Home » 2013 (Page 103)
Yearly Archives: 2013
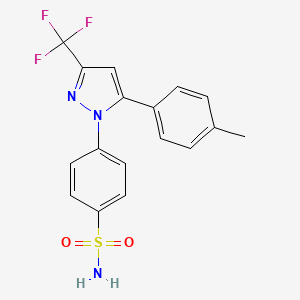
Drug spotlight, Celecoxib from G. D. Searle Company


CELECOXIB
4-[5-(4- methylphenyl)-3-(trifluoromethyl)-lH-pyrazol-l-yl]benzenesulfonamide
169590-42-5
mp…157-159 deg C
Celecoxib is a sulfonamide non-steroidal anti-inflammatory drug (NSAID) and selective COX-2 inhibitor used in the treatment of osteoarthritis, rheumatoid arthritis, acute pain, painful menstruation and menstrual symptoms, and to reduce numbers of colon and rectum polyps in patients with familial adenomatous polyposis. It is marketed by Pfizer. It is known under the brand name Celebrex or Celebra for arthritis and Onsenal for polyps. Celecoxib is available by prescription in capsule form.
Celecoxib was discovered developed by G. D. Searle & Company and was approved by the FDA on December 31, 1998. It was co-promoted by Monsanto Company (parent company of Searle) and Pfizer under the brand name Celebrex. Monsanto merged with Pharmacia, from which the Medical Research Division was acquired by Pfizer, giving Pfizer ownership of Celebrex. The drug was at the core of a major patent dispute that was resolved in Searle’s favor (later Pfizer) in 2004. In University of Rochester v. G.D. Searle & Co., 358 F.3d 916 (Fed. Cir. 2004), the University of Rochester claimed that United States Pat. No. 6,048,850 (which claimed a method of inhibiting COX-2 in humans using a compound, without actually disclosing what that compound might be) covered drugs such as celecoxib. The court ruled in favor of Searle, holding in essence that the University had claimed a method requiring, yet provided no written description of, a compound that could inhibit COX-2 and therefore the patent was invalid.

After the withdrawal of rofecoxib (Vioxx) from the market in September 2004, Celebrex enjoyed a robust increase in sales. However, the results of the APC trial in December of that year raised concerns that Celebrex might carry risks similar to those of Vioxx, and Pfizer announced a moratorium on direct-to-consumer advertising of Celebrex soon afterwards. After a significant drop, sales of Celebrex have recovered, and reached $2 billion in 2006.[6] Pfizer resumed advertising Celebrex in magazines in 2006, and resumed television advertising in April 2007 with an unorthodox, 2 1⁄2-minute advertisement which extensively discussed the adverse effects of Celebrex in comparison with other anti-inflammatory drugs. The ad drew criticism from the consumer advocacy group Public Citizen, which called the ad’s comparisons misleading. Pfizer has responded to Public Citizen’s concerns with assurances that they are truthfully advertising the risk and benefits of Celebrex as set forth by the FDA.
In late 2007, Pfizer released another U.S. television ad for Celebrex, which also discussed celecoxib’s adverse effects in comparison with those of other anti-inflammatory drugs.
Daniel L. Simmons of Brigham Young University, who discovered the COX-2 enzyme, is suing Pfizer to be credited with discovery of the technique in 1989 that eventually led to the drug, and for $1 billion USD. The company has made about $30 billion from the drug as of 2006. A settlement was finally reached in April 2012.
Celecoxib is licensed for use in osteoarthritis, rheumatoid arthritis, acute pain, painful menstruation and menstrual symptoms, ankylosing spondylitis and to reduce the number of colon and rectal polyps in patients with familial adenomatous polyposis. It was originally intended to relieve pain while minimizing the gastrointestinal adverse effects usually seen with conventional NSAIDs. In practice, its primary indication is in patients who need regular and long term pain relief; there is probably no advantage to using celecoxib for short term or acute pain relief over conventional NSAIDs, except in the situation where non-selective NSAIDs or aspirin cause cutaneous reactions (urticaria or “hives”). In addition, the pain relief offered by celecoxib is similar to that offered by paracetamol (acetaminophen).
Synthesis
https://www.google.com/patents/WO2010095024A2?cl=en
US 5,466,823, also discloses a process for the preparation of Celecoxib, which comprises reacting 4-methylacetophenone (II) with 1-ethyltrifluoroacetate (III) in the presence of methyl t-butyl ether and sodium methoxide, followed by recrystallisation from isooctane to produce l-(4-methylphenyl)-4,4,4-trifluorobutane-l ,3-dione (IV), which is further condensed with 4-hydrazinophenylsulfonamide hydrochloride (V) in the presence of ethanol to produce crude Celecoxib, which is recrystallised from ethyl acetate and isooctane to give Celecoxib (I),
The process is as shown in Scheme -I below:
HI rv
The synthesis of celecoxib was first described in 1997 by a team of researchers at Searle Research and Development. Celecoxib is synthesized by a Claisen condensation reaction of an acetophenone with N-(trifluoroacetyl)imidazole catalyzed by the strong base, sodium bis(trimethylsilyl)amide to produce a 1,3-dicarbonyl adduct. Condensation of the diketone with (4-sulfamoylaphenyl)hydrazine produces the 1,5-diarylpyrazole drug moiety.
Scheme-I The above process involves isolation of the intermediate l-(4-methylphenyl)-4,4,4- tiϊfluorobutane-l ,3-dione (IV) by crystallization, before condensing with 4- sulphonamido-phenylhydrazine, which adds to the cost and complexity of the synthesis.
Further, the above process proceeds with less selectivity to Celecoxib, which is having about 4 wt. % of regioisomer (VI) by-product under commercial conditions.
US 6, 150,534 discloses a process for the preparation of Celecoxib, which comprises, condensing l-(4-methylphenyl)-4,4,4-trifluorobutane-l ,3-dione (IV) with 4- sulphonamido-phenylhydrazine in presence of an amide solvent at controlled temperature to produce amide solvate of Celecoxib, which is further desolvated by recrystallization from isopropanol and water.
The above process also involves isolation of the intermediate l-(4-methylphenyl)- 4,4,4-trifluorobutane-l ,3-dione (IV) by crystallization, before condensing with 4- sulphonamido-phenylhydrazine,
US 5,892,053 discloses a process for the preparation of Celecoxib by condensing 4- methylacetophenone (II) with 1-ethyltrifluoro acetate (III) to produce l-(4- methylphenyl)-4,4,4-trifluoiObutane-l ,3-dione (IV), which is further reacted with 4- hydrazinophenylsulfonamide (V) in presence of aqueous mixture of alcohol and acid to produce Celecoxib.
US 6,579,988 discloses a preparation of Celecoxib via novel intermediate compound of formula VII. Formula VII
US 2007/0004924 Al discloses a process for the preparation of Celecoxib by condensing l-(4-methylphenyl)-4,4,4-trifluorobutane-1.3-dione (IV) with 4- hydrazinophenylsulfonamide (V) in presence of a solvent system containing an organic solvent, the salt of the 4-sulphonamidophenylhydrazine having a solubility in the organic solvent at least 0.05 M. ‘
US 2008/0234491 Al discloses the condensation of l-(4-methylphenyl)-4,4,4- trifluorobutane-l,3-dione (IV) with 4-hydrazinophenylsulfonamide (V) or its acid addition salts in the presence of a solvent medium comprising an alkyl ester, water or mixtures thereof to produce Celecoxib. Further, crystallization of crude Celecoxib is carried out in toluene alone.
l-(4-Methylphenyl)-4,4,4-trifluorobutane-l,3-dione (IV) is condensed with 4- hydrazinophenylsulfonamide (V) or its acid addition salt in a solvent selected from water, inert organic solvent to produce 4-[5-(4-methylphenyl)-3-(trifluoiOmethyl)-lH- pyrazol-l-yl]benzenesulfonamide (Celecoxib) of Formula I. The acid addition salts of compound of the formula IV includes, but are not limited to, hydrochloride, hydrobromide, sulfate, nitrate, oxalate, mesylate, methane sulfonate, and tartrate, preferably, hydrochloride salt. The suitable inert organic solvents for the above reaction include but are not limited to ketone solvents, such as acetone, methyl ethyl ketone, methyl isobutyl ketone, n-butanone, and tertiary-butyl ketone; nitrile solvents, such as acetonitrile. and propionitrile; halogenated solvents, such as dichloromethane, ethylene dichloride, and chloroform; esters, such as ethyl acetate, n-propylacetate, isopropyl acetate, and tertiary-butyl acetate; aprotic polar solvents, such as N,N- dimethylformamide, dimethylsulfoxide, and N,N-dimethylacetamide; ethers, such as diisopropyl ether, tetrahydrofuran and 1,4-dioxane; hydrocarbon solvents, such as cyclohexane, toluene and xylene; and mixtures thereof. The preferred solvent is water. The reaction may be performed at a temperature ranging from about 25°C to about reflux temperature of the solvent or mixture of solvents used for the reaction. The above reaction is conducted in presence of an acid selected from aqueous hydrochloric acid, aqueous sulfuric acid, p-toluene sulfonic acid, trifluoroacetic acid, and acetic acid to maintain the pH of the reaction mixture is below 7. More preferably, aqueous HCl is added. Crude Celecoxib (I) produced may be isolated by precipitation of compound from the reaction mixture, which may be performed by cooling the reaction mixture, followed by addition of an organic solvent selected from alcohols such as methanol, ethanol, isopropanol or aromatic hydrocarbons such as toluene, xylene, ethyl benzene and mixtures thereof solvents. The preferred solvent is mixture of methanol and toluene.
It has been observed that preparation of Celecoxib (I) using above reaction conditions results in regioisomer of compound (VI) to less than 2.5% by HPLC analysis.
EXAMPLE 1
Stage-1:
Preparation of l-(4-methylphenyl)-4,4,4-trifluorobutane-l,3-dione (IV)
4-Methylacetophenone (50 g, 0.373 mol) was dissolved in toluene (250 ml) and 30% methanolic sodium methoxide solution (80.6 g, 0.447 mol), followed by 1- ethyltrifluoro acetate (63.58 g, 0.447 mol) were added at 25-3O0C. Temperature of the reaction mass was raised to 55-600C and stirred ~ 4 hr to complete the reaction. The reaction mass was cooled to 20-250C and washed with 10% aqueous hydrochloric acid (200 ml). The layers were separated and concentrated the organic layer at 50-550C under reduced pressure to produce 80 g of l-(4-methylphenyl)-4,4,4-trifluoiObutane- 1,3-dione (IV) as an oily mass.
Stage-2:
Preparation of 4-[5-(4-methylphenyl)-3-(trifluorornethyl)-lh-pyrazol-l- yljbenzenesulfonamide (Celecoxib) (I) l-(4-Methylphenyl)-4,4,4-trifluorobutane-l,3-dione (IV) (80 g, 0.348 mol), 4- hydrazinophenylsulfonamide (V) (77.74 g, 0.348 mol) and concentrated hydrochloric acid (18.6 g) were added to DM water (500 ml) and heated to 98-1000C. The mass was stirred for 4 hr to complete the reaction. The reaction mass was cooled to 70-75 C and a mixture of toluene (600 ml) and methanol (10 ml) was added to the reaction mass. After 1 hr stirring at 70-750C, the reaction mass was cooled to 20-250C, the product was filtered and λvashed with toluene (100 ml) followed by DM water (200 ml). The product obtained was dried at 55-600C under reduced pressure to produce 1 15 g of Celecoxib crude. Chromatographic purity: 99%(by PTPLC, by area normalization)
……………………..
The synthesis of celecoxib was first described in 1997 by researchers at Searle Research and Development. It is synthesized by a Claisen condensation reaction of an acetophenone with N-(trifluoroacetyl)imidazole catalyzed by the strong base, sodium bis(trimethylsilyl)amide to produce a 1,3-dicarbonyl adduct. Condensation of the diketone with (4-sulfamoylaphenyl)hydrazine produces the 1,5-diarylpyrazole drug moiety.
Penning TD, Talley JJ, Bertenshaw SR, Carter JS, Collins PW, Docter S, Graneto MJ, Lee LF, Malecha JW, Miyashiro JM, Rogers RS, Rogier DJ, Yu SS, Burton EG, Cogburn JN, Gregory SA, Koboldt CM, Perkins WE, Seibert K, Veenhuizen AW, Zhang YY, Isakson PC (1997). “Synthesis and Biological Evaluation of the 1.5 Diarylpyrazole Class of Cyclooxygenase-2 Inhibitors: Identification of 4-[5-(4-Methylphenyl)-3-(trifluoromethyl)-1H-pyrazole-1-yl]benzenesulfonamide (SC-58634, Celecoxib)”. Journal of Medicinal Chemistry40 (9): 1347–1365. doi:10.1021/jm960803q. PMID 9135032.

