San Francisco, agreed: “This is a unique time period for glaucoma medications in that we have very good drugs, usually well tolerated. And they’ve gone generic. That’s important, because having such strong generic contenders out there makes it harder for drug companies to try to introduce new molecules into this arena. Specifically, the prostaglandin analogues have set a high bar. It’s hard to compete with them.
Given this barrier, what are the marketplace incentives for development? Sheer numbers, for a start: Ten thousand people a day turn 65, and this rate will continue for 18 years, Dr. Cantor said. “The number of people who are going to need treatment for glaucoma has already begun to increase substantially.
Even more important, “Despite all the advances, our medical therapy fails not only for compliance reasons, but just fails,” Dr. Cantor said. “We need to continue to have new alternatives for treatment that are more effective, that last longer, and that have simple dosing requirements.
Thus, any new drug that makes it from the bench to the clinic will be a welcome addition. “Obviously, we want new and better therapies. We still have no cure for glaucoma. And while half of all patients are treatable with one drug, half are not. So we still need additional therapies to treat glaucoma,” said Gary D. Novack, PhD, president of Pharmalogic Development.
……………………….
http://www.google.com/patents/EP1704141A1?cl=en
EXAMPLE 1 Synthesis of [1R- [l (Z) , 2α (R*) , 3α, 5α] ] -7- [3, 5-dihydroxy-2- (3-hydroxy-5-phenylpentyl) cyclopentyl] -5-heptenoic acid 4- (nitrooxy) butyl ester (compound 1)
I Synthetic Pathway ONO,
MW 72.11 MW 153.02 MW 198.02
MW 390.51 MW 507.62
II EXPERIMENTAL II.1 Preparation of 4-bromobutanol
Tetrahydrofuran (12.5 g – 173 mmol) was charged under nitrogen in a reactor cooled to 5-10 °C. Hydrogen bromide (7.0 g. – 86.5 mmol) was then added slowly and the reaction ■medium was stirred over a period of 4.5 hours at 5-10°C. The mixture was diluted with 22.5 g of cold water and the pH of this solution was adjusted to pH=5-7 by adding 27.65% sodium hydroxide (2.0 g) keeping the temperature at 5-10 °C. The solution was then extracted twice with dichloromethane (13.25 g) . The combined organic phases were washed with -25% brine (7.5 g) , adjusted to pH=6-7 with 27.65% sodium hydroxide and dried over magnesium sulfate. Dichloromethane was distilled off and crude 4-bromobutanol (10.3 g – 66.9 mmol) was obtained in a yield of about 77%. II.2 Preparation of 4-bromobutyl nitrate
In reactor cooled to -5 to 5°C, nitric acid fuming (8.5 g – 135 mmol) was slowly added to a solution of 98% sulfuric acid (13.0 g – 130 mmol) in dichloromethane (18.0 g – 212 mmol). 4-bromobutanol (10.2 g – 66.6 mmol) was then added to this mixture and the reaction medium was stirred at -5 to 5°C over a period of 2-5 hours. The mixture was poured into cold water (110 g) keeping the temperature between -5 °C and 3°C. After decantation, the upper aqueous phase was extracted with dichloromethane and the combined organic phases were washed with water, adjusted to pH=6-7 by addition of 27.65% sodium hydroxide, washed with brine and dried over magnesium sulfate. Dichloromethane was distilled off under vacuum and crude 4-bromobutyl nitrate (12.7 g – 64.1 mmol) was recovered in a yield of about 96%.
