Home » Posts tagged 'organic chemistry' (Page 34)
Tag Archives: organic chemistry
Flamel Technologies Announces FDA Approval of Bloxiverz

LYON, FRANCE — (Marketwire) — 06/03/2013 — Flamel Technologies today announced that the U.S. Food and Drug Administration (FDA) has approved the company’s New Drug Application (NDA) for Bloxiverz (neostigmine methylsulfate), a drug used intravenously in the operating room for the reversal of the effects of non-depolarizing neuromuscular blocking agents after surgery. Flamel expects to launch Bloxiverz in July 2013 in 0.5 and 1.0 mg/mL strengths
read all at
.http://www.drugs.com/newdrugs/flamel-technologies-announces-fda-approval-bloxiverz-3802.html
Sunovion Pharmaceuticals Inc. Announces FDA Approval of Latuda® (lurasidone HCl) as Monotherapy and Adjunctive Therapy in Adult Patients with Bipolar Depression

lurasidone
MARLBOROUGH, Mass. – Monday, July 1st 2013 [ME NewsWire]
First Atypical Antipsychotic Indicated for the Treatment of Major Depressive Episodes Associated with Bipolar I Disorder (Bipolar Depression) Both as Monotherapy and as Adjunctive Therapy with Either Lithium or Valproate
(BUSINESS WIRE)– Sunovion Pharmaceuticals Inc. today announced that the U.S. Food and Drug Administration (FDA) approved two new indications for the use of Latuda® (lurasidone HCl) as 1) monotherapy and 2) adjunctive therapy with either lithium or valproate, both to treat adult patients with major depressive episodes associated with bipolar I disorder (bipolar depression).
read all at
http://www.me-newswire.net/news/7829/en
Lurasidone (trade name Latuda) is an atypical antipsychotic developed by Dainippon Sumitomo Pharma and marketed by Sunovion in the USA. It was approved by the U.S. Food and Drug Administration (FDA) for treatment of schizophrenia on October 28, 2010 after a review that found that two of the four Phase III clinical trials supported efficacy, while one showed only marginal efficacy and one was not interpretable because of high drop-out rates. It is currently pending approval for the treatment of bipolar disorder in the United States. It was launched in Canada for the treatment of schizophrenia in September 2012, Health Canada giving their Summary Basis of Decision (SBD) as favourable on October 15, 2012
FDA Approves Brisdelle, paroxetine mesylate- First Non-Hormonal Treatment for Hot Flashes Associated with Menopause
June 28, 2013 –The U.S. Food and Drug Administration today approved Brisdelle (paroxetine) to treat moderate to severe hot flashes (vasomotor symptoms) associated with menopause. Brisdelle, which contains the selective serotonin reuptake inhibitor paroxetine mesylate, is currently the only non-hormonal treatment for hot flashes approved by the FDA.
There are a variety of FDA-approved treatments for hot flashes, but all contain either estrogen alone or estrogen plus a progestin.
read all at
more info
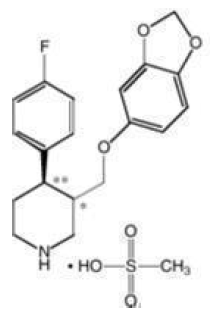
PEXEVA® (paroxetine mesylate) is an orally administered psychotropic drug with a chemical structure related to paroxetine hydrochloride (Paxil®). It is the mesylate salt of a phenylpiperidine compound identified chemically as (-)-trans-4R-(4′-fluorophenyl)-3S-[(3′,4′-methylenedioxyphenoxy) methyl] piperidine mesylate and has the empirical formula of C19H20FNO3•CH3SO3H. The molecular weight is 425.5 (329.4 as free base). The structural formula is: paroxetine mesylate
 |
Paroxetine mesylate is an odorless, off-white powder, having a melting point range of 147° to 150°C and a solubility of more than 1 g/ml in water.
Tablets
Each oval, film-coated tablet contains paroxetine mesylate equivalent to paroxetine as follows: 10 mg (white); 20 mg (scored, dark orange); 30 mg (yellow); 40 mg (rose). Inactive ingredients consist of dibasic calcium phosphate, hydroxypropyl methylcellulose, hydroxypropylcellulose, magnesium stearate, sodium starch glycolate, titanium dioxide, ferric oxide red (C.I. 77491) (20 mg and 40 mg only) and ferric oxide yellow (C.I. 77492) (20 mg, 30 mg, and 40 mg only).
EP1286965B1



Tobramycin

Tobramycin is an aminoglycoside antibiotic derived from Streptomyces tenebrarius and used to treat various types of bacteria infections, particularly Gram-negative infections. It is especially effective against species of Pseudomonas.[1]

Tobramycin works by binding to a site on the bacterial 30S and 50S ribosome, preventing formation of the 70S complex. As a result, mRNA cannot be translated into protein and cell death ensues. Tobramycin is preferred over gentamicin for Pseudomonas aeruginosapneumonia due to better lung penetration.