……………………………….


- The condensation of 4-methylacetophenone (I) with ethyl trifluoroacetate (II) by means of NaOMe in refluxing methanol gives 4,4,4-trifluoro-1-(4-methylphenyl)butane-1,3-dione, which is cyclized with 4-hydrazinophenylsulfonamide (III) in refluxing ethanol.
…………………
http://www.google.com/patents/US7759497
In U.S. Pat. Nos. 5,892,053 and 5,910,597, Zhi et al. describe a scalable two step process for the preparation of pyrazoles from the condensation of diketones and hydrazines. In the first step, a diketone is formed by the treatment of a ketone with base and ester in a suitable solvent. In the second step, the diketone is solubilized in an aqueous alcohol and condensed with a hydrazine to form the pyrazole product. This two step process has been used on a commercial scale for the preparation of celecoxib (4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazole-1-yl]benzenesulfonamide) sold under the trademark CELEBREX® by Pharmacia Corporation as shown in the following reaction:
While this synthetic approach proceeds with high selectivity to celecoxib, about 2-5 wt. % of regioisomer and hydroxyregioisomer by-products are formed under commercial conditions.
The regioisomer and hydroxyregioisomer by-products must be separated from celecoxib in a purification step to enable the celecoxib to meet purity requirements. The separation is typically done through a crystallization step in which celecoxib preferentially crystallizes while the regioisomer and hydroxyregioisomer by-products predominantly remain in solution. The celecoxib crystals are then removed from the resultant slurry and separated from impurities by solid-liquid separation techniques known to those skilled in the art, such as centrifugation or filtration.
Under commercial conditions used to date, of the two by-products, regioisomer is selectively formed over hydroxyregioisomer. This is problematic, however, since the regioisomer is generally more difficult to separate through crystallization from celecoxib than is the hydroxyregioisomer, and regioisomer concentrations of greater than about 1% typically require two crystallizations to achieve desired celecoxib purity. The second crystallization adds time to the manufacturing process and thus negatively impacts product throughput. Additionally, a second crystallization reduces yield as some celecoxib remains uncrystallized and is not recovered from the liquid phase.