II.3 Preparation of [1R- [lα-(Z) , 2β (R*) , 3α, 5α] ] -7- [3, 5- dihydroxy-2- (3-hydroxy-5-phenylpentyl) cyclopentyl] -5- heptenoic acid 4- (nitrooxy) butyl ester
Latanoprost acid (97.7%, S-isomer <1%) (213mg, 0.54 mmol) was dis.solved in 5.0 g anhydrous DMF. K2C03 (206′ mg, 1.49 mmol), KI (77 mg, 0.46 mmol)‘ and ‘4-bromobutylnitrate (805 mg, .25% w/w in methylene chloride, 1.02 mmol) were added. The reaction mixture was heated and stirred on a rotary evaporator at 45-50°C. fter 1.5. hour, TLC (Si, ■ CH2Cl2-MeOH, 5%) showed -no – starting acid. . . .. The reaction mixture was diluted with 100 ml ethyl acetate, washed with brine (3 x 50 ml), dried over MgS04 and evaporated to give yellowish oil (420 mg) .
5 1H NMR/13C NMR showed target molecule as a major product together with some starting 4-bromobutylnitrate and DMF. HPLC showed no starting acid. Residual solvent, 4- bromobutylnitrate and target ester were the main peaks. Butylnitrate ester showed similar UV spectrum as0 latanoprost and relative retention time was as expected.
Instrument: Bruker 300 MHz Solvent : CDC13 -5 H-NMR (CDC13) δ: 7.29-7.19 (5H, m, Ar) ; 5.45 (IH, m. CH=CH) ; 5.38 (IH, m, CH=CH) ;. 4.48 (2H, t, CH2-ON02) ; 4.18 (IH, m, CH-OH); 4.10 (2H, t, C00CH2) ; 3.95 (IH, m, CH-OH); 3.68 (IH, m, CH-OH); 2.87-2.60 (2H, ) ; 2.35 (2H, t) ; 2.25 (2H,m) ; 2.13 (2H,m) ; 1.90-1.35 (16H, m) .0 13C-NMR (CDCI3) ppm: 173.94 (C=0) ; 142.14; 129.55 (C5); 129.50 (C6) ; 128.50; 125.93 78.80 (Cu) ; 74.50 (C9) ; 72.70 (C-0N02) ; 71.39 (Ci5) ; 63.57; 52.99 (C12) 51.99 (C8); 41.30 (C10) ; 39.16 (Ci6) ; 33.66; 32.21; 29.73; 27.04; 26.70;5 25.04; 24.91; 23.72; 15.37.
 |
|
| Trabecular meshwork structure. The colors in this drawing delineate the layers of the TM. |
 |
|
| Hyperemia. A side effect that emerged in trials of ROCK inhibitors is hyperemia; researchers are exploring different strategies to reduce it. |
LEXANOPADOL, For Treatment of acute and chronic pain requiring opioid analgesia
LEXANOPADOL
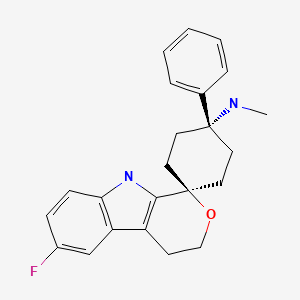
trans-6′-Fluoro-N-methyl-4-phenyl-4′,9′-dihydro-3’H-spiro(cyclohexane-1,1′-pyrano(3,4-b)indol)-4-amine
PRONUNCIATION lex” an oh’ pa dol
THERAPEUTIC CLAIM Treatment of acute and chronic pain requiring opioid analgesia
CHEMICAL NAMES
1. Spiro[cyclohexane-1,1′(3’H)-pyrano[3,4-b]indol]-4-amine, 6′-fluoro-4′,9′-dihydro-N-methyl-4-phenyl-, trans-
2. Trans-6′-fluoro-N-methyl-4-phenyl-4′,9′-dihydro-3’H-spiro[cyclohexane-1,1′-pyrano[3,4-b]indol]-4-amine
3. Trans -6’-fluoro-4’,9’-dihydro-N-methyl-4-phenyl-spiro[cyclohexane-1,1’(3’H)-pyrano[3,4-b]indol]-4-amine
MOLECULAR FORMULA C23H25FN2O
MOLECULAR WEIGHT 364.5
SPONSOR Grűnenthal GmbH
CODE DESIGNATIONS GRT6006, GRT13106G
CAS REGISTRY NUMBER 1357348-09-4
UNIIDZ4NDW1LZX
WHO NUMBER 9765
gbk
The heptadecapeptide nociceptin is an endogenous ligand of the ORL1 (opioid receptor-like) receptor (Meunier et al., Nature 377, 1995, p. 532-535), which belongs to the family of opioid receptors and is to be found in many regions of the brain and spinal cord, and has a high affinity for the ORL1 receptor. The ORL1 receptor is homologous to the μ, κ and δ opioid receptors and the amino acid sequence of the nociceptin peptide has a marked similarity to those of the known opioid peptides. The receptor activation induced by nociceptin leads, via coupling with Gi/o proteins, to an inhibition of adenylate cyclase (Meunier et al., Nature 377, 1995, p. 532-535).
