Home » Posts tagged 'TAIHO'
Tag Archives: TAIHO
Taiho’s Colon Cancer Drug Ups OS in Phase 3

TAS-102 (nonproprietary names: trifluridine and tipiracil hydrochloride)
Taiho’s Colon Cancer Drug Ups OS in Phase 3
Taiho Pharmaceutical Co. Ltd. announced results from its global Phase 3 RECOURSE trial on its oral combination anticancer drug TAS-102 in refractory metastatic colorectal cancer (mCRC). Read more…
FULL STORY
TAS-102 is an anti-cancer drug under development for colorectal cancer.[1]
Clinical trials
A phase II trial reported in 2011[2] and a phase III trial is due to end in 2014.[1][3]
Mechanism
TAS-102 consists of the cytotoxin trifluridine and the thymidine phosphorylase inhibitor (TPI) tipiracil.[4] Trifluridine is incorporated into DNA during DNA synthesis and inhibits tumor cell growth. Tipiracil protects trifluridine from being broken down when taken orally.[1]
References
- “New Drug for Colorectal Cancer Shows Promise in Phase II Trial”. 28 Aug 2012.
- “Novel Drug TAS-102 Makes Headway in Refractory Colorectal Cancer”. 4 Oct 2011.
- “Phase II study of TAS-102 for pretreated metastatic colorectal cancer”. 29 Aug 2012.
- “A novel combination antimetabolite, TAS-102, exhibits antitumor activity in FU-resistant human cancer cells through a mechanism involving FTD incorporation in DNA.”. Sep 2004.

Trifluridine
Trifluridine (also called trifluorothymidine or TFT) is an anti-herpesvirus antiviral drug, used primarily on the eye. It was sold under the trade name, Viroptic, by Glaxo Wellcome, now merged into GlaxoSmithKline. The brand is now owned by Monarch Pharmaceuticals, which is wholly owned by King Pharmaceuticals.
It is a nucleoside analogue, a modified form of deoxyuridine, similar enough to be incorporated into viral DNA replication, but the -CF3 group added to the uracil component blocks base pairing.
It is a component of the experimental anti-cancer drug TAS-102.
A Cochrane Systematic Review showed that trifluridine was a more effective treatment than idoxuridine or vidarabine, significantly increasing the relative number of successfully healed eyes in 14 days.[1]
References
- Wilhelmus KR (2010). “Antiviral treatment and other therapeutic interventions for herpes simplex virus epithelial keratitis”. Cochrane Database Syst Rev 12: CD002898. doi:10.1002/14651858.CD002898.pub4. PMID 21154352.
External links
- Costin D, Dogaru M, Popa A, Cijevschi I (2004). “Trifluridine therapy in herpetic in keratitis”. Rev Med Chir Soc Med Nat Iasi 108 (2): 409–12. PMID 15688823.
- Kuster P, Taravella M, Gelinas M, Stepp P (1998). “Delivery of trifluridine to human cornea and aqueous using collagen shields.”. CLAO J 24 (2): 122–4. PMID 9571274.
- O’Brien W, Taylor J (1991). “Therapeutic response of herpes simplex virus-induced corneal edema to trifluridine in combination with immunosuppressive agents.”. Invest Ophthalmol Vis Sci 32 (9): 2455–61. PMID 1907950.
- “Trifluridine Ophthalmic Solution, 1%” (PDF). Retrieved 2007-03-24.

Fig 4. Open Babel bond-line chemical structure with annotated hydrogens. Click to toggle size.