Example 7Preparation of Celecoxib with Hydrazine Reactant Containing Water
To a 250 mL reactor which had been purged with nitrogen and which had been fitted with a mechanical stirrer and a chilled condenser was charged while stirring, isopropyl alcohol (50.75 g), ethyltrifluoroacetate (37.95 g), sodium methoxide (25% in methanol, 53.28 g) and 4′-methylacetophenone (27.43 g). The reaction mixture was heated to 50-55° C. and held for at least 2 hours. To a separate 1 L reactor which had been purged with nitrogen and fitted with a mechanical stirrer and a chilled condenser, was charged 4-SAPH•HCl (45.96 g), isopropyl alcohol (101.2 g), water (74 g) and trifluoroacetic acid (23.43 g). The 4-SAPH•HCl was heated to 50° C. with agitation. At the completion of the 2 hour reaction period, the contents of the first reactor was transferred to the second reactor containing the 4-SAPH•HCl over a period of at least five minutes and the reaction mixture was then brought to 55° C. and maintained at that temperature for at least 30 minutes. The pH of the reaction mixture was then adjusted to be within the range of 3 to 9 followed by the addition of water (95 g). The contents were then heated to 65° C. and the pH was again adjusted to be within the range of 3 to 9. Water (90 g) was then added to the mixture over a time period of about 20 minutes while maintaining the temperature at about 65° C. The reaction mixture was then cooled to about 20° C. over a period of 12 to 14 hours to produce celecoxib (62-65 g) with less than 0.05% regio-isomer and undetectable regioisomer.
Example 8Preparation of Celecoxib with Anhydrous Hydrazine Reactant
To a 250 mL reactor which had been purged with nitrogen and which had been fitted with a mechanical stirrer and a chilled condenser was charged while stirring, isopropyl alcohol (50.75 g), ethyltrifluoroacetate (37.95 g), sodium methoxide (25% in methanol, 53.28 g) and 4′-methylacetophenone (27.43 g). The reaction mixture was heated to 50-55° C. and held for at least 2 hours. To a separate 1 L reactor which had been purged with nitrogen and fitted with a mechanical stirrer and a chilled condenser, was charged 4-SAPH•HCl (45.96 g), isopropyl alcohol (101.2 g) and trifluoroacetic acid (23.43 g). The 4-SAPH•HCl was heated to 50° C. with agitation. At the completion of the 2 hour reaction period, the contents of the first reactor was transferred to the second reactor containing the 4-SAPH•HCl over a period of at least five minutes and the reaction mixture was then brought to 55° C. and maintained at that temperature for at least 30 minutes. The pH of the reaction mixture was then adjusted to be within the range of 3 to 9 followed by the addition of water (95 g). The contents were then heated to 65° C. and the pH was again adjusted to be within the range of 3 to 9. Water (90 g) was then added to the mixture over a time period of about 20 minutes while maintaining the temperature at about 65° C. The reaction mixture was then cooled to about 20° C. over a period of 12 to 14 hours to produce celecoxib (62-65 g) with less than 0.05% regio-isomer. Analysis of the reaction mixture prior to initiation of crystallization indicated that the regio-isomer content was less than 0.5 mole percent of the reaction products.
Example 9Preparation of Celecoxib by Addition of Diketone Salt to 4-SAPH-HCl
To a 250 mL reactor, fitted with a mechanical stirrer and maintained under a nitrogen atmosphere, was added isopropyl alcohol (54.8 g, 0.912 moles), ethyl trifluoroacetate (38.0 g, 0.267 moles) and 25% sodium methoxide in methanol (53.3 g, 0.246 moles). To the agitated reactor was added 4-methylacetophenone (27.6 g, 0.206 moles). The reaction mixture was heated to 50° C. and maintained for 2 hours. To a second (1 liter) reactor was added 4-sulphamidophenyl hydrazine hydrochloride (46.0 g, 0.206 moles), isopropyl alcohol (101.3 g, 1.685 moles) and trifluoroacetic acid (11.7 g, 0.103 moles) with stirring. The reaction mixture was heated to approximately 45° C. Upon completion of the 2-hour reaction period in the 250 mL reactor, the contents was added to the second reactor over approximately 10 minutes. The reaction mixture maintained at 55° C. for 30 minutes. The pH was adjusted with 50% aqueous sodium hydroxide to a pH of 5-6. The reaction mixture was heated to 65° C. and water was added (95 g, 5.3 moles). The pH was again adjusted with 50% aqueous sodium hydroxide to a value of 5-6. Water (90 g, 5.0 moles) was added over 20 minutes while maintaining the temperature at 65° C. The reaction mixture was then cooled over 9 hours to 20° C. The reaction mixture was filtered, washed twice with 50% aqueous isopropyl alcohol and dried in a vacuum over for 16 hours to yield celecoxib (65.6 g) whose HPLC retention time was identical to that of authentic celecoxib. Regio-isomer was not detected by HPLC.
Example 10Preparation of Celecoxib by the Addition of 4-SAPH-HCl to Diketone
To a 1 L reactor fitted with a mechanical stirrer and maintained under a nitrogen atmosphere, was added isopropyl alcohol (54.7 g, 0.912 moles), ethyl trifluoroacetate (37.7 g, 0.267 moles), and 25% sodium methoxide in methanol (53.3 g, 0.247 moles). To the agitated reactor was added 4-methylacetophenone (27.32 g, 0.205 moles). The reaction mixture was heated to 50° C. and maintained for 2 hours. Trifluoroacetic acid (36.69, 0.321 moles) was added to the reaction mixture over a period of five minutes. 4-SAPH-HCl (46.0 g, 0.205 moles) was added through a power addition funnel over a period of 10 minutes. The reaction mixture was brought to 55° C. and maintained for one hour. Isopropyl alcohol (81.5 g, 1.36 moles) was added followed by the addition of 50% sodium hydroxide (18.5 g, 0.231 moles) to achieve a pH of 7. Water (87.8 g, 4.88 moles) was added and the reaction mixture heated to 65° C. Water (90.0 g 5.00 moles) was added over ten minutes. The reaction mixture was cooled to 20° C. over nine hours. The slurry was filtered and washed twice with 50% (weight) aqueous isopropyl alcohol (100 g). The solid was dried in a vacuum oven for 16 hours to yield celecoxib (67.2 g) whose HPLC retention time was identical to that of authentic material. Regio-isomer was not detected by HPLC.

4-[5-(4-Methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]benzenesulfonamide (Celecoxib)
E-mail: prataprp@ drreddys.com. Fax: 914044346285. Telephone: 9989997176


HPLC Conditions:
 29.2 min
29.2 min 30.9 min
30.9 min11
Bioorganic and Medicinal Chemistry, , vol. 15, # 4 p. 1802 – 1807

![]()
..celecoxib
………
http://www.google.com/patents/WO2014012074A2?cl=en
4,4,4-Trifluoro-l-(p-tolyl)butane-l,3-dione (C01)
25% sodium methoxide in methanol (51.3 ml, 223.5 mmol) and ethyl
trifluoroacetate (24.4 ml, 204.9 mmol) were dissolved in 110 mL methyl tert-butyl ether under N2, at room temperature. 4′-methyl acetophenone (25.0 ml, 186.3 mmol) was added and stirred at room temperature overnight. The reaction was washed with 3M HC1 and dried over magnesium sulfate. The solution was then evaporated and the resulting oil dried under vacuum overnight. The resulting light orange crystalline solid was washed with cold isooctane and dried under vacuum to yield an off white crystalline solid (37.3 g, 87% yield). LC tr=3.49 minutes (C- 18 column, 5 to 95% acetonitrile/water over 6 minutes at 1.7 mL/min with detection 254 nm, at 23 °C).
4-(5-(p-Tolyl)-3-(trifluoromethyl)-lH-pyrazol-l-yl)benzenesulfonamide (C02)
] C01 (23.55 g, 102.3 mmol) was refluxed with 4-sulphonamidophenyl
hydrazine HCl (23.95 g, 127.9 mmol) in 700 mL ethanol overnight. The reaction was evaporated, dissolved in 700 mL ethyl acetate, washed with water and brine, dried over magnesium sulfate and evaporated to -100 mL ethyl acetate. The product was crystalized by the addition of ~ 400 mL isooctane. After 15 minutes, the white crystalline solid was broken up, washed with isooctane and dried under vacuum (35.15 g, 90% yield). 1H NMR (400 MHz, CDC13) δ 7.94-7.91 (m, 2H), 7.51-7.49 (m, 2H), 7.21-7.20 (m, 2H), 7.15-7.13 (m, 2H), 6.77 (s, 1H), 2.41 (s, 3H). LC tr=4.27 minutes (C-18 column, 5 to 95% acetonitrile/water over 6 minutes at 1.7 mL/min with detection 254 nm, at 23° C). ES(neg)MS m/z 380 (M-H calcd for C17H14F3N302S requires 380).
![]()

