 .
.
Picture credit….Bethany Halford

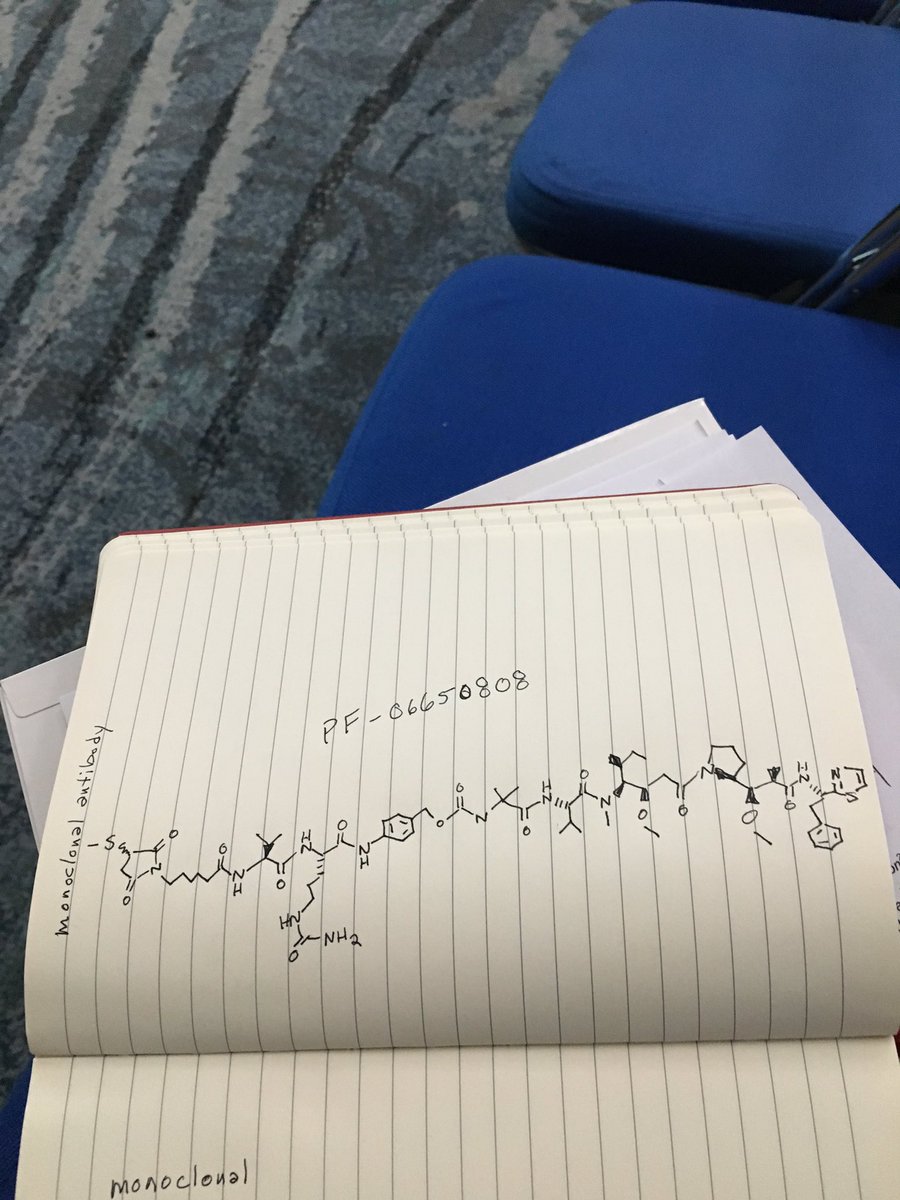
PF 06650808
CAS 1822383-80-1
A biologic for cancer treatment (Pfizer Inc.)
- Originator Pfizer
- Class Antineoplastics
- Mechanism of Action Notch-3 receptor antagonists
- No development reported Solid tumours
- 24 Jun 2018 Biomarkers information updated
- 28 Apr 2018 No recent reports of development identified for phase-I development in Solid-tumours(Late-stage disease) in USA (IV)
- 01 Jul 2017 Pfizer completes a phase I trial in Solid tumours (Late-stage disease) in USA (IV) (NCT02129205)
Company: Pfizer
Target: Neurogenic locus notch homolog protein 3 (NOTCH3): Activation and mutation of the NOTCH signaling pathway can lead to cancer.
Disease: Cancer
Notes: PF06650808 is an antibody-drug conjugate that delivers a cytotoxic payload molecule directly to tumor cells, explained Andreas Maderna, an associate research fellow at Pfizer. The payload molecule in PF06650808 was inspired by the marine natural product dolostatin 10, which is produced by cyanobacteria consumed by a type of sea slug.
https://cen.acs.org/articles/94/i15/New-drug-candidates-shine-San-Diego.html
PATENT
WO 2015171907
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2015171907
The present invention relates to stable isotopic identification of biologic products, methods of stable isotopic identification of such biologic products, and stable isotopic methods and systems for correlating biologic products to the processes by which they are made.