The nociceptin peptide shows a pronociceptive and hyperalgesic activity after intercerebroventicular administration in various animal models (Reinscheid et al., Science 270, 1995, p. 792-794). These findings can be explained as an inhibition of stress-induced analgesia (Mogil et al., Neuroscience 75, 1996, p. 333-337). In this connection, it has also been possible to demonstrate an anxiolytic activity of nociceptin (Jenck et al., Proc. Natl. Acad. Sci. USA 94, 1997, 14854-14858).
On the other hand, it has also been possible to demonstrate an antinociceptive effect of nociceptin in various animal models, in particular after intrathecal administration. Nociceptin has an antinociceptive action in various pain models, for example in the tail flick test in the mouse (King et al., Neurosci. Lett., 223, 1997, 113-116. It has likewise been possible to demonstrate an antinociceptive action of nociceptin in models for neuropathic pain, which is of particular interest inasmuch as the activity of nociceptin increases after axotomy of spinal nerves. This is in contrast to conventional opioids, the activity of which decreases under these conditions (Abdulla and Smith, J. Neurosci., 18, 1998, p. 9685-9694).
The ORL1 receptor is moreover also involved in regulation of further physiological and pathophysiological processes. These include, inter alia, learning and memory development (Manabe et al., Nature, 394, 1997, p. 577-581), audition (Nishi et al., EMBO J., 16, 1997, p. 1858-1864) and numerous further processes. A review article by Cabo et al. (Br. J. Pharmacol., 129, 2000, 1261-1283) gives an overview of the indications or biological processes in which the ORL1 receptor plays a role or with high probability could play a role. This mentions, inter alia: analgesia, stimulation and regulation of food intake, influence on μ-agonists, such as morphine, treatment of withdrawal symptoms, reduction in the addiction potential of opioids, anxiolysis, modulation of motor activity, impaired memory, epilepsy; modulation of neurotransmitter secretion, in particular glutamate, serotonin and dopamine, and therefore neurodegenerative diseases; influencing of the cardiovascular system, initiation of an erection, diuresis, anti-natriuresis, electrolyte balance, arterial blood pressure, water retention diseases, intestinal motility (diarrhea), relaxing effects on the respiratory tract, micturation reflex (urinary incontinence). The use of agonists and antagonists as anoretics, analgesics (also in co-administration with opioids) or nootropics is furthermore discussed.
The possible uses of compounds which bind to the ORL1 receptor and activate or inhibit this are correspondingly diverse. Alongside this, however, opioid receptors, such as the μ-receptor, but also the other sub-types of these opioid receptors, namely δ and κ, play a large role precisely in the area of pain therapy, but also in that of other indications of those mentioned. Accordingly, it is favourable if the compound also show an action on these opioid receptors.