http://www.slideshare.net/kennytirorx/celebrex-ms-report
………………………………….
http://www.google.co.in/patents/WO2011055233A2?cl=en
The preparation and use as COX-2 inhibitors of benzenesulfonamide derivatives such as celecoxib is described in US Patent No. 5,466,823. Processes for the preparation of celecoxib are also described in U.S. Patent Nos. 5,760,068 and 5,910,597.
As per the process exemplified in the U.S. Patent No. 5,760,068 (hereinafter referred to as the Ό68 patent), celecoxib is prepared by the reaction of l-(4-methylphenyl)- 4,4,4-trifluorobutane-l,3-dione with 4-sulphonamidophenyl hydrazine hydrochloride in absolute ethanol at reflux temperature under argon for 24 hours. The resulting mass is cooled to room temperature, followed by filtering and concentrating the reaction mixture to afford an orange solid, which is then recrystallized from a solvent system containing methylene chloride/hexane to produce the product as a pale yellow solid (melting point: 157° – 159°C).
The recrystallization process for preparing celecoxib described in the ‘608 patent suffers from disadvantages since the recrystallization process requires large volumes of solvents (more than 20 volumes each of methylene chloride and hexane solvents per gram of celecoxib), which is not commercially and environmentally, advisable for scale up operations. Moreover, the use of methylene chloride is hazardous to the environment and human health. The use of n-hexane is not advisable because it causes an ignition and fire risk due to its electrostatic charge accumulation property.
PCT Publication No. WO 01/42222 (hereinafter referred to as the ‘222 application) discloses three polymorphic forms (Form I, Form II and Form III) of celecoxib, pharmaceutical compositions, and methods of use thereof. The crystalline forms are characterized by powder X-ray diffraction (P-XRD), differential scanning calorimetry (DSC) and Infrared (IR) spectroscopy. The ‘222 application further teaches that the crystalline Form III of celecoxib is more thermodynamically stable than Form I and Form II. The ‘222 application also teaches that crystalline Form III of celecoxib is produced by crystallization of celecoxib from a solvent comprising isopropanol and water, for example, as described in U.S. Patent No. 5,910,597.
According to the ‘222 application, the polymorphic Form I is characterized by an X-ray powder diffraction pattern having peaks expressed as 2-theta at about 5.5, 5.7, 7.2 and 16.6 degrees; a melting point of about 162.5°C to about 163°C; a differential scanning calorimetry (DSC) endotherm maximum at about 163.3°C; and an Infra Red (IR) spectrum with peaks at about 3256 and 3356 cm-1. The polymorphic Form II is characterized by an X- ray powder diffraction pattern having peaks expressed as 2-theta at about 10.3, 13.8 and 17.7 degrees, a melting point of about 161°C to about 162°C; a differential scanning calorimetry (DSC) endotherm maximum at about 162°C. The polymorphic Form III is characterized by a melting point of about 160.8°C.
However, it has been observed by the present inventors that the celecoxib obtained after crystallization from isopropanol and water is fluffier resulting in low bulk density and poor flow properties. Moreover, it has also been observed that the particles of the crystalline form III of celecoxib obtained by the aforementioned crystallization processes are static or cohesive thereby increasing the difficulties of formulation scientists.
PCT Publication No. WO 01/42221 discloses an amorphous form of celecoxib, and processes for preparing amorphous celecoxib using crystallization inhibitors. Amorphous celecoxib exhibits an apparent glass transition at 111.4°C (onset).
EP Patent No. 1167355 (hereinafter referred to as the ‘355 patent) discloses a crystalline form, designated as Form I, of celecoxib, processes for the preparation, and pharmaceutical compositions thereof. The crystalline form is characterized by powder X-ray diffraction (P-XRD) and scanning electron microscopy (SEM). According to the ‘355 patent, the crystalline Form I is characterized by an X-ray powder diffraction pattern having peaks expressed as 2-theta at about 14.8, 16.05, 17.875, 19.615, 21.455, 22.080, 22.385, 23.425, 25.33 and 29.355 degrees. The crystalline Form I is further characterized by an X-ray powder diffraction pattern having additional peaks expressed as 2-theta at about 10.67, 10.97, 12.985, 13.855, 18.340, 18.685, 20.425, 20.67, 23.185, 24.51, 24.93, 25.73, 26.915, 27.63, 28.185, 29.955, 30.375, 31.405, 34.915, 35.585, 37.895, 44.070 and 45.250 degrees. The ‘355 patent teaches that the crystalline Form I has improved properties over prior art crystal form which is used for formulating celecoxib as disclosed in International Application No. WO 95/15316. The ‘355 patent teaches that the prior art crystal form (designated as Form II) has several disadvantages, caused by its crystal structure, since it has low bulk density and a crystal morphology that tends to form long cohesive needles.
According to the ‘355 patent, the celecoxib crystalline Form I is prepared by dissolving celecoxib in a solvent system comprising at least one amide solvent selected from the group consisting of N,N-dimethylformamide, Ν,Ν-dimethylacetamide, and mixtures thereof; and isolating the crystals of Form I by adding a non-solvent, especially water, to the solution.
The process for the preparation of the celecoxib crystalline Form I described in the ‘355 patent also suffers from drawbacks since the use of amide solvents in the purification/crystallization of celecoxib leads to the formation of solvates (for example, the formation of solvates of celecoxib with amide solvents such as dimethylacetamide and dimethylformamide can be found in the preparative examples of the WO 01/42222). It is well known that the removal of these residual amide solvents from the celecoxib crystalline form is very difficult and requires high temperatures.
Formation of solvates of celecoxib with amide solvents such as dimethylacetamide and dimethylformamide is also described in PCT Publication No. WO2005/014546. Moreover, celecoxib obtained by the crystallization process using amide solvents described in the ‘355 patent does not have satisfactory purity for pharmaceutical use.
The solvated forms of celexicob are not acceptable from regulatory point of view since they include substantial amounts of organic solvents, and thus are not acceptable for clinical use. It is well known that impurities and residual solvents in celecoxib or any active pharmaceutical ingredient (API) are undesirable and might be harmful. Purity standards are set by regulatory authorities with the intention of ensuring that an API is as free of impurities and residual solvents as possible, and, thus, are as safe as possible for clinical use. Furthermore, it is required to control the levels of these impurities in the final drug compound obtained by the manufacturing process and to ensure that the impurities and/or residual solvents are present in the lowest possible levels.
celecoxib characterized by a powder X-ray diffraction (P-XRD) pattern having peaks (designated as d-values) at about 16.0, 15.3, 12.3, 10.6 ± 0.2 A. According to the ‘340 application, the crystalline Form N of celecoxib is prepared by suspending celecoxib Form III in a hydrocarbon solvent selected from the group consisting of n-tetradecane, and n-decane, heating the suspension at high temperatures (about 165°C) while stirring, stirring the resulting emulsion at the same high temperature, followed by cooling to 145°C. The resulting mass is then reheated to about 165°C, followed by cooling to about 110°C, filtering the separated crystals, and drying at 100°C under the vacuo for 12 hours to produce celecoxib Form N.
The crystallization process for preparing celecoxib described in the ‘340 application also suffers from disadvantages since the processes involve tedious and cumbersome procedures such as the use of high boiling point solvents, large amounts of solvents (about 20 volumes of high boiling point solvents per gram of celecoxib), high temperatures (about 165°C), high drying temperatures (about 100°C), and prolonged periods of drying at a high temperature, resulting in formation of unwanted impurities, thereby making the process industrially unfeasible.
PCT Publication No. WO 05/089511 discloses a hydrate of celecoxib sodium salt characterized by a powder X-ray diffraction (P-XRD) pattern having peaks at 3.05, 8.91 and 10.77 degrees 2-theta.
PCT Publication No. WO 2006079923A1 discloses a crystalline Form IV of celecoxib characterized by a powder X-ray diffraction (P-XRD) pattern having peaks at about 4.46, 13.13, 18.29, 20.21, 21.83 and 26.24 degrees 2-theta. [0020] Based on the aforementioned drawbacks, the prior art crystallization processes may be unsuitable for the preparation of celecoxib, especially in crystalline Form III, in commercial scale operations.
| Patent | Submitted | Granted |
|---|---|---|
| 5-oxo-pyrrolidine-2-carboxylic acid hydroxamide derivatives [EP1004578] | 2000-05-31 | 2004-02-25 |
| ANALGESIC COMPOSITIONS COMPRISING ANTI-EPILEPTIC COMPOUNDS AND METHODS OF USING SAME [EP1011658] | 2000-06-28 | 2005-12-07 |
| SYNERGISTIC ANALGESIC COMBINATION OF OPIOID ANALGESIC AND CYCLOOXYGENASE-2 INHIBITOR [EP1014886] | 2000-07-05 | 2004-11-24 |
| BIARYLALKANOIC ACIDS AS CELL ADHESION INHIBITORS [EP1017382] | 2000-07-12 | 2006-03-01 |
| BIOADHESIVE COMPOSITIONS AND METHODS FOR TOPICAL ADMINISTRATION OF ACTIVE AGENTS [EP1021204] | 2000-07-26 | 2005-12-28 |
| Implantable medical device with enhanced biocompatibility and biostability [EP1023879] | 2000-08-02 | 2005-04-06 |
| NOVEL COMBINATION [EP1027052] | 2000-08-16 | |
| CYCLIC AMINO ACID DERIVATIVES AS CELL ADHESION INHIBITORS [EP1033983] | 2000-09-13 | |
| SUBSTITUTED BETA-ALANINE DERIVATIVES AS CELL ADHESION INHIBITORS [EP1034164] | 2000-09-13 | 2004-05-19 |
| Delayed-release oral formulation of dihydroxy open acid simvastatin and salts and esters thereof [EP1036563] | 2000-09-20 |
| Patent | Submitted | Granted |
|---|---|---|
| Crystalline hydrated dihydroxy open-acid simvastatin calcium salt [EP1036783] | 2000-09-20 | 2003-05-21 |
| CYCLOOXYGENASE-2 INHIBITION CYCLOOXYGENASE-2 INHIBITION [EP1039914] | 2000-10-04 | |
| Dioxocyclopentyl hydroxamic acids [EP1041072] | 2000-10-04 | 2003-07-16 |
| PHARMACEUTICAL ORAL DOSAGE FORM COMPRISING A COMBINATION OF AN OPIOID AGONIST AND NALTREXONE [EP1041987] | 2000-10-11 | 2006-04-19 |
| Method and compositions for the treatment and prevention of pain and inflammation [US2005101563] | 2005-05-12 | |
| Compositions of a cyclooxygenase-2 selective inhibitor and a neurotrophic factor-modulating agent for the treatment of central nervous system mediated disorders [US2005148589] | 2005-07-07 | |
| Compositions and methods of treatment involving peroxisome proliferator-activated receptor-gamma agonists and cyclooxygenase-2 selective inhibitors [US2003220374] | 2003-11-27 | |
| Methods for treating carbonic anhydrase mediated disorders [US2003220376] | 2003-11-27 | |
| Method of using a Cox-2 inhibitor and a 5-HT1A receptor modulator as a combination therapy [US2004147581] | 2004-07-29 | |
| Tyrosine kinase inhibitors [US2002137755] | 2002-09-26 |
| Cited Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| WO1995015316A1 | 14 Nov 1994 | 8 Jun 1995 | Stephen R Bertenshaw | Substituted pyrazolyl benzenesulfonamides for the treatment of inflammation |
| WO2001042221A1 | 6 Dec 2000 | 14 Jun 2001 | Michael J Hageman | Solid-state form of celecoxib having enhanced bioavailability |
| WO2001042222A1 | 1 Dec 2000 | 14 Jun 2001 | Leonard J Ferro | Polymorphic crystalline forms of celecoxib |
| WO2002000627A1 * | 26 Jun 2001 | 3 Jan 2002 | Mehmet Bahar | A crystalline form of celecoxib |
| WO2005014546A1 | 8 Aug 2003 | 17 Feb 2005 | Hetero Drugs Ltd | Novel crystalline forms of celecoxib |
| WO2005089511A2 | 17 Mar 2005 | 29 Sep 2005 | Transform Pharmaceuticals Inc | Novel pharmaceutical forms, and methods of making and using the same |
| WO2006051340A1 | 21 Jul 2005 | 18 May 2006 | Pliva Istrazivanje I Razvoj D | Novel form of celecoxib |
| WO2006079923A2 | 19 Jan 2006 | 3 Aug 2006 | Pharmacia & Upjohn Co Llc | Form iv crystalline celecoxib |
| EP1167355A1 | 15 Mar 2001 | 2 Jan 2002 | Fako Ilaclari A.S. | A crystalline form of celecoxib |
| US5466823 | 30 Nov 1993 | 14 Nov 1995 | G.D. Searle & Co. | Antiinflammatory agents; FDA Orange book listed patent for Celebrex |
| US5760068 | 14 Nov 1994 | 2 Jun 1998 | G.D. Searle & Co. | Substituted pyrazolyl benzenesulfonamides for the treatment of inflammation |
| US5910597 | 14 Oct 1998 | 8 Jun 1999 | G.D. Searle & Co. | Process for preparing 3-haloalkyl-1H-pyrazoles |
………..