Spectrum Plot
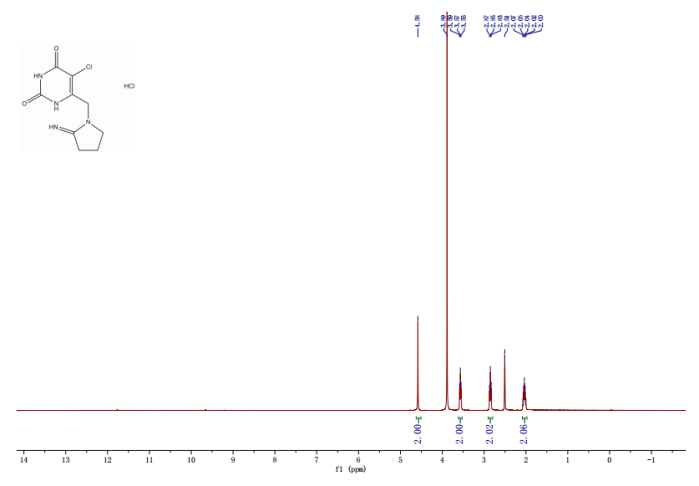
Fig 5. 1H NMR spectrum of C10H11F3N2O5 in CDCL3 at 400 MHz

Tipiralacil, also known as TPI, is a thymidine phosphorylase inhibitor (TPI). Tipiracil is one of the active components in TAS-102, which is an anticancer drug candidate currently in clinical trials. TAS-102 consists of the cytotoxin Trifluridine and the thymidine phosphorylase inhibitor (TPI) tipiracil. Trifluridine is incorporated into DNA during DNA synthesis and inhibits tumor cell growth. Tipiracil protects trifluridine from being broken down when taken orally.
183204-72-0 (Tipiracil -HCl); 183204-74-2(Tipiracil ).
|
References |
1: Peters GJ, Bijnsdorp IV. TAS-102: more than an antimetabolite. Lancet Oncol. 2012 Dec;13(12):e518-9. doi: 10.1016/S1470-2045(12)70426-6. PubMed PMID: 23182191.
2: Yoshino T, Mizunuma N, Yamazaki K, Nishina T, Komatsu Y, Baba H, Tsuji A, Yamaguchi K, Muro K, Sugimoto N, Tsuji Y, Moriwaki T, Esaki T, Hamada C, Tanase T, Ohtsu A. TAS-102 monotherapy for pretreated metastatic colorectal cancer: a double-blind, randomised, placebo-controlled phase 2 trial. Lancet Oncol. 2012 Oct;13(10):993-1001. doi: 10.1016/S1470-2045(12)70345-5. Epub 2012 Aug 28. PubMed PMID: 22951287.
3: Sobrero A. TAS-102 in refractory colorectal cancer: caution is needed. Lancet Oncol. 2012 Oct;13(10):959-61. doi: 10.1016/S1470-2045(12)70376-5. Epub 2012 Aug 28. PubMed PMID: 22951286.
4: Doi T, Ohtsu A, Yoshino T, Boku N, Onozawa Y, Fukutomi A, Hironaka S, Koizumi W, Sasaki T. Phase I study of TAS-102 treatment in Japanese patients with advanced solid tumours. Br J Cancer. 2012 Jul 24;107(3):429-34. doi: 10.1038/bjc.2012.274. Epub 2012 Jun 26. PubMed PMID: 22735906; PubMed Central PMCID: PMC3405214.
5: Suzuki N, Nakagawa F, Nukatsuka M, Fukushima M. Trifluorothymidine exhibits potent antitumor activity via the induction of DNA double-strand breaks. Exp Ther Med. 2011 May;2(3):393-397. Epub 2011 Mar 21. PubMed PMID: 22977515; PubMed Central PMCID: PMC3440718.
6: Shintani M, Urano M, Takakuwa Y, Kuroda M, Kamoshida S. Immunohistochemical characterization of pyrimidine synthetic enzymes, thymidine kinase-1 and thymidylate synthase, in various types of cancer. Oncol Rep. 2010 May;23(5):1345-50. PubMed PMID: 20372850.