PATENT
WO 2018045058
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2018045058&tab=PCTDESCRIPTION&maxRec=1000
CLIP
Rosen, L.S.; Wesolowski, R.; Gibson, B.; et al.
A Phase 1 dose escalation, safety, and pharmacokinetic study of PF-06650808, an anti-Notch3 antibody drug conjugate, in adult patients with advanced solid tumors
Eur Cancer Congr (September 25-29, Vienna) 2015, Abst 3OLBA
Maderna, A.
Therapeutic targeting the NOTCH3 receptor with antibody drug conjugates
251st Am Chem Soc (ACS) Natl Meet (March 13-17, San Diego) 2016, Abst MEDI 262
Hurvitz, S.A.; von Euw, E.; O’Brien, N.; et al.
Preclinical evaluation of targeting Notch-3 in breast cancer
107th Annu Meet Am Assoc Cancer Res (AACR) (April 16-20, New Orleans) 2016, Abst 1206
Chen, J.; Geles, K.; Silva, M.; Waterhouse, R.; Ma, D.; Charati, M.; Sapra, P.; Mccarthy, T.
Evaluate the impact of conjugation on targeting capacity, pharmacokinetics and tissue distribution of antibody drug conjugate, PF-06650808, in tumor bearing mice
22nd Int Symp Radiopharm Sci (ISRS) (May 14-19, Dresden) 2017, Abst P 052
///////////
PF 06650808
| Phase 1 |
$PFE compound inspired by auristatins
https://clinicaltrials.gov/ct2/show/NCT02129205
http://www.pfizer.com/sites/default/files/product-pipeline/8_7_2014_Pipeline_Update.pdf
ALL DATA COMING………
Notch-3 receptor antagonists
Neoplasms
Breast
| Pfizer |
Cancer
PF-06650808, is currently being examined in a Ph1 clinical trial (Protocol B7501001).
Notch3
Researchers are also exploring the use of Notch3 targeting. “The Notch pathway plays an important role in the growth of several solid tumours, including breast and ovarian cancer and melanoma,” explained Joerger. “In particular, Notch3 alterations such as gene amplification and upregulation are associated with poor patient survival. Research using Notch3 targeting as an innovative approach to treat solid malignancies included 27 patients unselected for Notch3 who received increasing doses of the anti-Notch3 antibody-drug conjugate PF-06650808. Responses were seen in two breast cancer patients (LBA 30). While preliminary, targeting Notch3 may become a new treatment approach in patients with selected solid tumours.”
The anti-Notch3 antibody-drug conjugate PF-06650808 is being developed by Pfizer.
- 31 Jul 2014 Phase-I clinical trials in Solid tumours (Late-stage disease) in USA (Parenteral)
- 30 Apr 2014 Preclinical trials in Solid tumours in USA (Parenteral)
- 30 Apr 2014 Pfizer plans a phase I trial for Solid tumours (late-stage disease, second-line therapy or greater) in USA (NCT02129205)
251st Am Chem Soc (ACS) Natl Meet (March 13-17, San Diego) 2016, Abst MEDI 262
/////////PF 06650808, PF-06650808, PF-6650808, monoclonal antibody, pfizer, phase 1, Solid tumours , Notch-3 receptor antagonists
C1(C(N(C(C1)=O)CCCCCC(=O)NC([C@H](C)C)C(=O)NC(C(=O)Nc2ccc(cc2)COC(=O)NC(C)(C)C(=O)N[C@@H](C(C)C)C(=O)[N@](C)C(C(CC)C)[C@@H](OC)CC(=O)N3CCC[C@H]3C(OO)C(C)C(=O)N[C@H](c4nccs4)CC)CCCNC(=O)N)=O)SC
 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE
 amcrasto@gmail.com
amcrasto@gmail.com
P.S
THE VIEWS EXPRESSED ARE MY PERSONAL AND IN NO-WAY SUGGEST THE VIEWS OF THE PROFESSIONAL BODY OR THE COMPANY THAT I REPRESENT, amcrasto@gmail.com, +91 9323115463 India.
I , Dr A.M.Crasto is writing this blog to share the knowledge/views, after reading Scientific Journals/Articles/News Articles/Wikipedia. My views/comments are based on the results /conclusions by the authors(researchers). I do mention either the link or reference of the article(s) in my blog and hope those interested can read for details. I am briefly summarising the remarks or conclusions of the authors (researchers). If one believe that their intellectual property right /copyright is infringed by any content on this blog, please contact or leave message at below email address amcrasto@gmail.com. It will be removed ASAP