Dr Raghupathi Reddy Anumula
Managing Director- Chemikos Laboratories Private Limited
Shire begins Phase II ‘Vascugel’ trials

April 05,2012
Shire has kicked off two mid-stage trials assessing Vascugel – which is being developed by Shire under the name SRM003 – for enhancing blood vessel repair in patients with end-stage renal disease (ESRD) receiving haemodialysis.
Before patients can undergo haemodialysis, an arteriovenous (AV) access site must be created where blood can be removed, filtered and returned to the body.
In most cases, AV access is achieved through either an AV fistula (AVF), where the vein is connected directly to the artery, or an AV graft, where the vein and artery are connected via a synthetic tube.
Shire notes that there are around 100,000 AV fistulas and 60,000 AV grafts occurring annually in the US. But complications – such as infection, blood clots, and narrowing of the vessel – are common and frequently lead to access failure.
In fact, an estimated 60% of AV grafts fail after one year, requiring an additional procedure to restore flow or to create another access site, Shire said.
AYURVEDA SPOTLIGHT- BRAHMI
Bacopa monnieri
| Bacopa monnieri | |
|---|---|
 |
|
| Scientific classification | |
| Kingdom: | Plantae |
| (unranked): | Angiosperms |
| (unranked): | Eudicots |
| (unranked): | Asterids |
| Order: | Lamiales |
| Family: | Plantaginaceae |
| Genus: | Bacopa |
| Species: | B. monnieri |
Bacopa monnieri (waterhyssop, brahmi, thyme-leafed gratiola, water hyssop) is a perennial, creeping herb whose habitat includes wetlands and muddy shores. Brahmi is also the name given to Centella asiatica, particularly in North India, and Kerala where it is also identified in Malayalam as muttil (മുത്തിള്) or kodakan. This identification of brāhmī as C. asiatica has been in use for long in northern India, as Hēmādri’s Commentary on Aṣṭāṅgahṛdayaṃ (Āyuṛvēdarasāyanaṃ) treats maṇḍūkapaṛṇī (C. asiatica) as a synonym of brahmi, although that may be a case of mistaken identification that was introduced during the 16th century.
Brahmi been used by Indian Ayurvedic medical practitioners for almost 3000 years. The earliest chronicled mention of Brahmi is in several ancient Ayurvedic treatises including the Caraka Samhita (6th century A.D.), in which it is recommended in formulations for the management of a range of mental conditions including anxiety, poor cognition and lack of concentration, and the Bravprakash Var-Prakarana (16th century A.D.).
Description