Like all aminoglycosides, tobramycin does not pass the gastro-intestinal tract, so forsystemic use it can only be given intravenously or intramuscularly. Ophthalmic (tobramycin only, Tobrex, or combined with dexamethasone, sold as TobraDex) and nebulised formulations both have low systemic absorption. The formulation for injection is branded Nebcin. The nebulised formulation (brand name Tobi) is indicated in the treatment of exacerbations of chronic infection with Pseudomonas aeruginosa in patients diagnosed with cystic fibrosis. A proprietary formulation of micronized, nebulized tobramycin has been tested as a treatment for bacterial sinusitis.[2] Tobrex is a 0.3% tobramycin sterile ophthalmic solution is produced by Bausch & Lomb Pharmaceuticals. Benzalkonium chloride 0.01% is added as a preservative. It is available by prescription only in the United States and Canada. In certain countries, such as Italy, it is available over the counter. Tobrex and TobraDex are indicated in the treatment of superficial infections of the eye, such as bacterial conjunctivitis. Tobramycin (injection) is also indicated for various severe or life-threatening gram-negative infections : meningitis in neonates, brucellosis, pelvic inflammatory disease, Yersinia pestis infection (plague).

Like other aminoglycosides, tobramycin is ototoxic: it can cause hearing loss, or a loss ofequilibrioception, or both in genetically susceptible individuals. These individuals carry a normally harmless genetic mutation that allows aminoglycosides such as tobramycin to affect cochlear cells. Aminoglycoside-induced ototoxicity is generally irreversible.

As with all aminoglycosides, tobramycin is also nephrotoxic, meaning it is toxic to thekidneys. This effect can be particularly worrisome when multiple doses accumulate over the course of a treatment or when the kidney concentrates urine by increasing tubular reabsorption during sleep. Adequate hydration may help prevent excess nephrotoxicity and subsequent loss of renal function. For these reasons parenteral tobramycin needs to be carefully dosed by body weight, and its serum concentration monitored. Tobramycin is thus said to be a drug with a narrow therapeutic index.
|
Mass-spectrum of tobramycin |
|
|
- “Tobramycin” (pdf). Toku-E. 2010-01-12. Retrieved 2012-06-11.
- “Nebulized Tobramycin in treating bacterial Sinusitis” (Press release). July 22, 2008. Retrieved 2009-12-06.


GSK Duchenne drug gets ‘breakthrough’ status

The US Food and Drug Administration has granted breakthrough therapy designation to GlaxoSmithKline’s drisapersen for the potential treatment of patients with Duchenne muscular dystrophy. read all at
http://www.pharmatimes.com/Article/13-06-28/GSK_Duchenne_drug_gets_breakthrough_status.aspx
Drisapersen (also known as PRO051 and GSK2402968 ) is an experimental drug under development by Prosensa for the treatment of Duchenne muscular dystrophy.
The compound is in a Phase III trial which is anticipated to complete by the end of 2013.
- ^ “PRO051/GSK2402968”. Prosensa. Retrieved 29 October 2012
- ^ “A Clinical Study to Assess the Efficacy and Safety of GSK2402968 in Subjects With Duchenne Muscular Dystrophy (DMD114044)”. ClinicalTrials.gov. Retrieved 29 October 2012.

Lyxumia approved in Japan for the treatment of type 2 diabetes

READ ALL AT
OLD ARTICLE

WORLD DRUG TRACKER group, linkedin, blog etc
World Drug Tracker
Labels: anthony crasto, catalyst, DRUGS, medicinal chemistry, New Drugs, organic chemistry, ORGANIC SYNTHESIS, PATENTS, process, world drug tracker …
WORLD DRUG TRACKER | LinkedIn
Follow World Drug Tracker on bloglovin
Follow World Drug Tracker using bloglovin, an easy way to follow your favorite blogs.
Navidea starts clinical trial for Alzheimer’s diagnostic drug

AZD4694, NAV4694 STRUCTURE
Navidea starts clinical trial for Alzheimer’s diagnostic drug
Business First of Columbus
The Phase 3 trial for the Alzheimer’s agent, at the moment named NAV4694, will compare how well the drugdisplays the buildup of a damaging protein in the brain of patients believed to have Alzheimer’s compared with what’s found in the autopsy. There …
read all at
http://www.bizjournals.com/columbus/news/2013/06/27/navidea-starts-clinical-trial-for.html
http://jnm.snmjournals.org/content/54/6/880.abstract
Navidea Biopharmaceuticals, a Dublin, Ohio biopharmaceutical company focused on precision diagnostics, earlier this week announced the completion of a study of its novel radiopharmaceutical NAV4694 as a biomarker for Alzheimer’s disease (AD).
 NAV4694 is designed to aid visual detection and quantification of cerebral beta amyloid in diagnosing Alzheimer’s disease (AD). One hallmark of AD is the accumulation of beta amyloid plaques between nerve cells in the brain.
NAV4694 is designed to aid visual detection and quantification of cerebral beta amyloid in diagnosing Alzheimer’s disease (AD). One hallmark of AD is the accumulation of beta amyloid plaques between nerve cells in the brain.
The study was designed and conducted by Navidea’s partner, AstraZeneca, to assess the safety and of the biomarker during PET scanning in subjects with AD and in healthy volunteers. Efficacy measures included binding parameters and overall image quality. The 16-patient trial was completed at Karolinska Institutet sites in Stockholm, Sweden.
FDA Approves Rixubis – First Recombinant Coagulation Factor IX For Use in Preventing Bleeding Episodes

Rixubis [Coagulation Factor IX (Recombinant)]
June 27, 2013 — The U.S. Food and Drug Administration yesterday approved Rixubis [Coagulation Factor IX (Recombinant)] for use in people with hemophilia B who are 16 years of age and older. Rixubis is indicated for the control and prevention of bleeding episodes, perioperative (period extending from the time of hospitalization for surgery to the time of discharge) management, and routine use to prevent or reduce the frequency of bleeding episodes (prophylaxis).
read all at


 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
