SYN
http://www.google.com/patents/US20110319440

SYNTHESIS ……………..ON THE WAY ….. WATCH OUT
The dimethyl analogue is

Jirkovsky et al., J. Heterocycl. Chem., 12, 1975, 937-940;
Campaigne et al., J. Heterocycl. Chem., 2, 1965, 231-235;
Efange et al., J. Med. Chem., 41, 1998, 4486-4491;
Ellingboe et al., J. Med. Chem., 35, 1992, 1176-1183;
Pearson et al., Aust. J. Chem., 44, 1991, 907-917;
Yokohama et al., Chem. Pharm. Bull., 40, 1992, 2391-2398;
Beck et al., J. Chem. Soc. Perkin 1, 1992, 813-822;
Shinada et al., Tetrahedron Lett., 39, 1996, 7099-7102;
Garden et al., Tetrahedron, 58, 2002, 8399-8412;
Lednicer et al., J. Med. Chem., 23, 1980, 424-430.
|
2-10-2012
|
Pharmaceutical dosage forms comprising 6′-fluoro-(N-methyl- or N,N-dimethyl-)-4-phenyl-4′,9′-dihydro-3’H-spiro[cyclohexane-1,1′-pyrano[3,4,b]indol]-4-amine
|
|
|
11-9-2011
|
Compositions containing spirocyclic cyclohexane compounds
|
|
|
10-12-2011
|
Spirocyclic Cyclohexane Compounds Useful To Treat Substance Dependency
|
|
|
5-32-2011
|
Spirocyclic Cyclohexane Compounds
|
|
|
1-21-2011
|
MIXED ORL1/MU-AGONISTS FOR THE TREATMENT OF PAIN
|
|
|
9-22-2010
|
SPIROCYCLIC CYCLOHEXANE COMPOUNDS
|
|
|
6-17-2009
|
Spirocyclic cyclohexane compounds
|
|
|
9-12-2008
|
Spirocyclic Cyclohexane Compounds Useful To Treat Substance Dependency
|
|
|
5-30-2008
|
Mixed ORL1/mu-agonists for the treatment of pain
|
| US8614245 | Jan 8, 2013 | Dec 24, 2013 | Gruenenthal Gmbh | Crystalline (1r,4r)-6′-fluoro-N,N-dimethyl-4-phenyl-4′,9′-dihydro-3′H-spiro[cyclohexane-1,1′-pyrano[3,4,b]indol]-4-amine |
| US8618156 * | Jul 6, 2012 | Dec 31, 2013 | Gruenenthal Gmbh | Crystalline (1r,4r)-6′-fluoro-N,N-dimethyl-4-phenyl-4′,9′-dihydro-3’H-spiro[cyclohexane-1,1′-pyrano[3,4,b]indol]-4-amine |
| US8765800 | Mar 15, 2013 | Jul 1, 2014 | Gruenenthal Gmbh | Crystalline (1r,4r)-6′-fluoro-N,N-dimethyl-4-phenyl-4′,9′-dihydro-3′H-spiro[cyclohexane-1,1′-pyrano[3,4,b]indol]-4-amine |
| US20130231381 * | Mar 15, 2013 | Sep 5, 2013 | Gruenenthal Gmbh | Crystalline (1r,4r)-6′-fluoro-N,N-dimethyl-4-phenyl-4′,9′-dihydro-3’H-spiro[cyclohexane-1,1′-pyrano[3,4,b]indol]-4-amine |
| US5356896 * | Dec 22, 1992 | Oct 18, 1994 | Sandoz Ltd. | Alkaline stabiling medium |
| US20060004034 * | May 11, 2005 | Jan 5, 2006 | Gruenenthal Gmbh | Treating conditions associated with the nociceptin/ORL1 receptor system, e.g. pain, drug withdrawal, anxiety, muscle relaxants, anxiolytic agents; e.g. 1,1-[3-dimethylamino-3-(pyridin-2-yl)pentamethylene]-3,4-dihydro-1H-2,9-diazafluorene |
MAKE IN INDIA
http://makeinindia.com/sector/pharmaceuticals/
Read all about Organic Spectroscopy on ORGANIC SPECTROSCOPY INTERNATIONAL 
keep watching for synthesis update on this drug

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....