Bacopa monnieri in Hyderabad, India.
The leaves of this plant are succulent and relatively thick. Leaves are oblanceolate and are arranged oppositely on the stem. The flowers are small and white, with four or five petals. Its ability to grow in water makes it a popular aquarium plant. It can even grow in slightly brackish conditions. Propagation is often achieved through cuttings.
Ecology
It commonly grows in marshy areas throughout India, Nepal, Sri Lanka, China, Taiwan, and Vietnam, and is also found in Florida, Hawaii and other southern states of the USA where it can be grown in damp conditions by the pond or bog garden.
Traditional uses
It has been used in traditional Ayurvedic treatment for epilepsy and asthma. It is also used in Ayurveda for ulcers, tumors, ascities, enlarged spleen, indigestion, inflammations, leprosy, anemia, and biliousness. This plant can be grown even as hydroponics using almost simple water.
Chemical constituents
Bacopa monnieri has many chemical constituents including alkaloids (brahmine and herpestine), saponins (d-mannitol and hersaponin, acid A, and monnierin), flavonoids (luteolin and apigenin). It also contains significant amounts of betulic acid, stigmasterol, beta-sitosterol, and bacopasaponins (bacosides A, bacosides B, bacopaside II, bacopaside I, bacopaside X, bacopasaponin C, bacopaside N2). The minor components include bacopasaponin F, bacopasaponin E, bacopaside N1, bacopaside III, bacopaside IV, and bacopaside V).
Pharmacology of chemical constituents
In rats, bacosides A enhance antioxidant defenses, increasing superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX) activity.Laboratory studies on rats indicate that extracts of the plant improve memory capacity. Some studies in mice suggest that ingestion of Bacopa for a 12 week period can significantly improve cognitive ability by accelerating the rate of learning and enhanced memory.The sulfhydryl and polyphenol components of Bacopa monnieri extract have also been shown to impact the oxidative stress cascade by scavenging reactive oxygen species, inhibiting lipoxygenase activity and reducing divalent metals.This mechanism of action may explain the effect of Bacopa monniera extract in reducing beta amyloid deposits in a mouse model of Alzheimer’s disease. B. monnieri has a demonstrated ability to reverse diazepam-induced amnesia in the Morris water maze test. The mechanism of this action is unknown. In some trials, bacopacide extract did not restore or enhance memory formation, but improved retention. In others, including a randomized clinical trial of 98 healthy older people (over 55 years), Bacopa significantly improved memory acquisition and retention. A 2012 systematic review found some evidence to suggest that Bacopa improves memory free recall, but there was a lack of evidence for enhancement of other cognitive abilities.
Brahmi may regulate antibody production by augmenting both Th1 and Th2 cytokine production.It may also cause a lower heart rate, and increase secretions in the stomach, intestines, and urinary tract. The increase in secretions may irritate ulcers and urinary tract obstructions.
No safety studies have been performed on brahmi’s use in humans. When a preparation of the plant was evaluated for safety and tolerability it showed no adverse effects but there were some reports of mild gastrointestinal symptoms.
However, participants in a 2001 double-blind study published in Psycho pharmacology experienced side effects including nausea, weakness and dry mouth while taking brahmi, notes the University of Michigan Health System. Brahmi could potentially cause elevated thyroid-hormone levels and decreased sperm counts. Therefore, taking brahmi should be avoided if you have a thyroid condition or are taking thyroid replacement therapies and other medications that affect thyroid function.
Aqueous extract of Bacopa monnieri (Brahmi) has been reported to reversibly suppress spermatogenesis and fertility in male mice with at a dose of 250 mg/kg body weight/day for 28 and 56 days(equivalent to 1.54 g/day for a 76kg male, when properly controlling for animal to human conversions ) Parameters of motility, viability, morphology, and number of spermatozoa in cauda epididymidis returned to baseline 56 days after treatment cessation.
The plant is known by many names in many international languages, including:
- ബ്രഹ്മി in Malayalam
- நீர்ப்பிரமி (Niirpirami)/ Valaarai in Tamil
- ผักมิ (Phak mi), พรมมิ (Phrommi) in Thai
- Lunuwila in Sinhalese (Sri Lanka)
- ae’ ae’ in Hawaiian (Hawaii)
- Rau Đắng in Vietnamese
- פְּשֵטָה שרועה (“psheta sru’a”) in Hebrew\
- conclusions
- Brahmi is a plant that has been used in traditional Indian medicine (Ayurveda). Be careful not to confuse brahmi (Bacopa monnieri) with gotu kola and other natural medicines that are also sometimes called brahmi.Brahmi is used for Alzheimer’s disease, improving memory, anxiety, attention deficit-hyperactivity disorder (ADHD), allergic conditions, irritable bowel syndrome, and as a general tonic to fight stress.People also take brahmi to treat backache, hoarseness, mental illness, epilepsy, joint pain, and sexual performance problems in both men and women. It is also sometimes used as a “water pill.”
-
Brahmi Benefits
Brahmi is considered a nootropic agent, which is the term given to a drug that improves mental functions such as cognition, memory, intelligence, motivation, attention, and concentration. Brahmi has been used in ayurvedic medicine and in traditional treatments for a number of disorders, particularly those involving anxiety, intellect, and poor memory.
Recent major scientific reviews of the plant suggest that it has prominent action on the central nervous system, where it improves understanding, memory, intellect, and speech, and corrects aberrations of emotions, mood, and personality in an individual. Based on the results of human clinical trials, the nootropic effects of Brahmi are thought to manifest after chronic dosing (i.e. 12 weeks) rather than acute (i.e. single day). Studies in humans have shown that chronic administration of Brahmi results in improvements in working memory, visual information processing, learning rate and anxiety.
In India, Brahmi is currently recognized as being effective in the treatment of mental illness and epilepsy. In certain parts of India, Brahmi is believed to be an aphrodisiac; in Sri Lanka, under the name of Loonooweella, Brahmi is prescribed for fevers; in the Philippines, it is used as a diuretic.
-
How does it work?
Brahmi might increase certain brain chemicals that are involved in thinking, learning, and memory. Some research suggests that it might also protect brain cells from chemicals involved in Alzheimer’s disease.
Phase III trial of lupus drug Benlysta (belimumab) in patients with ANCA (Anti-neutrophil Cytoplasmic Antibodies) positive vasculitis

3 mar 2013
GSK, the company said it has kicked off a Phase III trial of its lupus drug Benlysta (belimumab) in patients with ANCA (Anti-neutrophil Cytoplasmic Antibodies) positive vasculitis, a condition characterised by inflammation of the blood vessels.
The multicentre, multi-national, randomised, double-blind study will assess the drug’s efficacy and safety in combination with azathioprine for the maintenance of remission in patients with a particular type of vascultitic disease called ANCA associated Vasculitis (Granulomatosis with Polyangiitis (Wegener’s) or microscopic polyangiitis).

Belimumab (trade name Benlysta, previously known as LymphoStat-B) is a human monoclonal antibody that inhibits B-cell activating factor (BAFF), also known as B-lymphocyte stimulator (BLyS) B cells are responsible for part of the normal immune response, and also for the over-aggressive immune response in autoimmune diseases like systemic lupus erythematosus (SLE).
Belimumab is approved in the United States, Canada and Europe for treatment of SLE. However, the major phase III trials excluded the more severe cases of SLE with kidney and brain damage, so its effectiveness has not been demonstrated in those cases. A Phase III study for SLE patients with kidney disease is now recruiting.
U.S. F.D.A. reviewers were concerned that belimumab is only “marginally” effective, and that there were more deaths in the treatment group.
Phase II trials of belimumab for rheumatoid arthritis were unsuccessful. Phase II trials for Sjögren’s Syndrome were more successful.
Belimumab was developed by Human Genome Sciences (HGS) and Cambridge Antibody Technology. GlaxoSmithKline acquired HGS, took belimumab through Phase III clinical trials, and markets belimumab.

Phase 3-Volasertib for acute myeloid leukaemia in patients ineligible for intensive induction therapy

Volasertib (BI 6727) for Acute myeloid leukaemia. is a cell cycle kinase inhibitor of polo-like kinases 1, 2 and 3. Volasertib inhibits cancer growth by disrupting cell division and inducing cell death. Volasertib is administered as a 350mg one hour intravenous (IV) infusion on days 1 and 15 of a 28 day cycle in combination with low-dose cytarabine (LDAC), administered via subcutaneous injection (SC) at 20mg twice daily on days 1-10 of a 28 day cycle.
Acute myeloid leukaemia (AML): previously untreated; patients considered ineligible for intensive remission induction therapy – first line; in combination with low-dose cytarabine
volasertib (also known as BI 6727) is a small molecule inhibitor of the PLK1 (polo-like kinase 1) protein being developed by Boehringer Ingelheim for use as an anti-cancer agent. Volasertib is the second in a novel class of drugs called dihydropteridinone derivatives.
Health Canada approves NEUPRO (Rotigotine) patch for treatment of Parkinson’s disease and Restless Legs Syndrome