7: Temmink OH, Bijnsdorp IV, Prins HJ, Losekoot N, Adema AD, Smid K, Honeywell RJ, Ylstra B, Eijk PP, Fukushima M, Peters GJ. Trifluorothymidine resistance is associated with decreased thymidine kinase and equilibrative nucleoside transporter expression or increased secretory phospholipase A2. Mol Cancer Ther. 2010 Apr;9(4):1047-57. doi: 10.1158/1535-7163.MCT-09-0932. Epub 2010 Apr 6. PubMed PMID: 20371715.
8: Bijnsdorp IV, Kruyt FA, Fukushima M, Smid K, Gokoel S, Peters GJ. Molecular mechanism underlying the synergistic interaction between trifluorothymidine and the epidermal growth factor receptor inhibitor erlotinib in human colorectal cancer cell lines. Cancer Sci. 2010 Feb;101(2):440-7. doi: 10.1111/j.1349-7006.2009.01375.x. Epub 2009 Sep 29. PubMed PMID: 19886911.
9: Bijnsdorp IV, Peters GJ, Temmink OH, Fukushima M, Kruyt FA. Differential activation of cell death and autophagy results in an increased cytotoxic potential for trifluorothymidine compared to 5-fluorouracil in colon cancer cells. Int J Cancer. 2010 May 15;126(10):2457-68. doi: 10.1002/ijc.24943. PubMed PMID: 19816940.
10: Bijnsdorp IV, Kruyt FA, Gokoel S, Fukushima M, Peters GJ. Synergistic interaction between trifluorothymidine and docetaxel is sequence dependent. Cancer Sci. 2008 Nov;99(11):2302-8. doi: 10.1111/j.1349-7006.2008.00963.x. Epub 2008 Oct 18. PubMed PMID: 18957056.
11: Overman MJ, Kopetz S, Varadhachary G, Fukushima M, Kuwata K, Mita A, Wolff RA, Hoff P, Xiong H, Abbruzzese JL. Phase I clinical study of three times a day oral administration of TAS-102 in patients with solid tumors. Cancer Invest. 2008 Oct;26(8):794-9. doi: 10.1080/07357900802087242. PubMed PMID: 18798063.
12: Overman MJ, Varadhachary G, Kopetz S, Thomas MB, Fukushima M, Kuwata K, Mita A, Wolff RA, Hoff PM, Xiong H, Abbruzzese JL. Phase 1 study of TAS-102 administered once daily on a 5-day-per-week schedule in patients with solid tumors. Invest New Drugs. 2008 Oct;26(5):445-54. doi: 10.1007/s10637-008-9142-3. Epub 2008 Jun 5. PubMed PMID: 18528634.
13: Temmink OH, Emura T, de Bruin M, Fukushima M, Peters GJ. Therapeutic potential of the dual-targeted TAS-102 formulation in the treatment of gastrointestinal malignancies. Cancer Sci. 2007 Jun;98(6):779-89. Epub 2007 Apr 18. Review. PubMed PMID: 17441963.
14: Temmink OH, Hoebe EK, van der Born K, Ackland SP, Fukushima M, Peters GJ. Mechanism of trifluorothymidine potentiation of oxaliplatin-induced cytotoxicity to colorectal cancer cells. Br J Cancer. 2007 Jan 29;96(2):231-40. PubMed PMID: 17242697; PubMed Central PMCID: PMC2360012.
Taiho Pharma seeks Japanese nod to manufacture,market novel anti-tumour agent TAS-102
TRIFLURIDINE

TIPIRACIL
TAS-102 is an anti-cancer drug under development for colorectal cancer.[1]
| Combination of | |
|---|---|
| Trifluridine | cytotoxin |
| Tipiracil | thymidine phosphorylase inhibitor |
Clinical trials
A phase II trial reported in 2011[2] and a phase III trial is due to end in 2014.[1][3]
Mechanism
TAS-102 consists of the cytotoxin trifluridine and the thymidine phosphorylase inhibitor (TPI) tipiracil.[4] Trifluridine is incorporated into DNA during DNA synthesis and inhibits tumor cell growth. Tipiracil protects trifluridine from being broken down when taken orally.