Rotigotine
Novel dosage form represents new treatment for Parkinson’s with efficacy in motor symptoms of the disease
April 2,2013
UCB Canada Inc. announced today that Health Canada has approved NEUPRO® (rotigotine), the first and only non-ergolinic dopamine agonist available in a patch, to treat the signs and symptoms of idiopathic Parkinson’s disease (PD) and moderate-to-severe idiopathic restless legs syndrome (RLS), also known as Willis-Ekbom disease, in adults.
NEUPRO® is the first new treatment for Parkinson’s disease approved by Health Canada in five years and provides 24-hour delivery of rotigotine through the skin into the blood stream. NEUPRO® has demonstrated efficacy in managing motor symptoms associated with Parkinson’s disease.
“The ability to ensure a steady 24-hour delivery of medication with NEUPRO® may help to reduce debilitating on and off symptoms which many patients experience with Parkinson’s treatments,” says Dr. David Grimes, Director, Parkinson’s Disease and Movement Disorders Clinic at the Ottawa Hospital. “The impact of sustained symptom control in the morning and in the evening can have a substantial effect on a patient’s quality of life.”
UCB Canada Inc. is undertaking all measures required to supply the Canadian market with NEUPRO.
“Parkinson Society Canada is pleased to learn that Canadians living with Parkinson’s now have another treatment option to help manage the symptoms of this chronic disease,” says Joyce Gordon, President and CEO, Parkinson Society Canada. “With innovative therapies such as NEUPRO® and ongoing research into the causes of this disease, we will help to ensure a brighter future and better quality of life for Canadians living with Parkinson’s.”
Although the precise mechanisms of action of NEUPRO® as treatment for PD and RLS are unknown, as a PD treatment, the mechanism of action is thought to be related to increasing the activities of the dopamine receptors within the caudate-putamen, the region of the brain that regulates movement. Similarly, in RLS, the mechanism of action of NEUPRO® is thought to be related to its ability to stimulate dopamine receptors.
Data Demonstrated Significant Symptom Improvement for PD and RLS
The effectiveness of NEUPRO® (rotigotine) in the treatment of Parkinson’s disease was evaluated in a multinational drug development program consisting of four randomized, double-blind placebo-controlled phase 3 trials. A total of seven Canadian trial sites were involved in the international studies. In all trials, patients underwent a weekly titration of NEUPRO® in 2 mg/24 hour increments to the assigned or optimal dose.
In two trials, statistically significant improvements in the combined scores on the Unified Parkinson’s Disease Rating Scale (UPDRS) were observed in early-stage PD patients receiving NEUPRO® compared to patients receiving placebo. The UPDRS is a validated multi-item rating scale intended to evaluate mentation (mental activity), activities of daily living (ADL), motor performance, and complications of therapy. The two trials measured only the ADL and motor performance sections of the UPDRS. The UPDRS contains 13 questions relating to ADL, such as speech, dressing, and cutting food with utensils, and 27 questions related to the cardinal motor symptoms in PD patients—i.e., tremor, rigidity, bradykinesia, and postural instability.
Two trials of NEUPRO® in patients with advanced-stage PD examined change from baseline in “off” time, periods when the effectiveness of medication wears off and PD symptoms return. Statistically significant changes in off-times were observed in advanced-stage PD patients receiving NEUPRO® compared with those who received placebo.
The effectiveness of NEUPRO® in the treatment of Restless Legs Syndrome (RLS) was evaluated in two fixed-dose, randomized, double-blind, placebo-controlled phase 3 trials with maintenance periods of 6 months duration. Patients received NEUPRO® doses ranging from 0.5 mg/24 hours to 3 mg/24 hours, or placebo, once daily. Statistically significant improvements in sum scores on the International RLS Rating Scale (IRLS Scale) and the Clinical Global Impression – Improvement (CGI-I) assessment were observed in RLS patients receiving NEUPRO® compared with those receiving placebo. The IRLS Scale contains 10 items designed to assess the severity of sensory and motor symptoms, sleep disturbance, daytime somnolence, and impact on activities of daily living and mood associated with RLS. The CGI-I is designed to clinically assess RLS symptoms on a 7-point scale.
In clinical trials, treatment emergent adverse events reported in more than 10% of patients treated with NEUPRO® for Parkinson’s disease included nausea, vomiting, dizziness, somnolence, application site reactions and headache. Treatment emergent adverse events reported in more than 10% of patients treated with NEUPRO® for Restless Legs Syndrome, included nausea, application site reactions, fatigue and headache.
About Parkinson’s disease
Parkinson’s disease (PD) is a chronic, degenerative neurological disease which affects approximately 100,000 Canadians. PD develops with the loss of nerve cells in the brain that produce a chemical called dopamine. The symptoms of PD can have an impact on many dimensions of patients’ lives. As dopamine levels fall, movement (motor) symptoms—tremors (uncontrollable shaking), rigidity (stiffness or muscle tensing) and bradykinesia (slowness and loss of spontaneous movement) — can progress, along with the underlying symptoms of PD, which are less well recognized and may be under-treated.
About Restless Legs Syndrome
Restless Legs Syndrome (RLS) is a neurological disorder characterized by unpleasant sensations in the legs and an uncontrollable urge to move to gain relief. Over 80% of people with RLS also have periodic limb movement disorder (PLMD), which causes rhythmic limb movements during sleep. RLS affects between three and 10 per cent of the population to some extent. Some estimates are much higher because RLS is thought to be underdiagnosed, and in some cases, misdiagnosed. Most people with RLS have difficulty falling asleep and staying asleep. Daytime symptoms of RLS, such as inability to sit still and involuntary leg jerks, are increasingly recognized. While the underlying pathophysiology of RLS is not fully understood, it is thought to involve central dopamine systems. Recent neuroimaging data suggest that RLS patients may carry an abnormality in dopamine transport that can be visualized both day and night. RLS can cause exhaustion and daytime fatigue, and may affect work and personal relationships. Patients with moderate-to-severe RLS are often unable to concentrate, have impaired memory, or fail to accomplish daily tasks. These patients may require long-term treatment for their RLS symptoms.
About UCB Canada Inc.
Inspired by patients and driven by science, UCB Canada Inc. is a biopharmaceutical company focused on the discovery and development of innovative medicines and solutions to transform the lives of people living with severe auto-immune and central nervous system diseases. For more information, please consult www.ucb.com/worldwide/canada.
Rotigotine (Neupro) is a dopamine agonist of the non-ergoline class of medications indicated for the treatment of Parkinson’s disease (PD) and restless legs syndrome (RLS) in Europe and the United States. It is formulated as a once-daily transdermal patchwhich provides a slow and constant supply of the drug over the course of 24 hours.
Like other dopamine agonists, rotigotine has been shown to possess antidepressanteffects and may be useful in the treatment of depression as well.
Rotigotine was developed by Aderis Pharmaceuticals. In 1998, Aderis licensed worldwide development and commercialization rights for rotigotine to the German pharmaceutical company Schwarz Pharma (today a subsidiary of the Belgian company UCB S.A.).
The drug has been approved by the EMEA for use in Europe in 2006 and is today being sold in several European countries. In 2007, the Neupro patch was approved by the Food and Drug Administration (FDA) as the first transdermal treatment of Parkinson’s disease in the United States. However, as of 2008, Schwarz Pharma has recalled all Neupro patches in the United States and some in Europe because of problems with the delivery mechanism. The patch was reformulated, and was reintroduced in the United States in 2012.
Rotigotine has been authorized as a treatment for restless legs syndrome since August 2008.
Rotigotine is analogous to 7-OH-DPAT and UH-232, all three of which are aminotetralinderivatives. These compounds are similar in structure to dopamine, likely underlying theirpharmacology.
Cusack, N. J.; Peck, J. V.; Drugs Future 1993, 18, 1005.
FDA Approves Tris Pharma’s New Drug Application for Karbinal ER

Carbinoxamine Maleate
2-[p-Chloro-a-[2-(dimethylamino)ethoxy]benzyl]pyridine maleate (1:1) [3505-38-2].
April 3, 2013
Tris Pharma, a specialty pharmaceutical company focused on developing innovative drug delivery technologies, today announced that the U.S. Food and Drug Administration (FDA) has approved its New Drug Application (NDA) for Karbinal ER (carbinoxamine maleate) Extended-release Oral Suspension 4mg/5mL, the first sustained-release histamine receptor blocking agent indicated for the treatment of seasonal and perennial allergic rhinitis in children ages 2 and up.
“Karbinal ER is dosed only once every 12 hours, making it an attractive treatment option for the millions of allergy sufferers who don’t respond to second-generation antihistamines and aren’t satisfied with the cumbersome dosing schedules associated with the first-generation antihistamines,” said Ketan Mehta, founder, President, and CEO of Tris Pharma. “The approval of Karbinal ER marks our fourth liquid extended-release NDA approval based upon our proprietary OralXR+ technology. We are in the process of finalizing our marketing partner and plan to launch later this year in anticipation of the fall allergy season.”
Based on physician interviews approximately 30 percent of patients don’t get adequate relief from the non-sedating antihistamines. Carbinoxamine is a mildly-sedating antihistamine with years of proven safety and efficacy. Prior to 2006, carbinoxamine was widely used, with dozens of carbinoxamine-containing combination products including extended-release solid-dose products. However, nearly all of these were older products that hadn’t gone through the FDA’s rigorous approval process. Following the Drug Efficacy Study Implementation (DESI) review, the FDA removed all unapproved products with the exception of two immediate-release formulations, creating a void for patients and doctors who valued the benefits associated with an extended-release formulation.
Dr. Laura Garabedian, a New York-based pediatrician, said, “While I’ve always found carbinoxamine to be an effective option for treating the symptoms of allergies in children, the existing immediate-release formulations of carbinoxamine require dosing multiple times a day. This is especially inconvenient for children who are in school. Now, with the approval of Karbinal ER, I look forward to having an effective and great-tasting extended-release liquid formulation to offer patients as young as two years old.”
Karbinal ER Extended-release Oral Suspension is an H1 receptor antagonist indicated for the symptomatic treatment of:
- Seasonal and perennial allergic rhinitis
- Vasomotor rhinitis
- Allergic conjunctivitis due to inhalant allergens and foods
- Mild, uncomplicated allergic skin manifestations of urticaria and angioedema
- Dermatographism
- As therapy for anaphylactic reactions adjunctive to epinephrine and other standard measures after the acute manifestations have been controlled
- Amelioration of the severity of allergic reactions to blood or plasma
Tris Pharma is a specialty pharmaceutical company focused on the research and development of technologies-driven products. Tris has pioneered the delivery of sustained release in the liquid, chewable/ODT and strip dosage forms so patients do not have to swallow a pill. Tris’ Nobuse™ technology provides abuse deterrence for opioids and other abuse-prone drugs. Tris’ R&D and manufacturing facilities are located in Monmouth Junction, New Jersey, U.S.A. For more information, please visit http://www.trispharma.com.
Genmab: Daratumumab Granted Fast Track Designation By FDA