| February 28, 2013, |
|
Taiho Pharmaceutical Co., Ltd. has submitted an application to the Japanese Ministry of Health, Labour and Welfare for approval of the manufacture and marketing of the novel oral nucleoside anti-tumour agent TAS-102 (combination of trifluorothymidine [FTD] and tipiracil hydrochloride [TPI]). Taiho is seeking approval of TAS-102 for the indication of unresectable, advanced, recurrent colorectal cancer. The application for approval is based on the results of a phase II clinical trial (Study 10040030) conducted at 20 facilities throughout Japan. It was a randomized, double-blind comparative study of TAS-102 and a placebo involving 172 patients with unresectable, advanced, recurrent colorectal cancer that was refractory to the standard chemotherapy of at least two or more regimens containing fluoropyrimidine, irinotecan, and oxaliplatin. The results indicated that the group administered TAS-102 had improved overall survival rates (median overall survival: 9.0 months vs. 6.6 months) and a significantly reduced risk of mortality (HR: 0.56, p=0.0011). The most frequently reported adverse drug reaction with a CTCAE grade of 3 or higher was neutropenia. Grade 3 or higher diarrhea, fatigue, nausea, and other adverse reactions were no more than 10 per cent. Taiho Pharmaceutical is currently proceeding with a global phase III clinical trial of TAS-102 in a similar colorectal cancer population (RECOURSE) with the ultimate goal of global registration and commercialization of the agent. Taiho Pharmaceutical believes that TAS-102 will make a significant contribution to cancer patients and will continue its development efforts to broaden its use. TAS-102 is an anti-tumour agent composed of a combination of trifluorothymidine (FTD), a nucleoside that incorporates into DNA and inhibits a variety of genetic functions required for the proliferation of cancer cells, and tipiracil hydrochloride (TPI), an inhibitor of thymidine phosphorylase (which degrades FTD) that maintains an effective blood concentration of FTD. TAS-102 is administered twice daily to achieve a total daily dose of 70mg/m2 for five days followed by two days of rest and then repeated a second time. This is followed by a 14-day rest period to make a 28-day schedule for one course. |
- “New Drug for Colorectal Cancer Shows Promise in Phase II Trial”. 28 Aug 2012.
- “Novel Drug TAS-102 Makes Headway in Refractory Colorectal Cancer”. 4 Oct 2011.
- “Phase II study of TAS-102 for pretreated metastatic colorectal cancer”. 29 Aug 2012.
- “A novel combination antimetabolite, TAS-102, exhibits antitumor activity in FU-resistant human cancer cells through a mechanism involving FTD incorporation in DNA.”. Sept 2004.
 |
|
|---|---|
| TRIFLURIDINE | |
| 1-[4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-5- (trifluoromethyl) pyrimidine-2,4-dione |
Trifluridine (also called trifluorothymidine or TFT) is an anti-herpesvirus antiviral drug, used primarily on the eye. It was sold under the trade name, Viroptic, by Glaxo Wellcome, now merged into GlaxoSmithKline. The brand is now owned by Monarch Pharmaceuticals, which is wholly owned by King Pharmaceuticals.
It is a nucleoside analogue, a modified form of deoxyuridine, similar enough to be incorporated into viral DNA replication, but the -CF3 group added to the uracil component blocks base pairing.
It is a component of the experimental anti-cancer drug TAS-102.
TIPIRACIL
| NAME | 5-chloro-6-[(2-iminopyrrolidin-1-yl)methyl]pyrimidine-2,4-(1H,3H)-dione | |||
| CAS | 183204-74-2 | |||
| MOL F | C9H11ClN4O2 | |||
| STR |  |
|||
| USE | potentiator of antineoplastics; | |||
Taiho Pharmaceutical, a subsidiary of Otsuka Holdings Co., Ltd., is an R&D-driven specialty pharma focusing on the three fields of oncology, allergies and immunology, and urology.

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....