Monoclonal antibody
2/4/2013
Genmab A/S announced that the US Food and Drug Administration has granted Fast Track designation for daratumumab. This designation covers patients with multiple myeloma who have received at least three prior lines of therapy including a proteasome inhibitor (PI) and an immunomodulatory agent (IMiD) or are double refractory to a PI and an IMiD.
In August 2012, Genmab granted Janssen Biotech, Inc. an exclusive worldwide license to develop and commercialize daratumumab.
Daratumumab is an investigational anti-cancer drug. It binds to CD38.[1] Daratumumab was originally developed by Genmab, but it is now being jointly developed by Genmab along with the Johnson & Johnson subsidiary Janssen Biotech, which acquired worldwide commercialization rights to the drug from Genmab.[2]
Encouraging preliminary results were reported in June 2012 from a Phase 1/2 clinical trial in relapsed multiple myeloma patients.[3] Updated trial results presented in December 2012 indicate daratumumab is continuing to show promising single-agent anti-myeloma activity.[4]
- World Health Organization (2009). “International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 101” (PDF). WHO Drug Information 23 (2).
- “‘Janssen Biotech Announces Global License and Development Agreement for Investigational Anti-Cancer Agent Daratumumab'”. Janssen Biotech. Retrieved 2013-01-31.
- “ASCO: Drug Shows Promise in Myeloma”. MedPage Today.
- “‘Daratumumab Continues To Show Promise For Relapsed/Refractory Myeloma Patients (ASH 2012)'”. The Myeloma Beacon. Retrieved 2013-01-31.
AYURVEDA SPOTLIGHT- ASHWAGANDHA, THE STRESS BUSTER

Withania somnifera, also known as ashwagandha, Indian ginseng, poison gooseberry, or winter cherry, is a plant in the Solanaceae or nightshade family. Several other species in the genus Withania are morphologically similar. It is used as a herb inAyurvedic medicine.
| Ashwagandha root is known as “Indian Ginseng”. In Ayurvedic medicine it is considered an adaptogen that facilitates learning and memory. |
It grows as a short shrub (35–75 cm) with a central stem from which branches extend radially in a star pattern (stellate) and covered with a dense matte of wooly hairs (tomentose). The flowers are small and green, while the ripe fruit is orange-red and has milk-coagulating properties. The plant’s long, brown, tuberous roots are used for medicinal purposes.
Ashwagandha in Sanskrit means “horse’s smell” (ashwa– horse, gandha– smell), probably originating from the odour of its root which resembles that of a sweaty horse.
The species name somnifera means “sleep-inducing” in Latin
Cultivation

Ashwagandha plant with berries
Withania somnifera is cultivated in many of the drier regions of India, such as Mandsaur Districtof Madhya Pradesh, Punjab, Sindh, Gujarat,and Rajasthan.It is also found in Nepal
Withania somnifera is grown as late rainy-season (kharif) crop. Semitropical areas receiving 500 to 750 mm rainfall are suitable for its cultivation as a rainfed crop. If one or two winter rains are received, then root development improves.
The crop requires a relatively dry season during its growing period. It can tolerate a temperature range of 20 to 38°C and as low a temperature as 10°C. The plant grows from sea level to an altitude of 1500 meters.

Ashvagandha plant at Talkatora Gardens, Delhi
Withania somnifera is prone to several pests and diseases. Leaf spot disease caused byAlternaria alternata is the most prevalent disease, which is most severe in the plains ofPunjab, Haryana, and Himachal Pradesh. Biodeterioration of its pharmaceutically active components during leaf spot disease has been reported. The Choanephora cucurbitarumcauses a stem and leaf rot of Withania somnifera Oxyrachis tarandus (atreehopper/cowbug species) feeds on the apical portions of the stem, making them rough and woody in appearance and brown in colour. The apical leaves are shed and the plant gradually dies away. Carmine red spider mite (Tetranychus urticae) is the most prevalent pest of Withania somnifera in India.
The berries can be used as a substitute for rennet, to coagulate milk in cheese-making
The main chemical constituents are alkaloids and steroidal lactones. These include tropineand cuscohygrine. The leaves contain the steroidal lactones, withanolides, notably withaferin A, which was the first withanolide to be isolated from W. somnifera.
Traditional medicinal uses
In Ayurveda, the berries and leaves of W. somnifera are locally applied to tumors, tubercular glands, carbuncles, and ulcers.[5] The roots of W. somnifera are used to prepare the herbal remedy ashwagandha, which has been traditionally used to treat various symptoms and conditions.
Side effects
In two published clinical trials of W. somnifera, the side effects were not significantly different from those experienced by placebo-treated individuals.
Ashwagandha Scientific Research
Ashwagandha contains many useful medicinal chemicals, including withanolides, (steroidal lactones), alkaloids, choline, fatty acids, amino acids, and a variety of sugars. While the leaves and fruit have valuable therapeutic properties, the root of the ashwagandha plant is the part most commonly used in Western herbal remedies.
Medical researchers have been studying ashwagandha with great interest and as of this date have carried out 216 studies of its healing benefits, summarized below:
- confers immune system protection
- combats the effects of stress
- improves learning, memory, and reaction time
- reduces anxiety and depression without causing drowsiness
- stabilizes blood sugar
- lowers cholesterol
- reduces brain-cell degeneration
- contains anti-malarial properties
- offers anti-inflammatory benefits
Some studies have also found that ashwagandha inhibits the growth of cancer cells in small animals, but further research is needed to determine whether the herb prevents the development of tumors in human beings.
Ashwagandha in particular is known for its ability to calm, and some research indicates this herb can be used to promote sleep. In Texas, researchers noted the similarities in the sleep-inducing properties of ashwagandha and the calming effects of the well-known amino acid GABA. Likewise, ashwagandha has also been shown to ease anxiety or restlessness, as well as to reduce the symptoms of drug withdrawal. Its ability to stabilize moods and encourage adrenal recovery is highly valued by many herbalists.
But the benefits of ashwagandha extend far beyond mood. In India it is also used to help older patients with mental agility, cognitive ability, and memory. It is also known for its ability to fight off cold and cough symptoms. Preliminary studies give researchers reason to feel that ashwagandha also has the potential ability to decrease cancer cells without adversely affecting healthy cells.
Ashwagandha Practical and Precautions
The usual recommended dose is 600 to 1000 mg, twice daily. For people who suffer from insomnia and anxiety, having a cup of hot milk that contains a teaspoon of powdered ashwagandha before bedtime is beneficial. In extremely large doses, ashwagandha has been reported to induce abortions in animals. Although no similar studies have been carried out on humans, women should avoid the herb during pregnancy.You should consult your ayurvedic doctor or other health care professional before starting on any ayurveda treatment.
Other Name:
Ajagandha, Amangura, Amukkirag, Asan, Asana, Asgand, Asgandh, Asgandha, Ashagandha, Ashvagandha, Ashwaganda, Ashwanga, Asoda, Asundha, Asvagandha, Aswagandha, Avarada, Ayurvedic Ginseng, Cerise d’Hiver, Clustered Wintercherry, Ghoda Asoda, Ginseng Ayurvédique, Ginseng Indien, Hayahvaya, Indian Ginseng, Kanaje Hindi, Kuthmithi, Orovale, Peyette, Physalis somnifera, Samm Al Ferakh, Samm Al Rerakh, Sogade-Beru, Strychnos, Turangi-Ghanda, Vajigandha, Winter Cherry, Withania, Withania somnifera.

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....





















 +
+

 +
+









